Abstract
The different types of diabetes differ in disease pathogenesis but share the impairment or loss of β-cell function leading to chronic hyperglycaemia. While immune cells are present throughout the whole pancreas in normality, their number and activation is increased in diabetes. Different patterns and composition of inflammation could be observed in type 1, type 2 and type 3c diabetes. Immune cells, pancreatic stellate cells and fibrosis were present in the islet microenvironment and could add to β-cell dysfunction and therefore development and progression of diabetes. First studies investigating the use of anti-inflammatory drugs demonstrate their ability to rescue remaining β-cell function and their potential benefit in diabetes treatment. This article provides an overview of immune cell infiltrates in different types of diabetes, highlights the knowledge of their impact on β-cell function and introduces the potential of immunomodulatory strategies.
Introduction
Diabetes summarizes diseases of the pancreas leading to continuous hyperglycaemia and with that to side effects including microvascular and macrovascular complications. There are different types of diabetes with different disease patterns and causes; however, high blood glucose levels unify all of them. Different types of diabetes include type 1 diabetes (T1D), type 2 diabetes (T2D) and type 3c diabetes (T3cD). T1D is characterized by insulin deficiency caused by immune-associated destruction of pancreatic β-cells leading to chronic hyperglycaemia. 1 It accounts for around 23% of all diabetes cases with high global variation.1,2 It is believed to be caused by genetic as well as environmental causes with the gene predisposition possibly leading to the presence of activated immune cells in the pancreas already early in life. 1 The impact of the disease on the pancreas includes loss of β-cells and insulin expression and loss of pancreatic volume.1,3 T2D is the most common form of diabetes accounting for up to 90% of all cases.2,4 The disease is characterized by insulin resistance and/or impaired insulin secretion leading to chronic hyperglycaemia. 4 Both environmental and genetic risk factors impact on the development of T2D; however, there seems to be a strong correlation with environmental factors especially obesity. 4 T3cD, also known as pancreatogenic or secondary diabetes, describes a group of diseases of the exocrine pancreas leading to diabetes. These diseases include most commonly chronic pancreatitis (CP), but also cystic fibrosis (CF) and pancreatic cancer. 2 It is estimated that 1–9% of patients with diabetes have T3cD; however, the number might be highly underestimated due to difficulty with the diagnosis. 2 People with T3cD can present with impaired islet function often only evident during a glucose challenge and they can also present with ‘brittle’ diabetes showing through frequent hypoglycaemic events. 5
In general, inflammation is a process dedicated to eliminate infections in an acute response which can turn into a chronic process if pathogens are persistent.6,7 The pathway of inflammatory responses starts with inducers including microbes or intracellular proteins triggering the production of mediators for example cytokines to recruit leucocytes and resolve the impact of the inducer. 6 Chronic inflammation can be accompanied by fibrosis impacting on tissue morphology and function. 7 Klöppel et al. summarizing the staged process of fibrosis in the pancreas: initial injury leading to apoptosis/necrosis of cells during which cytokines get released; clear up of damaged cells by macrophages and activation of pancreatic stellate cells (PSCs) by the previously released cytokines; production and deposition of extracellular matrix (ECM). 8 Following I set out to summarize relevant literature to summarize the immune cell infiltration in the different types of diabetes, highlight key players, the impact of inflammation on β-cell function and potential implications of anti-inflammatory treatments for people with diabetes.
Methodology: Search strategy
To find relevant literature for this review, a search was conducted in the PubMed database as well as Google Scholar with the following terms: ‘type 1 diabetes’, ‘type 2 diabetes’, type 3c diabetes’, ‘inflammation’, ‘immune cell infiltration’, ‘insulitis’, ‘fibrosis’, ‘chronic pancreatitis’, ‘cystic fibrosis’, ‘pancreatic stellate cells’, ‘beta-cell function’, ‘cytokines’ and ‘anti-inflammatory treatment’. In addition, a search in the references of found publications was conducted.
