Abstract
Aims:
To study the prevalence of microvascular complications and renal changes associated with cystic fibrosis-related diabetes (CFRD).
Methods:
This retrospective cohort study was conducted at the West Midlands Adult Cystic Fibrosis centre, United Kingdom. Data regarding age, sex, microalbuminuria, retinopathy neuropathy, and biochemical results were collected for all people with CFRD who had an annual review from 1 January 2018 to 31 December 2018 at the centre. Descriptive statistics were analysed using STATAv15.1.
Results:
A total of 189 patients were included, of which 56.6% were male and median age (interquartile range) was 33 (27–39) years; 79.4% (150/189) had their annual review in 2018. Those with a biochemically impaired renal function numbered 7.2% (13/180) and 22.7% (32/141) had microalbuminuria; 17.2% (10/58) had diabetes related retinopathy. No one in our cohort had diabetic ulcers; however, 10.3% (13/126) had absent foot pulses.
Conclusion:
We found a higher prevalence of microalbuminuria compared with retinopathy in a large cohort of cystic fibrosis adults. This study demonstrates the need for regular specialist follow-up to facilitate early identification of such complications and a long-term prospective cohort to understand underlying mechanisms.
Research in context
1. What is already known about this subject?
Cystic fibrosis related diabetes affects approximately 50% of people with cystic fibrosis over 35 years of age and is increasingly prevalent as prognosis improves due to better treatment regimens, nutritional support and management by a multidisciplinary team. Several previous publications have examined prevalence of microvascular complications related to cystic fibrosis-related diabetes. However, limited data are available for the same from the UK.
2. What is the key question?
• What is the prevalence of microvascular complications in cystic fibrosis-related diabetes in the United Kingdom?
3. What are the new findings?
• In our study, 22.7% people with cystic fibrosis-related diabetes had microalbuminuria and 17% had background retinopathy.
• We found a higher frequency of microalbuminuria compared with retinopathy in our cohort.
• 10.3% had absent foot pulse.
4. How might these results change the focus of research or clinical practice?
• Our study demonstrates need for regular specialist follow-up to facilitate early identification of microvascular complications in cystic fibrosis-related diabetes and a long-term prospective cohort to understand underlying mechanisms.
Introduction
Cystic fibrosis (CF) is one of the most frequent autosomal recessive genetic conditions affecting Caucasians. It is caused by mutations in the cystic fibrosis transmembrane regulator gene, resulting in dysregulated chloride transport across epithelial cell membranes. Consequently, mucus secretions are thickened in multiple organ systems, including the lungs, pancreas, biliary tree and reproductive tract. 1 The main causes of morbidity in CF are respiratory failure, due to progressive lung disease, and malnutrition, due to pancreatic exocrine insufficiency and malabsorption. 2 However, longer term sequelae of CF are being increasingly recognised, including bone disease and CF-related diabetes (CFRD).
