Abstract
Context:
Neuregulin 4 (Nrg4) and neuregulin 1 (Nrg1) have been shown to play vital roles in several disorders of glucose metabolism. The pathophysiological role of Nrg4 and Nrg1 in gestational diabetes mellitus (GDM), however, remains poorly understood. We assessed the clinical relevance of the two cytokines in patients with GDM.
Methods:
The study recruited 36 GDM patients and 38 age-matched, gestational age (24–28 weeks of gestation)–matched, and BMI (during pregnancy)–matched controls in this study. Serum Nrg4 and Nrg1 were measured using ELISA. Inflammatory factors such as IL-6, IL-1β, leptin, TNF-α, and monocyte chemotactic protein 1 (MCP-1) were determined via Luminex technique.
Results:
Serum Nrg4 in GDM patients was significantly lower than that in the controls, while Nrg1 was significantly higher in the GDM group (p < 0.01). Inflammatory factors such as IL-6, leptin, and TNF-α were significantly increased in GDM patients, while MCP-1 and IL-1β were not significantly different between the two groups. In addition, serum Nrg4 was negatively correlated with fasting glucose (r = −0.438, p = 0.008), HOMA-IR (r = −0.364, p = 0.029), IL-6 (r = −0.384, p = 0.021), leptin (r = −0.393, p = 0.018), TNF-α (r = −0.346, p = 0.039), and MCP-1 (r = −0.342, p = 0.041), and positively correlated with high-density lipoprotein cholesterol (HDL-C) (r = −0.357, p = 0.033) in GDM group. Serum Nrg1 was positively correlated with BMI (r = 0.452, p = 0.006), fasting glucose (r = 0.424, p = 0.010), HOMA-IR (r = 0.369, p = 0.027), and triglyceride (r = 0.439, p = 0.007). The decrease of Nrg4 and the increase of Nrg1 were significantly related to the increased prevalence of GDM. Finally, ROC curve results indicated that Nrg1 combined with IL-6 and TNF-α might be an effective means for GDM screening.
Conclusions:
Lower circulating Nrg4 and higher circulating Nrg1 serve risk factors of GDM. Nrg1 combined with IL-6 and TNF-α might be a potential tool for GDM screening.
Introduction
Gestational diabetes mellitus (GDM) is one of the most common complications during pregnancy, affecting about 16% of pregnant women. 1 According to the latest report, about 18.4 million newborns are affected by GDM. 2 The increasing incidence of GDM has caused serious harm to both mothers and fetuses. Patients with GDM have a significantly increased risk of macrosomia and shoulder dystocia. 3 Later in life, women with GDM had a significantly increased risk of type 2 diabetes mellitus (T2DM), coronary artery disease, and stroke.4,5 Similarly, their offspring are more likely to develop T2DM, obesity, or cardiovascular disease and high blood pressure in early adulthood. 6 Given the harmfulness of GDM, the mechanism of GDM has aroused great interest. Chronic insulin resistance (IR), pancreatic β-cell dysfunction, and chronic subclinical inflammation are the main causes of GDM.7,8 The pathogenesis of GDM, however, has not been fully explained, and there is no reliable early diagnosis method and effective prevention strategy.
Neuregulins (Nrg1–4) are ligands for the receptor tyrosine kinases of the ErbB/HER family, which is involved in diverse biological processes. 9 Recent literatures strongly indicate Nrgs as endocrine regulators of metabolic physiology. Nrg4, an ErbB4-specific ligand, is mainly produced in white and brown adipose tissue and closely related to energy balance and glycolipid metabolism. 10 Wang et al. 10 found that Nrg4 can improve liver lipid metabolism and increase insulin sensitivity. Transfer of Nrg4 gene into mice can prevent weight gain, inhibit diet-induced chronic inflammation, and significantly improve IR. 11 In clinical study, Nrg4 was reported to be involved in several metabolic disorders, such as metabolic syndrome, 12 nonalcoholic fatty liver diseases (NAFLDs), 13 polycystic ovary syndrome (PCOS), 14 and T2DM.15,16 Yan et al. 16 found that Nrg4 was negatively correlated with inflammation markers in patients withT2DM. The role of Nrg4 in GDM, however, is less studied and controversial. Kurek Eken et al. 17 found that serum Nrg4 in GDM patients was significantly higher than that in normal pregnant women. In another small cross-sectional study, Kralisch et al. 18 came to the opposite conclusion. Moreover, there is no information regarding the relationship between Nrg4 and inflammatory factors in GDM.
