Abstract
Glucagon-like peptide-1 (GLP-1) has been evaluated for use in the treatment of type 2 diabetes mellitus (T2DM) due to its role in glucose regulation. Four GLP-1 receptor agonists (RAs) are currently indicated for T2DM in the USA. Exenatide and liraglutide are short-acting and require twice-daily and daily dosing, respectively. Two longer acting agents, exenatide long-acting release (LAR) and albiglutide, were formulated to allow for once-weekly dosing. All four GLP-1 RAs have demonstrated reductions in hemoglobin A1c, fasting blood glucose, and body weight both as monotherapy and in combination with first- and second-line diabetes agents including metformin, sulfonylureas, thiazolidinediones, and insulin. Greater glycemic control was seen with liraglutide compared with the other GLP-1 treatment options; however, the two long-acting agents were superior to exenatide twice daily. All agents were well tolerated with most adverse events being mild or moderate in nature. The most common adverse event was transient nausea which typically resolved 4–8 weeks after treatment initiation. Long-acting agents had lower rates of nausea but an increased incidence of injection site reactions. Trials have suggested GLP-1 RAs may improve cardiovascular risk factors including blood pressure, lipid parameters and inflammatory markers. Future trials are needed to confirm the clinical outcomes of these agents. Overall, GLP-1 RAs will provide benefit for patients with T2DM intolerable to or not reaching glycemic goals with first-line agents, especially in patients in need of weight loss.
Keywords
Introduction
Glucagon-like peptide 1 (GLP-1) is an endogenous hormone produced and released by intestinal epithelial endocrine L cells in response to food intake [Holst, 2007; Frias and Edelman, 2007; Nauck et al. 2004]. While the effects of GLP-1, including increased insulin secretion, decreased glucagon secretion, slowed gastric emptying, and maintenance of blood glucose (BG) levels, are maintained, GLP-1 secretion is often reduced in patients with type 2 diabetes mellitus (T2DM) [Holst, 2007; Frias and Edelman, 2007; Nauck et al. 2004; Kreymann et al. 1987; Nauck et al. 1986]. When exogenous GLP-1 is administered to patients with T2DM, fasting BG (FBG) is decreased while c-peptide and insulin secretion are increased until BG returns to normal [Nauck et al. 1993; Toft-Nielsen et al. 1999]. This glucose-dependent effect reduces the risk of hypoglycemia.
The use of GLP-1 therapy was limited by its rapid breakdown by dipeptidyl peptidase IV (DPP-IV) [Holst, 2006, 2007]. However, several GLP-1 receptor agonists have been discovered that are DPP-IV resistant, making them valuable in diabetes treatment. Exenatide and liraglutide were US Food and Drug Administration (FDA) approved in 2005 and 2010 and are administered subcutaneously twice daily and daily, respectively [Amylin Pharmaceuticals, Inc., 2011; Novo Nordisk A/S, 2013]. In 2012 and 2014, exenatide long-acting release (LAR) and albiglutide were approved. These agents were modified to increase the half life, allowing for once weekly subcutaneous administration [Amylin Pharmaceuticals, Inc., 2012; GlaxoSmithKline LLC, 2014].
The consensus treatment algorithm from the American Diabetes Association and the European Association for the Study of Diabetes recommends metformin first line for patients diagnosed with T2DM. If metformin does not reach glycemic goals, add-on therapy with either pioglitazone, a sulfonylurea (SU), a DPP-IV inhibitor, a GLP-1 RA, or insulin is recommended. Medication choices should be based on patient-specific conditions considering the effect on glycemic control and body weight, the risk of hypoglycemia and other potential advantages or disadvantages [Inzucchi et al. 2012]. The following sections discuss the efficacy and safety of exenatide, liraglutide, exenatide LAR, and albiglutide and characteristics that may influence their use.
Short-acting GLP-1 RAs
Efficacy
Exenatide 5 and 10 μg twice daily as well as liraglutide 0.6 mg, 1.2 mg, and 1.8 mg daily have been studied as monotherapy and in combination with metformin, SUs, thiazolidinediones (TZDs), and insulin. Liraglutide 0.6 mg showed minimal reduction in glycemic parameters and therefore results from the higher doses are reported here. Results from selected trials are summarized in Table 1.
