Abstract
Glucagon-like polypeptide (GLP-1) receptor agonist treatment has multiple effects on glucose metabolism, supports the β cell, and promotes weight loss. There are now five GLP-1 agonists in clinical use with more in development. GLP-1 treatment typically can induce a lowering of hemoglobin A1c (HbA1c) of 0.5–1.5% over time with weight loss of 2–5%. In some individuals, a progressive loss of weight occurs. There is evidence that GLP-1 therapy opposes the loss of β cells which is a feature of type 2 diabetes. The chief downside of GLP-1 treatment is the gastrointestinal motility disturbance which is one of the modes of action of the hormone; significant nausea, vomiting, and diarrhea may lead to discontinuation of treatment. Although daily injection of GLP-1 agents is successful, the development of extended release preparations allows for injection once weekly, and perhaps much longer in the future. The indication for GLP-1 use is diabetes, but now, liraglutide has been approved for primary treatment of obesity. When oral agents fail to control glucose levels in type 2 diabetes, there is a choice between long-acting insulin and GLP-1 agonists as additional treatments. The lowering of HbA1c by either modality is equivalent in most studies. Patients lose weight with GLP-1 treatment and gain weight on insulin. There is a lower incidence of hypoglycemia with GLP-1 therapy but a much higher incidence of gastrointestinal complaints. Insulin dosing is flexible while GLP-1 agents have historically been administered at fixed dosages. Now, the use of combined long-acting insulin and GLP-1 agonists is promising a major therapeutic change. Combined therapy takes advantage of the benefits of both insulin and GLP-1 agents. Furthermore, direct admixture of both in the same syringe will permit flexible dosing, improvement of glucose levels, and reduction of both hypoglycemia and gastrointestinal side effects.
Introduction
Incretins are hormones secreted by the digestive system in response to oral nutrient intake. Incretin action results in an augmented insulin secretion over and above the response solely attributable to the rise in plasma glucose concentration after a meal [Drucker, 2006]. Glucagon-like polypeptide (GLP-1) is the incretin hormone with the greatest effects on glucose metabolism. It is a 37-amino-acid peptide secreted from the L cells of the ileum and colon into the blood stream. GLP-1 is rapidly degraded in the circulation by the enzyme dipeptidyl dipeptidase 4 (DPP-4) [Nauck et al. 2011]. GLP-1 produces a glucose-dependent increase in insulin secretion by the β cell. Other significant effects of GLP-1 include suppression of glucagon secretion, slowing of gastric emptying time and promotion of satiety [Drucker, 2006]. GLP-1 also stimulates differentiation and proliferation of β cells and inhibits apoptosis [Gautier et al. 2005]. Although postprandial GLP-1 levels are equivalent in patients with type 2 diabetes and controls, the insulinotropic effect of GLP-1 is blunted in diabetes [Nauck et al. 2011]. Several studies have now shown that GLP-1 can lower glucose levels even in patients with severe β-cell impairment, presumably as a result of lowered glucagon levels and other noninsulin secretory mechanisms [Holst et al. 2011].
The physiologic properties of GLP-1 offer possible benefits which go beyond augmenting insulin levels with sulfonylureas or injected insulin or the reduction of insulin resistance by metformin and thiazolidinediones. GLP-1 effects can be provided therapeutically either by giving supplemental GLP-1 agonists to raise serum levels, or by slowing degradation of endogenous GLP-1 with inhibitors of the DPP-4 enzyme. Five GLP-1 analogues with resistance to DPP-4 degradation have been developed and are now in clinical use. They include exenatide, liraglutide, dulaglutide, lixisenatide, and albiglutide, with several more in development [Neumiller and Campbell, 2009; Norris et al. 2009; Petersen and Christensen, 2013; Trujillo and Nuffer, 2014; Amblee, 2014].
The GLP-1 agonists have many benefits. At the same time, their use is hindered by gastrointestinal intolerance due to motility changes. Nausea, vomiting, and diarrhea occur with initial use and usually diminish with continued treatment, but may force discontinuance of GLP-1 therapy in some individuals. The purpose of this review is to address the efficacy, tolerability, and safety of GLP-1 receptor agonists, to distinguish potential differences among the agents now available, and to evaluate the optimal use of these agents.
Chemistry, pharmacokinetics and pharmacodynamics
Exenatide
Exenatide is a 39-amino-acid peptide which has a 53% sequence identity to native GLP-1. Following subcutaneous administration, peak plasma concentration is usually reached in 2–3 h with plasma concentrations remaining elevated 4–8 h following a single subcutaneous injection [Nielsen et al. 2004]. No significant drug interactions were noted with acetaminophen, lovastatin, lisinopril, and warfarin when used concomitantly. Exenatide is not recommended for use in severe renal impairment (creatinine clearance < 30 ml/min). Due to its short half life, exenatide must be administered twice daily at a dose of either 5 or 10 µg.
Exenatide given twice daily demonstrated better glycemic control [both baseline and fasting plasma glucose (FPG) levels] than placebo in treatment-naive patients and patients inadequately controlled with oral antihyperglycemic agents as well as basal insulin [DeFronzo et al. 2010; DeRosa et al. 2010, 2011; Gallwitz et al. 2012] (Table 1). Exenatide treatment gave hemoglobin A1c (HbA1c) and FPG reductions similar to those with sulfonylureas and thiazolidinediones, but led to significant weight loss (p < 0.001), in contrast to these oral agents which were generally associated with weight gain [Gallwitz et al. 2012; DeFronzo et al. 2010]. The effect of exenatide is manifested more in reduction of postprandial blood glucose (PPG) than fasting glucose. In various studies lasting −52 weeks, there has been a significant loss of weight ranging from 1 to 4 kg compared with placebo. The tolerability of exenatide treatment is constrained by the effects on gastrointestinal motility. Nausea occurs in 30–50% of patients on initial treatment, with vomiting in up to 18%. Diarrhea occurs as well in about 10–15% of the patients. These undesirable effects usually diminish in the first few months of treatment but can be sufficiently severe to lead to discontinuation of therapy.
Summary of exenatide twice daily studies.

Each study is listed by first author. The comparative treatments in each study are listed along with the baseline therapy in the population studied and the duration of the study. The mean change in hemoglobin A1c (HbA1c), mean weight loss, and adverse events, chiefly gastrointestinal, are listed for the comparative treatments.
Exenatide is now available in an extended release microspheres formulation (exenatide ER). Exenatide ER consists of biodegradable polyglactin microspheres that are designed to release exenatide over an extended period of time. The extended release preparation given once weekly produces a relatively constant basal serum level resulting in about a 50% lower incidence of gastrointestinal (GI) side effects than the twice daily treatment. The benefits in terms of lowering of HbA1c and weight loss appear to be greater than with the daily preparation (Table 2). The exenatide ER preparation is more immunogenic than short-acting exenatide. Anti-exenatide antibodies were measured at prespecified intervals (4–14 weeks) in all exenatide ER treated patients (N = 918) in five comparator-controlled studies. In these five trials, 452 exenatide ER treated patients (49%) had low titer antibodies to exenatide at any time during the trials and 405 exenatide ER treated patients (45%) had low titer antibodies at study endpoint (24–30 weeks) (Bydureon package insert, Astra Zeneca, London: https://www.bydureon.com). The level of glycemic control in these patients was generally comparable to that observed in 379 exenatide ER treated patients (43%) without antibody titers. Higher titer antibodies were noted in 107 exenatide ER treated patients (12%) at endpoint. Of these patients, 50 (6% overall) had an attenuated glycemic response to the agent (<0.7% reduction in HbA1c). In the five comparator-controlled 24- to 30-week trials, injection site reactions were observed more frequently in patients treated with the ER formulation (17.1%) than in patients treated with daily exenatide (12.7%), titrated insulin glargine (1.8%), or those patients who received placebo injections [sitagliptin (10.6%), pioglitazone (6.4%), and metformin (13.0%) treatment groups]. These reactions for exenatide ER treated patients were more commonly observed in antibody-positive patients (14.2%) compared with antibody-negative patients (3.1%). One percent of patients treated with exenatide ER withdrew due to injection site adverse events (injection site mass, injection site nodule, injection site pruritus, and injection site reaction). In a separate 15-week study in which information on nodules was collected and analyzed, 24 out of 31 subjects (77%) experienced at least one injection-site nodule during treatment; 2 subjects (6.5%) reported accompanying localized symptoms.