Nrg1 plays fundamental roles in the growth and development of skeletal muscle, cardiac, and nerve tissues.19,20 Nrg1/ErbB pathway is considered a potential target for the treatment of neuromuscular and heart disorders. Recent studies suggest that Nrg1 plays roles in the regulation of glucose metabolism. The expression of glucose transporter 4 (GLUT4) is upregulated after treatment with Nrg1 in L6E9 muscle cells for 48 h, 21 suggesting that chronic Nrg1 treatment could regulate muscle glucose metabolism. The effect of Nrg1 on glucose uptake is additive to that of insulin and is mediated by PI3K-PDK1-PKCζ pathway. 22 Cote et al. 23 found that Nrg1 can induce glucose uptake in adult rat ventricular myocytes through Erk1/2 and Akt (Ser473) phosphorylation. Most recently, Heim et al. 24 clarified that recombinant human Nrg1 can induce glucose uptake by activating the PI3Kα-Akt-AS160 pathway and GLUT4 translocation in neonatal but not in adult cardiomyocytes. In vivo, both acute and chronic Nrg1 treatment could improve glucose tolerance in db/db mice.25,26 Arpaci et al. showed that circulating Nrg1 significantly decreased in patients with PCOS and was inversely correlated with Homeostatic Model Assessment of Insulin Resistance (HOMA-IR) and high-sensitivity C-reactive protein (hs-CRP). 27 Information regarding the role of Nrg1 in GDM, however, is rare.
In this study, we aimed to assess the clinical relevance of Nrg4 and Nrg1 in patients with GDM. We also intended to investigate the association between the two cytokines and inflammation.
Material and methods
Study participants and study design
This is a cross-sectional study. From July 2018 to February 2019, about 500 pregnant women with 24–28 weeks gestation participated in oral glucose tolerance test (OGTT) in the obstetric clinic of Huai’an First People’s Hospital. We screened out 45 GDM cases. Nine cases, however, that did not meet our inclusion criteria were excluded from our study. Eventually, we chose 36 cases as GDM group. Accordingly, we chose 38 age- and gestational-age-matched controls who agreed to participate in the study. All patients were diagnosed according to the GDM diagnostic criteria of American Diabetes Association (ADA) in 2012. During 2-h 75-g OGTT at 24–28 weeks of gestation, the blood glucose higher than any of the following items could be diagnosed as GDM: fasting glucose ⩾ 5.1 mmol/l, blood glucose ⩾ 10.0 mmol/l at 1 h, and blood glucose ⩾ 8.5 mmol/l at 2 h. All GDM patients did not receive any antidiabetic treatment, including diet control, exercise, and drug therapy. The exclusion criteria were as follows: age <18 or >40 years; smoking or drinking; abortion history; multiple pregnancy; immunosuppressive therapy; pregnancy-induced hypertension, preeclampsia, or both; and acute and chronic systemic inflammation. According to the expert consensus on prevention and treatment of obesity in Chinese adults in 2011, the GDM and control group were separately divided into two subgroups: normal weight group [18.5 ⩽ BMI (body mass index) < 24 kg/m2] and overweight or obese group (BMI ⩾ 24 kg/m2).
Anthropometric data collection
At 24–28 weeks of gestation, weight, height, and blood pressure were measured by well-trained nurses. The BMI is calculated using the following formula: BMI = weight (kg) / height squared (m 2 ).
