Abstract
Recent studies suggest a possible association of hyponatremia with osteoporosis, falls and bone fractures. The objectives of this narrative review were to further explore this association and the related pathophysiological mechanisms and to suggest a practical approach to patients with osteoporosis or chronic hyponatremia in clinical practice. We conducted an extensive PubMed search until October 2022 with the combination of the following keywords: ‘hyponatremia’ or ‘sodium’ or ‘SIADH’ and ‘fractures’ or ‘bone’ or ‘osteoporosis’, as MeSH Terms. Review of numerous observational studies confirms a significant independent association of, even mild, hyponatremia with two- to three-fold increase in the occurrence of bone fractures. Hyponatremia is a risk factor for osteoporosis with a predilection to affect the hip, while the magnitude of association depends on the severity and chronicity of hyponatremia. Chronic hyponatremia also increases the risk for falls by inducing gait instability and neurocognitive deficits. Besides the detrimental impact of hyponatremia on bone mineral density and risk of falls, it also induces changes in bone quality. Emerging evidence suggests that acute hyponatremia shifts bone turnover dynamics towards less bone formation, while hyponatremia correction increases bone formation. The key unanswered question whether treatment of hyponatremia could improve osteoporosis and lower fracture risk highlights the need for prospective studies, evaluating the impact of sodium normalization on bone metabolism and occurrence of fractures. Recommendations for clinical approach should include measurement of serum sodium in all individuals with fracture or osteoporosis. Also, hyponatremia, as an independent risk factor for fracture, should be taken into consideration when estimating the likelihood for future fragility fracture and in clinical decision-making about pharmacological therapy of osteoporosis. Until it is proven that normalization of sodium can lower fracture occurrence, correcting hyponatremia cannot be universally recommended on this basis, but should be decided on a case-by-case basis.
Keywords
Introduction
Hyponatremia, defined as serum sodium below 135 mmol/l, is the most common electrolyte abnormality encountered in clinical practice. In 2006, a landmark study demonstrated for the first time that chronic, even mild, hyponatremia is not really asymptomatic, as it was considered until then, and results in gait impairment, attention deficits and high risk of falls.1,2 Following this paradigm shift in the approach to chronic hyponatremia, research groups in the field of hyponatremia across the world started exploring the possible association of hyponatremia with fractures and osteoporosis.
In recent years, four reviews in this topic have been published. The first three, published between 2016 and 2019,3–5 were systematic reviews and meta-analyses which assessed the association of hyponatremia with fractures, osteoporosis and falls. The most recent one, published in 2019, 6 explored in depth the link between hyponatremia and bone fragility, reviewing data from human, animal and cellular studies. The present review article aims to summarize the current state of knowledge on the interaction of hyponatremia with fracture risk, bone mineral density (BMD) and falls as well as the underlying pathophysiological mechanisms, including emerging data from a plethora of observational studies in the field and studies on the interaction between serum sodium and bone turnover markers (BTMs), published since 2019. Most review articles up to date have focused on single aspects of hyponatremia-induced fracture risk, such as either on the bone fragility aspect or falls component. We aim to explore and include all the components of hyponatremia-induced bone loss and increased risk of falls and shed light on their complex interaction which ultimately leads to hyponatremia-related excess fracture risk. Furthermore, we outline current data on the pathophysiological basis of hyponatremia-related bone loss, focusing on the direct effect of hyponatremia on bone cells and its impact on bone turnover. Finally, in addition to identifying current gaps in the existing literature and outlining future research directions, this review article seeks to provide a practical and applicable-in-clinical-practice algorithm for fracture risk assessment and management in patients with hyponatremia.
Methods
We performed an extensive search in MEDLINE, including all articles published until October 2022, evaluating the association of hyponatremia with fractures and BMD. The specific keywords used were ‘hyponatremia’ OR ‘sodium’ or ‘SIADH’ OR ‘SIAD’ OR ‘vasopressin’ AND ‘fractures’ OR ‘bone’ OR ‘bone mineral density’ OR ‘bone turnover markers’ OR ‘osteoporosis’. All full-text of English-language publications reporting results of observational studies and experimental studies, reviews and meta-analyses were included. Exclusion criteria were: (i) case reports, (ii) case series, and (iii) abstracts. Studies were independently reviewed by PT and MPY and assessed for bias using the Newcastle-Ottawa scale.
Of note, this review article has some limitations since it is a narrative review rather than a systematic review and only one database was searched. Moreover, the quality of included studies was not assessed as rigorously as in systematic reviews or meta-analyses, introducing a potential risk for selection and reporting bias. In order to minimize this risk, search results were independently reviewed by both authors. Finally, as a general and not systematic review, critical appraisal and synthesis of data does not follow a stringent protocol and, therefore, it cannot offer a precise estimate of the relationship between variables, such as hyponatremia and osteoporosis.
Observational studies about the association of hyponatremia with low BMD and fractures
Several observational studies have evaluated the association of hyponatremia with occurrence of fractures, osteoporosis and falls, as summarized in Table 1. In 2008, the role of hyponatremia as a major risk factor for fall-related bone fractures in the elderly was firstly reported in two case-control studies.7,8 The association of hyponatremia with fracture occurrence was found to be independent of BMD. 9 In line with these findings, the prospective population-based Rotterdam study confirmed an increased risk for vertebral and non-vertebral fractures in patients with hyponatremia, independent of BMD and falls. 10 These data pointed towards a possible detrimental effect of hyponatremia on bone quality, which cannot be captured by BMD values.10–12 Another large cross-sectional longitudinal analysis of 5122 men aged ⩾65 years, enrolled in the Osteoporotic Fractures in Men (MrOS) study, found a similar, but even stronger, relationship between hyponatremia and fracture risk, with a threefold increase in the risk for hip and incident morphometric spine fractures. 13 Despite the higher incidence of falls and lower BMD at all sites in males with hyponatremia compared to normonatraemic ones, the strength of the association between hyponatremia and fractures was not altered after adjusting for BMD and falls. 13 The fact that neither osteoporosis nor falls seemed sufficient to explain the hyponatremia-related increased fracture risk might be explained by poor bone quality in patients with hyponatremia, 14 through either impaired ability to repair local microfractures 14 or alterations in collagen composition of bone. 10
Observational studies about the relationship of hyponatremia with BMD, falls and fractures.

