Abstract
Articular cartilage plays a crucial role in reducing friction between bones and enabling movements; however, it is frequently degraded due to persistent joint stress, aging, and osteoarthritis. As its self-repair ability is limited, various cell-based therapeutic strategies have been developed for cartilage regeneration. Conventional two-dimensional (2D) cell cultures inadequately replicate the complex intercellular interactions of native cartilage. In contrast, three-dimensional (3D) cell spheroid cultures can more accurately mimic in vivo cellular physiology, offering superior regenerative potential via improved cell-cell and cell-matrix interactions. These interactions can be enhanced with biomaterials to form composite spheroids, which exhibit substantial potential for improving cartilage regeneration and attenuating osteoarthritis progression in vivo by promoting cell survival and tissue integration. This review highlights current strategies for developing biomimetic composite spheroid systems, including spheroid encapsulation, scaffold incorporation, and 3D bioprinting. Furthermore, we discuss their advantages, translational potential for in vivo cartilage repair, and the challenges and future directions in cartilage tissue engineering.
Introduction
Articular cartilage is a specialized connective tissue that covers the ends of bones in synovial joints, enabling smooth joint movement and minimizing friction. However, due to its avascular and alymphatic nature, articular cartilage has a limited capacity for self-repair, making it particularly vulnerable to damage caused by acute trauma, repetitive microtrauma from mechanical stress, aging, and degenerative conditions (e.g. osteoarthritis). Persistent microtrauma triggers a series of pathological changes, including chondropenia, reduced proteoglycan synthesis, and disruption of the collagen framework, which ultimately impair cartilage integrity. While articular chondrocytes can repair the matrix under optimal conditions, their low cellular density and the absence of vasculature substantially limit their regenerative potential. Consequently, large or chronic cartilage lesions often fail to heal, contributing to the progression of osteoarthritis, a degenerative joint disease affecting over 40 million people globally.
Pathological changes involve the complex interactions between tissue damage and repair processes in bones and cartilages. 1 Thus, the regeneration of the complex microenvironment and extracellular matrix (ECM) in the cartilage tissue requires a specialized strategy to restore the biochemical composition of the cartilage and its biomechanical properties. 2 Articular cartilage defects present significant clinical challenges due to the tissue’s limited self-healing capacity. Recent advancements in tissue engineering and regenerative medicine have explored various therapeutic strategies to address these challenges. 3 Current surgical treatments for articular cartilage damage, including microfracture and osteochondral grafting, 2 aim to alleviate symptoms, and restore joint function but frequently fall to regenerate durable hyaline cartilage. Microfracture is commonly performed to stimulate the release of bone marrow-derived stem cells and promote cartilage formation. However, the resulting fibrocartilage lacks the mechanical and functional properties of native hyaline cartilage. Similarly, osteochondral grafting, which involves the transplantation of healthy cartilage tissue, is limited by donor-site morbidity and limited tissue availability. 4 To overcome current issues in cartilage regeneration, cell-based therapies utilizing expandable cells cultured in vitro have emerged as promising approaches. These therapies typically use autologous or allogeneic chondrocytes and mesenchymal stem cells (MSCs). 5 Especially, MSC-based therapies can benefit the modulation of the inflammatory environment within the joint, addressing the underlying pathology of osteoarthritis. 6 Although these therapies hold promise owing to their potential to modulate inflammation and promote ECM production, their effectiveness remains limited due to challenges, such as low cell survival after implantation, inadequate integration with host tissues, and the risk of dedifferentiation. 7
3D spheroid cultures have been developed to overcome the limitations of conventional two-dimensional (2D) cell culture systems. Unlike monolayer 2D culture, 3D spheroids allow for cellular aggregates similar to tissue-like structures. 8 Such 3D cellular organization promotes cell-cell (e.g. adherence junctions and gap junctions) and cell-matrix (e.g. integrin binding) interactions that preserve the chondrogenic phenotype and support ECM production.9–11 Accordingly, these spheroids can mimic the native cartilage microenvironment, making them highly suitable for in vitro expansion and in vivo cartilage regeneration.12,13 Recent advancements in cartilage tissue engineering have highlighted the superior regenerative potential of spheroid-based cell delivery compared to conventional dispersed cell approaches. For instance, Shim et al. 14 demonstrated that chondrocyte spheroids combined with hyaluronic acid microparticles significantly improved cartilage regeneration compared to dispersed cells, owing to improved maintenance of chondrocyte phenotype, and enhanced ECM deposition. Similarly, Decarli et al. 15 reported that bioprinted constructs using MSC spheroids exhibited accelerated chondrogenic differentiation and superior tissue maturation compared to those with dispersed MSCs. These findings suggest that spheroid-based approaches not only enhance the functional properties of engineered cartilage but also offer improve structural integrity, making them a promising strategy for effective cartilage repair. Nevertheless, 3D spheroids face several challenges, including inadequate nutrient and oxygen diffusion in large constructs, 16 limited vascularization, 17 and inappropriate mechanical properties of regenerated tissues. 18 Hence, advancing spheroid culture techniques is increasingly essential to enhance their therapeutic potential, particularly by improving cell viability and optimizing their mechanical properties.
Integrating biomaterials into spheroid cultures has been recognized as a promising strategy. Biomaterial-based spheroids, also referred to as composite spheroids, combine the biological advantages of 3D spheroid cultures with the structural and mechanical support provided by biomaterial. 19 Especially, scaffolds can enhance both cell adhesion and facilitate nutrient transport, which can address the limitations of spheroid-only approaches. 20 Recent studies also have highlighted the quantitative benefits of composite spheroids. For example, composite spheroids incorporating gelatin microparticles exhibited significantly enhanced cell proliferation, with total cell number increasing after 7 days, 1.5 times compared to monoculture spheroids. 21 Furthermore, co-culture composite spheroid promoted ECM production, contributing to improved mechanical integrity and hierarchical tissue organization. 14 These unique properties of biomaterials can be applied for 3D spheroids to enhance their performance in cartilage regeneration by providing desirable biological, and mechanical characteristics for cartilage repair. By mimicking the natural ECM and supporting cell growth and differentiation, composite spheroid systems offer a promising approach to enhance the efficacy of treatments for articular cartilage injuries. 14 Moreover, biofabrication techniques, such as 3D bioprinting and microfluidics, further enable the creation of composite spheroid systems with complex architectures that replicate the zonal organization and biomechanical properties of native cartilage.
To further clarify their distinct advantages, Table 1 compares five representative 3D cell culture platforms (composite spheroids, monoculture spheroids, organoids, scaffolds, and hydrogels) in terms of cell viability, ECM production, mechanical integrity, drug resistance, reproducibility, and cellular complexity.22–25 For instance, composite spheroids generally exhibit superior cell viability (90%–95%), ECM production, and drug resistance compared to other systems.
Comparison of characteristics of composite spheroids with other 3D cell culture models.

This review aims to provide a comprehensive overview of recent advancements in biomaterial-based composite spheroids for articular cartilage regeneration. We discuss the development and application of 3D spheroid culture techniques, explore strategies for incorporating biomaterials, and assess their efficacy in animal models. Finally, we discuss the challenges and future directions in optimizing composite spheroids for clinical translation, focusing on standardizing protocols, enhancing biomimicry, and leveraging advanced biofabrication technologies. To ensure a comprehensive and up-to-date overview, this review focused on literature published between 2018 and 2024, identified through structured searches in PubMed and Web of Science databases. Search terms included “composite spheroids,” “cartilage tissue engineering,” “3D spheroid culture,” and “biomaterial-embedded spheroids” were used to filter relevant articles. Priority was given to original research articles that examined spheroid formation, biomaterial integration, 3D bioprinting applications, or preclinical evaluation in cartilage repair.