Immune infiltration in diabetes
Type 1 diabetes
With T1D having an autoimmune component, immune cell infiltration in islets, called insulitis, has been studied in rodent and human pancreata. Research in the non-obese diabetic (NOD) mouse, a well-characterized model of T1D presenting with immune cell infiltration, allows the investigation of cell infiltration over the development of the disease. Studies highlighted the recruitment of different immune cells in the pancreas starting with dendritic-like cell and macrophage infiltration in vessels adjacent to islets moving towards para- and peri-insular infiltration with increasing age of the mice. 9 In a later stage, lymphocytes could be observed in the combination, the infiltration moved into islets and during these phases, β-cell destruction was observed.9,10 Additional studies showed that plasmacytoid dendritic cells are potent negative regulators of insulitis. 11 Although differences between rodent models and man exist, many similarities provide the possibility to investigate disease development, longitudinal studies or intervention experiments.3,9–11
In human tissue, analysis of fixed pancreas biopsies allows insight into immune cell infiltration which seemed to be less frequent in human pancreas samples compared to rodent models. 12 In addition, infiltrating immune cells can also be observed in non-diabetic control pancreata which led to different definitions of insulitis in different studies.3,13–16 In auto-antibody positive (AA+) donors, a state occurring prior to the T1D diagnosis, limited immune cell infiltration in islets was observed.14,17,18 Different studies investigating samples from patients with recent onset diabetes (<7 years) or donors with young age showed increased infiltrating immune cells in islets and their periphery compared to control patients or patients with longer standing diabetes.18–25 However, the number of affected donors and islets remains quite limited together with the number of islet infiltrating immune cells.3,15,21 Majority of studies are reporting an increased number of infiltrating immune cells in insulin containing islets compared to insulin-deficient islets indicating that insulitis might occur due to an auto-immune process.3,15,17,18,20,23,26,27 A variability of immune cell composition and number was observed in the different tissues indicating a heterogeneity between different patients even with similar disease stage.15,18,28 Studies investigating immune cell composition in more detail observed the majority of the infiltrates to be composed of cytotoxic T-cells followed by macrophages, B-cells and T-helper cells.15,21,22,28 Deeper analysis of islet-derived cytotoxic T-cells and T-helper cells from T1D donors revealed their reactivity to islet auto-antigens indicating their role in disease pathogenesis.28,29 In addition to insulitis, fibrosis as well as hyaluronan accumulation inside of islets could also be observed in some cases potentially impacting on stiffness of the tissue and contributing to the inflammatory process in the organ.3,19,20,30–32
The impact of T1D on the exocrine pancreas becomes obvious by the decrease in whole pancreas weight in AA+ and T1D organs.18,25,33 In AA+ pancreata, cases of acute pancreatitis and mild CP were observed in the exocrine parenchyma with T-helper cells as well as myeloid dendritic cells present.17,18 Some cases of T1D pancreata presented with focal acute pancreatitis, mild-to-moderate chronic pancreatitis or diffuse lymphocytic infiltrate in the acinar parenchyma, in and around ducts or around vessels.17,18,25,26 Rodriguez-Calvo et al. reported a significant increased number of T-helper cells, cytotoxic T-cells and myeloid dendritic cells in the exocrine tissue of T1D compared to control pancreata. 18
Taken together, these studies highlight the multiple inflammatory components impacting β-cell function in the pancreas of T1D. In the early stages or prior of the disease, immune cells are present in the exocrine tissue potentially being recruited towards the islets with the progression of the disease whilst dynamic changes in the composition can be observed. This highlights that the immune cascade could be targeted early on to dampen or prevent the progression towards the islets and therefore halter β-cell destruction in T1D. Further studies are needed for a deeper understanding of the activation patterns of tissue resident immune cells and their recruitment towards the islets. Additional multiplex staining or isolation of immune cells in the pancreas or islets following sequencing or proteomic analyses might lead to a better understanding in the role immune cells play in the development of diabetes.16,34
Type 2 diabetes
Increasingly more studies investigating inflammation in cases of T2D in animal models as well as man, found the presence of immune cells in the exocrine and endocrine parts of the organ. Interestingly, there is evidence indicating that insulitis can also be present in T2D with different diabetic and obese animal models presenting with immune cell infiltration into the endocrine compartment composed of mainly macrophages and granylocytes.7,35,36 A rodent non-obese model of T2D showed the progression of immune infiltration starting near ducts with granulocytes in younger animals moving towards islets with granulocytes and macrophages in later stages of the disease coinciding with presence of fibrosis. 36
Limited histological studies of islets in human T2D pancreata showed an increased infiltration of macrophages compared to non-diabetic controls.15,34,35,37 However, the number of infiltrating macrophages in T1D was observed higher than T2D in some islets. 15 In addition, a significantly increased number of cytotoxic T-cells was seen in the periphery of islets in T2D with no difference in islet infiltrating T-cells compared to islets of non-diabetic pancreata.18,34 Islet amyloid deposition, increasingly found in islets of T2D patients, was shown previously to increase β-cell apoptosis and decrease β-cell function possibly by the disturbance of cell-to-cell connections through the oligomers. 38 A transgenic mouse model showed the impact amyloid can have on increased expression of proinflammatory chemokines and cytokines as well as other inflammatory markers. 39 Resident islet macrophages can be stimulated by islet amyloid polypeptide aggregation to produce proinflammatory cytokines adding to the inflammatory environment.40,41 In addition significantly increased hyaluronan accumulation was observed in an amyloid mouse model accompanied by increased macrophage infiltration potentially impacting on inflammation as well as stiffness of the islet microenvironment.32,39