aHR, adjusted hazard ratio; aOR, adjusted odds ratio; BMD, bone mineral density; CKD, chronic kidney disease; DM, diabetes mellitus; FR, fracture risk; HF, hip fracture; HR, hazard ratio; HypoNa, hyponatremia; LS, lumbar spine; NDA, no data available; NormoNa, normonatraemia; NOF, neck of femur; NVF, non-vertebral fractures; RR, risk ratio; VF, vertebral fractures; yrs, years.
Several studies have also found that hyponatremia is linked to significantly increased odds of osteoporosis. This relationship was firstly demonstrated in 2010 by data analysis from the Third National Health and Nutrition Examination Survey (NHANES III), reporting that, after multivariable adjustments, mild hyponatremia was associated with higher odds ratio (OR) of osteoporosis at the hip (OR = 2.85, 95% CI: 1.03–7.86) and at the femoral neck (OR = 2.87, 95% CI: 1.41–5.81). 15 This study also addressed the question whether the relationship of hyponatremia with low BMD was found because increasing age is a strong independent risk factor for both hyponatremia 26 and osteoporosis. NHANES III data showed that the association of hyponatremia with osteoporosis was independent of age. 15 This, independent of age, association of hyponatremia with osteoporosis was also confirmed in a cross-sectional study of women below the average age of menopause, illustrating that low serum sodium had an independent contribution to anorexia nervosa-related osteopenia. 27 The association of hyponatremia with higher rates of osteoporosis was confirmed in a large cross-sectional observational study which, besides the hip and the femoral neck, also documented higher rates of osteoporosis at the lumbar spine. 16 This study added the novel observation that the longer the duration of hyponatremia, the higher the risk of developing osteoporosis. 16 It also showed that, after multivariable adjustments, hyponatremia was associated with 2.46-fold higher odds of total hip osteoporosis in age <55 years, 1.96-fold in age 55–67 years, and 1.55-fold in age >67 years, suggesting that age acts as a modifier of the independent association between hyponatremia and osteoporosis. 16 The attenuation of the hyponatremia-related osteoporosis risk in elderly patients could be due to aging being a competing stronger risk factor, offsetting the risk associated with hyponatremia and masking hyponatremia-induced bone loss. This observation that the contribution of hyponatremia to bone loss is more significant in younger individuals with no other risk factors, such as menopause or advancing age, may explain the lack of relationship between hyponatremia and osteoporosis in the Rotterdam study 10 which included predominantly elderly subjects. 16
A landmark case-control study compared data of 30,517 osteoporosis cases and 46,256 fragility fracture cases with age and gender-matched controls without fracture and osteoporosis, respectively, demonstrating a strong association of chronic hyponatremia with osteoporosis (OR = 3.97, 95% CI: 3.59–4.39) and fragility fractures (OR = 4.61, 95% CI: 4.15–5.11). 18 The odds of osteoporosis and fragility fracture increased incrementally with decrease in serum sodium levels, providing evidence that the association between hyponatremia and osteoporosis is dose-dependent (more severe hyponatremia results in higher risk) and time-dependent (chronic hyponatremia, defined as at least two low sodium values 1 year apart, carries higher risk than a single episode of hyponatremia). 18 These findings provide another possible explanation for the lack of relationship between hyponatremia and BMD in the Rotterdam study, 10 since this used a single low sodium value to define hyponatremia and included in the vast majority patients with hyponatremia between 130 and 134 mmol/l. 10 A cross-sectional study from Danish patient databases has confirmed that the presence of hyponatremia increases the risk of concurrent osteoporosis at both hip and lumbar spine, 19 while longitudinal data analysis from these databases reported that chronic mild hyponatremia primarily affected bone in the hip, with limited effect on the lumbar spine. 28 Another cohort study of 5610 females showed significantly lower T-score in total hip in those with hyponatremia, but not in lumbar spine, suggesting a predilection of hyponatremia to affect hip BMD. 14 Analysis of sodium values as a continuous parameter suggested that higher serum sodium resulted in statistically significant higher T-score at the total hip and femoral neck and was related to lower fracture risk. 14 Hyponatremia also led to a two-fold increase of the risk for major osteoporotic fracture, independent of osteoporosis. 14 The discrepancy between the impact of hyponatremia on hip and vertebrae may originate from the differences between cortical and trabecular bone. The higher content of sodium in the cortical bone 29 in combination with the high ratio of cortical to trabecular bone in the femoral neck and total hip could explain why the mobilization of sodium from the bone matrix in the context of hyponatremia disproportionately affects the hip compared to the vertebra. 28