Summary of exenatide extended release (ER) studies: the pivotal studies were termed the DURATION series.

The Exenatide ER studies were grouped in a series named “DURATION”. Each study is listed by first author. The comparative treatments in each study are listed along with the baseline therapy in the population studied and the duration of the study. The mean change in hemoglobin A1c (HbA1c), mean weight loss, and adverse events, chiefly gastrointestinal, are listed for the comparative treatments.
In addition to demonstrating improved glucose control, exenatide ER therapy was associated with a mean weight loss of 2–4 kg over 6 months, which was sustained in 66% of patients at 3 years. In the randomized, controlled trials, weight loss with exenatide ER treatment was similar to that of exenatide twice daily or metformin; significantly greater than that of sitagliptin, pioglitazone, or insulin glargine (glargine); and slightly but significantly less than that of liraglutide [Bergenstal et al. 2010; Blevins et al. 2011; Drucker et al. 2008; Russell-Jones, 2012]. Exenatide ER has been shown to improve blood pressure and lipid levels in addition to HbA1c and body weight. Mean systolic blood pressure reductions of 3–5 mmHg compared with pretreatment values were demonstrated in pooled extension data at 52 weeks [Bergenstal et al. 2010]. The systolic blood pressure reductions correlated weakly with weight loss, and the greatest reductions (–11.4 mmHg) were observed in patients with a high baseline systolic blood pressure. Patients who continued exenatide ER therapy for 3 years (n = 194) exhibited sustained reductions in systolic blood pressure (–2 mm Hg), triglycerides (–12%), total cholesterol (–9.9%), and low-density lipoprotein (LDL) cholesterol (–7.0%) [MacConell et al. 2013].
Liraglutide
Liraglutide is an acylated GLP-1 agonist, with a 97% amino acid sequence identity to endogenous human GLP-1. The mean peak (Cmax) for a subcutaneous single dose of 0.6 mg is achieved in 8–12 h post dosing, making it suitable for once daily administration [Meece, 2009]. The Cmax and area under the curve (AUC) increase proportionately over the therapeutic dose range of 0.6–1.8 mg. Approximately 99% of the liraglutide molecules are typically bound to plasma albumin, and the bound molecule has a half life of 11–13 h. Liraglutide is endogenously metabolized; only 5–6% of liraglutide-related metabolites are found in feces and urine. Although there is little renal excretion, experience with the use of liraglutide in patients with renal failure is limited. No significant drug interactions were noted when liraglutide was concomitantly administered with acetaminophen or atorvastatin, but there is a slight delay in Tmax of digoxin and lisinopril. Liraglutide treatment at the highest dose compared with placebo and to several oral hypoglycemics has resulted in reductions in HbA1c of about 1%, with weight loss of 2–3 kg over 26 weeks [Marre et al. 2009; Nauck et al. 2009; Garber et al. 2009; Zinman et al. 2009] (Table 3). Liraglutide is less immunogenic than exenatide, causing low levels of antibody formation [Buse et al. 2011b]. Injection site reactions have been reported in approximately 2% of patients receiving liraglutide in long-term (26 weeks or longer) controlled trials. These reactions have usually been mild [European Medicines Agency, 2009].
Summary of liraglutide studies: the pivotal studies were termed the LEAD series. The dosages of liraglutide were 0.6 mg, 1.2 mg, and 1.8 mg.

The Liraglutide studies were grouped in a series named “LEAD”. Each study is listed by first author. The comparative treatments in each study are listed along with the baseline therapy in the population studied and the duration of the study. The mean change in hemoglobin A1c (HbA1c), mean weight loss, and adverse events, chiefly gastrointestinal, are listed for the comparative treatments. HbA1c, hemoglobin A1c.
Dulaglutide
Dulaglutide, a biosynthetic fusion protein molecule, is a newly approved long-acting, once weekly GLP-1 receptor agonist with an N-terminal GLP-l receptor agonist sequence protected from DPP-4 and linked covalently to a human immunoglobulin G4-Fc heavy chain [US Food and Drug Administration, 2014; Jimenez-Solem et al. 2010]. Following subcutaneous administration, peak plasma concentration is usually reached in 24–72 h with a median of 48 h. The mean bioavailability of the drug is 65% for the 0.75 mg and 47% for the 1.5 mg dose respectively. Dulaglutide undergoes general protein catabolism with mean elimination half life of approximately 5 days. No significant drug interactions were noted with acetaminophen, lovastatin, lisinopril, and warfarin when used concomitantly. No dose adjustment is recommended in renal and hepatic impairment [Eli Lilly and Company, 2014].
The efficacy of once weekly dulaglutide was tested as a monotherapy and as a combination therapy in a series of trials [Assessment of Weekly AdministRation of Dulaglutide (AWARD)] [Dungan et al. 2014; Giorgino et al. 2014; Jendle et al. 2014; Nauck et al. 2014; Umpierrez et al. 2014; Wysham et al. 2014] (Table 4). In these trials, dulaglutide showed superiority to placebo and to several oral hypoglycemic agents. In a head-to-head comparison study (AWARD-3) with metformin, the mean reductions in HbA1c at the end of 26 weeks were 0.71%, 0.78%, and 0.56% for dulaglutide 0.75 mg, 1.5 mg and metformin, respectively (p < 0.025 for both dulaglutide groups versus metformin) [Umpierrez et al. 2014]. Dulaglutide treatment was also found to be superior in lowering HbA1c compared with sitagliptin (p < 0.001) in AWARD-5 [Nauck et al. 2014] and exenatide 10 µg twice daily in AWARD-1 [Wysham et al. 2014] in patients who were on maximum tolerated doses of metformin and pioglitazone. Dulaglutide was noninferior to liraglutide (AWARD-6) in lowering HbA1c and achieving target HbA1c less than 7% [Dungan et al. 2014]. Across four phase II and five phase III clinical studies, 64 (1.6%) dulaglutide-treated patients developed antidrug antibodies. Of these patients, 34 patients (0.9% of the overall population) had dulaglutide-neutralizing antibodies, and 36 patients (0.9% of the overall population) developed antibodies against native GLP-1 [Eli Lilly and Company, 2014]. The formation of antibodies did not reduce the glucose-lowering effect. Injection site reactions (injection site rash, erythema) were reported in 0.5% of dulaglutide-treated patients.
Summary of dulaglutide studies: the pivotal studies were termed the AWARD series. The dosages of dulaglutide studied were 0.75 mg and 1.5 mg.
