Abstract
Objective
To assess the effect of zoledronic acid (ZOL) on bone mineral density (BMD) and fracture risk at the L1–L4 vertebrae, femoral neck, hip and trochanter in Chinese women with osteoporosis.
Methods
A randomized controlled trial was conducted in female patients with osteoporosis, randomized to receive one 5-mg ZOL intravenous infusion per year or placebo equivalent. Facture risk and BMD were measured over a 2-year follow-up period.
Results
A statistically significant reduction in the risk of fracture was observed at the trochanter in the ZOL group (
Conclusions
This study indicated that ZOL could increase BMD and reduce fracture risk in women with osteoporosis over a 2-year follow-up period, and was not associated with any serious drug-related adverse effects.
Introduction
Osteoporosis is a skeletal disease characterized by compromised bone strength and increased risk of fracture. 1 In the USA, osteoporosis occurs in 55% of the population aged ≥50 years. 2 The incidence of osteoporosis is likely to rise as life expectancy increases. It is estimated, for example, that >25% of the population in Canada will be aged ≥65 years by 2041. 3 According to the World Health Organization, it is estimated that there are 75 million people with osteoporosis in Europe, the USA and Asia, with 9 million new fractures reported every year, worldwide.3,4 Osteoporosis-related fractures are usually associated with increased morbidity and mortality, and increased healthcare costs.5–7
Several therapies (including teriparatide, calcitonin, alendronate and strontium ranelate) have been shown to be well tolerated and effective in the prevention of osteoporosis.8–10 Clinical studies have indicated that nitrogen-containing bisphosphonates not only inhibit bone resorption but also preserve bone mass, thereby reducing the risk of osteoporotic fractures. 11 Most bisphosphonates are given orally but this results in low bioavailability and poor adherence, which consequently reduce the treatment benefit.12–13 Alendronate and risedronate have been reported to reduce nonvertebral and hip fractures in women with osteoporosis.14–18 Zoledronic acid (ZOL) is an aminobisphosphonate that has a prolonged dosing interval and a high affinity for mineralized bone. 19 Intravenous infusion of ZOL may rapidly localize to bone, where it reduces osteoclastic bone resorption through inhibition of farnesyl pyrophosphate synthase, which is a key enzyme in the mevalonate pathway.19,20
The effectiveness and tolerability of intravenous ZOL in decreasing fracture risk and influencing bone mineral density (BMD) has not previously been explored in a mainland Chinese female population. As such, there is a clinical need for randomized controlled trials of ZOL in this population. The present study assessed the effect of ZOL on BMD and fracture risk at the L1–L4 vertebrae, femoral neck, hip and trochanter, in women with osteoporosis enrolled in a 2-year, randomized, placebo-controlled trial at a large teaching hospital in China.
Patients and methods
Study population
Female patients with a primary diagnosis of osteoporosis were recruited from the Department of Endocrinology, First Affiliated Hospital of the General Hospital of the PLA, Beijing, China between May 2008 and November 2010. For inclusion in the study women had to be postmenopausal, have a BMD T-score ≤−2.5 at the femoral neck but no evidence of vertebral fractures, or a BMD T-score ≤1.5 with radiological diagnosis of two or more vertebral fractures. Exclusion criteria were: (i) patients with secondary osteoporosis or other diseases known to affect bone metabolism; (ii) patients taking sodium fluoride, parathyroid hormone, anabolic steroids or growth hormone within 6 months of study entry, or systemic corticosteroids within 12 months of study entry; (iii) patients with malignant, hepatic and renal diseases; (iv) a serum calcium concentration of >11.0 mg/dl and untreated hypocalcaemia.
The Ethics Committee of the First Affiliated Hospital of the General Hospital of the PLA reviewed and approved the study protocol. All patients enrolled in the study provided written informed consent.
Study treatment
Eligible patients were randomized at study entry to receive 5 mg ZOL (Yangtze River Pharmaceutical Group, Taizhou, Jiangsu Province, China) by intravenous infusion or placebo intravenous infusion (0.25 mg activated vitamin D3) at the start of the study and after 12 months. All patients also received a supplement of 600 mg elemental calcium and 400 IU vitamin D, orally, taken every day for the duration of the study.
Study assessments
Patients were followed up with telephone interviews and clinic visits at 6, 12, 18 and 24 months over the 2-year study period. Clinic visits consisted of an assessment of BMD, fracture incidence and adverse effects of treatment.