In light of this wealth of evidence, three meta-analyses of the relevant literature have been published.3–5 The first meta-analysis in this field, published in 2016, included 12 observational studies, comparing BMD, prevalence of osteoporosis, and fracture rates between patients with hyponatremia and normonatraemia. 3 In the meta-analysis for fracture occurrence, eight studies reported OR, with a statistically significant higher odds of fracture in hyponatraemic patients with a pooled OR of 1.99 (95% CI: 1.50–2.63), while four studies reported hazard ratio (HR) for fracture with a pooled HR of 1.62 (95% CI: 1.28–2.05, p < 0.001). 3 The meta-analysis of osteoporosis included only four studies, showing a statistically significant increase in the odds of osteoporosis in subjects with hyponatremia, as evidenced by a pooled OR of 1.23 (95% CI: 1.06–1.43, p = 0.04). Meta-analysis of studies analysing BMD at different anatomical sites showed that individuals with hyponatremia have significantly lower BMD only at the hip with a mean difference of −0.05 g/cm2 (95% CI: −0.06–0.04, p < 0.001). 3 The results of the meta-analysis were robust with little heterogeneity, suggesting that hyponatremia increases the risk of fracture and is associated with increased prevalence of osteoporosis. 3 Another systematic review, including 19 studies until 2017, showed that hyponatremia increases the odds of both fractures at all sites (pooled OR 2.34, 95% CI: 1.86–2.96) and osteoporosis (pooled OR 2.67, 95% CI: 2.07–3.43). 5 Finally, a meta-analysis of 15 studies concluded that hyponatremia is associated with a significantly increased risk of falls (pooled OR 2.14, 95% CI: 1.71–2.67). 4 This excess risk applies even using a cut-off sodium value below 135 mmol/l, but it is substantially higher for even lower sodium values. The same meta-analysis also confirmed that the presence of hyponatremia is associated with a higher risk of fractures, particularly hip fractures (pooled OR 2.00, 95% CI: 1.43–2.81). 4
Recent studies, since 2018, have focused on the relationship of hyponatremia with BMD and fracture risk in specific population subgroups, such as patients on thiazides, 21 those with epilepsy, 22 diabetes 23 and chronic kidney disease (CKD). 24 A large case-control study of patients on thiazide diuretics showed that thiazide-associated hyponatremia is related to a 1.5-fold increased risk of fracture, in both vertebrae and hip, compared to those on thiazides without hyponatremia, attenuating the fracture-protective effect of thiazides. 21 A cross-sectional study of patients with epilepsy established a significant association of hyponatremia with osteoporosis at any site (OR 2.91), with the strongest association being with lower BMD in lumbar spine. 22 A case-control study of patients with diabetes mellitus, matched for age, gender and median glycated haemoglobin, demonstrated that hyponatremia among patients with diabetes mellitus is associated with significantly increased risk for osteoporosis (OR 3.09) and fragility fracture (OR 6.41). 23 Of note, the OR for fracture was double in magnitude compared to that for osteoporosis, lending further support to the notion that hyponatremia, besides compromising BMD, may incur fracture risk by additional mechanisms, such as deteriorating bone quality and increasing gait instability and falls. 23 A case-control study of patients older than 60 years with stage 3 or greater CKD showed that chronic prolonged hyponatremia (lasting at least 90 days) is associated with 36% higher odds of hip fracture. 24 This association applied even to individuals with mild hyponatremia (serum sodium 131–134 mmol/l) and persisted regardless of the presence of osteoporosis and falls, indicating a potential mechanism of low bone quality, not captured by BMD measurements. 24
Since 2009, numerous observational studies have shown that, even mild, hyponatremia is significantly associated with fracture occurrence.3–5,7–10,13,14,17,18,20,21,23,24,30 Additionally, several epidemiological studies have demonstrated a relationship of hyponatremia with osteoporosis, mainly in the hip,3,5,14–16,18,19,22,23,25 as well as with falls.4,10,13,31 However, neither falls nor osteoporosis seem to be sufficient to explain the significant association between hyponatremia and fragility fractures.10,13,14,17,24 The mechanisms, through which hyponatremia can have a detrimental impact on bone quality, without being captured by BMD, warrant further exploration in order to be fully elucidated. In conclusion, chronic hyponatremia, even mild, represents an independent risk factor for both osteoporosis and fragility fractures.
Impact of chronic hyponatremia on gait and risk of falls
There is a paradigm shift in approaching chronic hyponatremia, according to which this is not a benign condition, replacing the old concept that patients with chronic hyponatremia are ‘asymptomatic’.1,2 In 2006, Renneboog et al. 1 undertook a landmark case-control study demonstrating that patients with chronic moderate hyponatremia fall dramatically more frequently than normonatraemic controls. Renneboog et al. firstly reported that hyponatraemic patients, considered to be clinically asymptomatic with normal neurological examination findings, were highly unstable in tandem gait with their eyes open. After correction of hyponatremia, the ‘total travelled way’ by the centre of pressure significantly decreased. 1 The same group detected also attentional impairments in association with chronic hyponatremia which consisted of significant increases in response latency and error rate. Chronic hyponatremia was related with a global decrease of attentional capabilities and a defect in both visual and auditory tests.1,2 Remarkably, patients with hyponatremia exhibited more severe balance and attention impairments than normonatraemic individuals when tested 30 min after ingestion of 0.55 g of alcohol per kg of body weight. 1 Of note, the threshold for gait deficits associated with hyponatremia was 134 mmol/l and for attention deficits was 132 mmol/l 2 . In line with these findings, a study in community-dwelling older individuals with even milder degree of hyponatremia (serum Na 130–135 mmol/l) showed a 5% decrease in a cognitive screening score in comparison with normonatraemic subjects, representing a cognitive deficit. 31 Of note, hyponatremia-induced alterations in attention and gait tests are much more pronounced in older (mean age 73 years) than in younger adults (mean age 48 years). 32 This increased sensitivity of older individuals in combination with their high incidence of falls suggests that, even mild, hyponatremia can have significant physiological effects in this age group. The strong independent association between, even mild, hyponatremia and high risk of falls has been confirmed in subsequent studies in older patients31,33,34 as well as in a meta-analysis of 15 studies (pooled OR 2.14, 95% CI: 1.71–2.67). 4
