Abstract
Background:
Metabolic syndrome (MetS) is defined by at least three of the following five criteria: blood pressure ⩾130/85 mmHg, fasting blood glucose ⩾5.6 mmol/l, triglycerides concentration ⩾1.7 mmol/l, waist circumference ⩾102 cm (for men), and high-density lipoprotein cholesterol concentration <1.03 mmol/l (for men). MetS has been associated with worse lower urinary tract symptoms (LUTS) and higher International Prostate Symptom questionnaire scores.
Materials and Methods:
MEDLINE, Cochrane, ClinicalTrials.gov, and SCOPUS were critically appraised for all peer-reviewed manuscripts that suitably fulfilled our protocol’s inclusion criteria established a priori. Meta-analytical and meta-regression calculations were performed in R using the Sidik–Jonkman and Hartung–Knapp random effects model and predefined covariates.
Results:
A total of 70 studies (n = 90,206) were included in qualitative synthesis. From these, 60 studies focused on MetS and LUTS: 44 reported positive correlations, 5 reported negative correlations, 11 reported no association, and 10 studies focused on MetS and total prostate volume (TPV). MetS positively correlated with moderate LUTS [odds ratio (OR) = 1.56, 95% confidence interval (CI) = 1.35–1.80], severe LUTS (OR = 2.35, 95% CI = 1.82–3.03), overactive bladder (OAB; OR = 3.2, 95% CI = 1.6–5.8), and nocturia severity (OR = 2.509, 95% CI = 1.571–4.007) at multivariate analysis. A total of 30 studies (n = 22,206) were included in meta-analysis; MetS was significantly associated with higher TPV (mean differences = 4.4450 ml, 95% CI = 2.0177–6.8723), but no significant predictive factors for effect sizes were discovered.
Conclusion:
Our meta-analysis demonstrates a significant association between the aggravating effects of MetS, which commonly coexists with obesity and benign prostate enlargement.
Keywords
Introduction
Metabolic syndrome (MetS) is defined by at least three of the following five criteria: blood pressure (BP) ⩾130/85 mmHg, fasting blood glucose (FBG) ⩾5.6 mmol/l, triglycerides (TG) concentration ⩾1.7, waist circumference (WC) ⩾102 cm for men and ⩾89 cm for women, and high-density lipoprotein cholesterol (HDL-C) concentration <1.03 mmol/l for men and <1.4 mmol/l for women. 1 One of the major contributing factors to MetS is obesity; the prevalence of those with obesity has almost since 1975. 2 In England, it affects 28% of adults and it was directly associated with 1117 hospital admissions in 2018/2019.3,4
Body mass index (BMI) ⩾35 kg/m2 has been positively correlated with moderate–severe lower urinary tract symptoms (LUTS) [odds ratio (OR) = 1.38, 95% confidence interval (CI) = 1.17–1.63]; 5 WC ⩾42 inches (106.7 cm) was also a significant factor. 6 In addition, low-density lipoprotein cholesterol (LDL-C) concentration >7.4 mmol/l caused a fourfold increased risk of benign prostatic hyperplasia (BPH; OR = 4.00, 95% CI = 1.27–12.63, p = 0.02). 7 LUTS encompass a variety of bladder conditions: BPH, urinary tract infection (UTI), overactive bladder (OAB), nocturia, interstitial cystitis (IC), and bladder pain syndrome (BPS). LUTS consist of storage symptoms (urinary incontinence, urgency, frequency, and nocturia), voiding symptoms (intermittency, slow stream, hesitancy, straining to void, terminal dribble, and splitting of stream), and post-micturition symptoms (incomplete bladder emptying).8,9 Obesity and more specifically patients with a BMI ⩾35 kg/m2 have been positively correlated with moderate–severe LUTS (OR = 1.38, 95% CI = 1.17–1.63).5,7 LUTS leads to worsening quality of life, sleep, and mental health in men and women. 9 LUTS severity may be quantified by the International Prostate Symptom Score (IPSS) that looks mild, moderate, and severe symptoms. 8
This systematic review and meta-analysis aims to review all existing evidence on the association between MetS and in LUTS – more specifically, the effect of MetS on prostatic inflammation and subsequent hyperplasia in patients with LUTS and BPH. MetS is a growing problem worldwide, and its role in LUTS is unclear; LUTS etiology is not entirely understood. While studies point toward an association between MetS and LUTS, several studies reported no association at multivariate analysis.10–13 Our aim is to provide new insight and propose therapeutic targets for MetS and LUTS.
Materials and methods
The protocol was developed according to the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) and followed methods outlined in The Cochrane Handbook for Systematic Reviews of Interventions. 14 This systematic review has been registered with PROSPERO (international prospective register of systematic reviews) with registration number CRD42020223412.
Search strategy
Two reviewers conducted systematic searches of the following databases: Medical Literature Analysis and Retrieval System online (MEDLINE), SCOPUS, Cochrane Central Register of Controlled Trials (CENTRAL), and ClinicalTrials.gov databases. The following MeSH (Medical Subject Heading) terms were used: (((((metaflammation) OR (metabolic cells)) OR (mitochondrial dna)) OR (inflammaging)) OR (metabolic syndrome)) AND (((((lower urinary tract symptoms) OR (luts)) OR (urinary tract infection)) OR (uti)) OR (interstitial cystitis)). In addition, reference lists of selected articles and other literature sources were browsed to ensure a comprehensive literature search was completed. Searches filtered results based on year of publication date (last 10 years), and the last search was carried out on 24 October 2020.
Study selection
Studies were imported into Covidence [Covidence (Veritas Health Innovation, Melbourne, Australia; http://www.covidence.org)]. 15 All studies were screened for selection by two reviewers independently (of a group of five) and any conflicts were resolved by a third reviewer. Selection was completed in two stages – first by title and abstract and then by full text. Studies were selected using specific criteria which removed duplicates. Five reviewers selected studies individually, and once completed, a second reviewer selected the studies. A third reviewer resolved conflicts. Studies were screened for title and abstracts and then full text screened. Studies were included if they met the inclusion criteria: cohort studies, case-control studies, randomized clinical trials, and cross-sectional studies (no limit on sample size, setting, follow-up period, or intervention); men and/or women aged 18 years or above; any component of MetS; any LUTS condition, for example, LUTS/BPH, OAB, detrusor overactivity (DO), and urinary incontinence (UI); and original articles. Exclusion criteria included the following: studies including children, pregnant women, bladder or prostate cancers/other forms of cancers, and animal models; editorials, letters, case reports, opinion pieces, commentaries, systematic reviews, and meta-analyses; and articles not in English.