Spheroid culture methods for articular cartilage tissues
Principles of spheroid-based approaches in cartilage tissue engineering
The functional capacity of spheroids in cartilage repair is critically determined by their cellular composition and the intricate interactions among cells within the cell aggregate. Typically, the selection of cells for spheroid production includes stem cells, chondrogenic cells, and osteogenic cells, which individually offer distinct advantages.26,27 Stem cells, particularly MSCs, are highly favored for their ability to differentiate into chondrocytes and osteoblasts, making them highly suitable for osteochondral tissue repair. 28 Chondrogenic cells, such as chondrocytes or pre-differentiated MSCs, can produce key ECM components, such as collagen type II and proteoglycans, both of which are essential for the mechanical strength and integrity of cartilage. 29 Osteogenic cells, frequently incorporated in biphasic or composite spheroids for osteochondral repair, contribute to subchondral bone regeneration by providing mechanical support to the overlying cartilage. 30
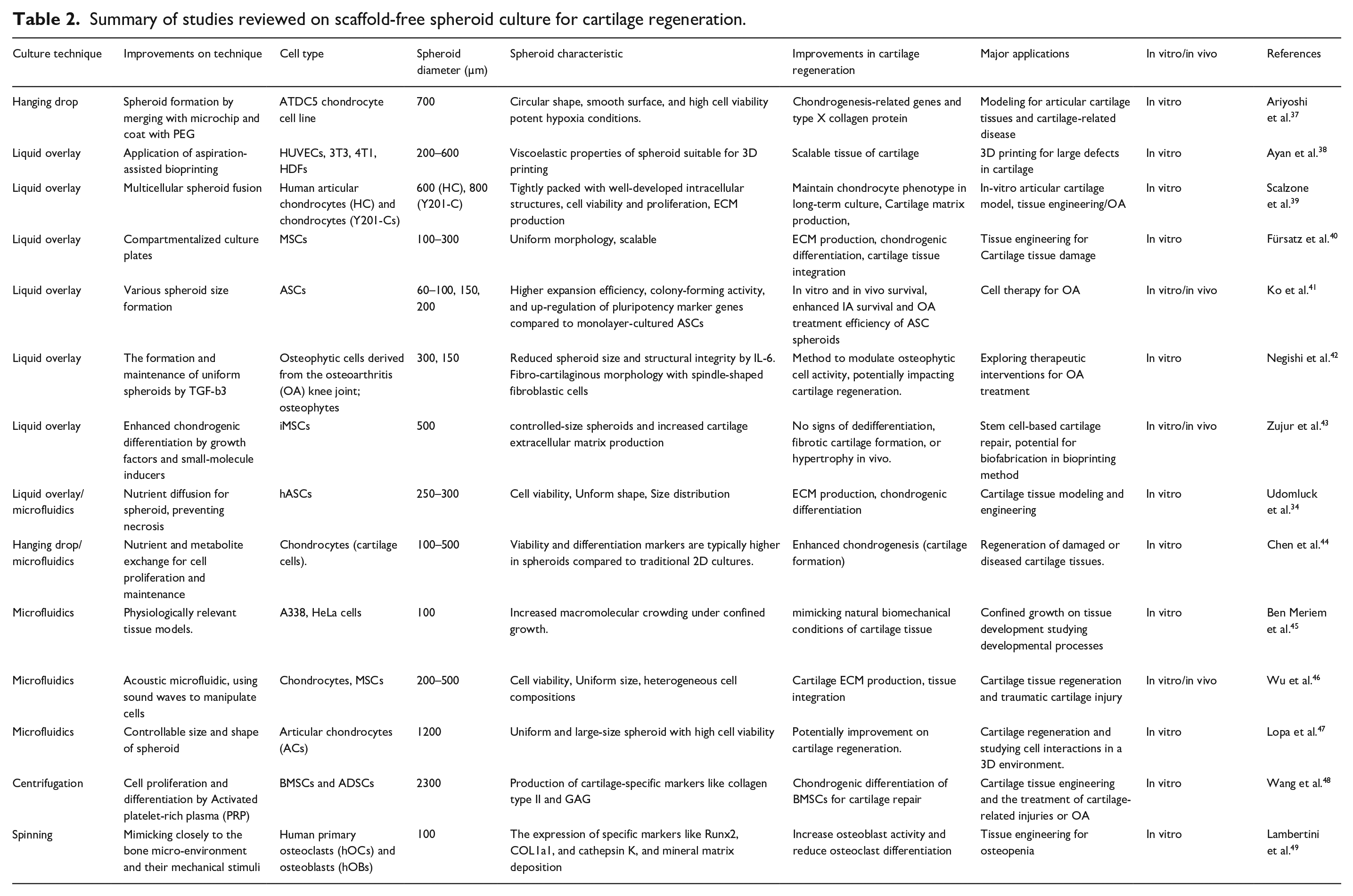
Scaffold-free spheroids represent a 3D cellular self-assembly without external scaffolds for their structural integrity. These self-supporting constructs facilitate cell-cell interactions that occur in the native tissue environment. Spheroids are formed via cell-cell adhesion mediated by cadherins, particularly N-cadherin and E-cadherin, leading to the establishment of the cohesive structures and cellular communication.31,32 This spheroid formation process relies on cell aggregation as the fundamental principle. Several techniques have been developed to produce scaffold-free spheroids, including a low-adhesion surface, rotational force, and microfluidic device as shown in Figure 1. Individual techniques utilize distinct physical mechanisms to promote cell aggregation. For instance, the hanging drop method, using small droplets of a cell suspension placed on the underside of a Petri dish lid, relies on gravity to facilitate cell aggregation. 33 Additionally, microfluidic-based techniques provide precise control over spheroid size and uniformity, using microscale channels to guide the cell assembly and enable high-throughput production of consistent spheroids. 34 Rotary suspension cultures utilize gentle agitation of cell suspension to prevent cell attachment to surfaces while inducing cell aggregation.35,36 Table 2 provides a summary of the highlighted studies on scaffold-free spheroids and their applications in cartilage tissue engineering.

Scaffold-free spheroid formation techniques categorized based on the mechanisms inducing cell aggregation: (a–c) low-adhesion surfaces methods, including (a) hanging drops, (b) liquid overlay, and (b) liquid overlay with microwells; (d–f) rotational force methods, including (d) centrifugation or pellet culture, (e) spinneret, and (f) rotational flask; and (g–i) microfluidic systems, including (g) droplet-based microfluidics, (h) side-chamber microfluidics, and (i) semicircular weir microfluidics.
Summary of studies reviewed on scaffold-free spheroid culture for cartilage regeneration.
Low-adhesion surface techniques
Low-adhesion surface techniques, such as hanging drop and liquid overlay methods (Figure 1(a)–(c)), are widely utilized for their simplicity, cost-effectiveness, and minimal need for specialized equipment. These methods rely on reduction of cell attachment to substrate surfaces, thereby promoting cell aggregation and spheroid formation. The hanging drop technique involves the placement of small droplets of cell suspension on the underside of Petri dish lid, in which cell aggregation is induced by surface tension and gravity. 50 However, this technique has limitations, particularly in culture media exchange, leading to insufficient nutrient supply and eventual cell death. As an alternative, a tapered tube design has been developed to enhance the media exchange rate in the hanging-drop system. Huang et al. 51 demonstrated that over 90% of MSCs remained viable after 3 days, and less than 40% were dead after 7 days.
The liquid overlay technique uses low-adhesion materials, such as polydimethylsiloxane, to fabricate hemispherical microwells that facilitate cell aggregation. This technique allows for medium exchange and control over spheroid formation in a wide size range. 52 Microwells are frequently produced by soft lithography, and their microstructures can be varied to mimic the native tissue structures. 34 For example, concave microwells with microfluidic-like structure were recently constructed using molds prepared using metal beads and central processing unit pins as molds. These novel microwells enhance nutrient exchange and support coculture platforms that replicate the structural and functional characteristics of osteochondral tissues. In addition, the low-adhesion technique can be further combined with a microneedle array. This methodology, known as the “Kenzan” technique, uses a microneedle array to align and shape microspheroids in a predefined 3D features by fusing microspheroids into a scaffold-free neotissue. The resulting construct can be removed while maintaining its defined size and shape (Figure 2(a)). 53 Recent innovations in low-adhesion techniques, such as the incorporation of microwells with microfluidic-like tunnels and the microneedle-based Kenzan system, have significantly advanced the potential of MSC spheroid cultures. These methods can improve spheroid viability and functionality and hold great promise for applications in cartilage repair and tissue engineering.