The reversibility of these abnormalities following restoration of normonatraemia was firstly found in an animal study using a rat model which exhibited hyponatremia-induced ataxic gait, increased anxiety levels, and impaired recognition and associative memory. 35 Subsequently, a prospective study in humans assessed the impact of hyponatremia correction on nerve conduction studies, recruiting 11 patients with mean baseline Na 127.7 mmol/l and correcting Na levels to 136.1 mmol/l following combination therapy with fluid restriction and normal saline or urea. 36 This study showed a statistically significant improvement in ‘Timed up and go test’ (TUG) from a mean 14.9 s at baseline to 12.5 s following correction of hyponatremia. Since TUG is a predictor of falls, this observation suggests that restoration of normonatraemia may lower the risk of falls. This study reported that severe hyponatremia caused slowing of motor and sensory nerve conduction velocities (NCVs) as well as increase in F-wave latencies which provide a means of examining transmission between stimulation sites in the limbs and related motor neurons in the spinal cord. Following correction of hyponatremia, there was significant improvement of nerve conduction studies, as evidenced by 14% increase of NCV and 21% decrease of F-wave latencies. 36 The INSIGHT (Investigation of the Neurocognitive Impact of Sodium Improvement in Geriatric Hyponatremia: Efficacy and Safety of Tolvaptan), a multicentre, randomized, double-blind, placebo-controlled, parallel group, titration-to-effect trial, randomized 29 patients with chronic euvolaemic/hypervolaemic hyponatremia to tolvaptan and 27 patients to placebo in order to assess the effect of tolvaptan on cognition and gait. 37 Tolvaptan-induced correction of natraemia from baseline levels of 129 to 136 mmol/l, correlated with significant improvement in various neurocognitive tests, particularly rapid motor movements. 37 This improvement in the psychomotor speed domain, considered as essential to maintain upright stability during walking, could improve gait stability and reduce risk of falls. 37
In total, several studies have found an association of chronic, even mild, hyponatremia with gait impairment, attention deficits, and a high risk of falls,1,2 suggesting that hyponatremia, regardless of severity, should be included in all fall risk assessment tools. 38 Correcting hyponatremia may be justified on the basis of lowering risk of falls, taking into consideration emerging evidence that normalization of sodium leads to improvement in gait and cognition.35–37
Effect of hyponatremia and its correction on bone turnover
There is paucity of data regarding the effect of hyponatremia on bone metabolism 39 as well as the impact of sodium normalization on BTMs.37,40,41 Only one study has examined the acute effects of hyponatremia on bone turnover in humans. This prospective observational study included 22 patients with acute, non-traumatic, aneurysmal subarachnoid haemmorhage who had normal sodium levels on presentation. 39 Eight participants developed hyponatremia, while 14 remained normonatraemic, with the main objective being to compare the changes in BTMs over 1-week period between the two groups. CTX-1 (C-terminal crosslinking telopeptide of type 1 collagen), a bone resorption marker, increased in both groups, but its changes were not significantly correlated with plasma sodium concentration. However, there was a significant positive correlation between P1NP (N-propeptide of type 1 collagen), a bone formation marker, and nadir plasma sodium concentration. A similar positive correlation was found between the bone turnover index (P1NP:CTX-1 ratio) and serum sodium levels. 39 This study demonstrated that changes in bone turnover may occur very rapidly after the onset of hyponatremia. Acute hyponatremia resulted in a shift towards negative bone turnover balance, as evidenced by blunting bone formation and a fall in the ratio of bone formation to bone resorption. 39 This small study is the only one which has tested and confirmed in humans the hypothesis that hyponatremia induces an acute shift in bone turnover dynamics towards less bone formation and subsequent bone loss. 39
So far, only three studies have examined the effect of hyponatremia correction on BTMs in humans. For the first time, in 2016, Verbalis et al. 37 included changes in BTMs as an exploratory efficacy endpoint in the randomized INSIGHT trial which showed that reversal of hyponatremia with tolvaptan led to improvement in neurocognitive performance. Compared to placebo, the tolvaptan group had non-significant increase in serum osteocalcin, a bone formation marker, and decrease in urine NTx (N-telopeptide):creatinine ratio, a bone resorption marker. Despite these changes not being statistically significant, there was a highly significant decrease in the bone resorption index, calculated as change from baseline in urine NTx:creatinine ratio divided by change in serum osteocalcin. 37 This improvement in bone resorption index is particularly striking, considering that it was observed after only 22 days of tolvaptan-induced hyponatremia correction. 37
The second study was a randomized trial of patients with epilepsy and chronic hyponatremia, assessing the impact of 3-month salt replacement therapy on BTMs and BMD. 40 At baseline, neither BMD nor BTMs were abnormal. After 3 months, there was no significant difference in BMD and BTMs, serum CTX and P1NP, between salt replacement (N-14) and placebo (N = 7) groups. However, at baseline, median serum sodium was borderline low 134 mmol/l and, after 3 months, serum sodium was similar in the intervention with salt tablets and in the placebo groups. 40 For all these reasons, this study could not truly assess the impact of sodium normalization on bone turnover.