Summary of clinical trials evaluating exenatide and liraglutide.
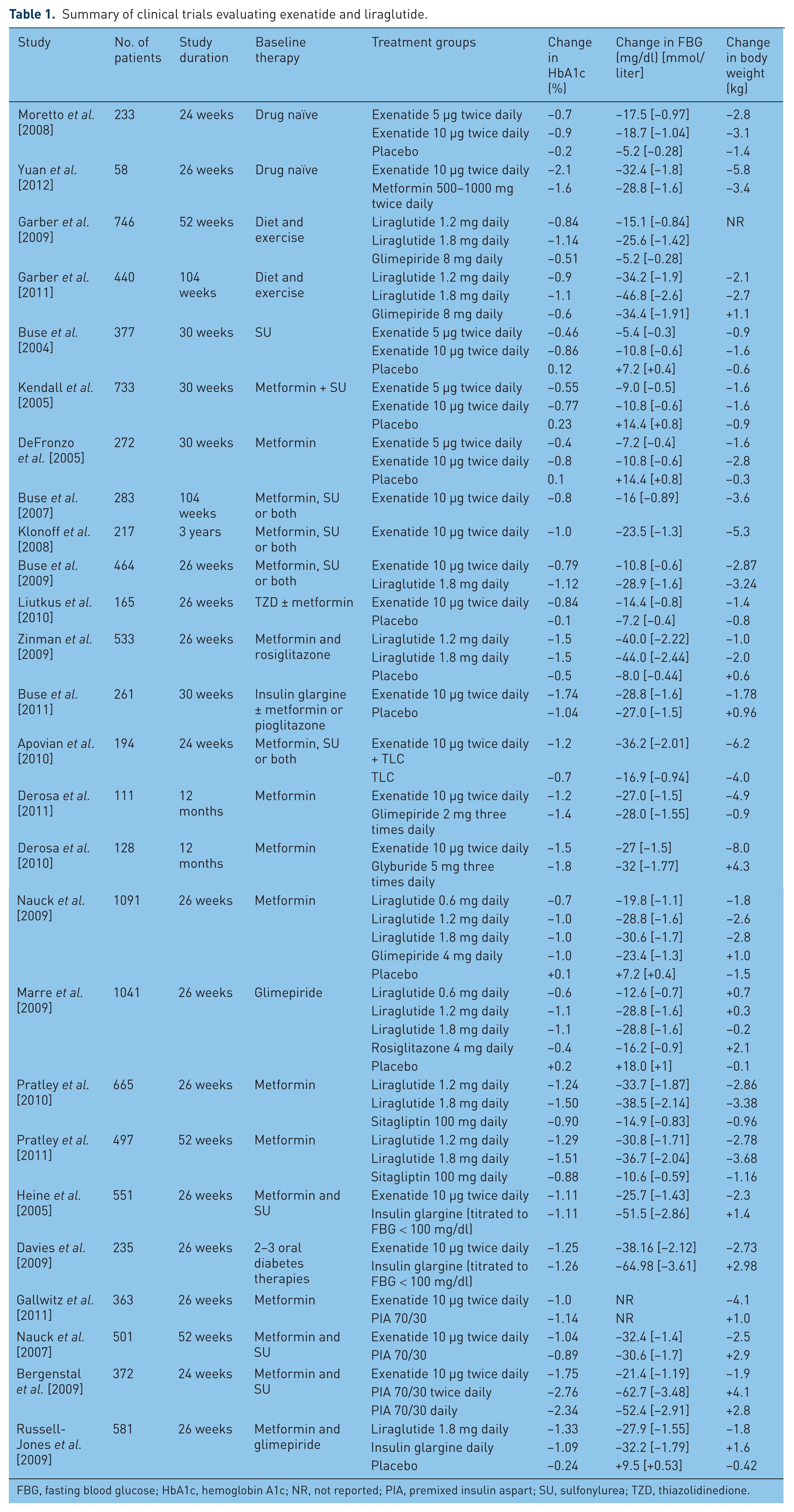
FBG, fasting blood glucose; HbA1c, hemoglobin A1c; NR, not reported; PIA, premixed insulin aspart; SU, sulfonylurea; TZD, thiazolidinedione.