CFRD affects approximately 50% of people with CF over 35 years of age 3 and is increasingly prevalent as CF prognosis improves 4 due to better treatment regimens, nutritional support and management by a multidisciplinary team. 5 CFRD is a distinct form of diabetes mellitus, but shares pathophysiological features with both type 1 and type 2 diabetes.6–8 It is principally caused by reduced insulin secretion, arising secondary to reduced beta cell mass and beta cell dysfunction. 9 The latter is caused by pancreatic ductal obstruction from viscous secretions and auto-digestion of pancreatic tissue from accumulation of exocrine enzymes. This results in fibrosis, fatty infiltration and beta cell loss. 9 It is well recognised in people with CF that the first stage of insulin secretion is delayed, resulting in postprandial hyperglycaemia, which leads on to impaired glucose tolerance and eventual CFRD.7,10 Screening is essential because of the typically insidious onset of CFRD. 6 Importantly, coexistence of diabetes with CF worsens pulmonary function, shortens lifespan and confers increased morbidity and mortality, particularly in females. 11 Pre-diabetes usually pre-dates a diagnosis of CFRD by up to 4–6 years.3,12 This highlights the importance of screening to minimise the deleterious consequences of hyperglycaemia and the detriment to lung function, nutrition and survival, before a diagnosis of CFRD is made. 13
Glycosylated haemoglobin (HbA1c) should be tested annually and used for monitoring but is not recommended as a screening test, because of its poor sensitivity.3,14 Consensus guidelines therefore recommend that people with CF should be screened for CFRD using the oral glucose tolerance test (OGTT) at least annually from the age of 10 years and at times of pulmonary infective exacerbations when treated with steroids or during work-up for lung transplantation. Although these guidelines acknowledge that OGTT is an imperfect test due to the variability of both the test and the underlying condition, they recommend OGTT as the screening test of choice.2,4
Guidelines recommend insulin as the preferred therapeutic agent 3 because of its effectiveness in managing hyperglycaemia in addition to its anabolic effects, which improves lung function and nutritional status. 15 Furthermore, it is recommended that people with CFRD should have multi-disciplinary team follow-up (preferably four times per year), be offered diabetes structured education programmes and that HbA1c should be monitored with a target of <7% (<53 mmol/mol).3,5
Although macrovascular disease is currently rare, 16 microvascular complications are increasingly being recognised as complications of CFRD, particularly with increased duration of diabetes and poorer glycaemic control. 17 Retinopathy, nephropathy and neuropathy are all recognised complications2,8 and prevalence of retinopathy has been reported from small studies as being equivalent to rates observed in type 1 diabetes. 18 Chronic kidney disease and microalbuminuria are also increasingly recognised in CF patients, for which CFRD is a strong risk factor. 19 Contributing to this renal damage are combinations of administration of intravenous aminoglycosides for treatment of infective exacerbations and long-term immunosuppressive therapy following lung transplantation. 19 Consensus guidelines therefore recommend annual screening for microvascular complications after 5 years of CFRD and frequent monitoring of risk factors: measuring blood pressure at each clinic appointment and annual lipid profiles. 3 Although microvascular complications are recognised, reported prevalence differs and the risk factors associated with their development remain poorly understood.
The objective of the study was to evaluate the prevalence of microvascular complications in CFRD in a large UK cohort.
Methods
This retrospective study was done in the United Kingdom in 2019. All people with CFRD who were registered in the West Midlands Adult Cystic Fibrosis Centre, University Hospitals Birmingham NHS Foundation Trust, Birmingham, UK were included for the study. We identified the study population from details in the outpatient attendance for the cystic fibrosis centre. We had full access to the database of the study population. We did not use any other specific code for identifying the study population. The study did not involve any linkage of databases.
Clinical and biochemical parameters were collected from 1 January 2018 to 31 December 2018. Demographic information collected included age (years), gender and ethnicity. Clinical parameters included the most recent systolic and diastolic blood pressure recorded using an automatic blood pressure cuff (mmHg), height (cm), weight (kg) and body mass index (BMI) (kg/m2). Clinical notes and letters were reviewed to obtain duration of diabetes (years), current insulin therapy, oral anti-diabetes agents, presence or absence of hypoglycaemic episodes, evidence of lipodystrophy (including lipohypertrophy or lipoatrophy) and foot examination including manual palpation of peripheral pulses. Hypoglycaemia was classified into mild (self managed) or severe (requiring third party assistance). Biochemical parameters were obtained from the most recent annual diabetes clinic review, including renal function [estimated glomerular filtration rate (eGFR) and creatinine], liver function testing (albumin, gamma-glutamyl transferase (GGT), bilirubin, Alanine Transaminase (ALT), Alkaline Phosphatase (ALP) and total protein), lipid profiles [total cholesterol, triglycerides, high-density lipoprotein (HDL) cholesterol, non-HDL cholesterol] and glycaemic control was measured using HbA1c (%, mmol/mol).