The third study was a pre-planned secondary analysis of a randomized controlled trial, using SGLT-2 (sodium-glucose transport protein 2) inhibitors for the treatment of hyponatremia induced by the syndrome of inappropriate antidiuretic hormone secretion (SIADH) in hospitalized patients. 41 Amongst 68 patients with SIADH-induced hypontraemia who were imposed on daily fluid restriction of <1000 ml, 34 received empagliflozin and 34 placebo for 5 days. The primary endpoint was the difference in BTMs between patients who reached normonatraemia on day 5 and those who remained hyponatraemic. Serum CTX increased during hospitalization in both groups, but without significant difference between patients with and without correction of hyponatremia. 41 Serum P1NP increased in patients who restored normonatraemia, whereas it remained stable in those with persistent hyponatremia, with a statistically significant difference between the groups (p = 0.005). As a result, patients who reached normonatraemia had significantly higher bone formation index, calculated by the ratio of P1NP to CTX, than those with persistent hyponatremia, suggesting that correction of hyponatremia led to an increase in osteoblastic activity after 5 days. 41
In total and despite short follow-up of 22 days 37 and 5 days, 41 two studies have reached the same conclusion that normalization of serum sodium exerts an early positive effect on osteoblastic activity. The lack of significant effect on bone resorption markers may be explained by the negative impact of immobility, which, because of its strong association with bone resorption, could mask the effect of sodium normalization. The long-term type and extent of effect of hyponatremia correction on bone turnover remains to be determined.
The pathophysiology of hyponatremia-induced bone loss
One-third of the total body sodium is stored in the bone matrix that serves as a sodium reservoir, being able to release sodium into the circulation during prolonged salt deprivation. Sodium homeostasis appears to be intrinsically related to bone physiology with increased bone resorption during hyponatremia occurring to preserve sodium homeostasis. Most experts in this evolving field agree that serum sodium concentration seems to be the signal by which osteoclasts sense extracellular fluid and total body sodium and may ‘misinterpret’ conditions without relative sodium deficiency, like SIADH, as total body sodium deficiency. Consequently, there would be ‘no brake’ to the stimulated bone resorption, as the serum sodium remains low no matter how much bone is resorbed. This would lead to further increased bone resorption as long as hyponatremia persists. 12 Although not fully clarified, the underlying molecular mechanisms of hyponatremia-induced bone loss have been studied both in vivo with hyponataemic rat models (e.g. SIADH) and in vitro with bone-cells cultures (e.g. osteoclasts’ and osteoblasts’ precursors).
In vivo studies
Verbalis et al. 15 first demonstrated in early 2000s that hyponatremia can cause a substantial reduction of bone mass in a rat model of SIADH. Analysis of excised femurs in rats using dual-energy X-ray absorptiometry showed that severe hyponatremia (mean serum Na 110 mmol/l) for 3 months significantly reduced BMD by 30% compared with normonatraemic rats. Micro-computed tomography and histomorphometric analyses indicated that hyponatremia markedly reduced both trabecular and cortical bone via increased bone resorption. The histology analysis revealed that hyponatremia was associated with increased number of osteoclasts per bone area. In addition, serum osteocalcin levels, which is a bone formation marker, was significantly decreased in the sera from hyponatraemic rats compared with normonatraemic controls. In total, these early results indicated that, at least prolonged and severe, hyponatremia is associated with increased bone resorption and decreased bone formation, leading to a negative balance of bone remodelling and ultimately bone loss. 15
However, as hyponatraemic patients have also elevated circulating arginine-vasopressin (AVP) levels, a potential contribution of increased AVP levels in hyponatremia-induced bone loss could not be excluded by these early data. Indeed, AVP injected into wild-type mice, exerted differential effects on bone cells by increasing osteoclastogenesis and decreasing osteoblastogenesis. 42 Conversely, genetically modified mice lacking AVP receptor 1a (AVPR1α−/−) displayed a remarkable high bone mass phenotype. 42 The rationale behind these results lies on the identified expression of both AVP receptors, AVPR1α and AVPR2 on osteoclasts and osteoblasts precursors. Upon stimulation AVPRs act through activation of extracellular signal regulated kinase (ERK), which in turn suppresses bone formation and increases bone resorption 42 [Figure 1(a)]. AVPR1α appears to be the main effector of AVP-induced skeletal changes, since the specific AVPR2 inhibitor, tolvaptan, did not affect significantly bone remodelling in wild type and ovariectomized mice. 43