Exenatide monotherapy lowered hemoglobin A1c (HbA1c) by 0.7–0.9%, FBG by 17.5–18.7 mg/dl (0.97–1.03 mmol/liter) postprandial BG (PBG) excursions by 21.3–24.7 mg/dl (1.18–1.37 mmol/liter). In addition, exenatide monotherapy resulted in body weight reductions of 2.8–3.1 kg [Moretto et al. 2008]. In a small study of 58 patients with obesity and newly diagnosed T2DM, exenatide monotherapy compared with metformin monotherapy showed similar reductions in FBG [–32.4 mg/dl (–1.8 mmol/liter)
Three 30-week trials evaluated exenatide added to metformin, a SU, and both. Exenatide 5–10 μg twice daily reduced HbA1c, FBG, and body weight by 0.4–0.86%, 5.4–10.8 mg/dl (0.3–0.6 mmol/liter), and 0.9–2.8 kg, respectively [Buse et al. 2004; Kendall et al. 2005; DeFronzo et al. 2005]. Extension trials demonstrated the durability of exenatide added to metformin or SU therapy with sustained reductions in HbA1c and FBG as well as progressive body weight reductions extending for up to 3 years of therapy [Ratner et al. 2006; Buse et al. 2007; Riddle et al. 2006; Blonde et al. 2006; Klonoff et al. 2008]. When stratified by baseline HbA1c, larger reductions in HbA1c were seen in patients with baseline HbA1c greater than 9% [Buse et al. 2004; Kendall et al. 2005; Riddle et al. 2006; Blonde et al. 2006]. When directly compared, 26 weeks of liraglutide significantly lowered HbA1c (–1.12%
Exenatide and liraglutide have recently been approved for use in combination with long-acting insulin. Two retrospective trials showed a significant decrease in both HbA1c (–0.5% to −0.7%) and body weight (–2.5 to −5.8 kg) when exenatide was added to insulin therapy. Changes in diabetes treatments varied, with one study showing significant decreases in insulin doses (–0.3 U/kg) as well as the proportion of patients taking metformin and SUs while the other showed increases in insulin doses (0.1 U/kg) and no change in oral diabetic therapies [Thong et al. 2011; Levin et al. 2012]. In a randomized, controlled trial including 261 patients, exenatide significantly reduced HbA1c (–1.74
When compared with oral, add-on therapies, exenatide and liraglutide continued to demonstrate benefit on glycemic control. In combination with metformin, a SU or both, exenatide plus intensive lifestyle modifications compared with lifestyle modifications alone significantly reduced HbA1c (–1.2
Both exenatide and liraglutide have been compared with basal insulin as an add on to oral therapy. When added to metformin and a SU in 551 patients, exenatide and insulin glargine reduced HbA1c comparably (–1.11% in both groups) over 1 year. Insulin glargine significantly lowered FBG [–51.5
Safety
Overall, side effects were minor and rarely required discontinuation of therapy. The primary side effect of both exenatide and liraglutide was nausea, which occurred in 8–51% and 10.5–45% of patients in trials, respectively [Moretto et al. 2008; Yuan et al. 2012; Buse et al. 2004, 2007; Kendall et al. 2005; DeFronzo et al. 2005; Ratner et al. 2006; Riddle et al. 2006; Liutkus et al. 2010; Zinman et al. 2007, 2009; Garber et al. 2009]. Nausea tended to be transient following treatment initiation and dose escalation, resolving over the first 8 weeks for exenatide and 4 weeks for liraglutide [Yuan et al. 2012; Buse et al. 2004, 2007 ; Kendall et al. 2005; DeFronzo et al. 2005; Ratner et al. 2006; Riddle et al. 2006; Zinman et al. 2007, 2009; Garber et al. 2009; Marre et al. 2009]. Nausea was less persistent in patients receiving liraglutide compared with exenatide [Buse et al. 2009].