Advanced research on scaffold-free spheroids generated using various techniques. (a) Kenzen techniques using a microneedle array to organize spheroids for large-scale tissue repair. 53 (a1) Microspheroids arranged on a microneedle array. MSCs spheroids (green) are positioned across three vertical layers, enabling the formation of high-density tissue constructs. (a2) Side view of the microneedle array with vertically aligned mature spheroid construct (black arrow), demonstrating precise layer-by-layer assembly. (a3) Top view of the detached infrapatellar fat pad-MSC (IPFP-MSC) construct after 4 days of culture on the array, illustrating a compact and integrated tissue structure. (a4) Safranin O staining of IPFP-MSC constructs cultured in chondrogenic medium for 3 weeks, indicating overall tissue architecture and proteoglycan distribution. (a5) High-magnification inset revealing mixed chondrocyte-like and fibroblast-like cell morphology and weak GAG staining, indicative of partial or heterogeneous chondrogenic differentiation. (b) Spheroid production using a microgravity bioreactor. Immunofluorescence staining for pluripotent markers (Oct4, Nanog, Sox-2, and Rex-1) on the spheroids cultured for 5 days. 54 (c) Acoustic levitation for self-organization of MSC sheets into spheroids and viability assessment. (c1) Experimental setup for the acoustic levitation system for spheroid formation. (c2) Time-lapse images of the transition from monolayers to stable spheroids. (c3) Axial and radial dimensions of cell aggregates stabilizing after 15 h of levitation. (c4) Confocal and fluorescence images of spheroids post-levitation, exhibiting live cells on the spheroid surface. (c5) Histological analysis of MSC spheroids embedded in fibrin after 24 h of levitation, visualized with Hematoxylin and Eosin (H&E) staining. (c6) Ki67 staining for cell proliferation in spheroids. 55
Rotational force techniques
Rotational force techniques employ liquid torque generated by centrifugal or agitation processes to induce cell aggregation into spheroids. These methods typically include centrifuges, spinnerets, and rotational flasks (Figure 1(d)–(f)). They are advantageous for high-density cell culture and large-scale spheroid production. The dynamic fluid movement enhance cell aggregation preventing cell attachment to surfaces. In addition, these methods permit dynamic culture that promotes nutrient transfer and waste removal. Spinner culture systems have been widely used for culturing various cells, including MSCs. 56 However, shear stress caused by fluid motion can cause cell damage. To minimize shear stress issues and maintain cell viability, microgravity bioreactors have been developed. For example, Zhang et al. 54 demonstrate that microgravity bioreactors could effectively preserve the stemness of human adipose-derived stem cells (hASCs) in spheroid cultures (Figure 2(b)).
Although the spinning or rotating bioreactor methods enable large-scale spheroid production, concerns remain regarding spheroid stability and cell viability in these rotational force techniques. Recently, He et al. 35 examined the aggregation kinetics of cells in spinner flasks to finely control over cell aggregation and spheroid dimensions using bovine articular chondrocytes (bACs), rabbit bone marrow-derived MSCs, or a combination of the two. Lower agitation rates and higher cell inoculation densities lead to increases in aggregation efficiency and spheroid size. They found that integrin β1 and cadherin molecules played critical roles in cell-cell adhesion and spheroid formation, respectively. Furthermore, co-aggregates different cell types (i.e. bACs and rMSCs) were successfully produced in spinner flasks. These co-aggregates exhibited a hierarchical structure, in which rMSCs and hACs were located in the core and on the periphery of spheroids, respectively, mimicking the zonal architecture of native cartilage. 57
Microfluidic techniques
Microfluidic systems have emerged as powerful tools for generating scaffold-free spheroids because of their ability to precisely control spheroid dimensions, fluid dynamics, and the spatial distribution of cells. Especially, these microfluidic systems utilize microscale channels and chambers to manipulate small fluid volumes, allowing for promoting controlled cell aggregation (Figure 1(g)–(i)). Moreover, the precise control over nutrient and oxygen delivery in the microfluidic systems enhances cell viability and functionality, making them suitable for the production of high-quality spheroids for cartilage tissue engineering. Microfluidic devices possess specialized characteristics, such as low-adhesion surfaces and flow patterns, which enhance cell-cell interactions and improve spheroid stability. For example, a perfusable microfluidic device was designed to generate size-controlled 3D spheroids from hACs obtained from osteoarthritis patients. 47 This system demonstrated successful long-term culture and stability of 3D spheroids suitable for cartilage tissue engineering applications. In continuous perfusion culture, AC spheroids remained viable and metabolically active for over 3 weeks. Additionally, immunohistochemical staining confirmed the deposition of cartilage-specific ECM components. Interestingly, the glycosaminoglycan (GAG) content in the spheroids were found to be dependent on the perfusion flow rate. 47
Another promising advancement in microfluidic techniques is the use of acoustic levitation for spheroid production with high biocompatibility and adaptability. Acoustic-based methods enable contactless cell manipulation by using acoustic forces. 55 During acoustic levitation, the acoustic forces exerted on suspended cells induce clustering and trapping the cells at different pressure nodes within a resonant cylindrical cavity at different pressure nodes within a resonant cylindrical cavity. Spheroids are formed via this controlled self-organization and can be further maintained and cultured in a perfused culture media system under levitation. A multi-trap acoustic levitation, termed CCMAL (contactless cell manipulation and acoustic levitation) system, enables the generation and growth of large, uniform, and reproducible spheroids (Figure 2(c)). This highly scalable CCMAL system allows for the simultaneous cultivation of numerous spheroids, making it a promising tool for large-scale cartilage tissue engineering. 55
The versatility and precision of microfluidic systems, including both perfusable microfluidic devices and acoustic levitation platforms, provide unique advantages for generating robust and biologically functional spheroids. These innovations offer significant potential for advancing cartilage regeneration by facilitating the creation of high-quality spheroids that replicate the native cartilage microenvironment. While microfluidic techniques offer precise control over spheroid size and cellular composition, their full potential is often realized when integrated with biomaterial-based strategies. This integration serves as the foundation for composite spheroid culture, providing more physiologically relevant and structurally robust platforms for cartilage regeneration.
Composite spheroid culture with biomaterials
Cellular components alone are insufficient for replicating the complexity of native tissues. The incorporation of biomaterials into spheroid cultures can enhance their biomimetic properties, creating composite spheroids that better resemble the native ECM and support functional tissue regeneration. 58 The scaffold materials used in composite spheroid production play important roles in cell growth, differentiation, and ECM synthesis. For effective cartilage regeneration, scaffolds should possess interconnected pores to facilitate cell signaling, nutrient and oxygen exchange, fluid permeability, and neo-vascularization within the surrounding tissues. 59
Scaffold materials can be broadly categorized into natural and synthetic scaffolds, which offer distinct advantages and limitations, respectively. Natural scaffolds (e.g. alginate, hyaluronic acid, and collagen) provide a biocompatible and bioactive environment that promotes cell adhesion and ECM production.60–62 These materials are particularly favored for cartilage regeneration because of their inherent biological abilities to support cellular signaling and matrix synthesis. However, natural materials frequently lack mechanical strength, limiting their uses in load-bearing applications. On the other hand, synthetic scaffolds (e.g. poly-L-lactic acid (PLLA) and polyethylene glycol (PEG)) offer superior mechanical properties, tunable degradation rates, and ease of chemical modification. These characteristics make them suitable for applications that require structural integrity and stability.15,63 However, synthetic materials lack inherent bioactivity and thereby necessitate functionalization with growth factors or ECM components to enhance their biological functions. Importantly, the physicochemical properties of scaffold materials are critical in modulating spheroid behavior and enhancing chondrogenic potential. Recent studies have shown that specific material characteristics, such as stiffness, degradability, and biochemical functionality, play essential roles in regulating cell-matrix interactions and ECM production within composite spheroids. For example, hydrogels with intermediate stiffness (~15–20 kPa) closely mimic the native cartilage microenvironment and promote optimal mechanotransduction. Gelatin methacrylate (GelMA) hydrogels tuned to ~17 kPa significantly enhanced COL2A1 and ACAN expression in MSC spheroids compared to softer and stiffer substrates. 64 Degradability is another key parameter. Biodegradable matrices, such as oxidized alginate or hyaluronic acid-based hydrogels, enable sustained ECM deposition, and facilitate tissue remodeling. Decarli et al. 15 reported a 2.3-fold increase in GAG content and improved spheroid fusion after 14 days in degradable hydrogels compared to non-degradable controls. Additionally, the biological activity of the scaffold strongly influences spheroid function. The incorporation of RGD motifs or cartilage-derived ECM components enhanced integrin-mediated cell adhesion, leading to more compact spheroids and increased expression of chondrogenic markers. 65 Koh et al. 66 showed that RGD-modified scaffolds resulted in a 1.8-fold increase in GAG content and improved spheroid cohesion after 14 days in vitro. These findings collectively suggest that the selection and tuning of specific material properties are essential to maximize the regenerative potential of spheroid-based cartilage constructs. Accordingly, the strategic combination of appropriate cells and functionally tailored scaffold materials allows researchers to optimize composite spheroids for effective cartilage tissue regeneration. 67
For the formation of functional composite spheroids, the interactions between cells and scaffold materials are essential. Especially, integrins (e.g. α5β1 and αVβ3) mediate cell-ECM interactions by binding to ECM proteins (e.g. fibronectin, collagen, and laminin). These interactions regulate cell adhesion, migration, and mechanotransduction, ultimately promoting cartilage-specific gene expression and tissue formation.61,68,69 This section explores three major techniques for scaffold-based spheroid cultures (Figure 2) – (i) spheroid-encapsulated scaffolds (encapsulation of spheroids within hydrogel-based scaffolds to enhance stability and control biomolecule release), (ii) biomaterial-incorporated spheroids (integration of microparticles or nanofibers into spheroids to improve mechanical properties and ECM production), and (iii) macroscale spheroids via 3D bioprinting (fabrication of large-scale tissue constructs by precisely arranging spheroids within bioprinted scaffolds). A summary of recent studies on composite spheroids, including their design principles and applications, is provided in Table 3.