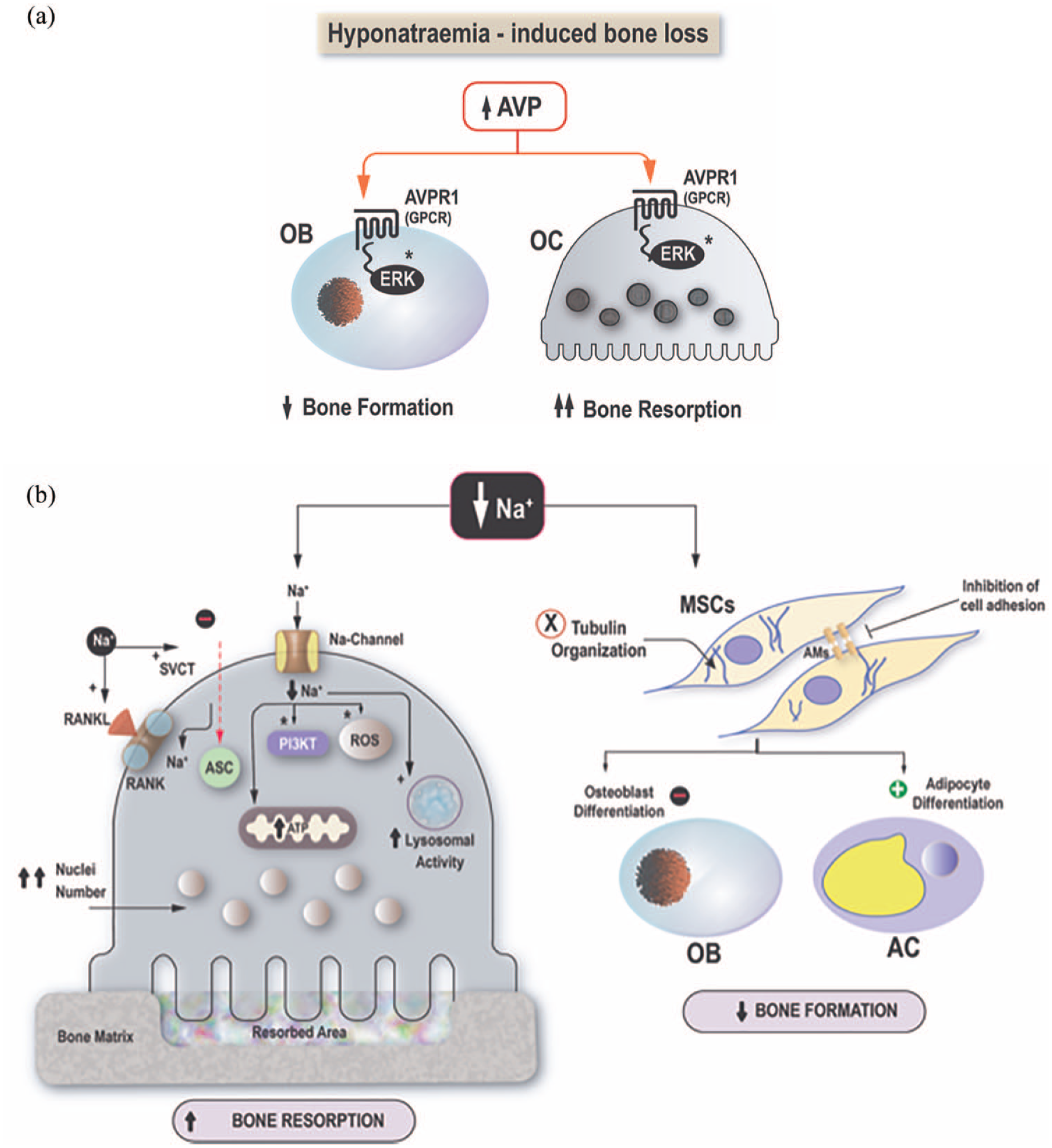
Proposed pathophysiological mechanisms of hyponatremia-induced bone loss. (a) High AVP levels act through the AVPR1a transmembrane G-protein coupled receptor in osteoclasts and osteoblasts activating intracellular ERK signalling pathways and leading to increased bone resorption and decreased bone formation, respectively. (b) Low sodium levels [Na+] exert direct effects on osteoclast precursors and MSCs. In osteoclasts: (i) inhibits ascorbic acid uptake through sodium dependent vitamin C transporter, (ii) activates ROS, RANKL/RANK and PI3K/Akt signalling pathways, (iii) increases lysosomal activity, (iv) increases mitochondrial energy production and (v) nuclei number, promoting osteoclast differentiation. In MSCs: (i) disturbs tubulin and cytoskeleton organization, (ii) inhibits cell adhesion and (iii) decreases osteoblastogenesis in favour of adipogenesis.
As the skeleton appears to be highly sensitive to the action of both posterior pituitary hormones AVP and oxytocin, and AVPR and oxytocin receptor (OXTR) exert opposing effects on bone mass,42,44 Sun et al. 43 investigated the potential interaction of AVP and oxytocin at the cellular level. In double mutants lacking both receptors (OXTR−/−:AVPR1α−/− mice) the bone mass was increased but to a lesser degree than in AVPR1α−/− mice, suggesting that the skeletal effects of elevated AVP levels in patients with chronic hyponatremia, may be exerted through both the AVP and oxytocin receptors. 43
In vitro studies
In vitro experiments with murine RAW 264.7 preosteoclastic cells and primary bone marrow monocytes have demonstrated a direct effect of low extracellular [Na+] on osteoclasts. In particular, low [Na+] in the culture media increased the number of mature osteoclasts enchasing their differentiation characteristics with higher nuclei number and increased interconnectivity compared to osteoclasts grown in normal [Na+], in a dose-dependent manner. Parallel with the increased osteoclasts’ number increased resorptive activity was also observed. Interestingly, the researchers have shown that correction of osmolality did not reverse the low [Na+] effects on osteoclasts, suggesting that both the increased osteoclastogenesis and resorptive activity are mostly attributed to sodium sensing, rather than osmolality sensing, although hypo-osmolality appears to exert a synergistic effect. The underlying molecular mechanisms involve sodium-dependent inhibition of ascorbic acid uptake, and subsequent activation of oxidative stress pathways [Figure 1(b)]. Additional intracellular signalling pathways, have been recently identified using ribonucleic acid (RNA) sequencing and gene ontology analysis and include receptor activator of nuclear factor kappa-B ligand, and PI3K/Akt pathways, lysosomal activity and dysregulation of autophagy, mitochondrial energy production, and osteoclast motility [Figure 1(b)], further supporting the central role of osteoclasts on the hyponatremia-induced bone loss. 45 Accordingly, hyponatremia also exerts direct effects on human mesenchymal stromal cells (hMSCs), which is the common progenitor for osteoblasts and adipocytes. In an experimental model of hMSCs isolated from the bone marrow of healthy donors, low Na+ levels inhibited cellular adhesion and cellular viability, and disrupted the tubulin organization of the cytoskeleton, leading to an overall disruption of cellular homeostasis. In addition, a preferential commitment towards the adipogenic phenotype at the expense of osteoblasts was also observed, 46 accompanied by low expression of osteoblastic markers on one hand and expression of factors stimulating osteoclast recruitment and activity on the other.
Collectively, data published so far from in vivo and in vitro studies show that the hyponatremia -induced bone loss has a complex and multifactorial pathophysiological basis with direct effects on the functionality and differentiation of bone cells that lead to an imbalanced bone remodelling with increased bone resorption and decreased bone formation (Figure 1).