Rates of hypoglycemia were relatively low, occurring in 4–12% of patients receiving exenatide and 3–12% of patients receiving liraglutide without concurrent SU [Moretto et al. 2008; Yuan et al. 2012; DeFronzo et al. 2005; Ratner et al. 2006; Buse et al. 2007; Liutkus et al. 2010; Zinman et al. 2007; Garber et al. 2009; Nauck et al. 2009]. Incidence of hypoglycemia with both therapies was higher in patients receiving concurrent SU therapy [Buse et al. 2004, 2009; Kendall et al. 2005; Apovian et al. 2010]. Compared with insulin, exenatide and liraglutide had lower or similar rates of overall hypoglycemia and reduced nocturnal hypoglycemia [Heine et al. 2005; Barnett et al. 2007; Davies et al. 2009; Gallwitz et al. 2011; Bergenstal et al. 2009; Russell-Jones et al. 2009].
Postmarketing reports of pancreatitis resulted in a black box warning (BBW) for both exenatide and liraglutide [Amylin Pharmaceuticals, Inc., 2011; Novo Nordisk A/S, 2013]. Four patients have reported pancreatitis with liraglutide in clinical trials; however, all recovered and two continued in the study [Garber et al. 2009; Nauck et al. 2009; Marre et al. 2009]. One patient receiving liraglutide was diagnosed with pancreatic carcinoma on day 11 of therapy; however, this was considered to be unrelated to liraglutide [Pratley et al. 2010]. Liraglutide also carries a black box warning for thyroid C-cell tumors due to occurrence in rat and mice studies. Carcinoma has not been seen in clinical trials but caution should be used in patients with a personal or family history of medullary thyroid carcinoma [Novo Nordisk A/S, 2013].
Development of antibodies occurs with both exenatide (27–49%) and liraglutide therapy (4.1–13%); however, the presence of antibodies has not affected clinical results [Moretto et al. 2008; Buse et al. 2004; Kendall et al. 2005; DeFronzo et al. 2005; Zinman et al. 2007, 2009; Marre et al. 2009; Russell-Jones et al. 2009]. The safety and tolerability of exenatide has been consistent across all nationalities [Lu et al. 2013; Gao et al. 2009; Kadowaki et al. 2009; Heine et al. 2005; Yang et al. 2011].
Long-acting GLP-1 RAs
Efficacy
Exenatide LAR 2 mg and albiglutide 30 and 50 mg are both dosed once weekly and have been studied as monotherapy, in combination with OAMs, and have been compared with various conventional diabetes treatment options [Amylin Pharmaceuticals, Inc., 2012; GlaxoSmithKline LLC, 2014].
Exenatide LAR monotherapy was compared with metformin, pioglitazone, and sitagliptin monotherapy in a 26-week study. HbA1c was reduced by −1.53% with exenatide LAR, –1.48% with metformin, –1.63% with pioglitazone, and −1.15% with sitagliptin from an average baseline HbA1c of 8.4–8.6%. Exenatide LAR was noninferior to metformin and superior to sitagliptin (
Add-on therapy with exenatide LAR once weekly was evaluated against sitagliptin or pioglitazone in patients nonresponsive to metformin monotherapy in a 26-week randomized, double-blind trial. The baseline average HbA1c of 8.5% was reduced to 7.2% in the exenatide LAR treatment arm, 7.7% in the sitagliptin treatment arm, and 7.4% in the pioglitazone treatment arm. Reductions in HbA1c were significantly greater with exenatide LAR than sitagliptin and pioglitazone (
In a 104-week randomized, placebo and active controlled trial, albiglutide was compared with placebo, sitagliptin, and glimepiride in patients inadequately controlled on metformin. Average baseline HbA1c was 8.1%. Albiglutide provided a significantly greater reduction in HbA1c (–0.63%) than all of the other treatment arms (–0.27% with placebo, –0.28% with sitagliptin, and −0.36% with glimepiride;
Exenatide LAR was compared with insulin glargine in patients with an insufficient response to metformin with or without a SU. After 26 weeks, HbA1c was significantly reduced with exenatide LAR compared with insulin glargine (–1.5
Safety
Over the 3-year follow up of the DURATION-1 study comparing exenatide LAR once weekly with exenatide twice daily, most adverse events with exenatide LAR were mild to moderate and included nausea, vomiting, diarrhea, upper respiratory tract infection, nasopharyngitis, and sinusitis [MacConell et al. 2013]. The incidences of nausea, vomiting, and diarrhea were highest during the first 12 weeks of exenatide LAR use and decreased to 1% during the last 12 weeks [MacConell et al. 2013]. Hypoglycemia was minor and occurred more often in patients on concomitant SU therapy than patients not treated with SUs (19.3%
There were no reported episodes of pancreatitis, pancreatic cancer, or thyroid cancer during the 3-year DURATION-1 study [MacConell et al. 2013]. Asymptomatic increases in pancreatic enzymes were observed in five subjects in the exenatide LAR group compared with none in the insulin glargine group at the conclusion of the DURATION-3 trial [Diamant et al. 2010].