Summary of studies reviewed on composite spheroid culture for cartilage regeneration.

Biomaterial-incorporated spheroids
The incorporation of biomaterials into spheroids offers a versatile approach to enhancing their functionality and structural complexity. Various biomaterials (e.g. spherical particles and fiber fragments) serve distinct roles within spheroid structures (Figure 3(a)–(c)). Integration of small sized biomaterials and spheroids can enhance oxygen and nutrient diffusion, especially in larger spheroids used for cartilage regeneration. For example, Kim et al. 21 incorporated gelatin microparticles into adipose-derived stem cell spheroids and found increases in cell-interactive surface area and diffusion space. Gelatin microparticle significantly enhanced cell proliferation and reduced the risk of necrosis in the spheroid core, thereby improving overall spheroid viability. Biomaterials incorporated within spheroids allow for direct cell-biomaterials interactions throughout the entire structure, facilitating uniform cellular responses and enhancing the therapeutic potential of the spheroid.

Composite spheroid culture with biomaterials classified by scaffold integration and spheroid arrangement. (a–c) Biomaterial incorporation within spheroids. Green bars represent cell-ECM interactions, while red bars represent cell-cell interactions. (a) Core-shell composite spheroids, (b) particle-incorporated spheroids, and (c) fiber-incorporated spheroids. (d–f) Spheroid encapsulation in biomaterials. (d) Spheroid encapsulated within hydrogels, (e) compartmentalized constructs of composite spheroids, and (f) spheroid growth and formation within hydrogels. (g–i) Spheroids as building blocks for macrostructures. (g) Spheroid fusion with biomaterial-based structures, (h) scaffolded spheroid bioassembly, and (i) spheroids combined with granular hydrogel.
Another key advantage of biomaterial-incorporated spheroids is their ability to deliver bioactive molecules in a controlled and localized manner. Encapsulation of growth factors (e.g. TGF-β3) and ECM proteins (e.g. matrilin-3) within microparticles enables the sustained release of these molecules and the enhancement of cellular responses and tissue regeneration. 81 This strategy not only improves spheroid functionality but also ensures consistent and prolonged delivery of regenerative cues to the encapsulated cells. In addition to microparticles, nanofibers have been successfully incorporated into composite spheroids to replicate the structural and functional complexity of native tissues, such as osteochondral tissue. For example, Lee et al. developed composite spheroids containing the nanofibers coated with BMP-2 and TGF-β3 to promote localized, controlled differentiation into osteogenic or chondrogenic lineages (Figure 4(a)). This approach allows for the creation of a biphasic construct that mimics the layered architecture of cartilage and subchondral bone, addressing key challenges in osteochondral tissue repair, such as delamination and inconsistent differentiation. 82 The incorporation of growth-factor-loaded fibers within the spheroids provides structural support and the sustained release of bioactive molecules, which promotes effective stem cell differentiation and tissue regeneration. This approach can overcome common issues, such as diffusion limitations and inconsistent growth factor delivery. 63 Despite these advancements, several limitations remained unaddressed, including long-term safety and biodegradability, and optimal scaffold design. Future studies are needed to develop biomaterial formulations and spheroid technologies to enhance their effectiveness in cartilage tissue engineering.

Representative advance research on composite spheroids using various techniques. (a) Polydopamine-coated fragmented fiber-incorporated spheroids designed to enhance chondrogenesis via sustained growth factor deliverys. 63 (a1) Schematic illustration of the formation of chondrogenic or osteogenic composite spheroids. (a2) Alizarin Red S staining of cross-sectioned spheroids of PS; polydopamine-coated fragmented fiber-incorporated hASCs spheroids), PS/B (PS cultured with BMP-2 containing media), and BS (BMP-2-immobilized fiber-incorporated hASCs spheroids. (a3) Alcian Blue staining of cross-sectioned spheroids of PS, PS/T (PS cultured with TGF-β3-containing media), and TS (TGF-β3-immobilized fiber-incorporated hASCs spheroids). Scale bar = 200 µm. (b) In situ spheroid formation in in-air bioprinted compartmentalized hydrogels. (b1) Schematic of spheroid formation within compartmentalized hydrogel. (b2) Brightfield micrographs of encapsulated chondrocytes at days 0, 1, and 21. 71 (c) Granular composite approach and component characterization. (c1) Schematic overview of granular composite design, in which MSC spheroids and norbornene-modified hyaluronic acid (NorHA) microgels were mixed to offer injectability for delivery to defects or molds, cell–cell contacts for enhanced chondrogenesis and spheroid fusion for tissue formation, and interparticle crosslinking to stabilize the composites. (c2) Schematic of granular composites over time, in which spheroid fusion and growth lead to cartilage tissue formation throughout the granular hydrogel. (c3) Representative histological images of granular composites for 20:80 and 35:65 spheroid to microgel volume ratios at day 56 stained for Alcian Blue (sGAG) and collagen II. Scale bars = 2 mm, and inset scale bars = 500 µm. 83
Spheroid-encapsulated scaffolds
Spheroid-encapsulated scaffolds for cartilage regeneration involve embedding spheroids, typically composed of chondrocytes or MSCs, within a 3D biomaterial scaffold in various ways (Figure 3(d)–(f)). Encapsulation within scaffolds supports the spheroids in maintaining their structural integrity while benefiting from the scaffold’s mechanical support and controlled degradation. 50 This technique preserves the chondrocyte phenotype, promotes ECM production, and supports cell-cell interactions. For example, van Loo et al. developed a biomaterial-based scaffold integrated with MSCs or chondrocytes spheroids. Specifically, alginate hydrogels were used as the compartmentalizing biomaterial to form a supportive matrix for spheroid production and culture (Figure 4(b)). The in-air microfluidic technique enabled rapid and scalable production of spheroid-forming compartmentalized hydrogels at ultra-high throughput rates. The method facilitates the formation of clinically sized cartilage tissues with high viability and enhanced expression of chondrogenic markers (e.g. SOX9, ACAN, and COL2A1), while simultaneously reducing the expression of fibrocartilage markers (e.g. COL1), suggesting more effective cartilage regeneration. 71 In addition, Muttigi et al. incorporated matrilin-3, a non-collagenous ECM protein, into the methacrylated hyaluronic acid hydrogel to create a biomimetic environment for the chondrogenic differentiation of the adipose derived-MSC (Ad-MSC) spheroids. The spheroids within the hydrogel exhibited cartilage-like ECM production, with deposition of Type II collagen and aggrecan. Furthermore, matrilin-3-primed Ad-MSC spheroids effectively prevented chondrocyte hypertrophy, a process associated with endochondral ossification and cartilage degeneration, as confirmed by histological analysis. 84 Despite these advantages, spheroid-encapsulated scaffolds for cartilage regeneration are necessary to further improve nutrient diffusion especially in large constructs.