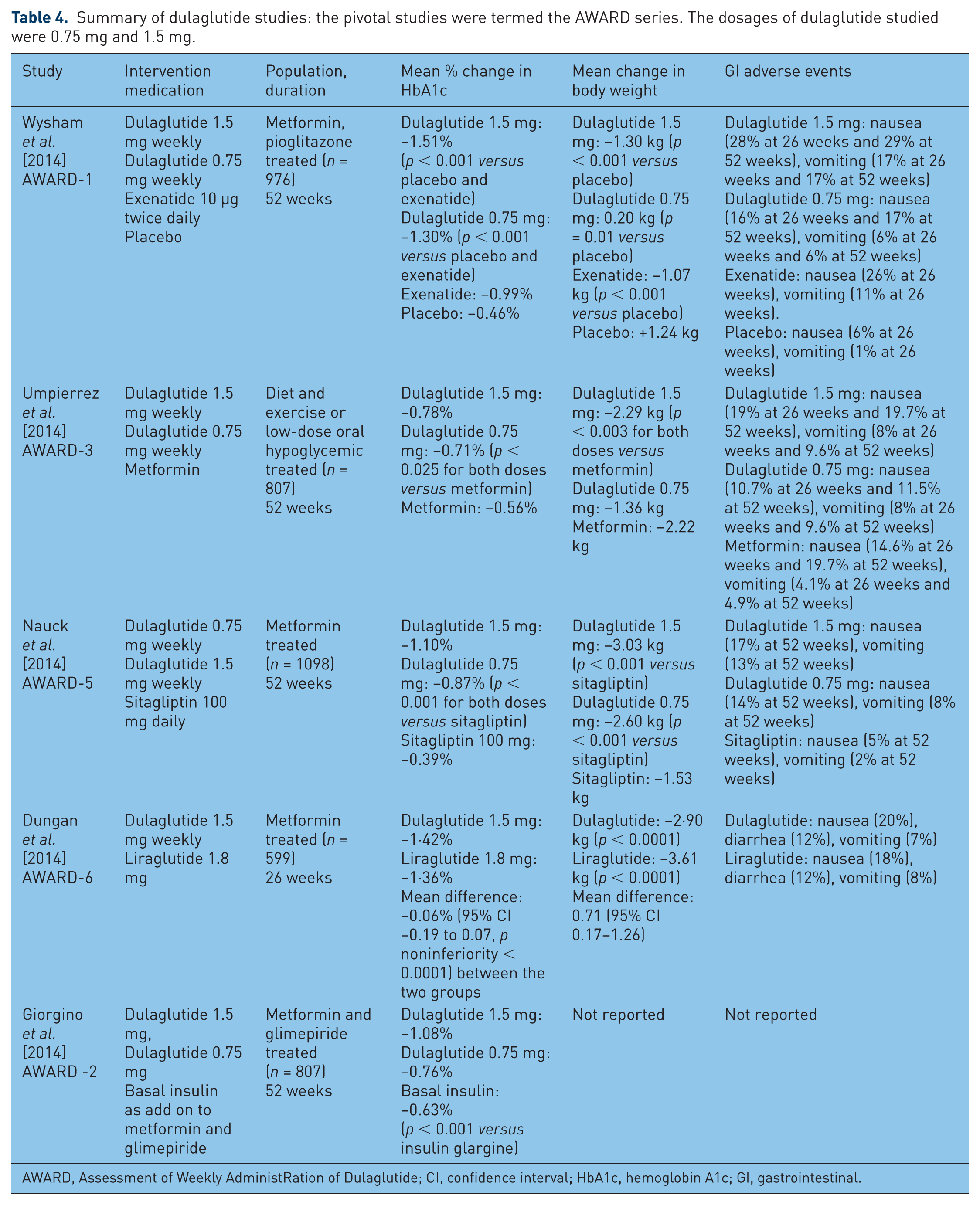
AWARD, Assessment of Weekly AdministRation of Dulaglutide; CI, confidence interval; HbA1c, hemoglobin A1c; GI, gastrointestinal.
Lixisenatide
Lixisenatide, a once daily GLP-1 agonist approved in the European Union and Australia in 2013, is a derivative of exendin-4 synthesized by omitting proline at position 38 and adding six lysine residues. Following subcutaneous administration, peak plasma concentrations (Cmax) are achieved in 1.5–2.5 h. Elimination of the drug is mainly through protein catabolism with a terminal half life of approximately 3 h [Distiller et al. 2008; European Medicines Agency, 2012]. Small studies suggest that no dose adjustment is required in patients with moderate renal failure [Liu and Ruus, 2009].
The efficacy of lixisenatide has been tested as a monotherapy and in combination therapy in randomized placebo and active comparator studies (GetGoal studies). The studies have included metformin, sulfonylurea, pioglitazone, and insulin-treated subjects [Ahrén et al. 2013; Bolli et al. 2014; Pinget et al. 2013; Riddle et al. 2013a, 2013b; Rosenstock et al. 2013] (Table 5). Reductions of 0.5–1.0 % HbA1c have been achieved in multiple 24-week trials with weight loss of 1–2 kg in these trials. Lixisenatide is administered once daily in a dose of 10 or 20 µg. There is no difference with morning or evening dosing. Lixisenatide is particularly effective in lowering glucagon levels after a meal and consequently has a pronounced effect on post-meal glucose [Ahrén et al. 2014a].
Summary of lixisenatide studies: the pivotal studies were termed the GET-GOAL series.

GI, gastrointestinal; HbA1c, hemoglobin A1c; LS, least squares.
In the Get-Goal studies, at the end of the main 24-week treatment period in placebo-controlled studies, 69.8% of lixisenatide patients had a positive antibody status. Injection site reactions were reported in 3.9% of the patients receiving lixisenatide while they were reported in 1.4% of patients receiving placebo. The majority of reactions were mild in intensity and usually did not result in discontinuation of the treatment. The change in HbA1c from baseline was similar regardless of the antibody status (positive or negative).
Albiglutide
Albiglutide is a recombinant fusion protein (molecular weight of ~73 kDa) consisting of two copies of a 30-amino-acid sequence of modified human GLP-1 linked in series to human albumin [Matthews et al. 2008; Bush et al. 2009]. The GLP-1 sequence has been modified with a glycine substituted for the naturally occurring alanine at position 8 in order to confer resistance to DPP-4-mediated proteolysis. These modifications greatly extend the half life (6–8 days), allowing once weekly dosing by subcutaneous injection. Albiglutide steady state is reached within 3–4 weeks. Albiglutide is degraded into small peptides and individual amino acids by proteolytic enzymes. No sig-nificant drug interactions were noted when albiglutide was concomitantly administered with warfarin, digoxin and oral contraceptives. Albiglutide decreased the AUC and Cmax of simvastatin when these drugs were administered concomitantly.
Albiglutide has undergone extensive testing both as monotherapy and in comparison and combination with several oral hypoglycemic agents [Rosenstock et al. 2014; Rendell et al. 2014; Ahrén et al. 2014b; Pratley et al. 2014; Wysham et al. 2014; Home et al. 2015; Weissman et al. 2014] (Table 6). Reductions in HbA1c are typically 0.3–1.0% with a 30 mg dose which is then uptitrated to 50 mg. At these dose levels, albiglutide is unique among the GLP-1 agonists in having a reduced GI side-effect profile, often approaching that of placebo. There appears to be no need for dose adjustment and the drug is well tolerated in patients with mild to moderate renal failure [Young et al. 2014; Wysham et al. 2014].
Summary of albiglutide: the pivotal studies were termed the HARMONY series.

CI, confidence interval; HbA1c, hemoglobin A1c; NS, nonsignificant.