Biochemical measurements
Fasting blood samples were taken and separated through centrifuge. Serum lipids were measured using a Hitachi 7600 analyzer (Hitachi, Ltd., Tokyo, Japan). Insulin levels were determined via an automated Immunoassay Analyzer (AIA-2000ST; TOSOH, toyama, Japan). High-performance liquid chromatography method was used for HbA1c determination (HLC-723G8; TOSOH). The rest serum samples were stored −80°C until Nrg1, Nrg4, and inflammatory factors measurement. The HOMA-IR is calculated using the following formula: HOMA-IR = [fasting glucose (mmol/l) × fasting insulin (mIU/l)] / 22.5. 28
Measurements of Nrg4 and Nrg1
Serum Nrg4 and Nrg1 were measured using ELISA Kit (Catalog No. CSB-EL016080HU, CSB-E17153h; CUSABIO, Wuhan, China). The sensitivity of Nrg4 assay is 31.25 pg/ml. The sensitivity of Nrg1 assay is 1.95 ng/ml. The intra-assay coefficient of variation (CV) was less than 8%, and the inter-assay CV was less than 10%.
Measurements of inflammatory factors and adipokines
Tumor necrosis factor α (TNF-α), interleukin 6 (IL-6), monocyte chemoattractant protein 1 (MCP-1), leptin, and interleukin 1β (IL-1β) were determined using Luminex technique. MILLIPLEX® Human Adipokine Magnetic Bead Panel (Catalog No. HADK2MAG-61 K-06, Millipore, Massachusetts, USA) was used to detect TNF-α, IL-6, MCP-1, leptin, and IL-1β.
Statistical analysis
GraphPad Prism 5 Software (GraphPad Software, Inc., California, USA) and SPSS version 23 (IBM, New York, USA) were performed for all statistical analyses. Q–Q plot and Kolmogorov–Smirnov test were used to test data distribution. Data with skewed distribution were shown as median [interquartile range (IQR), 25th–75th], and normally distributed data were presented with mean ± SD. Chi-square (χ2) test was performed to compare categorical data. Independent-samples t test and Mann–Whitney U test were used to compare continues variables between two groups. Spearman correlation analysis was conducted to analyze bivariate correlation. Next, all study participants were stratified into trisection according to Nrg4 or Nrg1 tertiles. Trend χ2 test was used to assess the correlation between Nrg4 or Nrg1 levels and the prevalence of GDM. Multivariate logistic regression models were performed to calculate odds ratios (ORs) and 95% confidence intervals (CIs) of GDM in Nrg4 or Nrg1 tertiles. Finally, several receiver operating characteristic (ROC) curves were drawn for GDM diagnosis. The p value of less than 0.05 (two side) was considered to be statistically significant.
Results
General characteristics of the study participants
As shown in Table 1, there is no significant difference between GDM group and normal group in general clinical biochemical data such as age, gestational age, BMI, blood pressure, lipid profile, alanine transaminase (ALT), aspartate aminotransferase (AST), creatinine, and uric acid (p > 0.05). The fasting glucose, 1-h blood glucose, and 2-h blood glucose during OGTT and HOMA-IR of GDM group are significantly higher than those of control group (p < 0.01).
General clinical and laboratory parameter in participants with and without GDM.

ALT, alanine transaminase; AST, aspartate aminotransferase; BMI, body mass index; DBP, diastolic blood pressure; GDM, gestational diabetes mellitus; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, Homeostatic Model Assessment of Insulin Resistance; LDL-C, low-density lipoprotein cholesterol; OGTT, oral glucose tolerance test; SBP, systolic blood pressure; TC, total cholesterol; TG, triglyceride.
Mann–Whitney U test was performed.
Independent-sample t test was performed.
Comparison of inflammatory factors measured by Luminex technique
We calculated the concentration of inflammatory factors in each sample according to the standard curve obtained by Luminex technique. As shown in Figure 1, IL-6, leptin, and TNF-α in GDM group are significantly higher than those in normal group (p < 0.05 or p < 0.01). There is no significant difference in MCP-1 and IL-1 β between the two groups (p > 0.05).

Comparison of serum inflammatory factors between GDM (n = 36) and normal controls (n = 38). (a) Comparison of IL-6; (b) Comparison of Leptin; (c) Comparison of MCP-1; (d) Comparison of TNF-α; (e) Comparison of IL-1. Independent-sample t test was performed for leptin comparison. Mann–Whitney U test was performed for the comparison of IL-6, MCP-1, TNF-α, and IL-1.