Large-scale spheroid formation through scaffolds combination
The combination of spheroids and scaffolds can improve cell-ECM interactions between spheroids and support their mechanical properties, facilitating the production of large spheroid-based constructs. To this end, various approaches have been developed, including spheroid fusion with biomaterial structures, scaffolded spheroid bio assembly, and spheroid integration with granular hydrogel (Figure 3(g)–(i)). Recently, 3D bioprinting with spheroid-scaffold combinations has garnered great attention to fabricate articular cartilage tissues. This technique involves printing chondrocyte spheroids and biomaterials that can be assembled into large tissue constructs. Spheroids, with their defined geometry and concentric organization of distinct cell populations, can closely mimic the native in vivo environment, making them ideal building blocks for tissue engineering. For example, Decarli et al. 15 demonstrated the bioprinted stem cell spheroids and subsequent chondrogenic differentiation. Human MSC spheroids were bioprinted into precisely designed geometries using a biomaterial ink composed of gelatin-methacryloyl and hyaluronic acid methacrylate. After bioprinting, the constructs containing the embedded hMSC spheroids were cultured in a chondrogenic differentiation medium. The combination of bioprinting and post-printing chondrogenic differentiation of hMSC spheroids enabled the generation of larger, more homogeneous cartilage tissue constructs in a controlled and reproducible manner.
Furthermore, biomaterials inks can provide spheroids with mechanical support and promote cell-matrix interactions appropriate for the development of functional cartilage tissues. In addition to 3D bioinks, injectable granular composites, which combine spheroids with similarly-sized particles, have been introduced to support the mechanical properties of constructs for cartilage tissue regeneration. Caprio et al. demonstrated the combination of aggregated adult porcine MSC cells spheroids and similarly-sized hyaluronic acid-based microgels for long-term chondrogenic culture. These spheroid-based composites exhibited mechanical properties comparable to native cartilage and effective tissue integration (Figure 4(c)). 83 Moreover, Kim et al. introduced the High-throughput Integrated Tissue Fabrication System for Bioprinting (HITS-Bio), a novel platform designed to enhance the scalability and efficiency of spheroid-based bioprinting. HITS-Bio utilizes a digitally controlled nozzle array to simultaneously position multiple spheroids, achieving a bioprinting speed 10 times faster than conventional systems while maintaining high cell viability (>90%). The system successfully fabricated 1 cm³ cartilage constructs comprising approximately 600 chondrogenic spheroids within 40 min. These constructs exhibited robust ECM deposition and cartilage-specific marker expression, indicating successful chondrogenic differentiation. 85 Complementing this approach, scaffolded spheroids (S-SPH) composed of MSCs have been applied as modular building blocks for bottom-up cartilage tissue engineering. These spheroids were assembled into anatomically relevant constructs using 3D printing techniques, resulting in improved spheroid fusion and ECM deposition. Notably, in vivo implantation in a rat cartilage defect model demonstrated enhanced tissue integration and hyaline-like cartilage formation, highlighting the translational potential of S-SPH-based strategies. 86 To bridge the gap between engineered construct fabrication and clinical application, future research should focus on optimizing biomaterial compositions that mimic the zonal architecture of native articular cartilage, enhance integration with host tissue, and improve the mechanical resilience of printed constructs. These advancements will be essential to fully realize the regenerative potential of composite spheroids in preclinical models and ultimately in cartilage repair strategies.
Regeneration of articular cartilage using composite spheroids
Composite spheroids enhance cartilage regeneration through a combination of biophysical structuring and biochemical signaling mechanisms that arise from the integration of cells and supportive biomaterials. The spheroid architecture facilitates high-density cell–cell interactions and mimics the early condensation phase of mesenchymal development, which is a critical step in initiating chondrogenesis. When embedded with bioactive biomaterials, such as GelMA, hyaluronic acid, or RGD-functionalized microgels, composite spheroids exhibit enhanced cell–matrix adhesion, improved retention and localized presentation of growth factors, and finely tunable mechanical cues. These microenvironmental cues activate mechano-transduction pathways (e.g. YAP/TAZ and integrin–FAK signaling), which subsequently upregulate key chondrogenic markers (e.g. SOX9, COL2A1, and ACAN) and promote ECM synthesis.87,88 The use of degradable materials further allows for dynamic remodeling and facilitates nutrient and oxygen diffusion throughout the spheroid, preventing hypoxic core formation and promoting uniform tissue maturation. The modular nature of composite spheroids enables spatial organization during 3D bioprinting, thereby allowing for the fabrication of constructs that replicate the zonal organization of native cartilage. 89 These multifaceted mechanisms collectively contribute to enhanced cartilage-specific tissue formation and establish a strong foundation for evaluating their therapeutic efficacy in vivo, as explored in the following section using murine, rabbit, and large animal models.
Animal model for articular cartilage regeneration
In preclinical studies, composite spheroids have been evaluated for their efficacy in repairing articular cartilage using various animal models. These models provide a platform to assess the effectiveness of different composite formulations and their ability to integrate into and regenerate damaged cartilage. Animal studies have demonstrated that composite spheroids can effectively repair cartilage defects, as evidenced by better histological and biomechanical results compared to other cell-based therapies. 90 Various animal models used for the evaluation of cartilage regeneration can be categorized into three groups based on animal size – murine, rabbit, and large animals (goat, sheep, and horse). 91 Murine models (mice and rats) are ideal for assessing biocompatibility and the initial regenerating function of composite spheroids due to their short breeding cycle and rapid tissue repair. Rabbit models, with their larger joint structures, provide a more relevant platform for evaluating the biomechanical properties of composite spheroids. 92 Large animal models (goat, sheep, and horse) are commonly used to assess the ability of composite spheroids to repair load-bearing joints as their cartilage thickness and joint size are more comparable to those of humans. 93 Among them, horse models are particularly useful for testing composite spheroids under mechanical stress because of their robust cartilage and joint mechanics useful for assessing functional integration. 6 Each type of model plays a distinct role in evaluating various functions of biomaterials within composite spheroids via different mechanisms. Optimization of spheroid composition and mechanical properties will allow researchers to develop more effective cartilage repair strategies, thereby offering advanced treatment options for arthritis patients. The applications of composite spheroids in murine, rabbit, and large animal group are discussed, highlighting the recent advancements.
