Abstract
Keywords
Introduction
Type 2 diabetes mellitus (DM) leads to markedly increased risk of eye, renal, and foot complications. The United Kingdom Prospective Diabetes Study (UKPDS) demonstrated that moderate levels of glucose control (mean end-of-study glycated hemoglobin [HbA1c] of 7.9% compared with 8.5%, and mean in-trial HbA1c of 7.1% versus 7.9%) significantly reduced onset and progression of eye and renal problems in patients with recently diagnosed type 2 diabetes. Based on these encouraging results, two large clinical trials (ACCORD and ADVANCE) were organized to test the hypothesis that normalization or near-normalization of glucose in patients with type 2 diabetes would lead to even greater improvement in both microvascular and macrovascular complications than what had been possible with the moderate levels of blood glucose reduction achieved in the UKPDS, whose treatment phase concluded in 1997 [UK Prospective Diabetes Study (UKPDS) Group, 1998c]. Moreover, both ACCORD and ADVANCE employed factorial designs to quantify the independent and possible conjoint effects of HbA1c and BP lowering on microvascular and macrovascular complications, and ACCORD also assessed the impact of masked fenofibrate in addition to statins on diabetes complications [Gerstein et al. 2008; Patel et al. 2008].
The hypothesis that reductions in eye, renal, and foot complications achieved through near-normalization of HbA1c would exceed the reduction in complications observed in the UKPDS was strongly supported by epidemiological data that consistently show an inverse relationship between level of HbA1c and occurrence of microvascular complications in type 2 diabetes. On the other hand, data from both clinical trials and epidemiological studies showed that rate of decline in microvascular complications is less at each 1% decrement in HbA1c as values approach the nondiabetes range [DCCT, 1993].
In this report, we quantify the impact of near-normalization of glucose control on specific eye, kidney, and foot complications in adults with established type 2 diabetes. We focus on the ACCORD and ADVANCE trials because these are the only large trials to date that successfully achieved and maintained near-normalization of glucose control (median HbA1c levels in the mid 6% range) throughout multiyear intervention periods. In addition, we consider information from randomized trials with factorial designs to compare the impact of glucose, BP, and lipid control on microvascular complications in type 2 diabetes [UK Prospective Diabetes Study (UKPDS) Group, 1998a, 1998b, 1998c].
Methods
Data
Comparison of selected characteristics of adults with type 2 diabetes randomized in various studies.

–, not available.
Analytic plan
This analysis focuses on the difference in incidence rates of microvascular complications in the intensive versus standard treatment groups in the ACCORD and ADVANCE trials. Initial publications from these study groups have emphasized the relative risk reductions associated (or not associated) with near-normalization of glucose. However, relative risk reductions do not fully account for the frequency of a given complication or outcome. The difference in microvascular complication rates in the intensive versus standard treatment groups (absolute risk reduction) can be easily converted mathematically into an estimate of the number needed to treat (NNT) using the following formula:
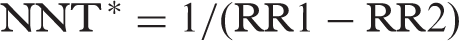
The 95% confidence intervals of the NNT point estimate are given by the reciprocal of the values defining the 95% confidence intervals of the difference in proportions (RR1 – RR2), which are (1/upper 95% CI for (RR1 – RR2)) and (1/lower 95% CI for (RR1 – RR2)).
A more detailed discussion of absolute risk reduction and the conversion of absolute risk data to NNT, and the advantages and limitations of this approach, are beyond the scope of this manuscript and can be found elsewhere [Ridker et al. 2009; Wu and Kottke, 2001; Rembold, 1996].
Results
Impact of near-normalization of glucose on microvascular complications in the ACCORD trial
Design of ACCORD
The ACCORD trial was designed to test the hypothesis that near-normalization of glucose will significantly reduce macrovascular (heart attack, stroke), microvascular (eye, renal, foot), and other (cognitive status, bone) adverse outcomes associated with type 2 diabetes. Every study subject met stringent eligibility requirements including (a) serum creatinine level <1.5 mg/dl, (b) having coronary heart disease (CHD) or two or more CHD risk factors, and being in certain middle to older age ranges, and (c) having A1c, blood pressure (BP), low-density lipoprotein (LDL), and high-density lipoprotein (HDL) in defined ranges, depending upon pre-ACCORD treatment for these conditions [Gerstein et al. 2007]. Table 1 provides data on characteristics of randomized subjects in ACCORD and other trials of interest to this analysis.