However, albiglutide injections are associated with up to a 20% incidence of injection site reactions, which can rarely force withdrawal of treatment. Injection site reactions (typically including rash, erythema, or itching at the injection site) occurred in 15% of patients treated with albiglutide compared with 7% with all comparators and led to discontinuation in 2% of albiglutide-treated patients. Antibody reactions developed in 4% of patients but were not associated with reduced ability to lower glucose levels. Injection site reactions were reported more frequently for antibody positive (41%, N = 116) than antibody negative patients (14%, N = 1927). Weight loss with albiglutide has not been as great as with the other GLP-1 agents.
Benefits of GLP-1 agonists
The GLP-1 agonists have been approved for the treatment of diabetes, the primary criterion being reduction of HbA1c. Yet, there are many hypoglycemic therapeutic agents which provide similar HbA1c improvement. The great interest in this class of drugs stems not from effect on HbA1c but, rather, from the weight loss which has been evident in the approval trials. The weight loss is modest, but it is the hope for a progressive long-term response, which has generated the excitement of physicians and, indeed, the public for GLP-1 agonists. GlP-1 agonists result in weight loss in patients with diabetes as well as those with obesity without diabetes [Vilsbøll et al. 2012]. Exenatide treatment for 12 months has led to weight reduction of as much as 13 kg, but the weight loss in extension studies of sustained release exenatide after the initial year of treatment has not been quite as impressive [Nayak et al. 2010; MacConell et al. 2013]. After 1 year of treatment exenatide ER yielded a weight loss of –3.7 kg which by 6 years was –4.3 kg [Henry et al. 2014].
Very recently, liraglutide given at doses up to 3 mg daily received approval for treatment for obesity as a separate indication [European Medicines Agency, 2015]. In a 2-year study of liraglutide in an obese nondiabetic population, mean weight loss at 2 years was 7.8 kg for patients receiving 3 mg of liraglutide daily, about 5 kg more than the placebo control group [Astrup et al. 2012]. Forty-eight percent of these patients reported nausea and 14% vomiting, but only 4 out of 93 (4%) withdrew from treatment. The mean 1-year weight loss on treatment with liraglutide 3.0 mg from randomization was 9.2 kg for participants reporting nausea/vomiting episodes versus 6.3 kg for those with none [a treatment difference of 2.9 kg; 95% confidence interval (CI) 0.5–5.3; p = 0.02] [Lean et al. 2014].
Similarly, in a comparison of exenatide ER with twice daily exenatide over a 30-week time period, patients on the weekly preparation who experienced nausea lost 5.4 kg compared with 3.4 kg for those who did not complain of nausea [Drucker et al. 2008]. In the same trial, those with nausea on the twice a day injections lost 4.1 kg compared with 3.4 kg for those without. It would appear that the GI side effects of GLP-1 agonists contribute to but are not solely responsible for the weight loss observed. Conversely, treatment with albiglutide, the GLP-1 agonist with the lowest incidence of GI side effects, has not yielded significant weight loss.
Cardiovascular effects of GLP-1 agonists
GLP-1 agonists have both receptor dependent and independent effects on cardiac muscle. These agents increase inotropic action, optimize glucose uptake in the myocardium, and cause vasodilator effects mediated through the nitric oxide/cyclic guanosine monophosphate pathway [Ban et al. 2008]. In a meta-analysis of 32 studies of exenatide and liraglutide, systolic blood pressure decreased by −1.79 mmHg (from −2.94 to −0.64) compared with placebo [Robinson et al. 2013]. Reduction in diastolic blood pressure failed to reach statistical significance (−0.54 mmHg; from −1.15 to 0.07). Heart rate increased by 1.86 beats/min (95% CI 0.85–2.87). This effect was more evident for liraglutide and exenatide ER than for exenatide twice daily. In an ambulatory blood pressure study, dulaglutide 1.5 mg was associated with a reduction in 24 h systolic blood pressure and an increase in 24 h heart rate [Ferdinand et al. 2014].
Compared with other classes of antihyperglycemic agents, exenatide twice daily was shown to decrease the likelihood of having a cardiovascular event over a period of 1–4 years [Best et al. 2010; Ratner et al. 2011]. In a study of patients with heart failure, infusion of exenatide for 2 days resulted in significant improvement in cardiac index (p = 0.003) and pulmonary capillary wedge pressure (p = 0.001) [Nathanson et al. 2012]. In a meta-analysis of 33 existing trials, the difference in the incidence of Major adverse cardiac events (MACE) between GLP-1 agonists and all comparators did not reach statistical significance [OR 0.78 (0.54–1.13), p = 0.18] [Monami et al. 2014], although the placebo and pioglitazone comparisons were significant. No effect was observed on mortality. Large-scale trials are currently under way designed to assess the effect of GLP-1 agents in populations at high risk of cardiac events.
Effect on lipid parameters and cardiovascular risk biomarkers
Modest reductions in postprandial triglyceride elevation, apo lipoproteins, lipoprotein cholesterol (all p < 0.001), and nonesterified fatty acids (p < 0.05) have been noted with exenatide and liraglutide treatment [Bunck et al. 2010; Schwartz et al. 2010; Fonseca et al. 2011]. Small improvements in LDL and triglyceride levels comparable to those of exenatide were obtained in a comparison study with dulaglutide [Wysham et al. 2014]. The multiple effects of GLP-1 agonists on glucose and lipid metabolism might be beneficial in patients with nonalcoholic steatohepatitis. Although animal models of this condition suggest possible benefit [Liu et al. 2012], there are few clinical data as yet to substantiate these findings in patients with liver disease.
Safety of GLP-1 agonists
The GLP-1 agents all have varying degrees of GI side effects. These issues do not by themselves imply significant health risks. The primary safety concerns have centered on medullary carcinoma of the thyroid and pancreatitis. GLP-1 receptors are expressed in thyroid tissue, particularly C cells [Gier et al. 2012]. In routine preclinical animal testing studies of liraglutide, an increase in the frequency of C-cell hyperplasia and thyroid tumors was observed in mice and rats [US Food and Drug Adminstration, 2009; Knudsen et al. 2010]. Although GLP-1 receptor stimulation induced calcitonin release and C-cell proliferation in rodents, these effects were not observed in nonhuman primates, implying possible species-specific differences in GLP-1 receptor expression in the thyroid. In clinical studies, analysis of calcitonin levels in over 5000 patients participating in liraglutide trials showed no effect [Hegedüs et al. 2011].
There has been great concern that the GLP-1 agonists may augment the incidence of pancreatitis in patients with diabetes. The US Food and Drug Administration Adverse Event Reporting System database recorded many reports of pancreatitis in exenatide- and sitagliptin-treated patients [Elashoff et al. 2011]. These findings led to a comprehensive regulatory review. Data from animal exposure studies as well as clinical studies with over 28,000 patients who received some form of incretin-based therapy were thoroughly reviewed by the US Food and Drug Administration and the European Medicines Agency. Their assessment of the totality of evidence was that there existed no causal association between use of GLP-1 agonists and DPP-4 inhibitors and possible pancreatitis and pancreatic cancer [Egan et al. 2014].