The impact of T2D on the exocrine pancreas can be seen in the decrease in pancreas weight by 15% compared to non-diabetic controls. 42 Immune cell infiltration was also observed in the exocrine tissue of some pancreata in different studies which included cytotoxic T-cells, T-helper cells and macrophages with the density of cytotoxic T-cells significantly higher in T2D than in control pancreata.17,18,34,43
In summary, these studies highlight the presence of inflammation in the exocrine and endocrine T2D pancreas and indicating potential impact in the development and progression of diabetes as well as potential targets for anti-inflammatory treatments. The limited studies available so far indicate differences in cell composition and exocrine/endocrine infiltration between T1D and T2D; however, additional studies are required to investigate the progression and impact of inflammation in T2D pancreata and the involved immune cell types in more detail.
Type 3c diabetes
Chronic pancreatitis
CP is characterized by progressive destruction of acinar tissue and its replacement by inflammatory and fibrotic tissue impacting on the function of the organ. 44
Macrophage infiltration was observed in a mouse model of CP accompanied with an increased expression of macrophage-associated cytokines and chemokines indicating their presence and signalling in the pancreas. 45 An anti-inflammatory and anti-fibrotic acting microRNA (miR-29a) was observed to be significantly reduced in the acute phase of a pancreatitis mouse model as well as locally in human CP tissue samples co-localizing with changed morphology. 46 This shows parallels between the rodent model and human samples and indicating the presence of fibrotic changes in the investigated pancreata.
A significant increase in connective tissue and mononuclear cells was observed in human CP pancreata positively correlated with patient reported pain intensity. 47 Immune cell infiltration with lymphocytes and macrophages was observed in fibrotic areas in the exocrine part of the pancreas, but very rarely in islets.15,43,45,48 The impact of pancreatitis on the whole pancreas was also notable by an increase in stiffness observed in acute and CP.49,50
In summary, the limited studies highlight the presence of focal inflammation in the exocrine pancreas but limited presence in the islet microenvironment. However, additional studies are needed to investigate the impact of inflammation on islets as well as the development of diabetes in people with CP to gain a better understanding what part of inflammation is impacting on islet dysfunction.
Cystic fibrosis
CF is caused by mutations in the CF transmembrane conductance regulator (CFTR) channel impairing anion transport across the cell membrane. In the pancreas, CF manifests by duct dilation and extensive replacement of acinar parenchyma by fibrosis and fat cells. 51 A well-studied animal model of CF, the CFTR-knock out ferret, suggests that apoptotic damage due to duct dilatation might be a trigger for the fibrous and inflammatory process with young CF ferrets presenting with increased fibrous tissue replacing acinar parenchyma. 52 Infiltration of neutrophils, macrophages and lymphocytes into exocrine tissue could already be observed in new-born CF ferrets which was not observed in non-CF controls. 52 Analysis of circulating mRNA expression of cytokines revealed a significant increase and peak expression of IL-8, IL-1β, IL-6 and CXCL10 during early life phases of CF ferrets during which these also experience a glycaemic crisis. 53 Tumour necrosis factor (TNF)-α and TGF-β1 showed their peak expression earlier and before the glycaemic crisis indicating their potential role in the start of the inflammation process in the pancreas. 53 Additional analysis of serum cytokine levels, which were not significant different between genotypes except in new-borns, indicated that pro-inflammatory changes develop local rather than systemic. 53 Isolated neonatal islets from this ferret model showed a significantly increased expression of some cytokines in CF compared to non-CF indicating the possible inflammatory signalling of islets themselves. 54
Studies investigating human CF pancreas, observed leucocytes infiltrating in islets in young CF donors and in decreased numbers in adult donors with a fibrotic disease pattern.51,55,56 In young donors, the infiltrates consisted mostly of T-cells as well as macrophages which were not visible in adult islet infiltrates. 55 The number of immune cell infiltrates did not correlate with insulin expression of the islets. 55 Another study reported no macrophage infiltration in islets; however, IL-1β expression was detected indicating a proinflammatory signalling in the islet area. 57 Isolated islet-infiltrating T-cells showed to be able to produce inflammatory cytokines which are able to negatively impact β-cell function. 56
Immune cells were also observed in the exocrine tissue of CF donors independent of age with leucocytes, T-cells and macrophages.55,57 However, in older cohorts, the numbers of immune cells was less and macrophages were not significantly different to control pancreata. 55 Exocrine parenchyma of adult CF pancreata with a fibrotic pattern contained more immune cells compared to a lipoatrophic pattern.51,55
Taken together, these studies show that in both the animal model and the human CF pancreas, inflammation can be observed in the exocrine and endocrine department. This indicates that islets can be negatively impacted by inflammatory cells and their signalling leading to the development of diabetes. Future studies are needed to investigate the immune cell infiltration in more detail to get a better understanding of the part inflammation plays in the development of CF-related diabetes.