Each ACCORD study subject was randomized twice at entry to the study. The first randomization (N = 10,251) was to either the intensive or standard glucose arms of the study. Intensive and standard glucose control arms differed in their HbA1c targets, which were <6% for the intensive arm and 7.0–7.9% for the standard arm. For both arms, clinicians could provide whatever glucose-lowering medications deemed appropriate to achieve the HbA1c targets. The median HbA1c at baseline was 8.1%; the intensive glucose arm achieved a median HbA1c of 6.4% while the standard glucose arm achieved a median HbA1c of 7.5% over a mean of 3.5 years of follow up [Gerstein et al. 2008],
The second randomization was to either (a) the intensive versus standard BP arms (N = 4733), or (b) the intensive versus standard lipid arms (N = 5518). The intensive BP arm had a systolic blood pressure (SBP) goal of <120 mmHg, and achieved a mean BP of 119.3/64.4 mmHg. The standard BP arm had a goal SBP of 130–139 mmHg, and achieved a mean BP of 133.5/70.5 mmHg [TAS Group, 2010b]. Participants in both lipid arms received open-label simvastatin; the intensive group also received 160 mg daily of masked fenofibrate while the standard group received a placebo [TAS Group, 2010a].
The active treatment phase for the ACCORD glucose trial ended in February 2008 when the ACCORD Data Safety and Monitoring Board (DSMB) of the National Institutes of Health (NIH)-sponsored study mandated that intensive glucose control patients be converted to the standard glucose control protocol due to a significant and persistent excess in overall and cardiovascular (CV) mortality in the intensively treated participants (NNT to cause one excess death was 94 patients for a mean of 3.5 years) [Gerstein et al. 2008]. Thus, the impact of near-normalization of glucose on microvascular complications must be considered in the context of the relatively short follow-up period, and in the context of the excess total and CV mortality caused by near-normalization of glucose in the ACCORD trial.
The ACCORD Eye Study enrolled 2856 ACCORD trial participants and was designed to assess the impact of intensive glucose, BP and lipid control on progression of diabetic nephropathy and on vision loss over a 4-year follow-up period. ACCORD Eye study participants had seven-field stereoscopic fundus photographs at baseline, 20, and 40 months [TASGaAES Group, 2010].
Microvascular outcomes in ACCORD
Summary of ACCORD data (to time of transition mean 3.4 years of follow up) showing the occurrence of specified microvascular events in those in the intensive versus the standard glucose control groups [Ismail-Beigi et al. 2010].

Significant at p < 0.05 or lower.
Over a 3.4-year follow-up period.
NNT, number needed to treat; n.s., not significant (for associations that are not significant, it is not appropriate to estimate NNT).
Prespecified secondary microvascular outcomes in ACCORD showed a mixed picture at 3.4 years, when the intensive glycemic intervention was stopped due to excess mortality in the intensive glycemia treatment group. Among secondary microvascular outcomes, 2/5 renal measures and 1/4 neuropathy measures were significantly improved in the intensive glucose group, while 1/5 renal measures was significantly worse in the intensive glucose group [Ismail-Beigi et al. 2010].
Impact of near-normalization of glucose on microvascular complications in the ADVANCE trial
Design of ADVANCE
ADVANCE tested the effect of intensive glucose-lowering regimen that aimed for A1c levels of ≤6.5% using the sulfonylurea gliclazide-MR on outcomes after a follow up of 5 years [Patel et al. 2008]. ADVANCE randomized 11,140 participants from 20 countries; mean age was 66 years and mean duration of type 2 diabetes was 8 years. The primary outcomes were a composite of macrovascular outcomes (comprising CV death, fatal and nonfatal myocardial infarction or stroke) and microvascular complications (comprising new or worsening nephropathy or retinopathy) assessed jointly and separately. Secondary microvascular outcomes included new or worsening nephropathy requiring renal replacement therapy, death from renal disease, development of macroalbuminuria, a doubling of serum creatinine to ≥200 µmol/l, new onset microalbuminuria (urinary albumin to creatinine ratio of 30–300 mg/g) and total renal events defined as the composite of new or worsening nephropathy or new onset of microalbuminuria, new, or worsening retinopathy.
Microvascular outcomes in ADVANCE
Summary of ADVANCE Data (mean 5 years of follow-up) showing occurrence of specified microvascular events in those in the Intensive versus the Standard glucose control groups. [Patel et al. 2008].

Significant at p < 0.05 or lower.
Over a 5-year follow-up period.
NNT, number needed to treat; n.s., not significant (for associations that are not significant, it is not appropriate to estimate NNT).