Safety of exenatide LAR was studied as a primary outcome in a 26-week extension trial conducted by Onishi and colleagues [Onishi et al. 2013]. Patients from Japan originally enrolled in a 26-week study comparing the safety and efficacy of twice-daily exenatide with once-weekly exenatide LAR in an Asian population were enrolled. Patients receiving twice-daily exenatide were switched to once-weekly exenatide LAR and patients receiving exenatide LAR continued once-weekly dosing [Onishi et al. 2013]. Researchers noted that of the patients initially on twice-daily exenatide, the majority of adverse events that led to study discontinuation occurred during the first 26 weeks before the patients were switched to once-weekly exenatide LAR. Typically, adverse events such as nausea, vomiting, diarrhea, and minor hypoglycemia decreased when patients were switched from twice-daily exenatide to once-weekly exenatide LAR. Conversely, the frequency of injection-site reactions increased in patients switching from twice-daily dosing to once-weekly dosing. Adverse event rates remained consistent in patients continuing on once-weekly exenatide LAR dosing. These findings are consistent with differences noted between the safety profiles of twice-daily exenatide
Similar rates of adverse events have been reported in trials comparing albiglutide with conventional therapy. Nausea, diarrhea, and injection site reactions continue to be the most reported adverse events in the albiglutide treatment groups, with no increased occurrence of serious adverse events when matched to comparator treatment arms [Ahren et al. 2014; Pratley et al. 2014].
Comparison of short- and long-acting agents
Exenatide LAR and liraglutide were compared head to head in a 26-week study. Both agents provided clinically significant lowering in HbA1c; however, liraglutide had a statistically greater reduction in HbA1c than exenatide LAR (–1.48%
Albiglutide was also compared with liraglutide in patients with T2DM inadequately controlled on metformin, TZD, SU, or a combination of the three in a 32-week noninferiority trial. Albiglutide lowered HbA1c but did not meet noninferiority criteria compared with liraglutide (
DURATION-1, a 30-week randomized, noninferiority study, compared exenatide LAR 2 mg once weekly with exenatide 10 μg twice daily in treatment-naïve patients and patients on one or more OAMs. Although both treatment groups had significant reductions in HbA1c, greater reductions were observed in the exenatide LAR group (–1.9
Once-weekly exenatide LAR was shown to be superior to twice-daily exenatide in a 24-week open-label study in patients who were either drug naïve or previously treated with one or more OAMs and had an average HbA1c of 8.5%. The average change in HbA1c was −1.6 in the once-weekly exenatide LAR arm and −0.9 in the twice-daily exenatide arm (
Ji and colleagues conducted a similar study in an Asian population. Patients were randomized to exenatide 10 μg twice daily or exenatide LAR 2 mg weekly. Again, exenatide LAR significantly lowered HbA1c compared with exenatide twice daily (–1.43
Additional benefits of GLP-1 therapy
Although results were mixed, the secondary outcomes of initial trials with exenatide and liraglutide suggested a potential benefit for cardiovascular risk factors. Exenatide 5–10 μg twice daily decreased systolic BP (SBP) by 3.5–9.4 mmHg and diastolic BP (DBP) by 0.8–4.1 mmHg [Moretto et al. 2008; Ratner et al. 2006; Buse et al. 2007; Liutkus et al. 2010; Apovian et al. 2010; Klonoff et al 2008]. A placebo-controlled trial randomized 54 patients controlled on metformin, a TZD, or both to receive exenatide 10 μg twice daily to specifically evaluate its effects on heart rate (HR) and BP. Exenatide showed no significant effect on HR or BP, although SBP was lowered by 5.6 mmHg compared with placebo [Gill et al. 2010]. Liraglutide 1.2–1.8 mg daily decreased SBP by 2.0–5.6 mmHg but tended to have no significant effect on DBP [Garber et al. 2009; Zinman et al. 2009; Marre et al. 2009; Russell-Jones et al. 2009]. Liraglutide 1.8 mg daily significantly reduced SBP compared with glimepiride [Garber et al. 2009; Nauck et al. 2009]. Exenatide LAR has also shown significant reductions in SBP and DBP [MacConell et al. 2013; Davies et al. 2013]. Some studies have seen significant improvement in low-density lipoprotein (LDL) [Buse et al. 2004; Klonoff et al. 2008; Zinman et al. 2009], triglycerides (TGs) [Ratner et al. 2006; Klonoff et al. 2008; Zinman et al. 2009], high-density lipoprotein (HDL) [Ratner et al. 2006; Klonoff et al. 2008], and apolipoprotein B (ApoB) [Buse et al. 2004; Ratner et al. 2006] with exenatide and liraglutide but others found no significant effects on lipids [Moretto et al. 2008; DeFronzo et al. 2005; Liutkus et al. 2010].