Example images of immune cell infiltration and fibrosis in different pancreata
Immuno-phenotyping of different pancreata was undertaken in the Quality in Organ Donation (QUOD) pancreas expand biobank collecting biopsies of whole donor pancreata in a standardized manner for research purposes from deceased donors after appropriate consent. A deeper analysis of the cohort is ongoing, but following are examples to visualize immune cell infiltration and collagen deposition in the microenvironment of islets in a non-diabetic control, T1D, T2D and CF pancreas (Figure 1). The example of a female and 39 years old non-diabetic control (panels C-1, C-2, C-3) pancreas shows a few leucocytes (panel C1, brown) as well as T-cells (panel C-2, red), but no B-cells (panel C-3, brown) in the microenvironment of islets (dotted line). The collagen is visualizing the basement membrane around the islets, around vessels and around acini (panel C-3). In the example of a male and 38 year-old-donor with the history of T1D (panel T1D-1, T1D-2, T1D-3), T-cells (panel T1D-1, red) as well as macrophages (panel T1D-2) both CD68+ (brown) and CD163+ (red) are present in the microenvironment of islets. No B-cells (panel T1D-1, brown) were visible in this case. Increased collagen deposition can be observed in the exocrine parenchyma also in close proximity to the pictured islets (panel T1D-3). In an example case for T2D of a male donor, 53 years of age (panel T2D-1, T2D-2, T2D-3), leucocytes (panel T2D-1, brown) and T-cells (panel T2D-2, red), but no B-cells (panel T2D-2, brown) were visible in the periphery of islets. Collagen (panel T2D-3) is present in and around the islets as well as in the exocrine tissue appearing similar to the control pancreas. The example of a male and 27-year-old donor with CF shows the impact CF can have on the pancreas with no acinar parenchyma visible (panel CF-1, CF-2). Leucocytes are present in the microenvironment of the islets (panel CF-1, brown) and a high amount of collagen (panel CF-2) is surrounding the pictured islets.

Example images of non-diabetic control (C), T1D, T2D and CF pancreata stained for immune cell marker and collagen distribution in the proximity of pancreatic islets (dotted lines). The sections were stained for immunohistochemistry by Novopath at RVI Newcastle with optimized and standardized protocols on a Ventana Discovery Ultra TM (Roche Diagnostics, Burgess Hill, United Kingdom). The non-diabetic control donor was female, 39 years old with no history of diabetes. C-1: CD45 in brown indicating the presence of immune cells in the exocrine tissue of control pancreata. Brown arrows indicate examples of CD45+ cells. C-2: CD20 in brown and CD3 in red highlighting no positivity for B-cell or T-cell marker in the control pancreas. Red arrows indicate examples of CD3+ cells, no CD20+ cells present in this image. C-3: SRFG staining shows collagen in red indicating the distribution of the basement membrane in the normal pancreas. The donor with a history of T1D was male, 38 years of age. T1D-1: CD20 in brown and CD3 in red indicating the presence of B-cells and T-cells in the T1D pancreas. Red arrows indicate examples of CD3+ cells, no CD20+ cells present in this image. T1D-2: CD68 in brown and CD163 in red demonstrating the presence of macrophages in the T1D pancreas. Brown arrows indicate examples of CD68+ cells, red arrows indicate examples of CD163+ cells. T1D-3: SRFG highlighting the increased collagen deposition in this T1D pancreas. The donor with T2D was male, 53 years of age. T2D-1: CD45 staining in brown indicating the presence of immune cells in the T2D pancreas. Brown arrows indicate examples of CD45+ cells. T2D-2: CD3 in red and CD20 in brown highlighting the presence of T-cells or B-cells in the periphery of islets in the T2D pancreas. Red arrows indicate examples of CD3+ cells, no CD20+ cells present in this image. T2D-3: SRFG staining highlighting collagen distribution in the T2D pancreas. The donor with a history of CF was male, 27 years of age with end stage CF in the pancreas. CF-1: CD45 in brown indicating the presence of immune cells in the CF pancreas. Brown arrows indicate examples of CD45+ cells. CF-2 SRFG highlighting severe changes in the exocrine tissue of the CF pancreas. The non-diabetic control, T1D and T2D pancreas were kindly provided by the QUOD pancreas expand biobank at Newcastle University. The CF pancreas was kindly provided by Prof Günter Klöppel.
