Abstract
A 71-year-old man without previous history of diabetes was hospitalized after suffering polyuria for 1 month and involuntary movement of the left arm for 1 week. His random serum glucose was 42.05 mmol/l and his hemoglobin A1C was 14% (129 mmol/mol). His serum osmolarity was normal and his urine ketone was negative. Cerebral CT revealed hyperdensity in the right basal ganglia. The patient was diagnosed with diabetic hemiballism-hemichorea (HH). Intravenous insulin was given and later shifted to continuous subcutaneous insulin infusion. During the hospital stay, insulin titration was guided mainly by flash glucose monitoring (FGM). Finger-prick glucose was occasionally checked to verify the accuracy of the FGM. Rapid correction of severe hyperglycemia was achieved without hypoglycemia. HH resolved within 1 week after euglycemia was achieved. This case emphasized the importance of being alert for HH as the initial presentation of diabetes and neuroimaging negative diabetic HH. In addition, interstitial glucose-monitoring technologies including continuous glucose monitoring and FGM can facilitate inpatient intensive insulin therapy in diabetic HH by avoiding hypoglycemia.
Keywords
Introduction
Diabetic hemiballism-hemichorea (HH) occurs predominantly in elderly patients with poorly controlled diabetes.1–3 Hypoglycemic therapy is the major treatment and HH resolves within days to weeks after the correction of hyperglycemia in most cases. 4 As previous cases were reported by neurologists, the emphasis has been on the clinical features and neuroimaging. Herein, we focused on how to use continuous subcutaneous insulin infusion (CSII) and the newer interstitial glucose monitoring technology, flash glucose monitoring (FGM), to achieve a rapid and smooth correction of severe hyperglycemia in this situation.
Case presentation
A 71-year-old right-handed man [height: 185 cm; body weight: 87 kg; body mass index (BMI) 25.4 kg/m2] presented at the emergency department following involuntary movement of the left arm for 1 week. He had no history of diabetes but experienced polydipsia, polyuria, and a weight loss of about 10 kg in the previous month. He also reported a 24-year history of well-controlled hypertension and a 4-year history of percutaneous coronary intervention. He was on regular medication for the above two diseases. He had been diagnosed with lung adenocarcinoma 4 years before presentation and had been receiving icotinib (125 mg tid) since then. On neurological examination, muscle tone and strength of extremities were generally normal. No cranial nerve impairment or sensory disturbance was identified. Routine laboratory evaluation revealed random serum glucose up to 42.05 mmol/l with normal serum osmolarity (304.5 mmol/l). The artery gas analysis was normal. His urine glucose was positive (+++) and ketone was negative. His A1C (HbA1c) was 14% (130 mmol/mol). Cerebral CT was arranged instead of MRI because of the metal coronary stent and revealed a hyperdense lesion in the right basal ganglia (Figure 1(a)). After neurology and endocrinology consultation, other etiologies for HH including cerebrovascular condition, infection, and neurodegenerative disorders were excluded. The patient was diagnosed with diabetic HH and referred to the endocrinology department for further therapy.

Neuroimaging of the patient. (a) Cerebral CT revealed a hyperdense lesion in the right basal ganglia (1 week from HH onset). (b) and (c) No abnormal lesion in the right ganglia was found on T1- or T2-weighted MRI (about 2 weeks from HH onset). (d) Follow-up brain CT revealed that the abnormal hyperdense lesion in the right basal ganglia had completely resolved (about 5 weeks from the onset of HH).
Investigation
1.5T cerebral MRI was performed 4 days later after the cardiology consultation on the metal coronary stent. No abnormal lesion in the right ganglia was found on T1- or T2-weighted MRI (Figure 1(b) and (c)). Meanwhile further laboratory tests were arranged for the etiological diagnoses of diabetes. No autoimmune antibodies for diabetes were detected. Cushing’s syndrome was excluded by the normal adrenocorticotropic hormone (ACTH) and cortisol level. Slight elevation of fasting plasma glucose (FPG) (6.21 mmol/l) with normal HbA1c was later found to have been documented 4 years previously before the application of icotinib. No deterioration of the lung cancer was reported in the regular evaluation 2 weeks previously. The patient was diagnosed with type 2 diabetes mellitus (T2DM). In light of the reported hyperglycemia caused by other epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (TKIs)5,6 and the similarity of mechanism, efficacy, and toxicity between icotinib and other EGFR TKIs, 7 the contribution of icotinib could not be fully excluded.