Initial studies of both exenatide and liraglutide found improvements in β-cell function and insulin sensitivity based on Homeostasis Model Assessment HOMA-B [Moretto et al. 2008; Buse et al. 2007; Liutkus et al. 2010; Zinman et al. 2007; Klonoff et al. 2008; Derosa et al. 2012; Zinman et al. 2009; Nauck et al. 2009; Marre et al. 2009; Pratley et al. 2010] and HOMA-S values [Buse et al. 2007] as monotherapy and in combination with metformin, SUs, or TZDs. Compared with exenatide, liraglutide had significantly greater improvements in β-cell function [Buse et al. 2009]. Compared with SUs, exenatide and liraglutide demonstrated significant improvements in insulin resistance (based on HOMA-IR) [Derosa et al. 2011; Garber et al. 2009, 2011] and β-cell function (based on HOMA-B) [Derosa et al. 2010] whereas therapy with SUs did not [Derosa et al. 2010, 2011; Garber et al. 2009]. In a 3-year study, exenatide significantly improved β-cell function based on disposition index. When exenatide was discontinued for 4 weeks after 1 year, β-cell function returned to baseline; however, after 3 years of therapy β-cell function remained elevated after exenatide discontinuation, suggesting long-term treatment may result in longstanding effects on β-cell function [Bunck et al. 2011].
Early studies of exenatide found evidence to suggest exenatide may decrease liver enzymes, aspartate aminotransferase and alanine aminotransferase (ALT), especially in patients with elevated baseline levels [Buse et al. 2007; Klonoff et al. 2008], which may suggest benefits for patients with liver impairment. In a study of 24 patients with T2DM on diet or metformin therapy, hepatic fat content was significantly reduced in patients randomized to receive a combination of pioglitazone and exenatide compared with pioglitazone alone (–4.5%
Secondary endpoints of early trials suggested significant improvement in inflammatory markers, including tumor necrosis factor α and CRP with exenatide twice daily and once-weekly therapy [Derosa et al. 2010, 2011; Chiquette et al. 2012; Davies et al. 2013]. A 16-week study also found a decrease in CRP with exenatide compared with placebo (–0.2 mg/liter
GLP-1 RAs in development
Four additional GLP-1 RAs, taspoglutide, lixisenatide, dulaglutide, and semaglutide are currently in development. Add-on therapy with taspoglutide 10 mg and 20 mg once weekly was compared with TZDs and basal insulin in patients whose condition was uncontrolled on metformin with or without a SU [Nauck et al. 2013; Pratley et al. 2013]. Taspoglutide 20 mg weekly was found to be noninferior to pioglitazone and insulin glargine, with average reduction in HbA1c of −1.36 and −0.98, respectively [Nauck et al. 2013; Pratley et al. 2013]. Compared with exenatide twice daily and sitagliptin in patients whose condition is inadequately controlled on metformin with or without a TZD, taspoglutide 20 mg weekly reduced HbA1c and FBG to a significantly greater extent (
Lixisenatide is available in Europe, Mexico, Australia, and Japan as the brand name Lyxumia; however, the US FDA new drug application was recently withdrawn by the manufacturer [Sanofi, 2013, Bridgewater, NJ]. Lixisenatide had positive results in HbA1c, FBG, and weight reductions compared with placebo in patients whose condition was uncontrolled on metformin with or without a SU [Bolli et al. 2014; Rosenstock et al. 2014]. The manufacturer, Sanofi, plans to resubmit the application to the US FDA after the current cardiovascular outcomes trial, ELIXA, has concluded [Sanofi, 2013].