The role of PSCs in inflammation of the pancreas
Next to immune cells, PSCs are thought to play an important role in the inflammation of the pancreas.58–60 PSCs are a myofibroblast-like cell type, possess are star-like shape and can be identified by α-smooth muscle actin and vimentin expression.58–61
Activated PSCs are thought to play an important role in pancreas fibrosis evident through a close proximity between these cells and affected areas.59–61 Cytokines including TGF-β and platelet-derived growth factor released by acinar cells in response to injury might induce activation on PSCs.59,60,62,63 It might also be possible that changes in the organ stiffness due to ECM deposition impact on the activation of PSCs similarly seen in hepatic stellate cells. 64 Once activated, PSCs produce fibril forming collagens including type I, type III and type XI as well as fibronectin and laminin contributing to the fibrotic changes in the organ.59–61,63,65,66 PSCs also express matrix metalloproteases (MMPs) including MMP-3 and MMP-9 which are proteins important for ECM degeneration. 65 The secretion can be inhibited in an autocrine manner by the secretion of TGF-β which can impact on the collagen deposition during injury. 65
Activated PSCs were observed in different pancreatic diseases including CP, CF and pancreatic cancer which also present with inflamed regions.54,61,66 In these tissues, the activation could start either due to direct impact of the stressor, for example alcohol, or following the sequence of injury, necrosis and fibrosis.8,48,67 In the initial stages of tissue injury in the pancreas, activated PSCs as well as macrophages were observed.48,54 The potential markers of stellate cell activation ACTA1 and ACTA2 were increased in islets obtained from early stages of a CF ferret model indicating their role in the early stages of the disease. 54 Isolated PSCs from a CP mouse model did secrete higher levels of cytokines including IL-4, IL-5, IL-10, IL-13 and TGF-β, whereas the expression of interferon (IFN)-γ, TNF-α and IL-1β was very low. 45 In addition, it was shown that these cytokines and additional secreted factors were able to promote macrophage polarization towards M2 which could also be confirmed with human PSCs. 45 With macrophages being key regulators of inflammation as well as fibrosis in a variety of tissues including the pancreas, this indicates a close interplay of different cell types during the course of inflammation in the pancreas. 68
Inflammatory environment and β-cell function
Next to direct T-cell-mediated impact on β-cell viability, cytokine-mediated pathways might impact on β-cell function. Cytokines and chemokines were shown to be secreted by different immune cells as well as PSCs and were shown to decrease functionality of β-cells. 7 Different studies showed that cytokines including IL-1β, INF-γ and TNF-α which are secreted by different immune cells as well as PSCs can negatively impact on viability and function of β-cells.69–71 It was demonstrated previously that PSCs can impair β-cell function and induce death which can be incremented by hyperglycaemia.72,73 Increased glucose/palmitate was leading to release of IL-6, IL-8, chemokine KC and G-CSF in mouse and human islets indicating the recruitment of immune cells and/or the activation of resident PSCs as a stress response.7,35 Surprisingly, IL-6 was shown to positively impact on α-cells promoting growth and function which is believed to possible compensate for impaired β-cell function in diabetes. 7 Furthermore, it was shown that the stiffness of surrounding microenvironment is impacting on β-cell function due to mechanosensing through Roh-associated protein kinase (ROCK) and myosin light-chain kinase pathways as well as β-catenin signalling. 74 Changes in pancreas stiffness through inflammation and fibrosis can add to the stress on β-cells possibly leading to dysfunction.