Data extraction
Five reviewers extracted data using Covidence. 15 A second reviewer checked the data extracted. Finally, the data were exported to Microsoft Excel from Covidence. Example of columns: reference, country, study design, start date, end date, method to classify LUTS, type of LUTS, sample size, gender, population description, MetS criteria, outcome measured, summary of association of Mets and LUTS, and quality assessment. Meta-analysis and meta-regression were conducted from February 2021 to 26 April 2021.
Quality assessment
Each study was assessed for bias using the Newcastle–Ottawa scale (NOS). Studies were evaluated on eight factors, categorized into three groups: selection (including whether the cohort is representative of the population), comparability (assessed on grounds of study design and the analysis performed), and outcome (i.e. the assessment of outcome, follow-up rate, and adequacy of follow-up period). Stars were awarded per category, with a maximum of four, two, and three stars possible for the ‘selection’, ‘comparability’, and ‘outcome’ categories, respectively. 16 Five reviewers assessed the studies to be of poor (three stars or less), fair (four–six stars), or good (seven–nine stars) quality (NOS). A risk of bias assessment using the Quality in Prognosis Studies (QUIPS) tool was also carried out for all 30 studies included in meta-analysis. 17 The QUIPS tool assessed study participation, study attrition, prognostic factor measurement, outcome measurement, study confounding, statistical analysis reporting, and overall risk of bias.
Data synthesis and statistical analysis
All meta-analytical calculations were carried out by an external statistician using R statistical software (v4.0.4) with meta package (v4.18-0). The drawn forest plots were contrived using this software. Pooled ORs were calculated with 95% CIs from the extracted count data, while continuous data were used to calculate pooled weighted mean differences (MD) with 95% CI. Pooled MD with 95% CI were calculated using the inverse variance method and random-effects model with Sidik–Jonkman estimation and Hartung–Knapp adjustment for random effects model. Presence of heterogeneity was tested using the χ 2 test and quantified with the I2 statistic (I2 > 75% considered significant). Heterogeneity was addressed by performing meta-regression analysis using mixed-effects model with predefined predictors (sample size, study rating, year of publication, and country of study). Meta-regression analysis was performed to address