We assessed for the following microvascular complications: microalbuminuria, retinopathy and neuropathy. Microalbuminuria was assessed by analysing the albumin–creatinine ratio (ACR) on an early morning urine sample from the most recent annual diabetes clinic appointment; the definition for microalbuminuria was in line with National Institute for Health and Care Excellence (NICE) guidelines. 20 Retinopathy screening attendance, images and reports were obtained from the UK national retinopathy annual screening programme. Diabetes-related retinopathy was graded as follows (in order of severity): no diabetes-related retinopathy, background retinopathy, pre-proliferative retinopathy, proliferative retinopathy and maculopathy, and the worst grade of either eye was recorded. Neuropathy was evaluated by physical examination, which included evaluation of sensation, motor function and autonomic changes. The physician who leads foot services in our hospital performed all foot examinations including manual palpation of peripheral pulses. The study was registered as a service evaluation audit with the department of clinical governance, University Hospitals Birmingham NHS Foundation Trust (audit approval number: 4571). Informed consent was waived as this was a retrospective data collection from patient case records. All data were anonymised at the stage of analysis and reporting.
Descriptive statistics were performed using STATA v. 15.1 and where analysis was conducted to assess the difference between two groups, chi-squared and Wilcoxon rank-sum tests were conducted depending on whether the variables considered were categorical or continuous (as data were non-parametric) respectively. The statistical significance level was set at p < 0.05.
Results
Baseline characteristics
A total of 189 adults with CFRD were cared for at our centre during 2018 and records of all were included in the study. Median age was 32 years (interquartile range 27–39) and 56.6% were male (n = 107/188). Demographics, clinical parameters and biochemical parameters are presented in Table 1. The annual review of 79.4% (150/189) took place in 2018. There were varying levels of missing data dependent on the parameters measured, which are also highlighted in Table 1.
Baseline characteristics of our cohort. Median age was 32 years (interquartile range 27–39). People in the cohort had body mass index within healthy range [median: 22 kg/m2 (21–26 kg/m2)]. Median systolic and diastolic blood pressure were also within normal range [123 mmHg (112–131 mmHg) and 78 mmHg (70–84 mmHg), respectively]. Their median lipid profiles were also within acceptable limits. We did not find any significant liver impairment in our cohort.

CFRD treatment
Median duration of diabetes (130/189) was 8 years (5–14 years) and median HbA1c (182/189) was 46 mmol/mol (40–61 mmol/mol) [6% (5.8–7.7%)]. At the point of this review, 56.1% (106/189) were recorded to be on some form of insulin therapy; 17.9% (34/189) were on dipeptidyl peptidase 4 inhibitors, 3.7% (7/189) on metformin and 2.1% (4/189) on sodium-glucose transport protein 2 inhibitors. Of those on insulin therapy, 62.3% (66/106) were on basal-bolus insulin regimen, 26.4% (28/106) on bolus insulin only, 8.5% (9/106) on mixed insulin solution and 2.8% (3/106) were on an insulin pump.
Complications associated with CFRD
Forty-four per cent (44/100) had experienced at least one episode of mild hypoglycaemia in the preceding year. All these patients who experienced hypoglycaemia were treated with insulin for CFRD. None of our patients reported severe hypoglycaemia in the study period. Evidence of lipodystrophy was present in 28.7% (31/108). We had records of retinopathy screening status on 138 members of our cohort. Of those, 52.2% (72/138) had evidence of attending screening and we had results for 58 people with CFRD in our cohort. We compared those who had retinopathy screening (n = 72) with the remainder of the population (n = 117) and found no significant differences between the groups in terms of age, gender, diabetes duration and control, and insulin therapy use (Table 2). Of those who had recorded retinopathy screening, 17.2% (10/58) had some form of diabetic eye disease (retinopathy or maculopathy). The severity of eye changes was graded using National Screening Committee Criteria for retinopathy (R0 – None, R1 – Background, R2 – Pre-proliferative, R3 – Proliferative), maculopathy (M0 – nil present, M1 – maculopathy), Photocoagulation (P) and Unclassifiable (U). 21 Seven patients had background retinopathy (R1 M0 worst eye), treated proliferative retinopathy (right eye: R1 M0, left eye: R3s M0) in one patient, pre-proliferative retinopathy and maculopathy (right eye: R2 M1, left eye: R2 M0) in one patient and only maculopathy (right eye: R0 M0, left eye: R0 M1) in one patient. People with retinopathy were younger and predominantly male compared with those without retinopathy (Table 3).