Comparing the impact of near-normalization of glucose on microvascular complications in the ACCORD and ADVANCE trials
ADVANCE found a significant benefit on nephropathy, driven by changes in onset of microalbuminuria and macroalbuminuria during the 5-year intervention period, but no impact on retinopathy during the intervention period. Conversely, ACCORD reported mixed effects on measures of nephropathy (less microalbuminuria but a greater rate of doubling of serum creatinine or decreased estimated glomerular filtration rate [eGFR]), but a benefit on progression of retinopathy within the intervention period.
Neither ACCORD nor ADVANCE detected a change in end-stage microvascular complications of type 2 diabetes, such as renal failure, blindness, or amputations. ACCORD reported no significant impact of near-normalization of glucose on either of its primary composite microvascular endpoints both of which included development of renal failure or retinal photocoagulation or vitrectomy to treat retinopathy. In the ACCORD Eye study, normalization of glucose slowed progression of retinopathy, but did not protect against moderate vision loss [Chew et al. 2010].
It is unclear why there was so much heterogeneity of renal and eye complication rates in ACCORD and ADVANCE. ACCORD drew its study patients mostly from the USA and some from Canada, while ADVANCE drew its participants from 20 nations (large numbers from Australia and China), not including the USA. Baseline serum creatinine for eligibility had to be <1.5 mg/dl at randomization in ACCORD, and <2.0 mg/dl in ADVANCE, which might contribute to differences in nephropathy results between the trials. ADVANCE had a run in period that led to the exclusion of about 10% of potential participants just prior to randomization.
Subgroup analysis of ADVANCE showed that the combined microvascular and macrovascular primary endpoint, which was driven to a great extent by a reduction in nephropathy, was significantly improved in those with no history of microvascular complications, but was not significantly improved in those with established microvascular complications at baseline. ACCORD has not yet analyzed differences in microvascular outcomes across patient subgroups.
Both trials had relatively short periods of follow up, a mean of 3.5 years in ACCORD and 5 years in ADVANCE. It is possible that longer follow up may have increased the differences in end-stage microvascular complications related to glucose control.
Other strategies to reduce microvascular complications in patients with type 2 diabetes
Although we have focused primarily on effects of near-normalization of glucose on microvascular complications in type 2 diabetes, it is worthwhile to briefly consider other strategies that may reduce microvascular complications in patients with type 2 diabetes, and to assess the interaction of these strategies with glucose control.
Impact of BP control in ADVANCE and interaction with glucose control
The ADVANCE trial used a double-blind factorial design to test the effects of a fixed combination of an angiotensin-converting enzyme (ACE) inhibitor (perindopril) and a diuretic (indapamide) used to lower BP irrespective of initial BP versus placebo on macrovascular and microvascular outcomes in a diverse group of patients with type 2 diabetes after 4.3 years of follow up [Patel et al. 2007].
Active BP treatment versus placebo in the ADVANCE trial resulted in a mean reduction of 5.6 and 2.2 mmHg in systolic and diastolic pressures, respectively [Patel et al. 2007]. This resulted in a significant 9% relative risk reduction in the composite primary outcome. However, the 8% and 9% relative risk reductions in macrovascular and microvascular outcomes, respectively, were not independently significant. There was a nonsignificant 18% relative risk reduction for new or worsening nephropathy, and a significant 21% relative risk reduction for development of microalbuminuria (incidence rates 23.6% vs. 19.6%; NNT = 25 for 4.3 years). Active BP treatment resulted in no significant reductions in new or worsening retinopathy, need for photocoagulation, visual deterioration, or new or worsening neuropathy. The relative risk of death from CV disease and all-cause mortality were significantly reduced in the active BP treatment arm of ADVANCE [Patel et al. 2007].
An analysis of the combined effects of the BP lowering and intensive glycemic control in the ADVANCE study has been reported [Zoungas et al. 2009]. Rates for two of the microvascular outcomes (all renal events and new onset of microalbuminuria) were significantly reduced by intensive BP control, or intensive glucose control, or both, with participants in both intensive arms having the lowest hazard ratio, but these interaction effects were not statistically significant. For two other renal outcomes, namely new or worsening nephropathy and new onset of macroalbuminuria, the hazard ratio (HR) for only the combined active treatment arm was significant, perhaps in an additive fashion. For new or worsening nephropathy, the HR in the combined active treatment arm was 0.67 (95% CI of 0.50–0.88) compared with the standard/placebo arm (incidence rates 2.9% vs. 4.3%; NNT = 71 for 4.3 years). The HR for new onset of macroalbuminuria in the combined active treatment arm was 0.46 (95% CI of 0.32–0.65) compared with the standard/placebo arm (incidence rates 1.58% vs. 3.41%; NNT = 55 for 4.3 years). The HR for the composite of major microvascular events was 0.81 (95% CI of 0.68–0.79) in the combined active treatment arm versus standard/placebo arm (incidence rates 7.6% vs. 9.3%; NNT = 59 for 4.3 years). The combined active treatment arm did not have significantly reduced HR for new or worsening retinopathy, or for major macrovascular events, major coronary heart events, or major cerebrovascular events. However, HR for CV mortality was reduced in the active BP treatment arm and in the arm receiving combined intensive treatment.