The most commonly observed adverse event with GLP-1 agonists is transient nausea, which may be the result of delayed gastric emptying. Depending on the trial, such nausea has been reported in up to 57% of patients treated with twice daily exenatide [Heine et al. 2005] and up to 56% of those treated with liraglutide [Zinman et al. 2007]. Although nausea resolves within 6–8 weeks in most patients, the incidence and severity can be reduced using a dose-escalation strategy. For exenatide, treatment is typically initiated with 5 µg twice daily given within 60 min before a meal. After this dose is well tolerated, it can be increased to 10 µg twice daily, usually within a month. Liraglutide is usually started at a dose of 0.6 mg daily and increased to 1.2 mg daily 1 week later. The dose may be increased subsequently to 1.8 mg daily. Liraglutide at a dose level of 3 mg has now been approved for treatment of obesity. Using these dose-escalation strategies, Buse and colleagues reported 28% of patients treated with exenatide and 26% of those treated with liraglutide experienced nausea and that the nausea was of shorter duration with liraglutide (p < 0.0001) [Buse et al. 2009]. In a 26-week trial comparing liraglutide with sitagliptin, 21–27% of GLP-1 versus only 5% of the DPP-4 treated cohorts experienced nausea. In the subsequent 26-week extension phase, the incidence of nausea fell in both groups, ranging from 1% to 3% of patients. The incidence of nausea is markedly lower with sustained release GLP-1. The highest reported incidence with exenatide ER is 26% [Drucker et al. 2008]. There is minimal difference in GI side effects comparing albiglutide with placebo in several studies.
Hypoglycemia is not a prominent feature of GLP-1 treatment, unless used in sulfonylurea and insulin-treated patients. Clearly, hypoglycemic events occur when there is no reduction in prior dosage of insulin or insulin secretagogues. In a 26-week comparison trial, minor hypoglycemia (blood glucose < 56 mg/dl successfully self treated) occurred in 34% of patients treated with exenatide and 26% of those treated with liraglutide [Buse et al. 2009]. In patients concomitantly treated with a sulfonylurea, 42% experienced an episode of minor hypoglycemia with exenatide and 33% had minor hypoglycemia with liraglutide. Severe hypoglycemia requiring the assistance of another person occurred in 2 of the 231 patients treated with exenatide and a sulfonylurea; no cases were observed in patients treated with liraglutide.
Injection site reactions are a differential feature among the agents. The long-acting agents exenatide ER and albiglutide have a significant incidence of such reactions (up to 17.6% for exenatide ER, up to 22% for albiglutide), some of sufficient intensity to lead to discontinuance of therapy. Liraglutide, lixisenatide, and daily exenatide, the daily GLP-1 agonists, do not have this problem. Interestingly, dulaglutide, which is administered weekly, does not have a significant level of injection site reactions.
GLP-1 agonists versus DPP-4 inhibitors
There are two alternative approaches to providing therapeutic levels of GLP-1: direct subcutaneous administration of GLP-1 agonists and oral DPP-4 antagonists. The DPP-4 inhibitors prevent the rapid breakdown of endogenous GLP-1 by DPP-4. The levels of GLP-1 reached with DPP-4 treatment are lower than those achievable by direct administration of GLP-1 agonists [Dalle et al. 2013].
Most comparison studies show greater reduction in HbA1c with the GLP-1 agonists than with the DPP-4 inhibitors. In addition, the GLP-1 agonists promote weight loss while the DPP-4 inhibitors are typically weight neutral. Conversely, the DPP-4 inhibitors have a much lower incidence of GI complaints than the GLP-1 agonists. In a comparison of exenatide ER to sitagliptin, the reduction of HbA1c was 1.5% with exenatide and 0.9% with sitagliptin (p < 0.001) [Bergenstal et al. 2010]. Exenatide-treated patients lost 2.3 kg compared with just 0.8 kg for sitagliptin. The exenatide-treated patients reported nausea and diarrhea at twice the rate seen with sitagliptin.
Although 2 weeks of treatment with twice-daily exenatide and once-daily sitagliptin resulted in similar reductions in FPG (−15 versus −19 mg/dl; p = 0.3234), the PPG reduction was significantly greater with exenatide (−112 mg/dl) compared with sitagliptin (−37 mg/dl; p < 0.0001) [DeFronzo et al. 2008]. Furthermore, patients who crossed over from sitagliptin to 2 weeks of exenatide achieved a further PPG decrease of 76 mg/dl compared with an increase of 73 mg/dl for those who crossed over from exenatide to sitagliptin. Patients treated with exenatide for 2 weeks before the crossover lost 0.8 kg, and those treated with sitagliptin lost 0.3 kg (p < 0.006).
In a 26-week study, there was a HbA1c reduction of 1.2% with liraglutide 1.2 mg daily; 1.5% with liraglutide 1.8 mg daily; and 0.9% with sitagliptin 100 mg daily (p < 0.0001 versus each liraglutide dose) [Pratley et al. 2010]. FPG decreased significantly more in the group taking 1.2 mg of liraglutide (−34 mg/dl) and in the group taking 1.8 mg of liraglutide (−39 mg/dl) compared with sitagliptin (−15 mg/dl; p < 0.0001). A weight loss of 2.9 kg was observed in the group taking liraglutide 1.2 mg, 3.4 kg in the group taking liraglutide 1.8 mg, and a loss of 1.0 kg in the sitagliptin group (p < 0.0001 versus both liraglutide dose groups). During the next 26 weeks, body weight stabilized in all three groups such that, after 52 weeks, the overall decrease in weight from baseline was 2.8, 3.7, and 1.2 kg, respectively (all p < 0.0001). Nausea (27%), vomiting (10%), and diarrhea (10%) were much more frequent in the liraglutide treatment cohort compared with the sitagliptin cohort (5%, 4%, and 5%, respectively).
In the AWARD-5 STUDY, the reduction in HbA1c was almost threefold greater with dulaglutide treatment than with sitagliptin, but nausea and vomiting also occurred at a threefold higher rate [Nauck et al. 2104]. In a study of albiglutide given weekly versus daily sitagliptin, there was a slightly greater improvement in HbA1c and greater weight loss with albiglutide [Leiter et al. 2014]. The incidence of GI complaints was no higher with albiglutide than with sitagliptin.
Comparison studies of the GLP-1 agonists
There have been numerous head to head studies directly comparing results of treatment with the various GLP-1 agents. At 24 weeks, Exenatide ER produced significantly greater reductions from baseline HbA1c than twice daily exenatide (1.60% versus 0.90%; p < 0.0001) [Blevins et al. 2011]. Similar reductions in mean body weight were observed in both groups. Mild to moderate nausea, the most frequent adverse event, was less common with exenatide ER (14%) than with short-acting exenatide (35%).
In a head to head study comparing liraglutide taken daily to once weekly exenatide ER, the change in HbA1c was greater in patients in the liraglutide group (–1·48%, standard error 0·05) than in those taking exenatide ER (–1·28%, 0·05) [Buse et al. 2013]. The most common adverse events were nausea (21%), vomiting (11%), and diarrhea (13%) in the liraglutide group, higher than with exenatide ER (9%, 6%, and 4%, respectively).
In an active comparator trial of lixisenatide in metformin-treated patients, the exenatide and lixisenatide groups had similar reductions in HbA1c with slightly lower mean weight loss, lower incidence of hypoglycemia, and better GI tolerability in the lixisenatide cohort compared with the exenatide arm [Rosenstock et al. 2013].
In a 28-day, multicenter study, patients (mean HbA1c 7.3%) received subcutaneous lixisenatide once daily (10 µg weeks 1–2, then 20 µg; n = 77) or liraglutide once daily (0.6 mg week 1, 1.2 mg week 2, then 1.8 mg; n = 71) 30 min before breakfast [Kapitza et al. 2013]. Lixisenatide reduced PPG significantly more than liraglutide (–12.6 versus –4.0 h·mmol/liter, respectively; p < 0.0001). Changes in FPG were greater with liraglutide (–0.3 versus –1.3 mmol/liter, p < 0.0001). Mean HbA1c decreased in both treatment groups (from 7.2% to 6.9% with lixisenatide versus 7.4% to 6.9% with liraglutide) as did body weight (–1.6 kg versus –2.4 kg, respectively). The overall incidence of adverse events was lower with lixisenatide (55%) versus liraglutide (65%).