Comparison of serum Nrg4 and Nrg1
As shown in Figure 2(a), serum Nrg4 of GDM group is significantly lower than that of controls (p < 0.001). As shown in Figure 2(b), serum Nrg1 of GDM group is significantly higher than that of controls (p = 0.004). Both in GDM and control group, there is no significant Nrg4 difference between the subgroups according to BMI (Figure 2(c)). As presented in Figure 2(d), in GDM group, serum Nrg1 in BMI ⩾ 24 kg/m2 subgroup is higher than that in 18.5 ⩽ BMI < 24 kg/m2 subgroup (p < 0.05).

Comparison of serum Nrg4 and Nrg1 between GDM (n = 36) and normal controls (n = 38). (a) and (b) show the comparison of serum Nrg4 or Nrg1 in GDM and normal controls. (c) and (d) show the comparison of serum Nrg4 or Nrg1 according to BMI. Mann–Whitney U test was performed for all the comparison.
Correlations of serum Nrg4, Nrg1, and clinical metabolic parameters
As shown in Table 2, in GDM group, Nrg4 is negatively correlated with fasting glucose, HOMA-IR, IL-6, leptin, MCP-1, and TNF-α, and positively correlated with high-density lipoprotein cholesterol (HDL-C), but not statistically correlated with other variables. In the control group, the correlation between Nrg4 and TC is statistically significant. As shown in Table 3, serum Nrg1 is only positively related to BMI, fasting glucose, HOMA-IR, and triglyceride (TG) in GDM group, but not with other variables. In the control group, only the correlation between serum Nrg1 and BMI was statistically significant.
Bivariate correlation between serum Nrg4 levels and other variables.
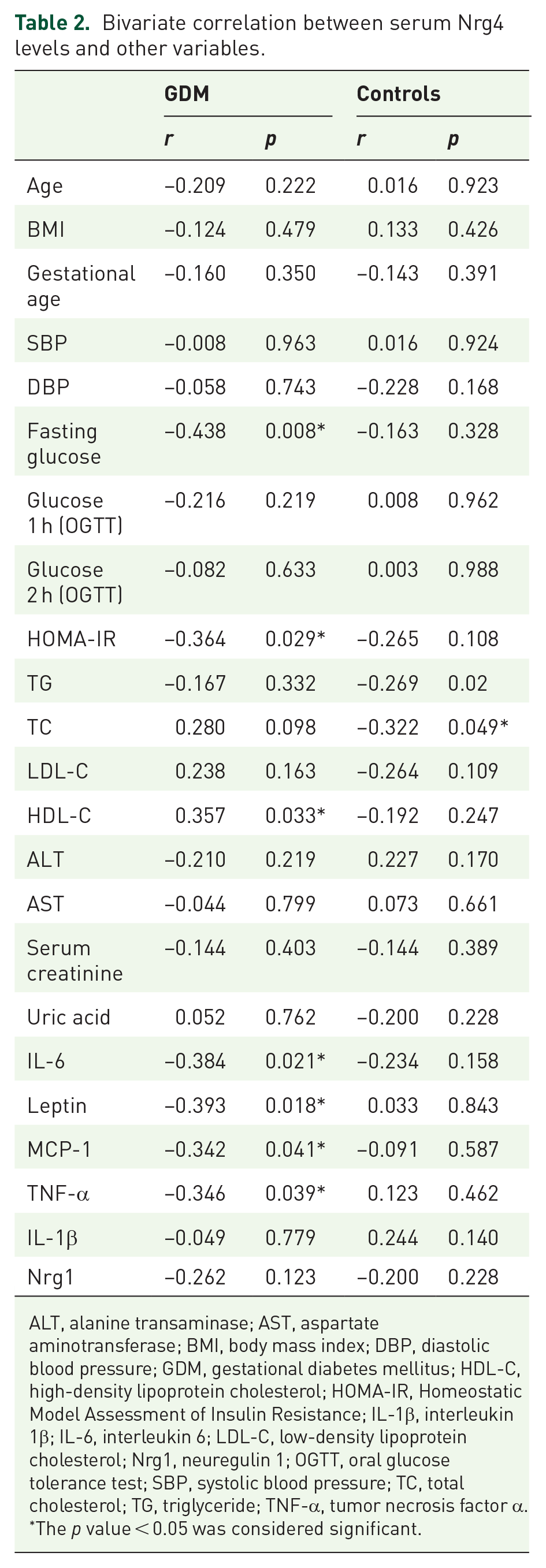
ALT, alanine transaminase; AST, aspartate aminotransferase; BMI, body mass index; DBP, diastolic blood pressure; GDM, gestational diabetes mellitus; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, Homeostatic Model Assessment of Insulin Resistance; IL-1β, interleukin 1β; IL-6, interleukin 6; LDL-C, low-density lipoprotein cholesterol; Nrg1, neuregulin 1; OGTT, oral glucose tolerance test; SBP, systolic blood pressure; TC, total cholesterol; TG, triglyceride; TNF-α, tumor necrosis factor α.