heterogeneity by checking for possible association of predefined factors (sample size, study rating, year of publication, and country of study) with effect size differences. Bubble plots were generated to visualize the results of meta-regression analysis. ORs were used to compare the relative odds of LUTS in relation to MetS. OR < 1 suggests the intervention or exposure is associated with reduced odds of said outcome occurring. OR = 1 suggests no association between the outcome and intervention. OR > 1 suggests higher odds of an outcome occurring as an association with an intervention. 14 Any potential publication bias was assessed with Egger’s test of intercept and visual evaluation of the funnel plot.
Results
In total, 1741 studies were imported into Covidence, which removed four duplicates. Four reviewers screened 1737 studies for title and abstracts, and 1518 were excluded. Five reviewers screened the full text of the remaining 219 studies; 149 studies were excluded. Seventy studies were included in qualitative synthesis and 30 in meta-analysis (Figure 1). Three studies used the same patient cohorts and were excluded.18–20 General characteristics of the included studies are presented in Table 1, while the outcomes measured and a summary of the association between MetS and LUTS are detailed in Table 2. A forest plot for total prostate volume (TPV) and MetS and mixed-effects model results are presented in Figure 2 and Table 3, respectively. Figure 3 represents meta-regression analysis (bubble plots) for age, study rating, and publication year. The results of the publication bias assessment – Egger’s test of the intercept – are presented in Figure 4. Figure 5 represents a QUIPS Risk of Bias Assessment for the 30 studies included in meta-analysis is presented as a graph (Figure 5) and table (Table 4).
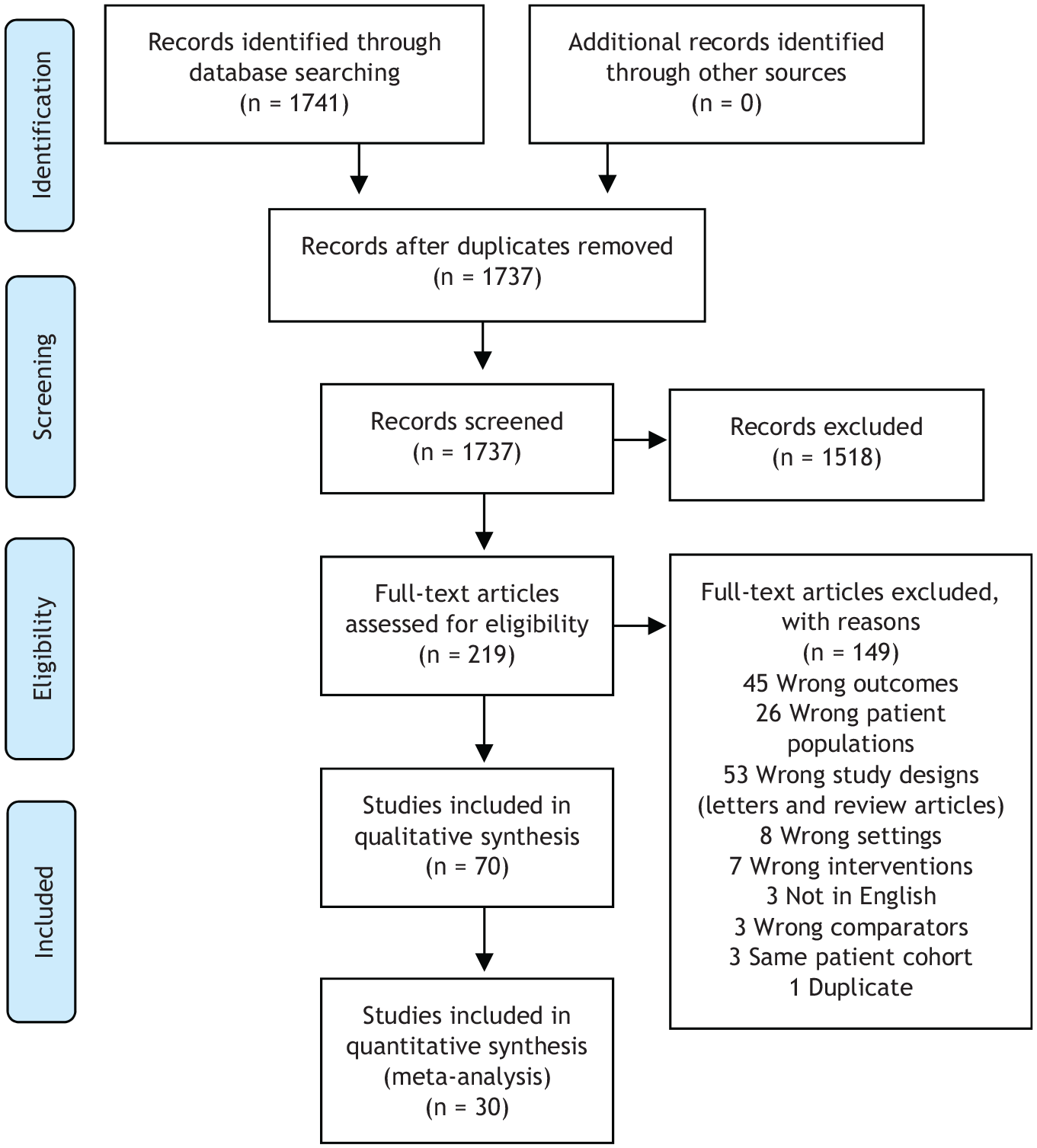
PRISMA flow diagram for studies assessed for eligibility from Moher et al. 21
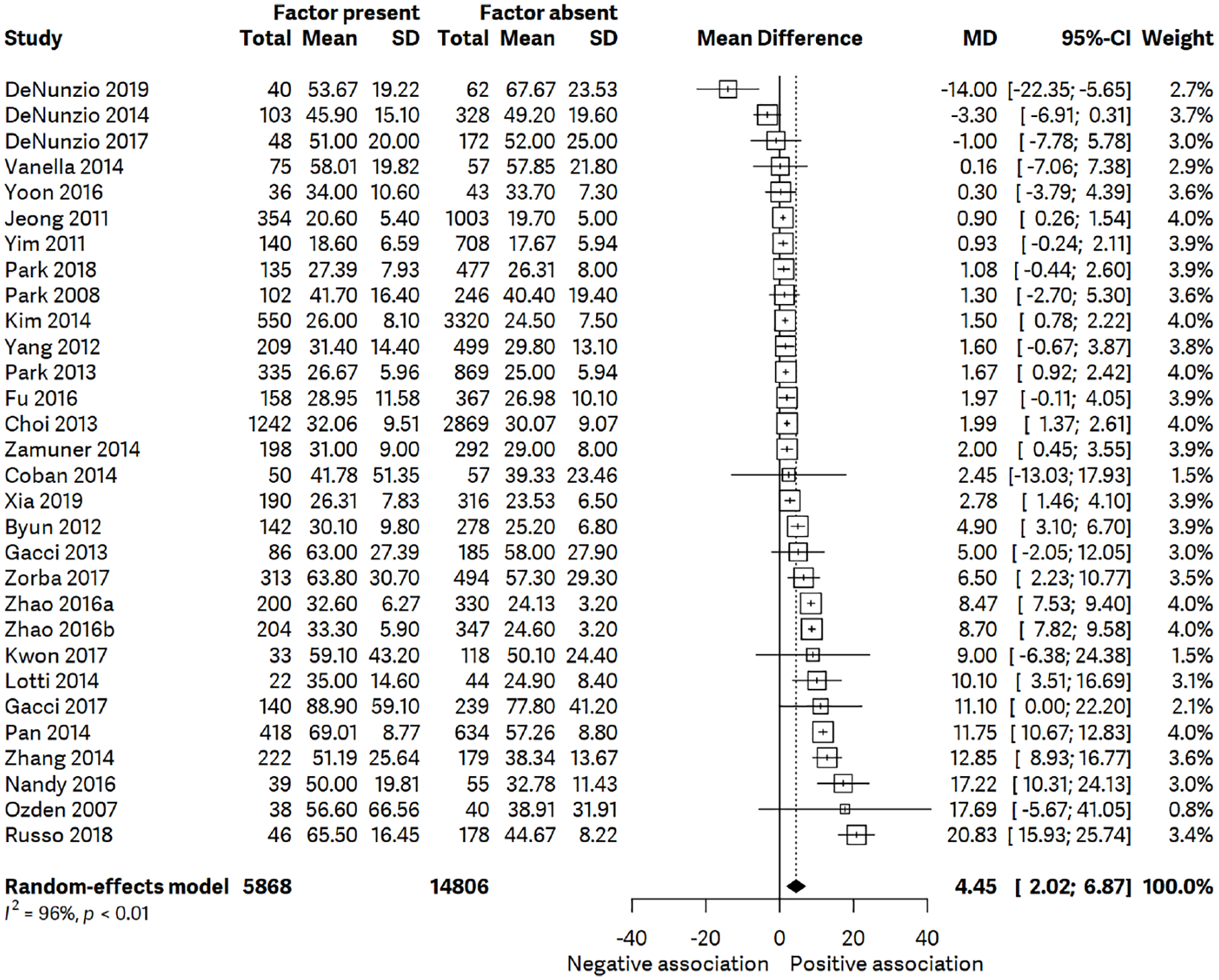
Forest plot for TPV and MetS. Number of studies combined: k = 30 (n = 22,206). MD = 4.4450; 95% CI = 2.0177–6.8723; t = 3.75; p = 0.0008. Quantifying heterogeneity: tau2 = 37.0851 [18.9614; 71.7320]; tau = 6.0898 [4.3545; 8.4695]. I2 = 96.3% [95.4%; 96.9%]; H = 5.17 [4.67; 5.72]. Test of heterogeneity: Q = 774.09; degrees of freedom (df) = 29; p < 0.0001. Details on meta-analytical method: inverse variance method; Sidik–Jonkman estimator for tau2; Q-profile method for confidence interval of tau2 and tau; Hartung–Knapp adjustment for random effects model.