Treatment
Icotinib was continued according to the oncological consultation. Intravenous insulin therapy (0.05–0.1 U/h*kg) was initiated in the emergency department. CSII was applied after correction of the severe hyperglycemia. As cerebral MRI was scheduled for day 4, the patient received capillary glucose monitoring in the beginning to avoid the potential damage of MRI on the FGM. After brain MRI, FGM (FreeStyle Libre 14-day device) was initiated with a 1-day overlap with the finger-prick (day 5).The basal rate and preprandial boluses were titrated mainly according to FGM. Capillary glucose was occasionally checked for discrepancies between interstitial and blood glucose levels in the period of rapid glucose change and at the lower glucose level (⩽3.9 mmol/l). As seen in Figure 2, discrepancies were detected at the lower glucose level. Intensive insulin therapy was shifted to oral hypoglycemic agents when the basal rate was reduced to 6.35 U/24 h with premeal boluses of 7–8 U. After 13 days of inpatient treatment, the patient was discharged with metformin (0.5 g tid) only. The detailed antidiabetic treatment is listed in Table 1. Ambulatory glucose profile indicated an average glucose value of 7.79 mmol/l, a standard deviation of 2.69 mmol/l, and a glucose variability of 34.5% (coefficient of variation (CV) = standard deviation of blood glucose/mean blood glucose × 100%). The glucose management indicator was 6.5%. The highest and lowest glucose levels were 13.9 mmol/l and 3.1 mmol/l, respectively. The time in rage (3.9–10 mmol/l) was 70%.The percentages of values ⩾7.8 mmol/l, ⩾10 mmol/l, and ⩾13.9 mmol/l were 46%, 26%, and 0%, respectively. The percentages of values ⩽3.9 mmol/l and 2.8 mmol/l were 4% and 0%, respectively.