A comparison of once weekly dulaglutide to once daily liraglutide found equivalent reduction in HbA1c (1.4%) [Dungan et al. 2014]. The most common GI adverse events were nausea (20% in the dulaglutide group versus 18% in the liraglutide group), diarrhea (12% versus 12%), dyspepsia (8% versus 6%), and vomiting (7% versus 8%), with similar rates of study drug discontinuation due to adverse events between the two groups (6% in each group). The hypoglycemia rate was 0.34 and 0.52 events per patient per year, respectively, and no severe hypoglycemia was reported.
In a study comparing weekly albiglutide with daily liraglutide the change in HbA1c from baseline to week 32 was –0·78% (95% CI –0·87 to –0·69) in the albiglutide group and –0·99% (95% CI –1·08 to –0·90) in the liraglutide group [Pratley et al. 2014]. Liraglutide patients lost 2.2 kg compared with only 0.6 kg in the albiglutide group (p < 0.05). Injection site reactions occurred in more patients given albiglutide than in those given liraglutide (12.9% versus 5.4%; treatment difference 7.5%; 95% CI 3.6–11.4; p = 0.0002), whereas the opposite was the case for GI events, which occurred in 49.0% of patients in the liraglutide group versus 35·9% in the albiglutide group (treatment difference –13.1%; 95% CI –19.9 to –6.4]; p = 0.00013).
GLP-1 agonists and insulin
The two potential diabetes therapies which require injection are the GLP-1 agonists and insulin. Many comparison studies have been performed to try to definitively answer this question. In a 32-week study comparing exenatide with glargine in patients treated with metformin or sulfonylureas, exenatide and titrated glargine gave similar improvement in HbA1c both 1.36%; p < 0.001 [Barnett et al. 2007]. The mean HbA1c at endpoint was above the American Diabetes Association (ADA) target with both treatments exenatide 7.57%; glargine 7.58% (0.09%). Similar proportions of patients achieved an HbA1c up to 7% (37.5% and 39.8%, respectively; p nonsignificant) or up to 6.5% (21.5% and 13.6%). Patients lost weight during exenatide treatment, whereas they gained weight during insulin treatment; (mean difference –2.2 kg; 95% CI –2.8 to –1.7; p < 0.001). The reduction from baseline in FPG was greater with glargine (–4.1 mg/dl) than with exenatide (–2.9 mg/dl) (p < 0.001). Compared with glargine, exenatide treatment resulted in significantly lower 2 h (PPG) excursions (p < 0.016) and total daily mean glucose excursion (p < 0.001). The proportions of patients reporting nausea during exenatide and glargine treatment were 42.6% and 3.1%, respectively; vomiting occurred in 9.6% of the exenatide patients and 3.1% of the glargine patients The incidence of hypoglycemia in the two groups was 14.7% for glargine and 25.2% for exenatide (p nonsignifiant).
In a 26-week study, patients with type 2 diabetes whose condition was inadequately controlled on two or three oral antidiabetic drugs were randomized to add-on exenatide 5–10 µg twice daily (n = 118) or glargine (titrated to target FPG ⩽ 5.6 mmol/liter; n = 117) for 26 weeks [Davies et al. 2009]. Mean baseline HbA1c was 8.65% (0.68%) in the exenatide group and 8.48% (0.66%) in the glargine group. Exenatide and glargine gave similar HbA1c improvements: –1.25% (0.09%) and –1.26% (0.09%), respectively (p nonsignifcant), but had divergent effects on body weight [–2.73 (0.31) versus +2.98 (0.31) kg, respectively, p < 0.001].
In a similar study, Heine and colleagues found comparable lowering of HbA1c with both exenatide and glargine, weight loss of 2–3 kg on exenatide contrasted with weight gain on glargine, a much higher incidence of GI complaints on exenatide, and similar rates of symptomatic hypoglycemia [Heine et al. 2005]. Exenatide reduced the PPG levels much more than glargine (p < 0.05) whereas the reduction in FPG was much greater in the basal insulin cohort. The rate of withdrawal of patients from these comparison studies due to adverse events has typically been up to two times greater on exenatide.
There were similar findings in studies comparing exenatide treatment to biphasic insulin [Nauck et al. 2007]. The addition of aspart insulin premeal did not improve the lowering of HbA1c (–0.89 with biphasic insulin compared with –1.04 with exenatide). In a very significant small study, β-cell secretion was measured by glucose clamp over a 3-year period in patients treated with exenatide compared with those treated with glargine [Bunck et al. 2011]. There was a 39% increase in endogenous insulin secretion with exenatide treatment contrasted to no effective change in the glargine patient cohort. In a comparison of exenatide therapy to glibenclamide, the Homeostatic Model Assessment β (HOMA-β) index increased after 9 and 12 months with exenatide, whereas there was no improvement with the sulfonylurea. In addition, there was a statistically significant decrease in the Homeostatic Model Assessment-Insulin Resistance (HOMA-IR) index with exenatide but no change with glibenclamide.
In a study comparing liraglutide with glargine, there was a slightly greater reduction in HbA1c with liraglutide as well as weight loss compared with weight gain on glargine [Russell-Jones et al. 2009]. The incidence of hypoglycemia was similar for the two cohorts.
Results with the long-acting GLP-1 agonists have paralleled those with the daily GLP-1 agonists. Exenatide ER was compared with glargine in a 3-year study [Diamant et al. 2010]. At 3 years, the HbA1c change was −1·01% (SE 0.07) in the exenatide group versus −0.81% (0.07) in the glargine group (least squares mean difference −0.20%, SE 0.10, 95% CI −0.39 to −0.02; p = 0·03). The exposure-adjusted rate of overall hypoglycemia was threefold higher in patients given glargine (0.9 events per patient per year) than in those given exenatide (0.3 events per patient per year). The reduction in weight with exenatide ER was 2.5 kg compared with a 2 kg weight gain in glargine-treated patients.
Dulaglutide treatment resulted in superior lowering of HbA1c compared with glargine, with greater weight loss and no difference in rates of hypoglycemia [Giorgino et al. 2014]. Subjects who were on baseline metformin and glimepiride therapy were exposed to dulaglutide 0.75 mg weekly, 1.5 mg weekly and basal insulin (titrated to fasting glucose levels). At the end of 1 year, the mean reductions in HbA1c were 0.76, 1.08, and 0.63% in dulaglutide 0.75 mg, 1.5 mg, and glargine groups, respectively (p < 0.001 versus glargine for the dulaglutide 1.5 mg once-weekly group).
In a 52-week study comparing albiglutide and glargine, HbA1c decreased from 8.28 ± 0.90% (67.0 ± 9.8 mmol/mol) (mean ± SD) at baseline to 7.62 ± 1.12% (59.8 ± 12.2 mmol/mol) at week 52 in the albiglutide group with a similar reduction in the glargine group (from 8.36 ± 0.95% to 7.55 ± 1.04%) [Pratley et al. 2013]. Body weight increased in the glargine group and decreased in the albiglutide group, with a mean treatment difference of –2.61 kg (95% CI –3.20, –2.02; p < 0.0001). Documented symptomatic hypoglycemia occurred in a higher proportion of patients in the glargine group than in the albiglutide group (27.4% versus 17.5%, p < 0.04).