Meta-regression analysis for predictors: (a) age, (b) study rating, and (c) publication year. Results were not significant.

Publication bias assessment. Egger’s test of the intercept: intercept 1.073; 95% CI = 1.71–3.86; t = 0.754; p = 0.4570147. Egger’s test does not indicate the presence of funnel plot asymmetry.

QUIPS risk of bias assessment graph for the 30 studies included in meta-analysis. Risk of bias for the following components: study participation, study attrition, prognostic factor measurement, outcome measurement, and study.
General characteristics of studies included in systematic review.
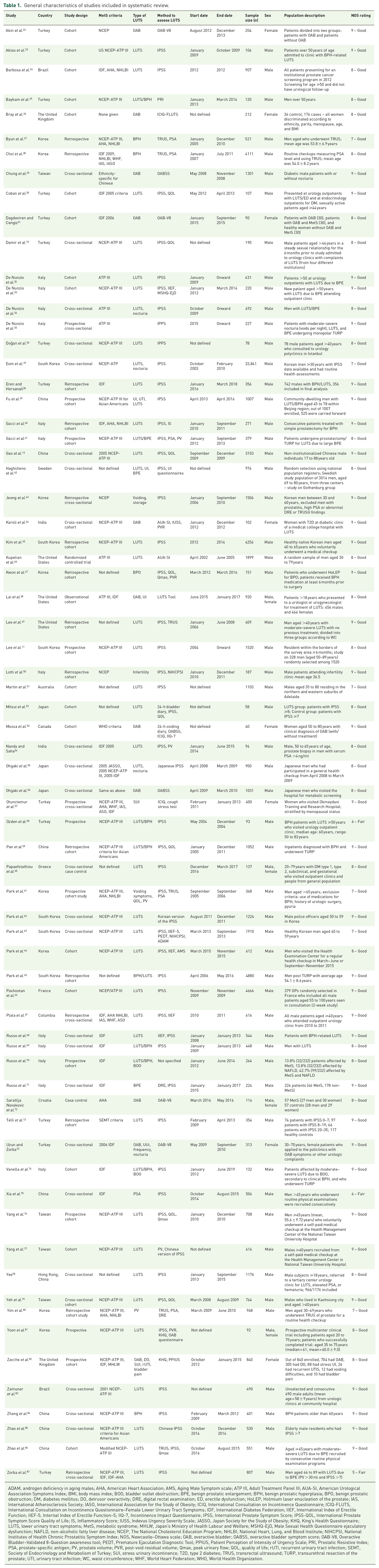
ADAM, androgen deficiency in aging males; AHA, American Heart Association; AMS, Aging Male Symptom scale; ATP III, Adult Treatment Panel III; AUA-SI, American Urological Association Symptoms Index; BMI, body mass index; BOO, bladder outlet obstruction; BPE, benign prostatic enlargement; BPH, benign prostatic hyperplasia; BPO, benign prostatic obstruction; DM, diabetes mellitus; DO, detrusor overactivity; DRE, digital rectal examination; ED, erectile dysfunction; HoLEP, Holmium laser enucleation of the prostate; IAS, International Atherosclerosis Society; IASO, International Association for the Study of Obesity; ICIQ, International Consultation on Incontinence Questionnaire; ICIQ-FLUTS, International Consultation on Incontinence Questionnaire–Female Lower Urinary Tract Symptoms; IDF, International Diabetes Federation; IIEF, International Index of Erectile Function; IIEF-5, Internal Index of Erectile Function–5; IIQ-7, Incontinence Impact Questionnaire; IPSS, International Prostate Symptom Score; IPSS-QOL, International Prostate Symptom Score Quality of Life; IS, Inflammatory Score; IUSS, Indevus Urgency Severity Scale; JASSO, Japan Society for the Study of Obesity; KHQ, King’s Health Questionnaire; LUTS, lower urinary tract symptoms; MetS, metabolic syndrome; MHLW, Japan’s Ministry of Health Labour and Welfare; MSHQ-EjD, Male Sexual Health Questionnaire ejaculatory dysfunction; NAFLD, non-alcoholic fatty liver disease; NCEP, The National Cholesterol Education Program; NHLBI, National Heart, Lung, and Blood Institute; NIHCPSI, National Institutes of Health Chronic Prostatitis Symptom Index; NOS, Newcastle–Ottawa scale; OAB, overactive bladder; OABSS, overactive bladder symptom score; OAB-V8, Overactive Bladder–Validated 8-Question awareness tool; PEDT, Premature Ejaculation Diagnostic Tool; PPIUS, Patient Perception of Intensity of Urgency Scale; PRI, Prostatic Resistive Index; PSA, prostate-specific antigen; PV, prostate volume; PVR, post-void residual volume; Qmax, peak urinary flow; QOL, quality of life; rUTI, recurrent urinary tract infection; SEMT, Society of Endocrinology and Metabolism of Turkey; SUI, stress urinary incontinence; T2D, type 2 diabetes; TRUS, transrectal ultrasound; TURP, transurethral resection of the prostate; UTI, urinary tract infection; WC, waist circumference; WHF, World Heart Federation; WHO, World Health Organization.
Outcomes measured and summary of MetS and LUTS association.