Dulaglutide has been studied against placebo and conventional therapy options in several clinical trials. Dulaglutide 1.5 mg and 0.75 mg weekly provided significant reductions in HbA1c over 26 weeks compared with placebo. Dulaglutide monotherapy was also compared with metformin monotherapy and was found to be noninferior to metformin for HbA1c lowering, with no differences in side-effect profile and weight reduction. Dulaglutide was also shown to be noninferior to sitagliptin in HbA1c reduction from baseline to 52 weeks [Lilly, 2013]. Several trials are still underway comparing dulaglutide with insulin glargine and evaluating cardiovascular outcomes with dulaglutide use.
Semaglutide is an investigational once-weekly GLP-1 RA. In a dose-finding, phase II trial, semaglutide 0.8 mg and 1.6 mg once weekly produced greater reductions in HbA1c at 12 weeks than liraglutide 1.2 mg and 1.8 mg daily and greater weight reductions compared with placebo. Participant withdrawals were related to gastrointestinal adverse events [Nauck et al. 2012]. A phase II pharmacokinetic study in subjects with or without renal impairment suggested dose adjustment may not be needed for patients with renal insufficiency, although more studies are needed [Marbury et al. 2014]. Semaglutide is currently undergoing phase III trials.
Place in therapy
All four FDA-approved GLP-1 RAs, exenatide, liraglutide, exenatide LAR, and albiglutide, have demonstrated efficacy in regard to glycemic control with significant reductions in HbA1c, FBG, and PBG both as monotherapy and in combination with common first- and second-line agents. HbA1c reductions ranged from 0.5% to 1.8%, indicating GLP-1 RAs are most appropriate in patients with an HbA1c less than 9% in order to reach HbA1c targets. Long-acting GLP-1 RAs seem to be superior in glycemic control compared with exenatide but not compared with liraglutide. Only exenatide and liraglutide are currently approved and have been shown to provide additional lowering of PBG for use in combination with long-acting insulin. Long-acting GLP-1 RAs still require additional studies to determine if benefit is seen. Body weight reductions were also significant with all GLP-1 RAs, which may make them a superior option for patients who are overweight or obese compared with treatments which may increase weight, including SUs, TZDs, and insulin. In general, GLP-1 RAs were well tolerated with most adverse effects being mild and gastrointestinal in nature. Nausea was the most common adverse effect and tended to resolve after the first weeks of treatment. Incidence of nausea was lower and less persistent in longer-acting agents. All agents carry a warning for the potential to cause pancreatitis. In patients with a history of pancreatitis, other options may be more prudent. Liraglutide, exenatide LAR, and albiglutide all carry a BBW for C-cell thyroid tumors. These agents should not be used in patients with a personal or family history of medullary thyroid carcinoma. Given the increased risk of cardiovascular disease with diabetes, the effects of medications on CV risk factors should be considered. While studies have suggested potential benefits on BP, lipids, and inflammatory markers with GLP-1 RAs, larger trials are needed to determine the clinical relevance of these effects.
Conclusion
GLP-1 RAs provide improved glycemic control and weight reduction along with a good safety profile. The addition of GLP-1 RAs may be considered in patients not tolerating first-line medications, patients receiving metformin, a SU, a TZD, or insulin therapy and not meeting glycemic goals, or patients in need of weight loss.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