Bone mineral densities of the lumbar vertebrae (L1–L4), total hip, femoral neck and trochanter were measured by dual-energy X-ray absorptiometry (Hologic®, Waltham, MA, USA) at each study visit.
Vertebral fracture was defined as a reduction of ≥20% (minimum 4 mm) from baseline in the height of any vertebra, as measured by radiography. All fractures were confirmed by the Genant
Safety assessments included recording of all adverse events, physical examination, measurement of vital signs, haematological toxicity monitoring, blood biochemistry and urinalysis at each clinic visit during follow-up. Adverse events were recorded and classified according to the Medical Dictionary for Regulatory Activities. 22
Statistical analyses
Statistical analyses were carried out using SPSS® statistical software, version 16.0 (SPSS, Inc. Chicago, IL, USA) for Windows®. Efficacy and safety parameters were compared between ZOL and placebo groups by Student’s
Results
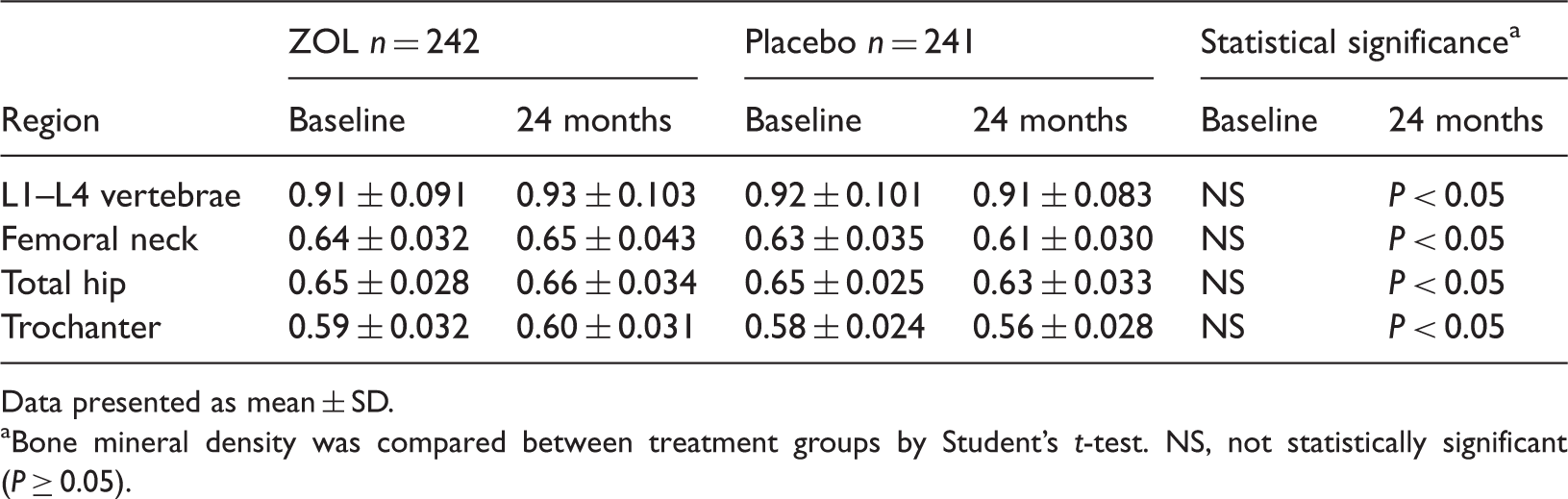
A total of 694 patients with osteoporosis were screened for inclusion in the study; 211 of these patients were ineligible for study participation according to the prespecified inclusion and exclusion criteria, therefore 483 were randomized to treatment. Of the randomized patients, 242 received ZOL and 241 received placebo. The mean body mass index (BMI), previous osteoporosis drug use, T-score at the femoral neck and baseline BMD in the L1–L4 lumbar vertebrae, femoral neck, hip and trochanter were similar in the ZOL and placebo groups. Demographic characteristics and T-scores at the femoral neck for the patient population are summarized in Table 1; baseline BMD is presented in Table 2. Fracture incidence at the L1–L4 vertebrae, femoral neck, hip and trochanter was lower in patients receiving ZOL compared with patients receiving placebo (Figure 1); the only significant differences between treatment groups were at the trochanter ( Mean incidence of fractures during a 24-month follow-up period in Chinese women with osteoporosis, treated with 5 mg zoledronic acid ( Change in bone mineral density (BMD) at (A) L1–L4 vertebrae, (B) total hip, (C) femoral neck and (D) trochanter, in Chinese women with osteoporosis treated with 5 mg zoledronic acid ( Demographic and baseline characteristics of 483 Chinese women with osteoporosis, randomized to receive 5 mg zoledronic acid (ZOL), administered once every 12 months, or placebo equivalent. Data presented as mean ± SD or Vertebral fracture was defined as a reduction of ≥20% (minimum 4 mm) from baseline in the height of any vertebra as measured by radiography. No statistically significant between-group differences were observed ( Bone mineral density at the L1–L4 vertebrae, femoral neck, total hip and trochanter in 483 Chinese women with osteoporosis, randomized to receive 5 mg zoledronic acid (ZOL), administered once every 12 months, or placebo equivalent. Data presented as mean ± SD. Bone mineral density was compared between treatment groups by Student’s 


No statistically significant differences were observed between the two treatment groups with respect to serious adverse events or deaths: eight (3.3%) patients in the ZOL group and nine (3.7%) patients in the placebo group experienced serious adverse events or died during the study. Serious cardiac symptoms were reported in six (2.5%) patients in the ZOL group and eight (3.3%) patients in the placebo group. There were no significant differences in the incidences of the most common adverse events, which were first-dose acute-phase reactions: headache, chills, pyrexia, myalgia, arthralgia and influenza-like symptoms were reported in 67 (27.7%) patients receiving ZOL and 61 (25.3%) patients receiving placebo. There were no significant differences in the incidences of the more serious events of atrial fibrillation, cardiac arrhythmia and renal dysfunction.