AP, Antero-posterior; AUA-SI, American Urological Association Symptoms Index; BMI, body mass index; BMR, basal metabolic rate; BP, blood pressure; BPE, benign prostatic enlargement; BPH, benign prostatic hyperplasia; BPO, benign prostatic obstruction; BPS, bladder pain syndrome; CI, confidence interval; CRP, C-reactive protein; CVS, cardiovascular system; DBP, diastolic blood pressure; DM, diabetes mellitus; DO, detrusor overactivity; ED, erectile dysfunction; FBG, fasting blood glucose; FLI, fatty liver index; HbA1c, hemoglobin A1C; HC, hip circumference; HDL-C, high-density lipoprotein cholesterol; HO, heme oxygenase; HoLEP, Holmium laser enucleation of the prostate; HOMA-IR, Homeostatic Model Assessment - Insulin Resistance; HT, hypertension; IIEF, International Index of Erectile Function; IIEF-EF, International Index of Erectile Dysfunction – Erectile Dysfunction; IIEF-5, Internal Index of Erectile Function–5; IL-8, Interleukin 8; IPP, Inflatable Penile prosthesis; IPSS, International Prostate Symptom Score; IPSS-T, International Prostate Symptom Score Total; IR, insulin resistance; IS, inflammatory score; KODAMA, knowledge discovery by accuracy maximization; LDL-C, low-density lipoprotein cholesterol; LUTS, lower urinary tract symptoms; MetS, metabolic syndrome; MPV, Mean Platelet Volume; NAFLD, non-alcoholic fatty liver disease; NC, neck circumference; NGF, nerve growth factor; OA, osteoarthritis; OAB, overactive bladder; OAB-Q, overactive bladder-questionnaire; OABSS, overactive bladder symptom score; OP, Open Prostatectomy; OR, odds ratio; OSA, obstructive sleep apnea; PAM, partition around medoids; PRI, prostatic resistive index; PSA, prostate-specific antigen; PV, prostate volume; PVR, post-void residual volume; Qmax, peak urinary flow; QOL, quality of life; RA, Rheumatoid Arthritis; RI, Resistive Index; SBP, systolic blood pressure; SHBG, Sex Hormone Binding Globulin; SUI, stress urinary incontinence; T2D, type 2 diabetes; T2DM, type 2 diabetes mellitus; TG, triglycerides; TPV, total prostate volume; TRUS, transrectal ultrasound; TT, total testosterone; TURP, transurethral resection of the prostate; TZV, transition zone volume; UI, Urinary Incontinence; UUI, urinary urgency incontinence; WC, waist circumference; WHR, waist-hip ratio.
Mixed-effects model results.
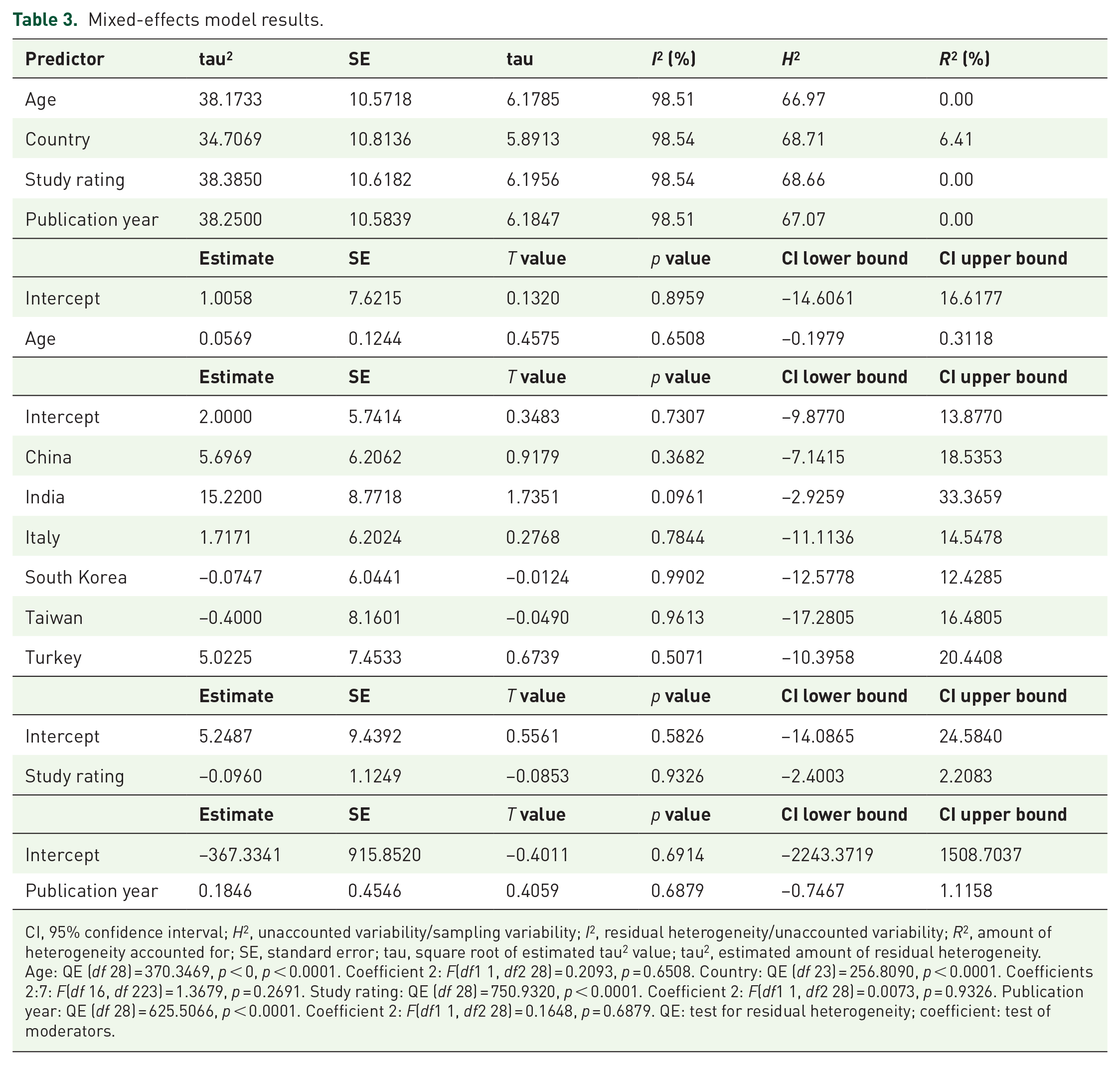
CI, 95% confidence interval; H2, unaccounted variability/sampling variability; I2, residual heterogeneity/unaccounted variability; R2, amount of heterogeneity accounted for; SE, standard error; tau, square root of estimated tau2 value; tau2, estimated amount of residual heterogeneity.