Ambulatory glucose profile discrepancies between finger-prick glucose (red) and interstitial glucose (blue) levels at lower glucose levels (arrow).
Antidiabetic treatment of the patient.
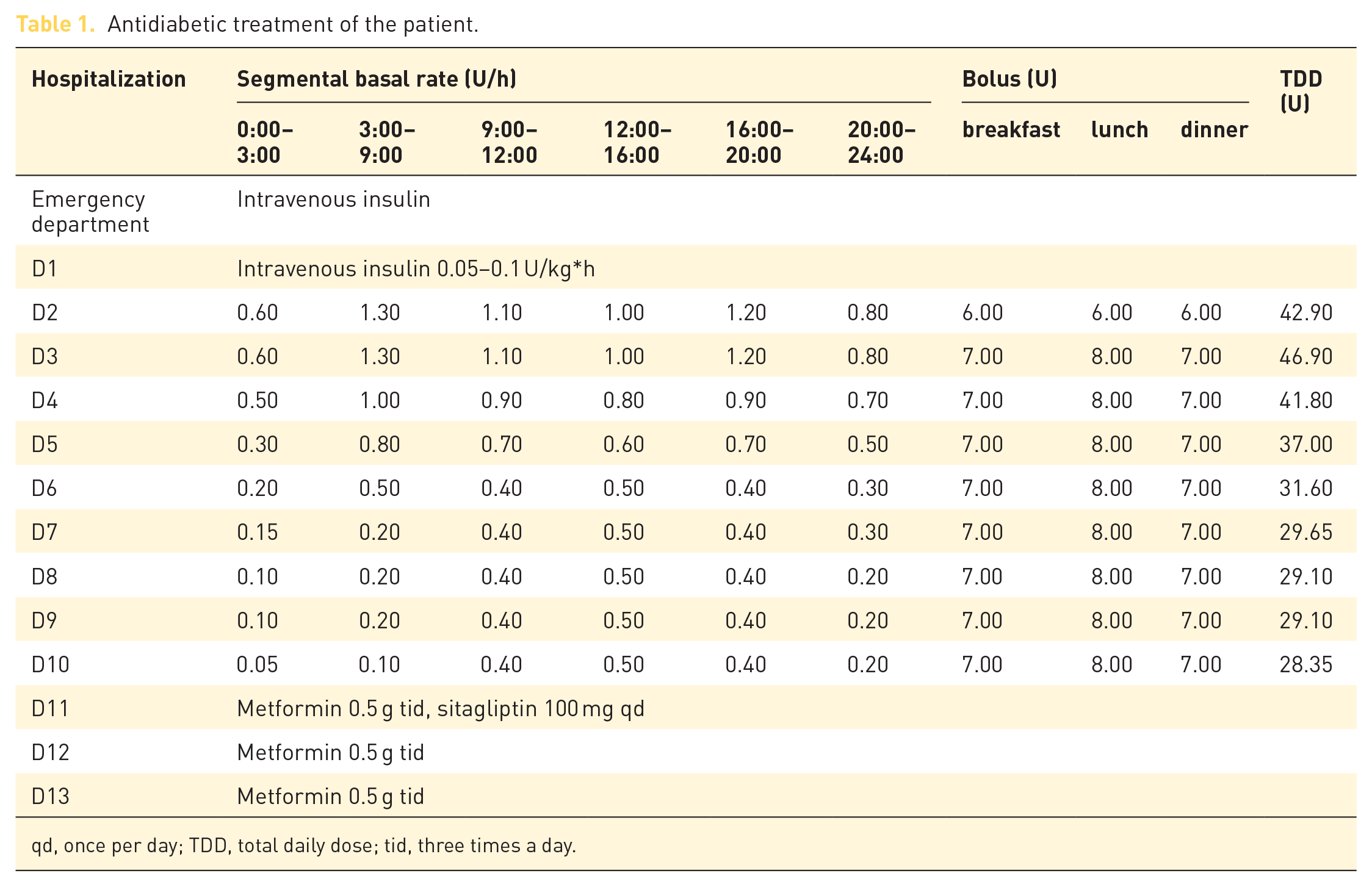
qd, once per day; TDD, total daily dose; tid, three times a day.
Outcome and follow up
HH alleviated on day 3 and resolved on day 5. Two weeks after discharging, the patient reported an FPG of 5.6–7.4 mmol/l and postprandial plasma glucose (PPG) of 8.5–10.4 mmol/l. In addition, the abnormal hyperdense lesion in the right basal ganglia had completely resolved in the cerebral CT (about 5 weeks from HH onset) (Figure 1(d)).
Discussion
Diabetic HH occurs predominantly in elderly patients with diabetes with a reported average age of 72 years. 3 An Asian predominance is apparent while a female predominance is controversial.1–3 Diabetic HH is associated with both T2DM and T1DM, and is usually provoked by an episode of nonketotic hyperglycemia and less frequently by ketosis. 3 Severe hyperglycemia is the most obvious metabolic disorder while plasma osmolarity can be normal. According to a latest systematic review, serum glucose levels [median and interquartile range (IQR)] were 23.3 (17.8–28.3) mmol/l for the nonketotic cases and 29.7 (20.0–43.9 mmol/l) for the ketotic ones. The HbA1c (median and IQR) were 13 (12–15)% [119 (108–140) mmol/mol] and 15 (13–16)% [140 (119–151) mmol/mol] for the nonketotic and ketotic patients, respectively. 3 To be noted, although most patients have a history of poorly controlled diabetes, HH can be the initial presentation of diabetes.