Impact of BP control on microvascular complications in the UKPDS study
The UKPDS study tested the effects of tight versus less tight glycemic control on macrovascular and microvascular outcomes among 4320 recently diagnosed adult patients (mean duration of diabetes of 4 years) who were followed for a median of 10.4 years. Microvascular complications (defined as a composite of retinopathy requiring photocoagulation, vitreous hemorrhage, and fatal and nonfatal renal failure) were significantly reduced among patients undergoing intensive glycemic control (achieved HbA1c levels in the 7.1–7.9% range) during the active intervention period and were maintained during an additional observational follow-up period [Holman et al. 2008; UK Prospective Diabetes Study (UKPDS) Group, 1998c].
A subset of 1148 patients who enrolled in the UKPDS study with hypertension at entry (mean BP of 160/94 mmHg) were allocated to tight (758 patients) and less tight (390 patients) BP control and followed for a median of 8.4 years [UPDSU Group, 1998]. Participants in the intensive BP arm were treated with an ACE inhibitor (captopril) or a beta-blocker (atenolol). The mean age of these participants was 56 years and the median duration of diabetes in this subset was 2.6 years. The mean BP level achieved in the intensive and standard arms were 144/82 and 154/87 mmHg, respectively. Patients in the intensive BP control arm had a significant 37% reduction in the composite microvascular endpoint defined above (incidence rate 19.2 vs. 12.0 per 1000 patient years), and the effect was mostly due to decreased risk of retinal photocoagulation. In addition, there was a 34% reduction in the risk of deterioration of retinopathy by two steps (incidence rate 51.3% vs. 34.0%; NNT = 9 for 8.4 years), and a 47% reduction in the risk of deterioration of visual acuity by three lines in the early treatment of diabetic retinopathy study (ETDRS) chart (incidence rate 19.4% vs. 10.2%; NNT = 10.9 for 7.5 years). There was also a 29% reduction in the proportion of patients who developed microalbuminuria (>50 mg/l) in the sixth year of follow up; this effect was no longer significant at study end. There was no significant effect on the risk of development of macroalbuminuria (>300 mg/;), or indices of peripheral or autonomic neuropathy [UPDSU Group, 1998].
The potential risk reduction in those randomized to both the UKPDS glucose and BP trials for microvascular outcomes was not reported, perhaps due to lack of power necessary for the analysis. However, the incidence of ‘any diabetes-related endpoint’ was lower in those simultaneously allocated to intensive glycemia and BP arm of the trial, and similar trends were seen for all-cause mortality and diabetes-related deaths [Stratton et al. 2006].
Results of the above studies indicate that the incidence of some, but not all, microvascular complications of type 2 diabetes among those with recently diagnosed (UKPDS) or established diabetes (ADVANCE) may be independently reduced by intensive treatment of glycemia and BP. Furthermore, the beneficial effects of these treatments for some measures of microvascular outcome in ADVANCE appear to be partially additive. Additional data forthcoming from the ACCORD trial may also address the conjoint effects of BP and glucose control on microvascular outcomes in that trial.
Impact of BP control and fenofibrate use on retinopathy in ACCORD
The ACCORD Eye study further evaluated 2856 patients in the glucose, BP, and lipid studies of ACCORD. Near-normalization of glucose led to a significant reduction in progression of retinopathy defined by a three-step or more deterioration in the ETDRS Severity Scale (odds ratio [OR] = 0.67; 95% CI of 0.51–0.87; p = 0.003) but did not reduce moderate vision loss (HR = 0.95; 95% CI of 0.80–1.13; p = 0.56). Intensive BP control led to a nonsignificant worsening in progression of diabetic retinopathy (OR = 1.23; 95% CI of 0.84–1.79; p = 0.29) and nonsignificant increase in moderate vision loss (HR = 1.27; 95% CI of 0.99–1.62; p = 0.06). Use of fenofibrate versus placebo was associated with reduced progression of diabetic retinopathy (OR = 0.60; 95% CI of 0.42–0.87; p = 0.006) but no improvement in moderate vision loss (HR = 1.04; 95% CI of 0.83–1.32; p = 0.73). The NNT for 4 years to prevent progression of retinopathy in one subject was 32.2 for near-normalization of glucose, and 27.0 for fenofibrate treatment. The retinopathy benefit related to fenofibrate use was not significantly different in those in the intensive and standard glucose arms of ACCORD (p = 0.46).