The addition of rapid-acting to long-acting insulin does not appear to significantly change the comparisons to GLP-1 agonist treatment. One study compared mixed Neutral Protein Hagedorn (NPH)–aspart biphasic insulin with twice daily exenatide treatment. Glycemic control achieved with exenatide was noninferior to that achieved with biphasic insulin aspart (HbA1c change: exenatide –1.04% ± 0.07%, biphasic insulin aspart –0.89% ± 0.06%; difference –0.15%; 95% CI –0.32% to 0.01%). Exenatide-treated patients lost weight, while patients treated with biphasic insulin aspart gained weight (between-group difference –5.4 kg; 95% CI –5.9 to –5.0 kg). Both treatments reduced FPG (exenatide –1.8 ± 0.2 mmol/liter, p < 0.001; biphasic insulin aspart –1.7 ± 0.2 mmol/liter, p < 0.001). Greater reductions in PPG excursions following morning (p < 0.001), midday (p = 0.002), and evening meals (p < 0.001) were observed with exenatide. The withdrawal rate was 21.3% (54/253) for exenatide and 10.1% (25/248) for biphasic insulin aspart. Nausea (33% incidence, 3.5% discontinuation) was the most common adverse event observed with exenatide.
In a study of dulaglutide compared with glargine with additional premeal insulin lispro given to both arms, greater reductions in HbA1c were observed with dulaglutide at 26 and 52 weeks [Jendle et al. 2014]. Weight decreased with dulaglutide and increased with glargine. The rates of hypoglycemia were slightly higher with glargine. Nausea and diarrhea were frequent with dulaglutide for both the 1.5 mg (25.8%, 16.6%) and 0.75 mg (17.7%, 15.7%) doses compared with glargine (3.4%, 6.1%).
The trials comparing GLP-1 agonists with basal insulin to treat patients who no longer achieve glycemic control on oral agents have not demonstrated clear superiority of one or the other modality; rather, there is a complementarity of effects, lower FPG on basal insulin, lower PPG with GLP-1 treatment, weight loss with GLP-1 agents while there is weight gain but much fewer GI side effects with insulin. The addition of rapid-acting insulin to long-acting insulin treatment does not appear to change these comparison effects.
The complementary benefits of GLP-1 agonists and insulin have led to attempts to use both modalities together. In a short study, the glucose excursion after a standardized meal for glargine, glargine plus sitagliptin, and glargine plus exenatide in metformin-treated patients was directly compared [Arnolds et al. 2010]. The unadjusted 6 h PPG excursions of both glargine plus exenatide and glargine plus sitagliptin were significantly less than that for glargine alone (606 ± 104 versus 612 ± 133 versus 728 ± 132 mg/dl/h; p = 0.0036 and 0.0008). This was reflected in a greater reduction in HbA1c with glargine plus exenatide (–1.9%) and glargine plus sitagliptin (–1.5%) than with glargine alone (–1.2%) (p < 0.01). The American Diabetes Association HbA1c target of less than 7.0% was reached by 80.0, 87.5, and 62.5% of subjects, respectively. The glargine plus exenatide cohort had the highest number of adverse events, mostly GI (56%). Hypoglycemia (blood glucose < 50 mg/dl) rates were low and comparable among groups. Weight decreased with glargine plus exenatide (–0.9 ± 1.7 kg) and increased slightly with glargine alone (0.4 ± 1.5 kg; p < 0.04).
Buse and colleagues explored the alternative of using twice daily exenatide in subjects already treated with glargine compared with management by simply raising the insulin dose [Buse et al. 2011a]. The HbA1c level decreased by 1.74% with exenatide and 1.04% with placebo (between-group difference –0.69%; 95% CI –0.93% to –0.46%; p< 0.001). Weight decreased by 1.8 kg with exenatide and increased by 1.0 kg with placebo (between-group difference –2.7 kg; 95% CI –3.7 to –1.7). Average increases in insulin dosage were 13 units daily for exenatide compared with 20 units for placebo. The estimated rate of minor hypoglycemia was similar between groups. Thirteen exenatide recipients and one placebo recipient discontinued the study because of adverse events (p < 0.010); rates of nausea (41% versus 8%), diarrhea (18% versus 8%), vomiting (18% versus 4%), headache (14% versus 4%), and constipation (10% versus 2%) were higher with exenatide than for the cohort treated solely by increasing the glargine dose.
In a comparison of albiglutide treatment of patients poorly controlled on glargine to addition of premeal lispro [Rosenstock et al. 2014], HbA1c decreased from baseline by –0.82% ± SE 0.06% (9.0 mmol/mol) with albiglutide and –0.66% ± 0.06% (7.2 mmol/mol) with lispro by 26 weeks; treatment difference –0.16% (95% CI –0.32 to 0.00; 1.8 mmol/mol; p < 0.0001). Weight decreased with albiglutide but increased with lispro (–0.73 ± 0.19 kg versus +0.81 ± 0.19 kg). The mean glargine dose increased from 47 to 53 IU (albiglutide) and from 44 to 51 IU (lispro). Adverse events for albiglutide versus lispro included severe hypoglycemia (zero versus two events), documented symptomatic hypoglycemia (15.8% versus 29.9%), nausea (11.2% versus 1.4%), vomiting (6.7% versus 1.4%), and injection site reactions (9.5% versus 5.3%).
A real world 24-month study using chart review of cases with either exenatide added to glargine-treated patients or glargine added to exenatide-treated patients showed an average reduction of 0.7% in HbA1c with either addition [Levin et al. 2012]. There was no weight gain when glargine was added to exenatide and a 2.5 kg weight loss when exenatide was added to glargine.
In a follow on of a long-term insulin degludec trial, 88 subjects who had completed 104 weeks of treatment with degludec plus metformin with HbA1c greater than 7.0% were treated with liraglutide and compared with 89 subjects treated with aspart for 26 weeks [Mathieu et al. 2014]. The liraglutide-treated cohort had a 0.74% reduction in HbA1c, significantly more than for the aspart-treated group (−0.39%). More liraglutide-treated patients achieved HbA1c less than 7.0% without confirmed hypoglycemia and without weight gain; estimated odds ratio (liraglutide/aspart) 13.79 (95% CI 5.24–36.28); p < 0.0001. Liraglutide treatment resulted in a 2.8 kg weight loss compared with a 0.9 kg weight gain for aspart.
A meta-analysis of three 24-week trials in which lixisenatide was added to basal glargine treatment showed an average improvement of HbA1c of 0.3%–0.8% compared with insulin alone [Charbonnel et al. 2014]. There was no weight gain or modest weight loss in the three studies.
With the coming of the long-acting GLP-1 agonists, daily injections of insulin can be supplemented by a weekly or even less frequent injection of GLP-1 agonist. It is true that daily administration of a GLP-1 agent such as liraglutide seems to have greater effects on HbA1c than long-acting exenatide or albiglutide. An alternative to administer such agents separately from insulin is to mix the two in the same injection device. This has proven possible with liraglutide and long-acting insulin degludec. The results of coadministering degludec and liraglutide (degludec–liraglutide) in combination have demonstrated that the benefits of each individual agent can be achieved with lower doses of each. Consequently the adverse effects of insulin including hypoglycemia and weight gain and the GI side effects of liraglutide are all reduced. Despite the complex polymeric character of insulin degludec, it is possible to add liraglutide in a fixed ratio and maintain the pharmacokinetic properties of both agents. Although the ratio of degludec to liraglutide is fixed at 100 units/3.6 mg, the ability to modify and titrate the volume administered allows for true flexibility in dosage of liraglutide. This flexibility has paid great dividends in terms of significant reduction of hypoglycemia and GI adverse events.