Difference in baseline characteristics and management of cystic fibrosis-related diabetes between people who had retinopathy screening and the rest of the cohort. There were no significant differences between the groups in terms of age, gender, diabetes duration and control, and insulin therapy use.
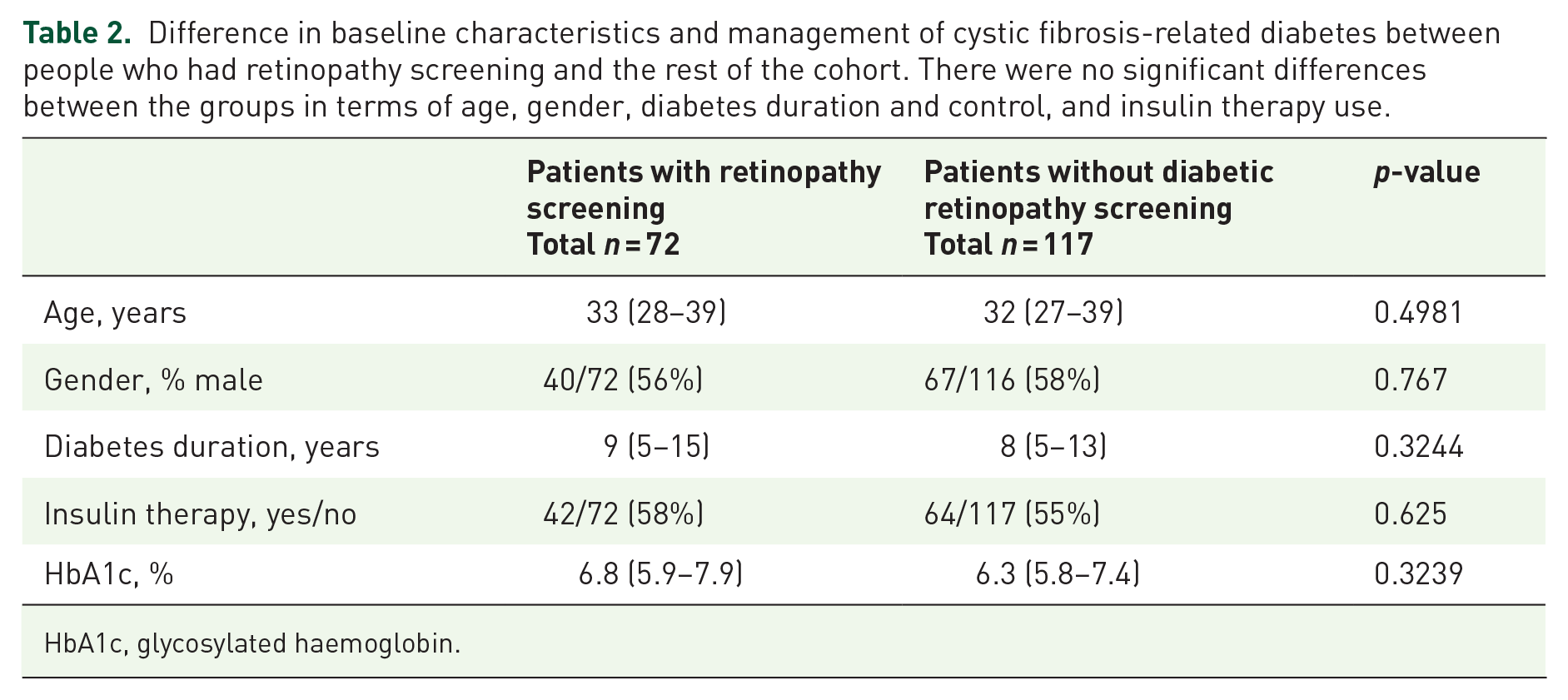
HbA1c, glycosylated haemoglobin.