Murine animal models
Murine models are useful for determining whether biomaterials can support spheroid cell survival and initial cartilage matrix formation. The contribution of murine models to in vivo behavior includes the determination of composite spheroid integration into cartilage defects at an early stage.76,84,94 Due to their fast-healing capacity, mice allow researchers to observe initial cartilage regeneration and tissue formation. In another study, nude mice were used as the animal model to evaluate the in vivo survival rate and functionality of composite spheroids produced from chondrocytes encapsulated with hyaluronic acid microparticles. Composite spheroids consisting of chondrocytes and hyaluronic acid microparticles, when implanted into nude mice, exhibited a 2.5-fold increase in GAG content compared to control groups. These constructs also demonstrated sustained retention at injection sites for up to 4 weeks, whereas spheroids without hyaluronic acid persisted for only 2 weeks post-injection. 14 Murine models can also be used to assess the stability of regenerating cartilage. For example, immune-deficient nude mice were used to test the efficacy of decellularized sturgeon cartilage extracellular matrix (dSCECM) scaffolds with chondrocyte spheroids in inhibiting chondrocyte hypertrophy and promoting cartilage regeneration. After 4 weeks of subcutaneous implantation, dSCECM scaffold-derived specimens formed cartilaginous tissues without bone-like tissue in histological staining results (Safranin O-Fast Green and Alizarin Red S staining). In addition, dSCECM constructs showed reduced expression of chondrocyte hypertrophy markers, such as COL10A1 and RUNX2. In contrast, the specimens from type I collagen scaffolds used as a control showed bone-like tissue with substantial calcium deposition (Figure 5(a)). 75 Overall, the ability of spheroids to stimulate the production of ECM components can be assessed rapidly through murine models. However, the small size of mice joints limits the applicability of large composite spheroids, and their short lifespan makes it difficult to assess long-term performance and the repair of chronic cartilage damage. Moreover, while mice are genetically tractable and suitable for high-throughput studies, their extremely small joint sizes and ultra-thin cartilage layers (~30–100 µm), making them poor biomechanical analogs of human knee. 95 Additionally, their relatively immature immune system and rapid cartilage turnover may obscure long-term inflammatory responses that are critical for evaluating biocompatibility of implants. 6

Representative results of composite spheroids in animal models for cartilage regeneration. (a) Mice model: (a1) Gross appearance of COL I scaffolds and decellularized sturgeon cartilage extracellular matrix (dSCECM) scaffolds compared to chondrocyte-seeded COL I and dSCECM constructs after 4 weeks of subcutaneously implantation in mice in mice under normal or hypertrophic conditions. (a2) Histological analysis of specimens from COL I and dSCECM scaffolds after 4 weeks of subcutaneous implantation. Staining includes H&E, Safranin O-Fast Green, and Alizarin Red S staining. Blue arrows indicate bone like tissue formation and calcification zone. 75 (b) Rabbit model: (b1) Optical images of osteochondral tissues harvested from Chamber group (control), polydopamine-coated fragmented fiber-incorporated hASCs spheroids (PS), and BMP/TGF-β3-immobilized fiber-incorporated hASCs spheroids (BS/TS) groups. (b2) X-ray images of osteochondral defects (red boxes indicate the defect area). (b3) 3D reconstruction from micro-CT (μCT) analysis of the untreated defects (Defect), Chamber, PS, and BS/TS groups. (b4) Quantification of bone volume fraction (BV/TV) from μCT analysis (n = 7).
Rabbit animal models
Rabbits provide a useful model for assessing the abilities of the biomaterial scaffolds to promote the cell retention in the defect site to guide the formation of hyaline-like cartilage. For example, composite spheroids composed of hASCs and polydopamine (PD)-coated fibers immobilized with bone morphogenetic protein-2 (BMP-2), as an osteogenic spheroid, or immobilized with transforming growth factor-beta 3 (TGF-β3), an a chondrogenic spheroid, have been used in male New Zealand white rabbits to regenerate osteochondral tissue at a cylindrical defect site on femoral trochlear groove (Figure 5(b)). 63 Micro-computed (μCT) analysis revealed a 65% improvement in bone volume/total volume (BV/TV) in osteochondral defects treated with BMP-2/TGF-β3 composite spheroids compared to untreated controls. Histological evaluation exhibited uniform COL2 expression and enhanced GAG deposition, indicating the formation of hyaline-like tissue. The spheroids facilitated organized tissue regeneration, with cartilage formation in the TGF-β3-treated layer and strong trabecular bone formation in the BMP-2-treated layer. 63 These findings support the utility of the rabbit model in evaluating spatially organized biphasic regeneration by composite spheroids.
Moreover, the rabbit joint environment provides a more comprehensive platform for evaluating how the spheroids interact with surrounding tissues and whether the biomaterial enhances cellular integration into the defect site. For instance, New Zealand white rabbits have been employed to test composite spheroids made of GelMA hydrogels embedded with microspheres loaded with TGF-β3, insulin-like growth factor 1, and platelet-derived growth factor-BB for sequential growth factor release for enhanced cartilage regeneration. These composite spheroids promoted endogenous stem cell recruitment and macrophage polarization toward a regenerative M2 phenotype, which is essential for reducing inflammation and facilitating cartilage repair. 85 Despite the thinner cartilage in rabbits compared to humans, this model remains valuable for evaluating biomaterial biocompatibility, spheroid-mediated healing, and early-stage cartilage formation, making it a critical preclinical tool before transitioning to larger animal models or clinical trials. Rabbit models offer moderate improvements, with cartilage thicknesses of ~300–500 µm and easier surgical manipulation. However, they typically regenerate primarily with fibrocartilage rather than hyaline cartilage and exhibit species-specific immune responses to xenogeneic materials that differ from those in humans. Furthermore, their quadrupedal gait results in joint loading profiles that do not accurately reflect human biomechanics, which can potentially affect the mechanical assessment of engineered constructs. 96
Large animal models
Large animal models, such as goats, sheep, and horses, are increasingly used in preclinical testing of composite spheroids due to their closer mimicry of human joint sizes and cartilage thickness compared to small animals. Composite spheroids in goat models have demonstrated the ability to restore cartilage tissue and support the formation of hyaline cartilage over longer periods. For example, goats were used to test the long-term efficacy of a bi-layered scaffold with hydrophilic-hydrophobic structure made from polycaprolactone (PLGA-g-PCL) and PEG, which was packed with goat bone marrow stem cells (gBMSCs) spheroids to regenerate temporomandibular joint (TMJ) condyle cartilage. The composite spheroids, formed by gBMSCs in the bottom layer of PLGA-PCL-PEG, presenting a hydrophilic environment created by a high content of PEG, promoted the chondrogenic differentiation necessary for hyaline cartilage development. After 2 months of implantation at the TMJ condyle defect site, the scaffold facilitated defect repair with the formation of newly formed neo-cartilage. Histological scoring revealed a 70% improvement in matrix uniformity and cartilage thickness compared to scaffold-only groups, closely mimicking native TMJ cartilage, as confirmed by positive staining for Col II, Col I, and GAG (Figure 6(a)). 73

Representative results of composite spheroids in large animal models for cartilage regeneration. (a) Goat model: Digital images of goat TMJ condyle tissues from Health (native TMJ condyle tissue from healthy goats), 1# (Defects created without treatment), 2# (Defects treated with bilayered scaffolds only), and 3# (Defects treated with bilayered scaffolds containing gBMSCs cultured for 14 days in fibrocartilage differentiation medium). Histological staining includes H&E, Col II, Col I, and GAG. Black triangles indicate the boundary between native condyle cartilage and defect site. 73 (b) Horse model: (b1) Preparation of at 3D construct and surgical procedure. Synovial membrane-derived MSCs (SM-MSCs) formed a spheroid with a diameter of approximately 600 μm. A cylindral construct (6.3 mm in diameter, 5 mm in height) was produced for implantation. (b2) A cylindrical osteochondral defect was created in each medial condyle of each hind limb before implantation. The construct was autografted into the defect in the right hind limb, while the left limb served as a control without implantation. The white region under the construct represents the construct sill (asterisk) and the clear region beneath the sill represents the mold (double asterisks). 97
The sheep model allows researchers to assess biomaterial-based spheroids treatments for osteochondrogenic tissue, which is essential for functional articular cartilage regeneration. For instance, composite spheroids made of MSCs and a hydrogel matrix composed of alginate and hyaluronic acid were implanted at osteochondral defects in the medial condyle of skeletally mature sheep (“Ile de France”). The study demonstrated significant cartilage and subchondral bone regeneration in the sheep model. MRI and micro-CT scans indicated that the treated joints had a significantly larger area of regenerated articular cartilage compared to untreated controls. After 6 months of post-implantation, the formation of hyaline-like cartilage and proper integration with subchondral bone was observed in histological analysis. 98
Horse model is particularly advantageous for evaluating spheroids in osteochondral regeneration due to its joint environment that can closely mimic human joints under load-bearing conditions, thereby allowing for better assessment of cartilage and subchondral bone integration. For example, a scaffold-free 3D construct made of synovial membrane-derived MSC (SM-MSC) spheroids for osteochondral regeneration were implanted at a cylindrical osteochondral defect in femoral medial condyle of ponies. The results demonstrated that the SM-MSC constructs significantly promoted cartilage and subchondral bone regeneration over a 6-month period. Compared to control sites, the implanted defects showed better structural integration, including improved cartilage coverage and subchondral bone formation, as confirmed by arthroscopic (macroscopic) assessment, CT, MRI, and histological assessments (Figure 6(b)). 97 Despites these advantages, large animal models are expensive and require specialized surgical procedures to implant biomaterial-based spheroids into joints. Moreover, cartilage regeneration in large animal model takes longer to assess, requiring more extended periods of monitoring to determine the full efficacy of the biomaterial. Although, large animals, such as goats, sheep, and horses, provide closer anatomical and biomechanical analogies to human joints, with cartilage thicknesses of 1–2 mm and physiological joint loading, they also show high inter-individual variability and more pronounced immune responses to human-derived or synthetic implants. 99 Moreover, prolonged evaluation periods (>6 months) and logistical and ethical issues pose significant challenges. 99 These limitations highlights the need to carefully select models based on specific translational endpoints and to interpret immunological and mechanical responses in the framework of species-specific anatomy and physiology.