Treatment of hyponatremia-related osteoporosis
In view of recent evidence about improvement of BTMs following hyponatremia correction,37,41 the key question is whether normalization of serum sodium can reverse osteoporosis. Clinical evidence in favour of the reversibility of hyponatremia-induced bone loss is limited to case reports. The first case described was a male patient with malignant chronic SIADH and severe osteoporosis who showed remarkable improvement in Z-score at the lumbar spine from −3.8 to −2.8 within 7 months after restoration of normal sodium and AVP levels. 47 Another case report was that of an adolescent male with valproate acid-induced SIADH, osteopenia and fragility fractures, whose BMD normalized 19 months after resolution of hyponatremia. 48 The efficacy of currently available anti-osteoporotic pharmacological agents in the management of hyponatremia-induced bone loss has not yet been investigated. An interesting observation in a longitudinal study was that, despite adjustment for the use of oral and intravenous bisphosphonates, chronic mild hyponatremia was still significantly associated with worse longitudinal BMD changes in the hip region. 28 This suggests that bisphosphonates may have limited efficacy in the treatment of hyponatremia-induced bone resorption, 28 highlighting the need for studies to address this issue.
Areas for future research
The significant association of hyponatremia with osteoporosis and fractures raises several questions. First and foremost, the key, but still unanswered, question remains whether correction of hyponatremia can improve patient outcomes, such as BMD, risk of falls and fracture occurrence. For this reason, long-term prospective studies, ideally randomized controlled trials, should be undertaken with the aim to evaluate the impact of correcting hyponatremia on bone health and fracture rate. In keeping with the US Food and Drug Administration requirement for a primary outcome of fracture for phase 3 osteoporosis drug trials, studies using the hard clinical endpoint of fracture occurrence are warranted to assess the effect of hyponatremia correction. Fractures are the ideal primary endpoint, but they can make these studies prohibitively large, long and costly because of the need to recruit a large number of participants with a long follow-up. Alternatively, prospective studies could examine the potential reversibility of osteoporosis through normalization of serum sodium. Using changes in BMD as the primary endpoint would reduce the sample size, duration and cost, but its appropriateness as a reliable surrogate marker for fracture risk reduction is still debated.
Second, preliminary data, showing early improvement of BTMs after correction of hyponatremia, highlight the need to evaluate the short- and long-term impact of sodium normalization on bone metabolism with the use of BTMs. These studies using BTMs, not only will require shorter follow-up than BMD studies because of their ability to detect changes much earlier, but also will shine more light on the bone remodelling processes. We suggest that these studies should recruit ambulatory outpatients with chronic hyponatremia in order to minimize the effects of confounding variables, such as immobility and illness-related hypercortisolaemia, which have a well-known strong negative impact on bone metabolism with a shift towards bone resorption.
Third, it is also worth exploring and comparing the effect of various pharmacotherapies for hyponatremia due to SIADH on BMD and BTMs. Besides their bone effect through changes in serum sodium, these agents per se may impact bone metabolism. Additionally, emerging data about the role of AVP signalling in bone mass regulation through V1a and V2 receptors in osteoblasts and osteoclasts raise the possibility that a treatment modality for SIADH may affect bone metabolism, at least partially, through AVP changes. Therefore, these studies should include measurement of serum copeptin, an excellent surrogate marker for AVP. These studies should include in one arm tolvaptan, a selective AVP antagonist with direct action on AVP receptors and high efficacy in restoring hyponatremia, which is the only medication currently approved by the regulatory authorities for the treatment of euvolaemic hyponatremia. Studies should also include the other main treatment modalities: fluid restriction which has been the first-line treatment of SIADH for decades; urea which is recommended as the second-line therapy for SIADH by the European guidelines 49 ; empagliflozin, an SGLT-2 inhibitor, which is widely available as antidiabetic medication and has recently emerged as a promising treatment option for both short-term use in hospitalized patients with SIADH 50 and long-term use in outpatients with chronic SIADH. 51
Fourth, another key question applies to the efficacy of common antiosteoporotic agents, such as bisphosphonates and denosumab, in hyponatremia-related osteoporosis. Limited observational data that bisphosphonates, the most commonly prescribed drugs for osteoporosis, 52 may have limited aniresorptive effect in patients with mild chronic hyponatremia highlight the need to evaluate the effect of bisphosphonates on BMD in this context and test whether it differs from their effect on osteoporosis in general.
Fifth, taking into account the association between hyponatremia and osteoporosis, it remains to be answered whether BMD measurement is indicated in all, or at least a subgroup of, patients with chronic hyponatremia. It is essential to determine the number needed to screen (NNS) in order to prevent one adverse event; a fragility fracture. The NNS will mainly depend on the predictive value of hyponatremia to detect low BMD and the effectiveness of current therapies in lowering fracture risk. Evaluation of clinical effectiveness should be supplemented by a cost-effectiveness analysis.
Sixth, clinical studies on the reversibility of hyponatremia-related osteoporosis and fracture occurrence should be supplemented by mechanistic studies, exploring the pathophysiology of hyponatremia-induced bone loss. In vitro and in vivo studies are warranted to identify the potential mediators and the extent of their contribution to bone loss, with a focus on the role of sodium sensing, AVP and oxytocin.