The p value < 0.05 was considered significant.
Bivariate correlation between serum Nrg1 levels and other variables.
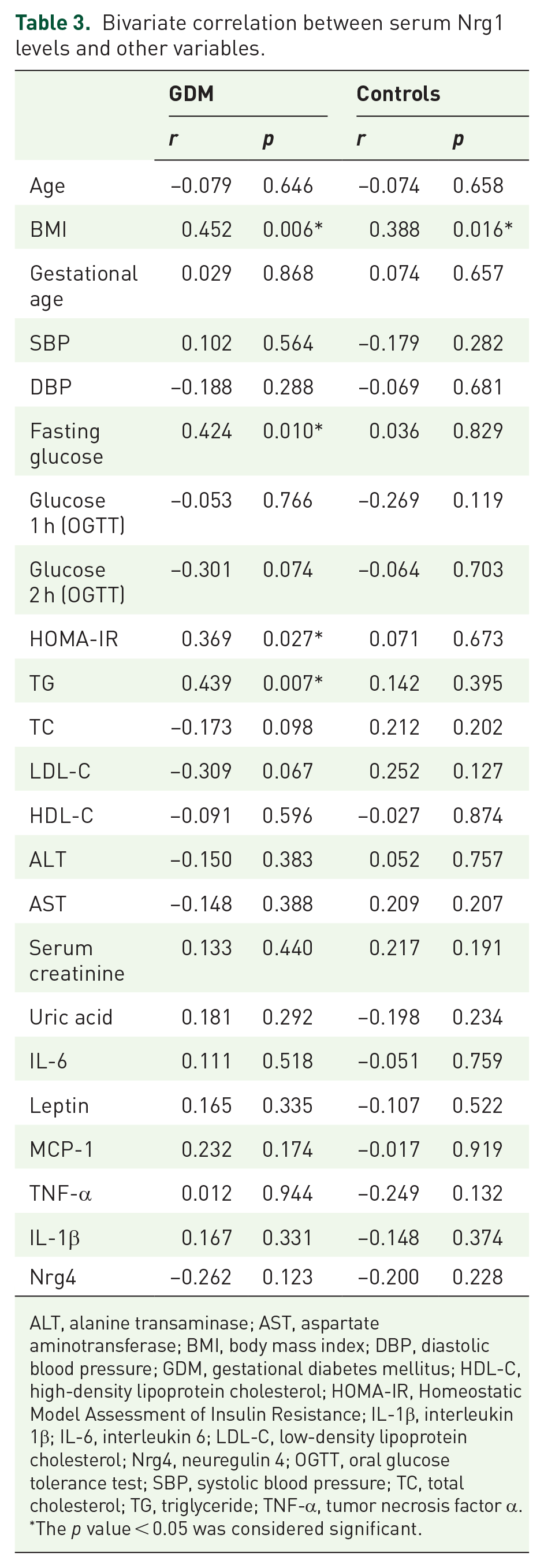
ALT, alanine transaminase; AST, aspartate aminotransferase; BMI, body mass index; DBP, diastolic blood pressure; GDM, gestational diabetes mellitus; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, Homeostatic Model Assessment of Insulin Resistance; IL-1β, interleukin 1β; IL-6, interleukin 6; LDL-C, low-density lipoprotein cholesterol; Nrg4, neuregulin 4; OGTT, oral glucose tolerance test; SBP, systolic blood pressure; TC, total cholesterol; TG, triglyceride; TNF-α, tumor necrosis factor α.
The p value < 0.05 was considered significant.