Comparing the demographics and clinical characteristics between those with and without retinopathy. People with retinopathy were younger and predominantly male compared with those without retinopathy.

CFRD, cystic fibrosis-related diabetes; HbA1c, glycosylated haemoglobin.
Median eGFR, creatinine and ACR were 85 (71–90) ml/min per 1.73 m2, 67 (57–79) μmol/l and 0.9 (0–2.6) mg/mmol respectively. Renal function was biochemically impaired in 7.2% (13/180) (eGFR <60 ml/min per 1.73 m2). Interestingly, 22.7% (32/141) had microalbuminuria. One study subject had macroalbuminuria (410 mg/mmol). Table 4 compares the demographic and clinical information between those with and without microalbuminuria.
Comparing the demographics and clinical characteristics between those with and without microalbuminuria. People with microalbuminuria were older than those without microalbuminuria; otherwise there was no significant difference between the two groups.

CFRD, cystic fibrosis-related diabetes; HbA1c, glycosylated haemoglobin.
None of our study cohort had neuropathy and there were no diabetes-related foot ulcers (189/189 examined and notes reviewed). However, 10.3% (13/126) had absent peripheral foot pulses on palpation.
Discussion
In our cohort, 79.4% of people with CFRD had their annual review performed but datasets for the biochemical and retinal screening were incomplete. A longitudinal study by Scheuing et al. 22 compared received care with guideline standards in people with CFRD 3 and showed that HbA1c, BMI and lipid profile were tested at least once annually 88.8%, 86.5% and 79.5% of the time. People with CFRD were screened for retinopathy or microalbuminuria 29.9% and 33.2% of the time respectively, but overall only 7.9% had a complete set of examinations which would meet all guideline standards. 22 This demonstrates the low adherence to guidelines for people with CFRD, which can be explained by the context of burdensome treatment regimens and frequent hospital attendances for people with CF. Nevertheless, CFRD warrants optimal care, a multidisciplinary team approach and close monitoring for complications in order to prevent harm.
Frequent hypoglycaemic episodes can suppress hypoglycaemic awareness, increasing risk of severe hypoglycaemic episodes, and adversely affect quality of life.23,24 Therefore, monitoring and characterisation of hypoglycaemia in CFRD patients is important, but challenges remain because continuous glucose monitoring is not routinely available for this cohort. Nevertheless, our finding that 44% of our CFRD cohort reported non-severe hypoglycaemia at their annual review highlights the importance of a multidisciplinary team approach to optimise diabetes management in CFRD.
The majority of our cohort did not attend for annual eye screening in the period studied and of those who did, 17% had evidence of retinopathy or maculopathy. Roberts et al. 17 performed a cross-sectional analysis in a Welsh cohort of people with insulin treated CFRD to assess retinopathy (using digital photography) and retinal screening attendance. In their cohort, 36% of patients did not attend for their retinal screening appointment in the year assessed, but of those who did, 42% had evidence of retinopathy, ranging from mild to proliferative, and associated with maculopathy. 17 Choudhury et al. 25 performed a retrospective seven-year longitudinal study in this same Welsh cohort and demonstrated that 44% of screened people with CFRD had retinopathy. 25 Yung et al. 26 found retinopathy in 16% and 23% of a small cohort of patients at 5 years and 10 years of CFRD respectively, but HbA1c was not significantly different between people with and without retinopathy. 26 Andersen et al. also reported retinopathy in 36% of insulin-treated people with 10 years’ duration of CFRD. 18 The lower frequency of retinopathy observed in our cohort could be due to inclusion of non-insulin-users and lower median HbA1c compared with the cohorts assessed in these other series [46 mmol/mol (6.4%) versus 61 mmol/mol (7.7%) in Roberts et al. 17 ], despite similar duration of diabetes. Our study therefore reflects better glycaemic control, but retinal disease remained prevalent despite this. Further studies with larger cohorts and serial follow-up are needed to confirm these findings.