In a 26-week trial, patients with type 2 diabetes treated with metformin with or without pioglitazone were randomly assigned (2:1:1) to daily injections of degludec–liraglutide, degludec, or liraglutide (1.8 mg per day) [Gough et al. 2014]. After 26 weeks, mean HbA1c decreased by 1.9% to 6.4% with the combination, by 1.4% to 6.9% with insulin alone, and by 1.3% to 7.0% with liraglutide alone. GI adverse events in the degludec–liraglutide group were less common than in the liraglutide group (nausea 8.8% versus 19.7%). The insulin-only group had the fewest events (nausea 3.6%). The number of confirmed hypoglycemic events per patient year was 1.8 for the combination, 0.2 for liraglutide alone, and 2.6 for insulin alone.
In a 26-week, double-blind trial, patients with type 2 diabetes with HbA1c 7.5–10.0% on basal insulin (20–40 units) and metformin with or without sulfonylurea/glinides were randomized (1:1) to once-daily degludec–liraglutide or degludec alone titrating to a FPG between 4 and 5 mmol/liter. Both groups reached a mean insulin equivalent of 45 units [Buse et al. 2014]. Yet, HbA1c decreased by 1.9% with the combination product and by only 0.9% (10 mmol/mol) with insulin alone. Mean weight reduction with degludec–liraglutide was 2.7 kg versus no weight change with insulin alone (p < 0.00010). Hypoglycemia incidence was comparable (24% for the combination versus 25% for insulin alone. The incidence of nausea was low in both groups (6.5% for the combination versus 3.5% for insulin alone).
Glargine and lixisenatide can also be mixed effectively. A similar approach is being tested with this combination. The development plan for this combination product thus far includes the possibility of multiple different dose ratios, so that higher doses of glargine can be administered with lower doses of lixisenatide for those patients who have GI intolerance.
Discussion
Although there are many hormones released by the digestive system in response to food intake, of these incretins, GLP-1 has proven to have the greatest therapeutic potential. Prevention of breakdown of endogenous GLP-1 with inhibitors of DPP-4 has been effective in lowering HbA1c. However, the administration of GLP-1 agonists in excess of levels reached by DPP-4 inhibition has greater effects on glucagon suppression, gastric emptying, and satiety. The overall benefit achievable with GLP-1 agonists is not then limited to the hypoglycemic effect, which is similar to that obtained with metformin and sulfonylureas, but also includes reduction of blood pressure, possible preservation of β cells, and weight loss. It is this last property which has generated the most interest in GLP-1 therapy. Type 2 diabetes is usually associated with obesity. Although many agents can improve blood glucose levels, there is usually minimal effect on adiposity, which often increases with control of diabetes. The sulfonylureas, thiazolidinediones, and insulin are particularly likely to promote weight gain.
The goal of GLP-1 treatment is to lower glucose levels while achieving other benefits, in particular, weight loss. Liraglutide at high dose has received an indication for the treatment of obesity even without accompanying diabetes. The weight loss seen with GLP-1 therapy is relatively modest, but is significant in preventing the progressive gain seen in patients with diabetes after suppression of high glucose levels with other agents. There is no doubt that part of the weight loss achieved with GLP-1 agents is a result of GI intolerance at high GLP-1 levels. These GI complaints have been the primary obstacle to even greater use of GLP-1 agents. Nausea, vomiting, and diarrhea are typically seen in the early stages of GLP-1 treatment and diminish over time, but there is also less weight loss when the GI side effects diminish.
The benefits of GLP-1 therapy are significant. At this time, GLP-1 agonists are administered by injection, although semaglutide, which can survive digestive activity, is in development for oral use. It is reasonable to compare GLP-1 treatment with insulin injection as the most appropriate modality for patients who no longer can maintain glycemic control on metformin with or without other oral agents. A straightforward comparison suggests that basal insulin and GLP-1 agents provide equivalent lowering of HbA1c, with weight loss a benefit of GLP-1 treatment, yet accompanied by significant GI side effects. Agents such as albiglutide with fewer tolerability issues seem to also offer less weight loss.
When oral hypoglycemic agents alone do not achieve sufficient suppression of hyperglycemia, historically, the next step has been the addition of long-acting insulin injections. The coming of the GLP-1 agents offers an alternative, particularly attractive given the development of the long-acting GLP-1 agonists. At this time, a weekly injection is the norm. Further developments promise agents which last far longer. The GLP-1 agents offer glucose lowering comparable to insulin with weight loss. Side effects appear limited to GI motility disturbance resulting in nausea, vomiting and weight loss. The more serious concerns relating to medullary carcinoma of the thyroid and pancreatitis have been suppressed somewhat by the US Food and Drug Administration and European Medicines Agency joint statement. Nonetheless GI symptoms are hard to ignore when a patient earnestly complains. The algorithms created by writers of guidelines are difficult to apply to an unhappy patient. There must be a balance. It is here that the art of medicine must come into play for each individual patient. We must assess the benefits of GLP-1 therapy counterbalanced by the tolerability of unpleasant side effects. The decision has to be made based on full discussion and with input from each patient. Some patients will endure unpleasant GI events to achieve the Holy Grail of weight loss, others will not.
Given the issues, the choice between using a GLP-1 agonist versus basal insulin no longer has to be all or none. It is much more reasonable to use both to get the combined benefits of the two modalities rather than to frame these as alternatives. A major step forward is possible with the recognition of the combined benefits of insulin and GLP-1 agonists. The concept of coadministration of basal insulin and GLP-1 agonist was first voiced by Nauck in an opinion piece aptly named ‘GLP-1 analogues and insulin: sound the wedding bells?’ [Nauck and Meier, 2011].
Certainly the beneficial effects of GLP-1 agents and of insulin can be achieved by separate administration of the individual components, but the advantage of administration of a combination in the same device is clearly the ability to titrate dosage, particularly of the GLP-1 agonist. The development of exenatide established a concept of fixed dose administration, either 5 or 10 µg, and subsequent agents have all adhered to a limited set of fixed dose syringes. This approach is in contrast to the flexible titration of insulin.
The success of degludec–liraglutide in the initial approval trials certainly supports the concept that the introduction of insulin therapy in type 2 diabetes should not be limited to insulin as a single component, but rather, should be as an insulin GLP-1 combination. The coming development of glargine–lixisenatide combinations should further the concept of flexible dosing.
The discovery of GLP-1 and subsequent analysis of its physiological properties has been a great scientific success. We now fully understand the complex relationships which comprise the incretin effect. Conversely, the exploitation of the therapeutic potential of GLP-1 analogues is still ongoing and broadening. The use of GLP-1 agonists as primary treatment of obesity is an advance. So is the realization that insulin and GLP-1 agonists should be concurrently administered. There is much still to learn. GLP-1 has known β-cell supportive effects, including stimulation of neogenesis and proliferation and prolongation of survival [Brubaker and Drucker, 2004; Buteau, 2008]. It remains to be proven conclusively that GLP-1 treatment opposes the progressive loss of β-cell mass which contributes to the worsening of type 2 diabetes over time. There are also long-term studies underway which may reveal a cardiovascular advantage to GLP-1 treatment. The results of these studies are awaited as the fundamental role of GLP-1 treatment of type 2 diabetes solidifies.
Footnotes
Conflict of interest statement
Dr Rendell has been an investigator in several trials on GLP-1 agents.
Funding
Rose Salter Medical Research Foundation and Association of Diabetes Investigators both provided infrastructural support.