Nrg4, Nrg1, and the risk of GDM
All study participants were divided into trisection according to Nrg4 and Nrg1 concentrations (Nrg4, T1: <158.33 pg/ml, T2: 158.33−237.50 pg/ml, T3 ⩾ 237.50 pg/ml; Nrg1, T1: <8.06 ng/ml, T2: 8.06−13.52 ng/ml, T3 ⩾ 13.52 ng/ml). As shown in Figure 3(a), following the increasing Nrg4 levels, the prevalence of GDM gradually decreases (p for trend < 0.01). Figure 3(b) suggests that the prevalence of GDM gradually rises as Nrg1 increases (p for trend < 0.01).

The prevalence of GDM in different Nrg4 or Nrg1 concentration ranges. (a) With the increases of Nrg4, the prevalence of GDM decreases gradually. (b) With the increases of Nrg1, the risk of GDM increases gradually. Trend χ2 test was used to assess the correlation between Nrg4 or Nrg1 levels and the prevalence of GDM.
In order to further assess the relationship between Nrg4, Nrg1, and the risk of GDM, we established multiple logistic regression models based on its concentration to calculate OR and 95% CI of GDM. As shown in Table 4, after adjustment for age, BMI, blood pressure, gestational age, liver function, renal function index, lipid profile, HOMA-IR, and inflammatory factors, the highest Nrg4 concentration has the lowest OR of GDM. As shown in Table 5, even after adjusting for age, BMI, systolic blood pressure (SBP), diastolic blood pressure (DBP), gestational age, liver function, renal function, lipid profile, and inflammatory factors, T3 of Nrg1 levels has a higher risk of GDM than T1.
ORs and 95% CIs for GDM risk according to serum Nrg4.

ALT, alanine transaminase; AST, aspartate aminotransferase; BMI, body mass index; CI, confidence interval; DBP, diastolic blood pressure; GDM, gestational diabetes mellitus; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, Homeostatic Model Assessment of Insulin Resistance; IL-1β, interleukin 1β; IL-6, interleukin 6; LDL-C, low-density lipoprotein cholesterol; OGTT, oral glucose tolerance test; OR, odds ratio; SBP, systolic blood pressure; TC, total cholesterol; TG, triglyceride; TNF-α, tumor necrosis factor α.
Model 1: crude; model 2: adjusted for age, BMI, SBP, DBP, and gestational age; model 3: adjusted for model 2 + ALT, AST, lipid profile, serum creatinine, and uric acid; model 4: adjusted for model 3 + HOMA-IR, MCP-1, IL-1β, TNF-α. IL-6 and leptin were excluded because of multicollinearity.
ORs and 95% CIs for GDM risk according to serum Nrg1.

ALT, alanine transaminase; AST, aspartate aminotransferase; BMI, body mass index; CI: confidence interval; DBP, diastolic blood pressure; GDM, gestational diabetes mellitus; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, Homeostatic Model Assessment of Insulin Resistance; IL-1β, interleukin 1β; IL-6, interleukin 6; LDL-C, low-density lipoprotein cholesterol; OGTT, oral glucose tolerance test; OR, odds ratio; SBP, systolic blood pressure; TC, total cholesterol; TG, triglyceride; TNF-α, tumor necrosis factor α.
Model 1: crude; model 2: adjusted for age, BMI, SBP, DBP, and gestational age; model 3: adjusted for model 2 + ALT, AST, lipid profile, serum creatinine, and uric acid; model 4: adjusted for model 3 + MCP-1, IL-1β, TNF-α, IL-6, and leptin. HOMA-IR was excluded because of multicollinearity.