Microalbuminuria has long been associated with increased risk of developing chronic kidney disease (CKD).27–29 In a seven-year longitudinal study, Quon et al. 30 found 2.4-fold increased risk for developing CKD in CFRD patients treated with insulin for 1–4 years. 30 Berg et al. 31 in a cross-sectional analysis showed that concomitant moderate CKD in CF was not only significantly associated with higher prevalence of CFRD (p < 0.001), but also increased cumulative intravenous aminoglycoside usage (p < 0.005), advancing age (p < 0.001) and longer duration of chronic pulmonary infections (p < 0.008). 31 As we demonstrate that microalbuminuria represents a frequent complication of CFRD, this warrants annual monitoring but must be interpreted with caution because microalbuminuria may be falsely elevated with antibiotics, severe illness or physical stress. 32
We observed a relatively higher frequency of microalbuminuria compared with retinopathy in our cohort. This is in keeping with a study performed by Van den berg et al., 33 who compared microvascular complications in CFRD with people with type 1 diabetes in a combined UK and Netherlands cohort. They showed that people with CFRD were significantly more likely to develop microalbuminuria (21% versus 4.1%, p = 0.003) and less likely to develop retinopathy (10% versus 24%, p = 0.044) compared with type 1 diabetes matched controls. 33 Lind-Ayres et al. 34 showed in their study that the odds of developing permanent microalbuminuria were seven times higher in CFRD and 48 times higher with concomitant lung or liver transplantation. 34
Although there was no evidence of diabetes-related foot ulcers in our cohort, 10.3% had absent peripheral foot pulses, a finding which, to our knowledge, has not been previously reported. All these patients were referred to vascular surgical review for further assessment. We did not record the results for this review. Vascular insufficiency was not objectively verified as this finding could indicate macroangiopathy, which warrants further investigation. Clubbing is pathognomonic of CF and hypertrophic osteoarthropathy of the hands and toes arises secondary to hypoxia, inflammation and abnormal vascularisation in CF patients.35,36 In future work, we would need to compare our CFRD cohort with matched non-CFRD controls to determine whether macroangiopathy is additionally accountable for the vascular insufficiency observed.
Limitations
Despite the large sample size studied here, there are a few limitations for this study. None of the people in our cohort had a renal biopsy, and therefore it was not possible to diagnose diabetes-related nephropathy on pathology. In addition, retinal screening and microalbuminuria were incomplete, rendering it difficult to establish accurate frequencies of microvascular complications. Also, of note is that the difference in the sample sizes of our cohorts for microalbuminuria and retinopathy are different, which may have some influence on the results. We also have recorded microalbuminuria based on analysing the ACR on an early morning urine sample, in line with NICE guidelines. 20 We note that CFRD guidelines recommend that microalbuminuria should be made if two out of three tests are abnormal within a 3–6-month period 3 and hence our results may need further validation. We do not have history of any organ transplantation for our cohort. Additionally, we were not able to explore blood glucose measurements and variability, or adherence to treatment, transplantation status, haemoglobin level, number of hospital admissions, intravenous aminoglycoside treatment (frequency or duration), drug history, exercise, or lung function testing. These factors are important because they can contribute to transient microalbuminuria. Further, lack of confirmatory tests for vascular insufficiency limits the extrapolation of these findings.
Conclusion
We found a higher frequency of microalbuminuria compared with retinopathy in a large cohort of CF adults. Exposure to aminoglycosides as part of CF treatment might explain the higher frequency of microalbuminuria, yet not in entirety. We suggest that future research should assess additional potential risk factors for microalbuminuria. Longitudinal studies are also needed to assess the development of long-term diabetes-related comorbidities in people with CFRD, given the prevalence. Overall, our study demonstrates that people CFRD are at risk of microvascular disease, confirming the need for a multi-disciplinary team approach for judicious screening and monitoring to minimise the development of microalbuminuria and retinopathy in people with CFRD.
Footnotes
Author contribution(s)
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