Age: QE (df 28) = 370.3469, p < 0, p < 0.0001. Coefficient 2: F(df1 1, df2 28) = 0.2093, p = 0.6508. Country: QE (df 23) = 256.8090, p < 0.0001. Coefficients 2:7: F(df 16, df 223) = 1.3679, p = 0.2691. Study rating: QE (df 28) = 750.9320, p < 0.0001. Coefficient 2: F(df1 1, df2 28) = 0.0073, p = 0.9326. Publication year: QE (df 28) = 625.5066, p < 0.0001. Coefficient 2: F(df1 1, df2 28) = 0.1648, p = 0.6879. QE: test for residual heterogeneity; coefficient: test of moderators.
QUIPS risk of bias assessment table for each study included in meta-analysis (k = 30).

QUIPS, Quality in Prognosis Studies.
Risk of bias for following components: study participation, study attrition, prognostic factor measurement, outcome measurement, study confounding, statistical analysis reporting, and overall risk of bias.
Summary of qualitative data
A total of 70 studies were included in qualitative synthesis. From these, 60 studies focused on MetS and LUTS: 44 reported positive correlations, 5 reported negative correlations, 11 reported no association, and 10 studies focused on MetS and TPV (Table 2). MetS positively correlated with moderate LUTS (OR = 1.56, 95% CI = 1.35–1.80; p < 0.001), severe LUTS (OR = 2.35, 95% CI = 1.82–3.03; p < 0.001), 66 OAB (OR = 3.2, 95% CI = 1.6–5.8, p = 0.01), 44 and nocturia severity (OR = 2.509, 95% CI = 1.571–4.007, p = 0.001) 34 at multivariate analysis. Demir et al. 10 reported positive correlations between MetS and LUTS (OR = 2.4, 95% CI = 1.24–4.59, p = 0.009); however, significance was lost at multiple logistic regression analysis. Baykam et al. 25 found no association between LUTS and BMI (kg/m2); only FBG was significant at multivariate analysis (β = 0.001, t = 3.491, p = 0.001). Gao et al. 13 found that MetS was not associated with the severity of LUTS (multivariate: OR = 0.97; 95% CI = 0.67–1.39).
Summary of meta-analysis
Initially, data from 70 studies was extracted and a meta-analysis on MetS and LUTS, which included 33 studies, was conducted; this generated 16 forest plots. the following outcomes versus MetS were evaluated: International Prostate Symptom Score Total (IPSS-T), IPSS voiding, IPSS storage, International Prostate Symptom Score Quality of Life (IPSS-QOL), TPV (ml), prostate-specific antigen (PSA; ng/ml), uroflowmetry Qmax (ml/s); and post-void residual volume (PVR; ml). Furthermore, forest plots for IPSS severity and each MetS component were generated; results were not significant; however, heterogeneity was relatively low in some plots. Given that TPV proved significant, we explored this further and systematically searched for studies on TPV and MetS (10 additional studies were identified). We generated another forest plot for TPV and MetS (total of 30 studies), which proved highly significant, albeit heterogeneity was high: I2 96.3% [95.4%; 96.9%]. Results are presented in Figure 2. Due to the high heterogeneity, a meta-regression analysis was performed to test the impact of covariates on heterogeneity. Meta-regression analysis was performed for predictors, age, country, study rating, and publication year; results were not significant (p > 0.05); therefore, predictors had no effect on heterogeneity (Figure 3; Table 3). An Egger’s test of the intercept was performed to test for publication bias; the test revealed a symmetric inverted funnel shape indicating a ‘well-behaved’ data set, in which publication bias is unlikely (intercept 1.073, 95% CI = –1.71 to 3.86, t = 0.754, p = 0.4570147; Figure 4). A risk of bias assessment was also performed, as shown in Figure 5 and Table 4, with an overall high risk of bias in most studies.
Discussion
Associations between LUTS and MetS have long since been contentious with clinical mechanisms and remain poorly understood. This meta-analysis sought to review all current published data in order to highlight any significant findings to date. Our meta-analysis (k = 30, n = 22,206) on TPV and MetS indicated significant results confirmed a significant association (MD = 4.4450, 95% CI = 2.0177–6.8723, t = 3.75; p = 0.0008). However, heterogeneity was high (tau2 = 37.0851 [18.9614; 71.7320], I2 = 96.3% [95.4%; 96.9%], H = 5.17 [4.67; 5.72]). Meta-regression produced non-significant results suggesting that predictors (age, country, study rating, publication year) had no effect on heterogeneity. Our study found no association between MetS and IPSS or its subgroups, PSA, Qmax, and PVR. Several studies have demonstrated that MetS causes inflammation and prostatic hyperplasia in men with BPH/LUTS. The results of our meta-analysis are consistent with other literature. Zou et al. 88 conducted a meta-analysis on 16 studies (BPH patients, n = 1895) on MetS and BPH in Chinese patients; TPV (MD = 10.15 ml; 95% CI = 7.37–12.93) and annual prostate growth rate (MD = 0.49 ml/year; 95% CI = 0.24–0.73) were significantly higher in BPH patients with MetS compared with patients without MetS. A meta-analysis by Gacci et al. 89 reported similar findings; TPV was significantly higher in BPH patients with MetS (+1.8 ml, 95% CI = 0.74–2.87, p < 0.001). In addition, no association was found between MetS and IPSS. 89 Wu et al. 90 also reported a significant association between MetS and TPV (OR = 2.34, 95% CI = 1.25–3.42) after performing a meta-analysis on six comparative studies (n = 61,826). Again, similar to our study, Wu et al. found no significant association between MetS and IPSS or PVR. 90 Wang et al. 91 (k = 8, n = 3093) reported that BPH patients with MetS had significantly higher prostate growth rates (MD = 0.67 ml/year, p < 0.001) and prostate volumes (MD = 6.8 ml, p = 0.010). No significant association between MetS and IPSS, and Qmax was found; however, there was an almost significant association with PSA (MD = 0.24 ng/ml, p = 0.056). 91 Li et al. 92 also significantly associated MetS with higher annual prostate growth rate and prostate volume; no association was found between MetS and IPSS/IPSS subgroups. In contrast to our study, Li et al. 92 significantly associated MetS with reduced Qmax (MD = –0.48, p = 0.001) and increased PVR (MD = 8.28, p < 0.001). Russo et al. 93 demonstrated that a significant association between MetS and prostate volume (MD = 2.18, p = 0.03) was found; no association was reported with IPSS. Differences in results may be due to the number and type of studies included in meta-analysis. Our meta-analysis included retrospective, cross-sectional studies and randomized controlled trials (RCTS; k = 30, n = 22206); not all studies used transrectal ultrasonography (TRUS) to measure TPV. Wu et al. 90 included retrospective studies and one prospective study (k = 6, n = 61,826); studies used TRUS; one study used suprapubic ultrasound. Wang et al. 91 included cohort or case-control studies (k = 8, 3093), all of which used ultrasound or TRUS; heterogeneity (I2) was also high (90.1%). Li et al. 92 included prospective and retrospective studies (k = 21, n = 15,317); 17 studies used TRUS to measure TPV. Forest plot results indicated a significantly lower heterogeneity of 49%, while our heterogeneity was 96%. 92 Russo et al. 93 (k = 19, n = 18,476) included six studies in the forest plot for prostate volume and heterogeneity was 85%; BPH definitions varied, and studies used TRUS and/or digital rectal examination (DRE) or IPSS alone.