Discussion
In the present randomized controlled clinical trial, treatment of Chinese women with osteoporosis with an annual injection of 5 mg ZOL significantly increased BMD in the L1–L4 vertebrae, total hip, femoral neck and trochanter, and lowered the incidence of fractures, compared with the rates observed in the placebo group. The differences in BMD between the two groups remained significant during 24 months’ follow-up.
Findings from the present study are consistent with those of previous trials.23–25 Findings of a study in Taiwan indicated that, compared with placebo, ZOL was associated with an increase in BMD of 4.9%, 4.3% and 7.0% in the total hip, femoral neck and trochanter, respectively, in Chinese women with osteoporosis. 24 Similarly, it has been reported that an annual single 15-min infusion of 5 mg ZOL significantly improved BMD over a 3-year period and was not associated with serious adverse events, compared with placebo. 23 Published data on the effects of ZOL in osteoporosis are, however, inconsistent. Another large sample study with 7765 postmenopausal osteoporosis-affected women indicated that 5 mg ZOL had no significant influence on BMD in the femoral neck compared with placebo. 26 Such discrepancies in clinical outcome may be due to differing case backgrounds, sample size or other factors. The role of ZOL in women with osteoporosis, therefore, needs to be validated in larger sample-size studies (including women of different ethnicities or including more ethnically diverse groups).
In the present study, the overall fracture risk over 24 months in osteoporosis-affected women treated with ZOL was lower compared with placebo, which confirms the findings of previous studies.24–26 It has been reported that ZOL treatment significantly lowered the incidence of clinical vertebral and nonvertebral fractures compared with placebo (hazard ratio [95% CI] of ZOL for fracture during the 3-year follow-up period 0.34 [0.21, 0.55]).
25
In their study, Hwang
The most common adverse events associated with ZOL treatment in the present study were first-dose acute-phase reactions such as pyrexia, myalgia, arthralgia, headache, chills and influenza-like symptoms. Similar symptoms have also been reported elswhere.23,27–30 The incidence of serious adverse events in the present study was, however, markedly lower than that determined in some investigations24,30 and this may be due to discrepancies in the average BMI between the populations studied.
In conclusion, results of the present randomized, placebo-controlled clinical trial indicate that a single yearly dose of 5 mg ZOL can increase BMD and decrease the risk of fracture among female patients with osteoporosis over a 24-month period. Furthermore, ZOL was not associated with any serious drug-related adverse effects. The present findings suggest that ZOL could be used as an effective treatment for women with osteoporosis.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgements
The authors would like to thank the First Affiliated Hospital of the General Hospital of the People’s Liberation Army for its help and all the patients who participated in this study.