Evaluation of cartilage repair and functional outcomes in animal models
In preclinical studies of cartilage regeneration, animal models must closely replicate the physiological conditions of human joints, including cartilage thickness, biomechanical properties, and joint loading conditions. These parameters are essential for accurately assessing the effectiveness of regenerative approaches and ensure their clinical translation. The evaluation of cartilage repair and functional recovery in animal models focuses on two primary aspects: tissue regeneration and joint functionality. For tissue engineering, histological analyses are commonly performed to assess key parameters, such as ECM composition, collagen formation, GAG deposition, and cellular organization. These factors indicate the quality of the regenerated cartilage and its similarity to native hyaline cartilage. In addition, MRI and CT imaging provide quantitative assessments of cartilage thickness, matrix integrity, and structural integration with the underlying subchondral bone. For joint functionality, functional recovery is evaluated through gait analysis (joint mobility, movement symmetry, and load-bearing capacity) and arthroscopy (direct visualization of cartilage repair and joint health). These techniques provide valuable insights into the restoration of joint function in the regenerated tissue. Studies consistently demonstrate that composite spheroids significantly improve cartilage repair, leading to greater cartilage thickness, enhanced matrix retention, and better subchondral bone formation. Collectively, composite spheroids are widely recognized as an effective strategy for treating arthritis-related cartilage damage and improving joint function.
Histological analysis
Histological analysis enables the assessment of the quality of cartilage repair and the integration of newly formed tissue in composite-spheroid applications. After implantation of composite spheroids – typically composed of MSCs or chondrocytes embedded within a biomaterial scaffold – tissue samples are harvested at various time points, ranging from a few weeks to several months post-implantation. These samples are fixed, sectioned, and stained with specific dyes to evaluate cartilage and bone regeneration. Several histological staining techniques are commonly used to assess the structural and biochemical properties of regenerated tissue. Safranin O stains GAGs, a key component of the cartilage ECM. Alcian Blue stains proteoglycans, providing information on the composition and quality of the cartilage matrix. 100 H&E staining evaluates overall cell morphology, tissue structure, and matrix organization.101,102 Masson’s trichrome staining highlights collagen distribution within the regenerated cartilage and bone. Immunohistochemistry detects specific collagen types, such as collagen type II, indicative of hyaline cartilage, and collagen type I, indicative of fibrocartilage and bone.101,102 For example, histological analysis was performed to evaluate cartilage and subchondral bone regeneration after the implantation of scaffold-free 3D constructs derived from SM-MSCs. The staining techniques mentioned above demonstrated that composite spheroids facilitated ECM production and promoted subchondral bone formation. However, despite these promising observations, the regenerated tissue exhibited lower proteoglycan content than native hyaline cartilage, suggesting that further optimization is necessary to achieve ideal cartilage repair. 97 Histological analysis is essential for evaluating composite spheroids in cartilage repair. Combination of staining methods and molecular markers enable researchers to gain valuable insights into biomaterial-based cartilage engineering.
MRI and CT scans
MRI and CT imaging have been widely used to assess the effectiveness of composite spheroid treatments in cartilage repair. These imaging modalities provide complementary information, enabling a comprehensive assessment of both cartilage and subchondral bone regeneration. MRI is particularly valuable for evaluating the quality of the repaired cartilage, as it provides high-resolution images of soft tissues (e.g. cartilage), allowing for the detection of changes in cartilage compositions (e.g. water content and proteoglycan levels). Variations in T2-weighted signal intensity indicate different stages of regeneration. T2 hyper-intense signals suggest inflammation or fibrosis, whereas a shift to a hypo-intense signal over time suggests cartilage maturation and reduced inflammation. 103 MRI can also track the integrity and thickness of a cartilage layer, making it an ideal tool for monitoring the progress of tissue repair.104,105 On the other hand, CT scans are especially useful to evaluate subchondral bone regeneration, as they provide precise measurements of bone volume, density, and surface area. CT imaging enables the evaluation of bone integration beneath the repaired cartilage, precise measurement of cartilage thickness, and detection of potential bone remodeling or calcification.106,107
MRI and CT together offer complementary insights into the structural and functional impact of composite spheroids on regeneration of both cartilage bone components in the joint. For example, MRI and CT scans were used to non-invasively assess the volume and structural integration of regenerated cartilage and subchondral bone over a 6-month period after composite spheroid implantation. CT imaging visualized bone regeneration and detected changes in the radiolucent volume, while MRI assessed the quality and thickness of the repaired cartilage based on the modified MOCART grading system. 97 In another study, MRI and CT were employed to evaluate cartilage repair after the implantation of a bi-compartmented scaffold combined with MSC spheroids in a sheep model. MRI monitored in vivo healing process by tracking changes in signal intensity over time to assess inflammation and tissue regeneration. Meanwhile, CT scans measured the thickness and surface area of the newly regenerated cartilage. These imaging techniques revealed that treated sites showed significantly larger cartilage areas and better subchondral bone integration compared to untreated controls, demonstrating the effectiveness of composite spheroids in osteochondral regeneration. 98
Evaluation of functional recovery in cartilage regeneration
The assessment of composite spheroids for functional recovery in animal studies primarily focuses on restoring joint mobility and mechanical integrity of repaired cartilage. Although functional assessments are less common compared to histological and biochemical evaluations, they have gained importance in directly reflecting the practical efficacy of the treatment. Typically, these functional assessments are performed using gait analysis, range of motion (ROM) testing, and biomechanical testing. Joint mobility is commonly assessed by locomotor analysis and electromyography (EMG).6,104 A Vicon motion capture system can be used to track the movement of joints, such as the hip, knee, and ankle. Stride length, joint flexion, and stance-to-swing phase ratios are analyzed to quantify improvements in mobility. Additionally, EMG recordings were used to monitor muscle activity during movement. These analyses indicate possible improvements in joint mobility and muscle function. 108
The mechanical integrity of repaired cartilage is often assessed through biomechanical tests measuring key parameters, such as compressive modulus and hardness, which indicate the tissue ability to withstand mechanical loads. For example, Zhang et al. examined the effectiveness of Fk-CS@HA-Fb composite spheroids by evaluating cartilage repair through biomechanical analysis. After 12 weeks of treatment, they measured the compressive modulus (2.22 GPa) and hardness (148 kPa) of the repaired cartilage. These values in the treated group were significantly higher than those in control groups treated with simple hydrogels or PBS and were comparable to normal cartilage, indicating the potential of composite spheroids for restoring biomechanical function. 109 Current studies demonstrate promising improvements in joint function and cartilage biomechanics; however, further research is required to clearly assess the long-term durability and integration of regenerated cartilage with native tissues.