Conclusions
Epidemiological data consistently demonstrate a strong inverse association of plasma glucose levels and microvascular complications [Klein, 1995; Klein et al. 1994; Barrett-Connor et al. 1991]. This observation, coupled with encouraging results from the UKPDS study, raised hopes that near-normalization of glucose through intensive pharmacological treatment might successfully prevent or delay progression of both end-stage and early microvascular complications in patients with type 2 diabetes.
The results of ACCORD and ADVANCE provide inconsistent evidence that near-normalization of blood glucose reduces onset and progression of microvascular complications. Neither study documented an impact of near-normalization of glucose on protocol-specified composite measures of end-stage microvascular complications. Near-normalization of glucose had a beneficial impact on progression of retinopathy in ACCORD, but not in ADVANCE. Conversely, a benefit of near-normalization of glucose on nephropathy progression was observed in ADVANCE, but was less consistent in ACCORD. Nonetheless, the short duration of the ACCORD and ADVANCE trials leaves open the possibility that near-normalization of glucose may confer as yet undocumented future reductions in end-stage microvascular complications during longer-term follow up. In comparison, the UKPDS and Steno-2 studies, which both achieved only more moderate glucose control, both had significant reductions in end-stage microvascular complications within their active treatment period [Gaede et al. 2008, 2003; Turner et al. 1999].
The modest incremental benefits of near-normalization of blood glucose on microvascular complications in ACCORD and ADVANCE are mitigated to a considerable degree by the documented risks of near-normalization of glucose observed in those trials, and by other considerations. These include: (a) significantly increased risk of severe hypoglycemia with more intensive glucose control; (b) significantly increased weight gain with more intensive glucose control; (c) failure of near-normalization of glucose to significantly reduce overall or CV mortality or prespecified measures of macrovascular events in either ACCORD or ADVANCE [Gerstein et al. 2008; Patel et al. 2008]; (d) near-normalization of glucose significantly increased all cause mortality (one excess death for every 94 patients treated intensively for 3.5 years; p = 0.04) and CV mortality (p = 0.02) in ACCORD; this effect was found in all prespecified patient subgroups; (e) alternative clinical strategies (such as the use of fenofibrate or BP control) that may be safer than near-normalization of blood glucose may effectively reduce microvascular complications [TASGaAES Group, 2010]; (f) the recent identification of genetic markers of microvascular complication risk that may enable clinicians to tailor HbA1c targets and other clinical strategies to reduce microvascular complications in especially high-risk patients [Ma et al. 2010]; (g) patient willingness to adopt a clinical strategy that may decrease certain risks, such as progression of albuminuria, while increasing other risks, such as mortality; (h) the high marginal cost per year of life gained from intensive glucose control, which increases with age and which is considerably higher than the cost per year of life gained from moderate levels of lipid or BP control [CDC Diabetes Cost-effectiveness Group, 2002].
It is intriguing that earlier studies that achieved more moderate levels of blood glucose control, including the UKPDS and Steno-2 trials, also achieved significant reductions in both CV or total mortality as well as in end-stage microvascular complications during their active treatment and observational follow-up phases [Gaede et al. 2008; Holman et al. 2008]. The UKPDS trial achieved HbA1c values in the 7.1–7.9% range in the intensively treated arm, and the multifactorial-treatment Steno-2 trial controlled HbA1c to a mean of 7.7% with concomitant aggressive BP and lipid control [Gaede et al. 2008; Holman et al. 2008]. It remains to be seen in patient-level meta-analyses of ACCORD and ADVANCE whether background levels of BP and lipid control modify the effect of near-normalization of glucose on microvascular complications.
In conclusion, the available data from ACCORD and ADVANCE, seen from the perspective of the UKPDS and Steno-2 follow-up studies, suggest that moderation in glucose treatment goals, coupled with a strong emphasis on a broad-based tailored CV risk reduction strategy may be the cornerstones of maximally effective diabetes care.
Footnotes
Acknowledgement
We would like to acknowledge Stephen E. Asche of HealthPartners Research Foundation who provided statistical consultation on the analysis.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
None declared.