The diagnostic value of serum Nrg4, Nrg1, and their combination with inflammatory factors in GDM
To test the diagnostic value of Nrg4 and Nrg1 to GDM, we constructed several ROC curves. As presented in Figure 4(a), Nrg4 alone has higher diagnostic efficiency [area under the curve (AUC) area: 0.737]. To detect the diagnostic efficacy of Nrg4 combined with inflammatory factors in GDM, ROC curves were constructed with Nrg4 + IL-6, Nrg4 + leptin, Nrg4 + TNF-α, Nrg4 + IL-6 + leptin, and Nrg4 + TNF-α + leptin as variables. Data in Figure 4(b) indicate that Nrg4 combined with IL-6 and TNF-α has the greatest diagnostic ability for GDM (AUC area: 0.759) and is better than Nrg4 single diagnosis shown in Figure 4(a). Similarly, to test the diagnostic efficacy of Nrg1 combined with inflammatory factors in GDM, ROC curves were constructed with Nrg1 + IL-6, Nrg1 + leptin, Nrg1 + TNF-α, Nrg1 + IL-6 + TNF-α, Nrg1 + IL-6 + leptin, and Nrg1 + TNF-α + leptin as variables. As shown in Figure 4(c), when Nrg1 combined with IL-6 and TNF-α as variables, AUC area is 0.787, which has the highest efficiency in all diagnostic models.

The predictive value of serum Nrg4, Nrg1, and their combination with inflammatory factors in GDM. (a) Nrg4, Nrg1 alone, and their combination for GDM diagnosis. (b) Nrg4 combined with inflammatory factors for GDM diagnosis. (c) Nrg1 combined with inflammatory factors for GDM diagnosis.
Discussion
With the help of Luminex technique, this study investigated the expression of Nrg4 and Nrg1 in GDM and their relationship with inflammatory factors. Our results indicate that lower circulating Nrg4 and higher circulating Nrg1 serve risk factors of GDM. Nrg1 combined with IL-6 and TNF-α might be a potential tool for GDM screening.
The GDM is defined as different degrees of glucose tolerance abnormalities that occur or are first recognized during pregnancy. IR during pregnancy is one of the major causes of GDM. 7 Our data suggest that serum Nrg4 decreases in GDM and negatively correlates with fasting glucose and HOMA-IR, in line with the findings from Kralisch et al., 18 which suggests Nrg4 might be involved in the progress of GDM. Recent studies suggest that Nrg4 is involved in the process of energy balance and glucose metabolism. Wang et al. 10 found that Nrg4 can activate STAT5 signal, inhibit the activity of liver X receptor (LXR), reduce the expression of sterol regulatory element binding protein-1c (SREBP-1c), improve liver lipid metabolism, and increase whole-body insulin sensitivity. Chen et al. 29 found that Nrg4 overexpression increased energy consumption, accelerated glucose metabolism in mice, and increased the expression of protective adipokines in obese mice. Zeng et al. 30 reported that recombinant Nrg4 could increase GLUT4 transport to the cell membrane of adipocytes, thus increasing glucose uptake, but the effect was weakened after ErbB4 receptor silencing. In clinical research, Cai et al. 12 demonstrated that circulating Nrg4 in patients with metabolic syndrome was lower than that in healthy controls. Similar results were clarified in patients with other IR-related disorders, such as NAFLD, 13 PCOS, 14 and T2DM,15,16 suggesting the protective role of Nrg4. Nevertheless, information regarding circulating Nrg4 in GDM remains controversial. Our data are contrary to the results of another cross-sectional study. 17 Sample size, the choice of ELISA kit, and racial difference may account for the inconsistency of these findings.
This study observed that Nrg1 is significantly higher in the GDM group and shows positive correlation with fasting glucose, HOMA-IR, and TG, which suggest that Nrg1 may be involved in the progress of GDM. Several literatures in vitro and in vivo have established the positive role of Nrg1 in the regulation of glucose intake.21 –26 The function and expression of Nrg1, however, changed under pathological state. Nrg1-induced Akt phosphorylation can be attenuated by pretreatment with palmitate in cardiomyocytes, indicating that overnutrition or hyperlipidemia may lead to Nrg1 resistance. 31 We speculate the increase of Nrg1 in GDM is a compensatory mechanism for the increase of blood glucose. But this hypothesis needs more basic research to clarify.