Studies included in our meta-analysis used the same laboratory parameters and equipment for blood and urine analysis. Prostate volume (PV) was used as a reliable measurement of LUTS, and TRUS was considered more accurate than DRE. 94 Confounding factors were identified and adjusted for age, sex, smoking, alcohol consumption, sexual activity, UTIs or infections, constipation, exercise, drug intake, race, and menopause. Confounders were adjusted for using logistical regression analysis,10,63,66,68 multivariate analysis,24,25,34,46,51,52,77,82 and sensitivity analysis. 22 Restrictions in design were also performed for age and sex; patients were also stratified according to age, 22 menopause, 57 or smoking status. Akin et al. 22 used receiver operating characteristics (ROC) curve and calculated area under the curve (AUC) for OAB and WC (AUC = 0.72 cm 2 , 95% CI = 0.65–0.79, p < 0.001); this produced highly sensitive and specific cutoff values to determine OAB presence (WC = 98.5 cm). MetS criteria often included gender-specific and race-specific BMI and WC cutoffs for obesity. The exclusion criteria included patients with neurological disorders, depression, antidepressant use, anticholinergic medication use, diuretics, bladder or prostate cancer, UTI, stress urinary incontinence (SUI), and urinary symptoms since childhood.10,22,63,66,68
The strengths of our study include a clear objective and inclusion/exclusion criteria, not limited by sample size, follow-up period, length of intervention, or setting. We performed an extensive search of MEDLINE, SCOPUS, CENTRAL, and ClinicalTrials.gov; reference lists of selected articles and other literature sources were also searched to ensure a comprehensive search of sources. Each study was screened by two independent reviewers; conflicts were resolved by a third reviewer. Data extraction was reviewed by a second reviewer. We have included a PRISMA flowchart with reasons for exclusion of studies; the list of excluded studies (and conflicts) is available on Covidence. We included a table of eligible studies, detailed summaries, and characteristics. We performed a quality assessment (NOS) for each study included in our study (Table 1). Our current meta-analysis on TPV and MetS (k = 30, n = 22,206) indicated significant results, albeit heterogeneity was relatively high (Figure 3). Furthermore, a robust method with Sidik–Jonkman estimation and Hartung–Knapp adjustment was used to avoid type I error (false positives) in obtained results and to control for possible uncertainty due to heterogeneity. In addition, a meta-regression analysis was conducted to address the resultant high heterogeneity; there was no significance in predictors being associated with effect sizes (Figure 4(a)–(c); Tables 3 and 4). Furthermore, an Egger’s test of the intercept indicated no funnel plot asymmetry (Figure 4(d)); publication bias was not present. We performed a risk of bias assessment using the QUIPS tool and generated a graph (Figure 5).
Most previous studies did not record and adjust for all confounders. Not all studies excluded covariates, for example, neuropathy.44,60 In diabetic patients, hyperglycemia can result in small nerve fiber damage, known as neuropathy. This disorder can lead to an array of urological conditions, including urgency, incontinence, incomplete emptying, UTIs, and ED. Diabetes can also cause uropathy, which is when there is an obstruction in the urinary tract; this results in bladder disorders, recurrent UTIs, and sexual dysfunction. 95 Oxidative damage can also cause a loss of bladder sensation. 96 Patients with neuropathy would be more likely to report worse LUTS symptoms and quality-of-life scores. In women, diabetic neuropathy was significantly associated with LUTS. 97 In men, prostatic growth is stimulated by elevated activity of the sympathetic nerve, which is caused by elevated insulin levels. 98 Studies did not always collect data on comorbidities such as cardiovascular disease or T2D. 35 Patients with diabetes have been shown to have higher incidences of DO and patients also tend to be older, which is another factor that increases the likelihood of developing LUTS.99,100 In addition, the following confounding factors could also lead to a variation in results. At binary logistic regression, OAB significantly correlated (p < 0.001) with duration of menopausal >5 years (OR = 25.7, 95% CI = 5.82 = 113.72), parity more than twice (OR = 27.94, 95% CI = 8.25–94.6), and previous gynecological surgery (OR = 33.04, 95% CI = 8.78–124.38). 101 Moderate-to-severe LUTS incidence was increased twofold in men aged 70 to 79 years (OR = 2.11, 95% CI = 1.32–3.38) compared with other age groups. 102 OAB was linearly associated with asthma (p = 0.001), bladder or prostate cancer (p = 0.001), and neurological conditions (stroke, Parkinson’s disease, multiple sclerosis; p < 0.001). 103 Major adverse cardiac events (MACE), such as acute myocardial infarction, were positively associated with moderate–severe LUTS (OR = 2.38, 95% CI = 2.56–3.07, p < 0.001). 104 Alcohol consumption > 72 g/day caused close to a threefold increased risk of moderate–severe LUTS (OR = 2.96, 95% CI = 1.61–5.44). History of STIs was also a risk factor (OR = 1.50, 95% CI = 1.08–2.07). Vigorous physical activity negatively correlated with incidence of moderate–severe LUTS (OR = 0.61, 95% CI = 0.44–0.85). 102 Zhu et al. 105 negatively correlated OAB with employment status (OR = 0.64, 95% CI = 0.46–0.90). However, a meta-analysis by Zhu et al. 105 also found no significant association between OAB and the following: menopause, sex, vaginal delivery, educational level, parity, race, marital status, smoking, and alcohol consumption.