The typical clinical features of diabetic HH are involuntary movement of unilateral extremities with severe hyperglycemia. Bilateral limbs or/and the head are affected in some cases. 8 The typical imaging finding is hyperintense lesions in the basal ganglia on T1-weighted brain MRI and the putamen is most frequently involved.2,9–12 The findings on T2-weighted MRI vary from hyper- and iso- to hypointensity. 3 Hyperdense lesions can also be identified on CT in some cases. To be noted, some cases are imaging-negative, and some bilateral cases are associated with unilateral imaging abnormality. Further, with an average age of over 70 years, some patients are unable to undergo brain MRI because of their metal dentures or coronary stents. The diagnosis should be based on clinical manifestation rather than excessively relying on imaging.
In most diabetic HH cases, movement disorder resolved within days to weeks after the correction of hyperglycemia. 1 Although hypoglycemic treatment is the fundamental treatment for diabetic HH, it has seldom been discussed as previous cases have been reported mainly by neurologists. In fact, multiple daily injections (MDIs) are widely used in diabetic HH treatment and oral antidiabetic drugs are also applied in some cases. Herein, we addressed the rapid and smooth correction of severe hyperglycemia in diabetic HH with novel technologies including the insulin pump and FGM. There is general agreement that one leading barrier to intensive insulin therapy is hypoglycemia. Most patients with diabetic HH are even more vulnerable because of their age and concomitant cardiovascular diseases. It is important to mimic the physiological endogenous insulin secretion of healthy individuals to reduce the risk of hypoglycemia. The latest generation of long-acting insulin analogs, degludec and glargine U300, are better options for patients who are experiencing nocturnal hypoglycemia with glargine and detemir. In addition, the adjustable segmental basal rates during the day and night, different patterns of premeal insulin boluses 13 make CSII clearly superior to MDI, especially for those patients who suffer from hypoglycemia unawareness, frequent episodes of hypoglycemia, or severe hypoglycemia. Another advantage of CSII is correction boluses can be given without additional injections, which reduces pain and inconvenience.
Frequent glucose measurement is a prerequisite for intensive insulin therapy. Capillary glucose measurement is traditionally used during hospitalization but fails to detect fluctuations in nocturnal or asymptomatic hypoglycemia in some cases. Newer technologies including continuous glucose monitoring (CGM) and FGM not only reduce the need for the painful and inconvenient finger-prick test, but also provide more comprehensive data and glucose trend arrow. 14 They facilitate the fine tuning of insulin dosage, reduce hypoglycemia episodes (especially nocturnal hypoglycemia), and increase time in the target range. FGM is preferable in most cases for self-calibration and longer duration, and has demonstrated a 38% and a 43% reduction in hypoglycemia in T1DM and T2DM patients, respectively. 15 However, CGM is more appropriate for those patients without hyperglycemia awareness as it provides alarm signals,. 14 Since CGM and FGM measure glucose level in interstitial fluid instead of in blood, discrepancies occur during periods of rapid glucose change and at extreme glucose levels. 16 A confirmatory finger-stick test is mandatory in these circumstances. The FreeStyle Libre 14-day device is not officially approved as a nonadjunctive device for the mean absolute relative difference (MARD) above 10% and capillary glucose should be checked in some situations including the first 10 h of wearing the sensor. 17
Although in most cases, diabetic HH resolved after glucose correction. Some reported patients with prolonged diabetic HH were prescribed with dopamine receptor blocking agents, neuroleptics, or dopamine deletion agents. 12 In very rare refractory or persistent cases, surgical interventions such as deep brain stimulation are applied. 4 Neurology consultation is needed for differential diagnosis of other etiologies for HH, to assess the severity of HH, and also to provide a more comprehensive therapy.
Conclusion
In summary, this case emphasized the importance of being alert for HH as the initial presentation of diabetes and the neuroimaging of negative diabetic HH. In addition, interstitial glucose monitoring technologies including FGM and CGM can facilitate inpatient intensive insulin therapy to avoid hypoglycemia.
Footnotes
Author contribution(s)
Funding
This research was supported by the National Key R&D Program of China (2018YFC1311500), the National Natural Science Foundation of China (No.81802829), and the Natural Science Basic Research Plan in Shaanxi Province (Grant No. 2020JQ-501).
Conflict of interest statement
The authors declare that there is no conflict of interest.
Informed consent
Informed consent for the publication of clinical data has been obtained from the patient.