Obesity is the most common risk factor for GDM and is related to the short-term and long-term adverse outcomes of pregnant women and their offspring.32,33 To analyze the relationship between Nrg4, Nrg1, and obesity, we stratified all women into two subgroups according to BMI. In accordance with our previous results in T2DM and the data from Kralisch et al. obtained in GDM,15,18 serum Nrg4 between study participants with BMI ⩾24 and 18.5 ⩽ BMI < 24 kg/m2 shows no significant discrepancy. Interestingly, several studies found a positive association between Nrg4 and obesity parameters in T2DM or GDM.17,34 The discrepancy might be attributed to several reasons. Nrg4 is not only expressed in adipose tissue, but also secreted by other organs into the blood, and body fat is not the sole decisive factor of Nrg4.34,35 The different clinical features of the study participants among different research can also affect the results. Therefore, the association between Nrg4 and BMI may be diverse. Differ to Nrg4, Nrg1 is higher in the subgroup of BMI ⩾ 24 kg/m2, especially in women with GDM. Former studies demonstrated that Nrg1 reduced weight gain and suppressed food intake when administered into mice.36,37 Similar to leptin resistance, we speculate that Nrg1 resistance occurs in obesity. But this hypothesis needs more work to prove.
Due to the chronic low-grade inflammation of adipose tissue, adipokines such as TNF-α, IL-6, and leptin are inappropriately secreted, which is closely related to the occurrence of obesity and serves a link between obesity and IR in GDM. 38 Our results demonstrate that serum IL-6, leptin, and TNF-α are significantly increased in GDM patients, while MCP-1 and IL-1β show no significant difference. In addition, Nrg4 is negatively correlated with IL-6, leptin, TNF-α, and MCP-1, suggesting the potential anti-inflammatory effect. Consistent with our findings, circulating Nrg4 negatively correlated with inflammatory factors in diabetic peripheral neuropathy patients. 39 In addition, recent work has shown that Nrg4 can inhibit the expression of IL-6, TNF-α, and advanced glycation end products, and also can improve renal function damage, tubulointerstitial fibrosis, and inflammation by downregulating TNF receptor 1. 40 Unlike Nrg4, although Nrg1 is implicated in the regulation of immune system response and inflammation, 41 serum Nrg1 showed no significant association with these inflammatory factors in GDM.
At present, there is no unified diagnostic standard for GDM in the world. In recent years, GDM screening has aroused great interest. The ROC is widely used for comparison of two or more different diagnostic tests. The AUC of each test can also be calculated and compared. In this study, several ROC curves were constructed, and it was found that Nrg1 combined with IL-6 and TNF-α was the most effective model in GDM screening. Unfortunately, cutoff values in combined diagnosis of ROC curves were not available. We can only observe the diagnostic ability of these models by AUC.
The strengths of this study are as follows: First, the age and BMI of the study participants were matched to eliminate the bias caused by grouping; second, using Luminex technique to detect multiple factors at the same time, the error caused by multistep operation can be reduced. There are also some weaknesses of our work. First, due to the low incidence of GDM in Huai’an city, the sample size is relatively small. The conclusions need to be confirmed in other ethnic and regional cohort studies; second, this study is limited to serology, and the specific mechanism needs to be further clarified in more basic studies. Third, Nrg1 has many subtypes, which presented different functions. The ELISA kit we used, however, did not indicate which specific subtype of Nrg1 be detected, which may affect our results. Fourth, due to the low compliance of GDM patients, data after delivery are not available.
Conclusions
In summary, this study assessed the clinical relevance of Nrg4 and Nrg1 in GDM and found that lower circulating Nrg4 and higher circulating Nrg1 serve risk factors of GDM. Nrg1 combined with IL-6 and TNF-α might be a potential tool for GDM screening.
Footnotes
Acknowledgements
We sincerely thank the volunteers of this study. LZ, BL, and WW contributed equally to this work.
Author contributions
LZ, BL, and WW contributed to conceptualization, formal analysis, methodology, supervision, writing-review, and editing. SM and SZ contributed to investigation, methodology, writing-review, and editing. XC contributed to formal analysis, methodology, writing-review, and editing. JZ contributed to investigation, methodology, and writing-original draft. CL contributed to conceptualization, funding acquisition, investigation, supervision, and writing-original draft.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Research and Development Fund of Kangda College of Nanjing Medical University (KD2018KYJJZD006).
Ethical approval and consent to participate
This work is in line with the principles of Helsinki Declaration and approved by the ethics committee of Xinghua People’s Hospital (Approval ID: 2016-0023). All participants were aware of the research purpose and signed an informed consent form.
Availability of data and materials
All data generated or analyzed during this study are included within this published article.