Moreover, multiple studies were cross-sectional, which cannot account for temporal relationships between MetS and LUTS. Retrospective studies rely on data previously collected; assessment of MetS and LUTS could not be controlled (Table 1). Furthermore, nocturia is self-reported; data rely on patients accurately recording their symptoms. 35 IPSS also relies on self-reporting of symptoms, an assessment which, although validated, can be subjective; the LUTS group may have been able to recall and report their symptoms better compared with control subjects (memory bias). IPSS also has high variability; 106 BPH/LUTS symptoms are not constant. Most studies selected patients from a single institution, and samples were relatively small.
Selecting patients from a specialist urology clinic can result in more severe presentations of LUTS. This is clearly at variance compared with the general population prevalence of severe LUTS. This was likely due to a referral bias as patients included in this meta-analysis were referred to a specialist urology clinic from wider region; cases with milder symptoms were probably managed more locally (referral bias). Patients attending these clinics were older, which is a risk factor for LUTS and MetS. Aging increases the risk of developing obesity, T2D, hypertension, insulin resistance, and dyslipidemia. Participants were mostly men. In addition, asymptomatic control groups were not always included, and many studies did not include follow-up data. LUTS and MetS criteria were also highly heterogeneous; this made it difficult to compare studies. According to World Health Organization (WHO), American Heart Association (AHA); National Heart, Lung, and Blood Institute (NHLBI); and International Diabetes Federation (IDF), the WC cutoffs for MetS for Caucasian men and women are ⩾102 and ⩾88 cm, respectively. WHO and IDF have lower cutoffs for Asian men and women: ⩾90 and ⩾80 cm, respectively. The Japanese Obesity Society has an even lower cutoff for Asian men (⩾85 cm) and a slightly higher cutoff for Asian women (⩾90 cm). 1
Results rely on the population included in a study; the prevalence of MetS, obesity, and LUTS in a sample; and the smoking status of individuals. In RCTs, the effect of MetS components on LUTS is unclear because taking a random sample of men and women in the community does mean disorders of the uropoietic system will be present in the sample.13,42,46 Furthermore, all RCTs are hypothetically designed for sample following a power calculation with 95% CI (p = 0.05). Even if results are significant, there is a 5% chance they are due to chance. Even though PV is associated with LUTS, some studies did not collect data concerning PV.66,67,83 Most studies defined general obesity as BMI ⩾30 kg/m2, while some studies included overweight participants (BMI = 25–29 kg/m2). According to WHO (1999), BMI ⩾25 kg/m2 indicates overweight and BMI ⩾30 kg/m2 indicates obesity. 107 This classification was intended for international use; however, the classification was revised given that high rates of T2D and cardiovascular risk factors were reported in Asian populations with an average BMI below 25 kg/m2, below the WHO cutoff for ‘overweight’. 108 BMI does not take into account muscle mass, and percentage body fat and BMI can differ according to age, sex, and ethnicity. In addition to using IPSS to measure symptoms of LUTS and BPH, TRUS should be used to accurately measure TPV. MetS should be carefully managed when treating larger TPVs in individuals with LUTS and BPH. More studies are required to determine the role of MetS in prostate inflammation and enlargement. Improved study designs and homogenized samples led by hypothesis-driven ideas are required. Future research should focus on the development of multicenter, multinational controlled trials with accurate definitions of MetS and LUTS. Recruiting from specialist centers and clinics is a better option than RCTs as it ensures that the sample contains individuals with LUTS and MetS. Specialists will also diagnose LUTS and MetS more accurately. Specialist urologists should administer questionnaires to reduce error. In addition, all MetS components should be investigated, and asymptomatic groups should be included. A more patient-specific method of measuring LUTS severity is also needed. Combining measurements of LUTS, QOL, and overall health status may increase specificity and sensitivity. 109 TRUS should be used to measure TPV and LUTS. CIs above 95% would be ideal. More research into other uropoietic disorders especially on a genetic and molecular level is needed. More data on the inflammatory markers involved are essential in confirming the role of MetS on inflammatory uropoietic disorders.
Conclusion
The present meta-analysis indicated no significant association between MetS, or its components, and LUTS. This is likely due to significant heterogeneity of methods used to evaluate LUTS symptoms in the studies we included. Regarding TPV and MetS, a significant association was noted in our study and is consistent with other studies in this field. Obesity, large WC, hypertension, hyperinsulinemia, dyslipidemia, hypercholesterolemia, and hypertriglyceridemia have been associated with worse symptoms of uropoietic disorders at multivariate analysis. Interventions aimed at weight loss including behavioral modification, obesity pharmacotherapy, and obesity surgery are recommended and should be at the forefront of management of patients with MetS and disorders of the uropoietic system.
Footnotes
Acknowledgements
The authors are grateful to Francesco Rubino and Dudley Robinson for their support. They are also grateful to Alexander Miras for providing access to Covidence systematic review software. The data that support the findings of this manuscript are available by the corresponding author upon reasonable request.
Author contributions
A.O. involved in data curation, formal analysis, methodology, project administration, writing-original draft, and writing-review and editing. B.M.L. involved in data curation, formal analysis, methodology, and project administration. E.O. involved in data curation, formal analysis, investigation, methodology, project administration, resources, software, and validation. N.G. involved in data curation, methodology, project administration, and software. A.S.D.S. involved in data curation, methodology, project administration, and software. Z.M.Z. involved in data curation, methodology, project administration, software, and validation. A.D.M. helped in software and writing-review and editing. C.W.l.R. and R.P.V. contributed to resources, software, supervision, and writing-review and editing. L.C. contributed to resources, software, supervision, validation, and writing-review and editing. G.K.D. involved in conceptualization, formal analysis, funding acquisition, investigation, methodology, project administration, supervision, validation, writing-original draft, and writing-review and editing.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institute for Health Research (NIHR), South London Clinical Research Network (CRN) ‘Green shoots’ Investigator Award supported Dr Dimitriadis in the writing of this manuscript.