Practical approach to patient with hyponatremia
Hyponatremia, even mild, is a readily identifiable risk factor for falls, osteoporosis and bone fracture occurrence. 9 This independent association should inform clinical decision-making in day-to-day clinical practice with respect to measuring serum sodium in individuals with fractures or osteoporosis as well as testing BMD in patients with hyponatremia. The first scenario is patients presenting with fractures. Several studies have recorded a high prevalence of 9–13% for hyponatremia in patients aged 65 years or older admitted with any type of bone fracture.7,8,53 This prevalence is even higher, estimated at around 19%, among elderly patients admitted with hip fractures.54,55 These data highlight the need to reinforce the recommendation for serum sodium measurement in all patients presenting with bone fracture (Figure 2). The second scenario is an individual with osteoporosis. Taking into consideration the high rate of hyponatremia among people with osteoporosis in combination with the ease and low cost of screening for hyponatremia, we recommend measuring sodium concentration in patients with low BMD. The third scenario is that of an individual with hyponatremia and whether BMD should be measured. In general, any screening programme could be justified only on the basis of high-quality evidence that it can reduce morbidity or mortality. Obviously, a universal screening programme with BMD measurement in patients with hyponatremia cannot be recommended since it has not been proved that correcting hyponatremia reduces fracture risk. A large proportion of patients with hyponatremia, such as all women above the age of 65 and postmenopausal women younger than 65 years with high fracture risk, should, anyway, undergo BMD testing according to the US Preventive Services Task Force et al. 56 The decision about offering BMD testing to a patient with hyponatremia should be individualized, taking into consideration various parameters. Since the association of hyponatremia with fracture risk is dependent on the duration and magnitude of hyponatremia, 18 the longer the duration of hyponatremia and the lower the sodium value, the larger the potential negative effect of hyponatremia on bone health. We suggest considering hyponatremia as a risk factor for fracture, provided it is chronic with minimum duration of 6 months, since brief episodes of hyponatremia are expected to have minimal long-term impact on bone health. We recommend that hyponatremia should trigger BMD testing in the presence of two or more risk factors, such as history of fragility fracture, parental history of hip fracture, history of falls, significant exposure to glucocorticoids, causes of secondary osteoporosis, low body mass index, smoking and significant alcohol intake (Figure 2).

Proposed algorithm for fracture risk assessment and management in patients with hyponatremia.
The inclusion of hyponatremia into the Fracture Risk Assessment Tool (FRAX), 57 the most commonly used tool to calculate a person’s future fracture risk, has been proposed on the basis of its high prevalence, the ease and reliability of serum sodium measurement and its association with occurrence of falls and fractures.6,58 However, the incorporation of hyponatremia into the FRAX algorithm would require data collection from large patient cohorts in order to quantify the contribution of hyponatremia to excess fracture risk as well as examine the influence of age and gender on hyponatremia-induced fracture risk. Since FRAX score does and will not, at least, for the foreseeable future include serum sodium, it may underestimate the fracture risk in patients with hyponatremia. Therefore, two different strategies have been proposed in order to take into account the hyponatremia-related excess fracture risk. The first option is to strongly consider offering pharmacotherapy in patients with chronic hyponatremia and FRAX score below, but close to, the intervention threshold. As such, a postmenopausal woman with 18% probability of major osteoporotic fracture over the next decade, who otherwise would not be a candidate for antiosteoporotic treatment in view of the intervention threshold being ⩾20%, 59 could be offered pharmacotherapy in the presence of hyponatremia. The second, appealing but untested, prospect is ticking the box of rheumatoid arthritis in calculating the FRAX score, as it has already been suggested for other risk factors. For example, the additional fracture risk in patients treated with aromatase inhibitors for breast cancer seems to be similar with that of rheumatoid arthritis, suggesting the use of rheumatoid arthritis input in these patients. 60 Similar suggestions have been made for patients with other conditions, related with increased fracture occurrence and not included in FRAX, such as multiple sclerosis and type 2 diabetes.61,62
Lack of data on the role of correcting serum sodium for fracture prevention cannot justify treatment of hyponatremia with a sole purpose of lowering fracture risk. However, emerging data have demonstrated that resolution of chronic mild hyponatremia can lead to improvement in various neurocognitive aspects; thus, factors, including older age, history or high risk of falls and presence of gait or cognition disturbances, increase the likelihood that reversal of hyponatremia will be beneficial for these patients. In addition, the probability that normalization of serum sodium will lead to significant BMD improvement and fracture risk reduction is higher in subgroups, such as premenopausal females and younger individuals without major risk factors for osteoporosis. This suggestion is based on the rationale that the higher the likely contribution of hyponatremia to osteoporosis, the more likely that its restoration will improve bone metabolism. In total, the decision about correcting hyponatremia should be taken on a case-to-case basis, based on a benefit-risk ratio and always in tandem with seeking the underlying cause of hyponatremia. Determining the cause of hyponatremia is of paramount importance because it will facilitate its successful treatment and it may also reveal an unknown underlying medical condition with potentially significant sequalae. In view of the scarcity of data about the impact of hyponatremia on response to antiosteoporotic treatment, treatment of osteoporosis in patients with hyponatremia should be similar with that in normonatraemic patients in terms of the choice of treatment modality for osteoporosis, the duration of treatment, decisions about switching therapy and frequency and mode of monitoring.
Concluding remarks
In conclusion, considerable volume of recent epidemiological data support that hyponatremia is an independent risk factor for fractures. The etiology of this relationship seems to be multifactorial. Hyponatremia has a negative impact on gait stability, resulting in an increased risk of falls, and is also associated with osteoporosis by inducing increased bone resorption to mobilize sodium stores in bone. Nonetheless, neither osteoporosis nor falls are sufficient to fully explain the mechanisms by which hyponatremia leads to increased risk of fractures, suggesting additional pathways through which hyponatremia affects bone quality.
Hyponatremia, as a risk factor for fracture, should be taken into consideration in estimating probability for future fracture. The key question remains whether hyponatremia is a modifiable risk factor for osteoporosis and fractures, highlighting the need for prospective studies examining the effect of sodium normalization on BMD and fracture incidence. As long as there is no proof that hyponatremia correction can prevent fractures, treatment of hyponatremia cannot be generally recommended on the basis of bone benefits. A decision about restoring normonatraemia should be taken on an individual basis, taking into account potential benefits in terms of neurocognition, risk of falls and fracture risk.