Anti-inflammatory treatment options
Different studies with anti-inflammatory treatments in patients with T1D or T2D showed the potential benefit of these treatments on β-cell function.75,76 These therapies have different target opportunities with either impacting on immune cells directly or intervening on their secreted signals including cytokines.75,76 Various studies targeting immune cells with anti-CD20 or anti-CD3 antibodies in recent onset T1D showed that these can partially preserve β-cell function even with short-term treatments of 1 year.75,77–79 In targeting cytokines including TNFα, a small pilot study demonstrated a preserved β-cell function. 80 Treatment of children, newly diagnosed with T1D, with anti-inflammatory serum protein α1-antitrypsin in a small cohort lead to improved or maintained β-cell function in some of the patients. 81 However, a multi-centre trial with IL-1 inhibition via antibody or receptor antagonist for 9–12 months showed no impact on β-cell function compared to placebo treatments. 82 Furthermore, another trail investigating the inhibition of IL-1β did not reduce the incident of new onset T2D and demonstrated no long-term improvement of glycaemic levels. 83 In addition, well-known anti-inflammatory drugs including salicylates demonstrated improved β-cell function with improved glycaemic control in short-term trials.84–86 These trials show the potential to rescue remaining β-cell function with anti-inflammatory treatments making it possible to improve treatments for different types of diabetes. 75 However, to date, these treatments fail to cure diabetes as well as prevent onset of diabetes, highlighting the need for further research in this aspect of disease pathogenesis.75,83
Conclusion
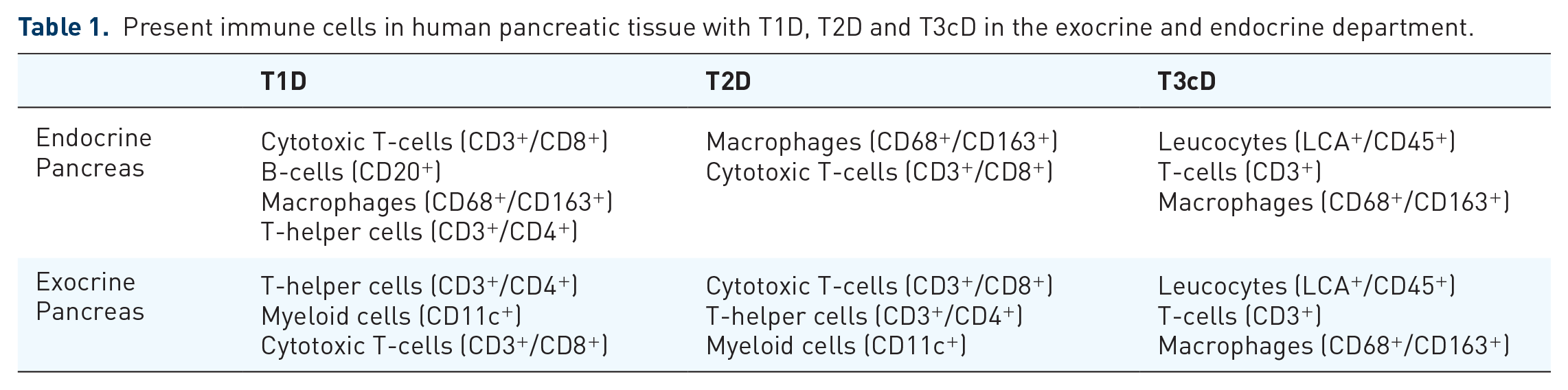
Taken together, it is evident that immune cells are present in the normal pancreas and an increased infiltration in endocrine and exocrine departments can be observed in all types of diabetes (Table 1). The studies present found a difference in the immune cell composition in the different types of diabetes, potentially due to different disease pathogenesis. The process of inflammation and fibrosis seems to be dynamic with changes in cell composition at different points of the disease progression. The impact of the inflammatory mechanisms on islets and specific β-cells cannot be denied and can potentially add to the stressors already present in diabetes including hyperglycaemia. Anti-inflammatory treatments are able to rescue remaining β-cell function indicating their potential in improving care in different types of diabetes. Further research will be required to better understand the involvement of inflammation in the development and progression of diabetes working towards improved therapies.
Present immune cells in human pancreatic tissue with T1D, T2D and T3cD in the exocrine and endocrine department.