Challenges and future directions
While composite spheroids show great promise for cartilage regeneration, several challenges must be addressed to sufficiently achieve their therapeutic efficacy. Key challenges for composite spheroid systems in cartilage regeneration include biomaterial optimization, regulatory approval, in vivo integration, and scalable manufacturing (Figure 7). The standardization of composite spheroid culture protocol remains a major hurdle because variations in composition and culture conditions can significantly affect their regenerative properties. Establishment of a consistent and reproducible protocol – covering biomaterials, cell type, seeding density, culture medium, and duration – is essential for clinical applications. 110 To establish effective standardization, identification of the most suitable cell types and optical cell ratios is important for spheroid formation. MSCs, chondrocytes, and other progenitor cells have demonstrated their regenerative potential; however, determination of their optimal composition in combination with biomaterials requires further investigation. For instance, a study by Sayed et al. reported that spheroids formed with 2.5 × 10⁴ cells in chondrogenic differentiation medium exhibited robust cartilage marker expression. Interestingly, auricular spheroids increased in size over time, whereas nasal aggregates tended to shrink, highlighting the influence of cell source and density on spheroid morphology and ECM production. 111 Similarly, the culture medium formulations and biochemical supplements play vital roles in spheroid development by supporting viability, proliferation, and differentiation. Growth factors, such as transforming growth factor-beta (TGF-β) and bone morphogenetic proteins (BMPs), are usually supplemented to promote chondrogenesis. However, their concentrations and timing must be carefully calibrated to prevent undesired differentiation pathways or inhibitory effects. 112 Another key factor is culture duration, which affects spheroid maturation and ECM deposition. While extended culture periods allow for robust matrix formation and cell-matrix interactions, prolonged cultivation can compromise spheroid stability and cell viability, primarily due to diffusion limitations. Accordingly, the optical culture duration must be determined to balance adequate maturation and spheroid integrity. 113 For instance, Chiesa et al. found that spheroids (~200 ± 20 µm) maintained >95% viability and strong GAG and Col II expression after 14 days. However, by day 28, increased mineralization and central necrosis were observed, highlighting the importance of optimizing culture duration to preserve structural integrity. 113 Addressing these challenges will significantly enhance the reproducibility and clinical reliability of composite spheroids for articular cartilage regeneration. In addition, future developments should focus on enhancing both the structural and biochemical complexity to more accurately replicate the native articular cartilage microenvironment. One major limitation of current composite spheroids is the difficulty in replicating the heterogeneous ECM composition and zonal architecture of articular cartilage. 114 Different regions in zonal cartilage exhibit distinct cellular arrangements and matrix compositions. To address this issue, advanced biofabrication techniques, such as 3D bioprinting and microfluidic systems, are explored to spatially organize cells and biomaterials within spheroids. These technologies allow for precise construction of composite systems that mimic the superficial, middle, and deep zones of native cartilage in both form and function. In addition, mechanical stimulation is a promising factor in cartilage development and regeneration. Incorporation of dynamic mechanical stimulation, such as cyclic compression, shear stress, or hydrostatic pressure, into culture conditions can further enhance ECM synthesis, promote cell alignment, and improve the biomechanical properties of regenerated tissues. Integration of multi-zonal bioprinting with immune-responsive materials and real-time bioreactor feedback systems may facilitate the clinical translation of composite spheroid-based therapies for osteochondral repair.
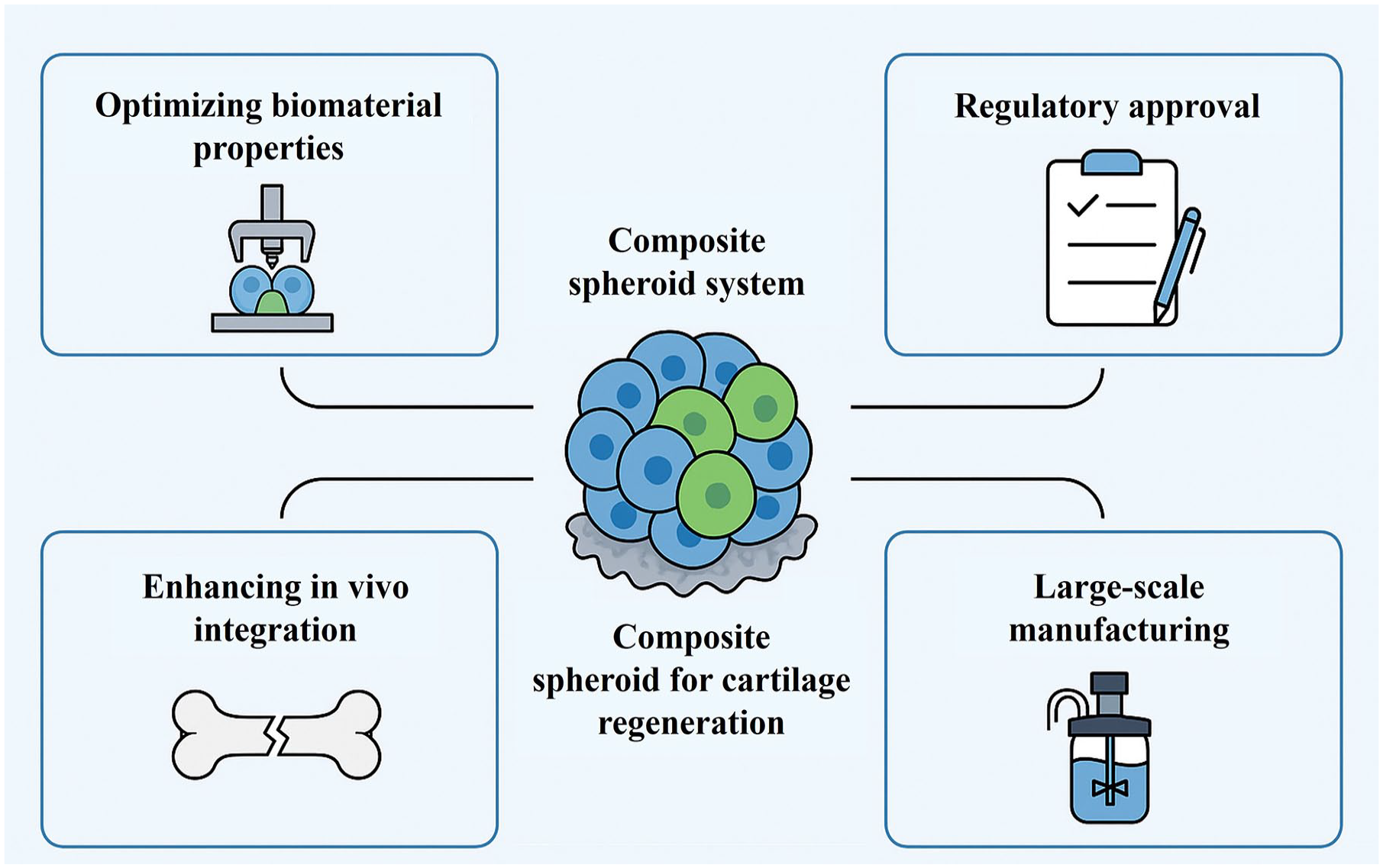
Future direction and key challenges for composite spheroids in cartilage tissue regeneration.
Translating composite spheroids into clinically applicable therapies for cartilage regeneration faces several critical challenges, including GMP-compliant scalability, immune compatibility, and regulatory complexity. While bioreactor systems offer scalability for mass production, excessive shear stress during dynamic culture can impair spheroid integrity. In contrast, in-air microfluidics recently demonstrated high-throughput spheroid production capability (>10,000 spheroids per min) while maintaining uniform size and over 90% viability. 71 To address immunogenicity concerns associated with allogeneic sources, recent study suggested the use of HLA-matched or hypoimmunogenic iPSC lines. 115 Moreover, modulating amino acid composition and metabolic pathways within MSC spheroids has emerged as a potential strategy to regulate immune responses and affect their proliferation, differentiation, and chondrogenic potential. 116 Regulatory oversight of tissue-engineered constructs lacks standardization, with classification often influenced by their structural components, biological function, and clinical application. 117 This variability poses challenges for defining clear pathways toward clinical approval. To overcome these hurdles, several actional solutions can be proposed: advanced cryopreservation protocols to retain stem cell viability and function118,119; stem cell banking to enable standardized, traceable starting materials; and modular bioreactors to support reproducible and scalable culture conditions and scalable manufacturing. Collectively, these approaches are essential for ensuring consistency, safety, and efficacy in the clinical applications of spheroid-based cartilage regeneration therapies.
Conclusion
Although the importance of biomaterials is well recognized in cartilage tissue engineering, growing attentions has been directed toward advancements in materials and fabrication techniques, particularly composite spheroid approaches. This review highlights recent developments in composite spheroid culture and their application in cartilage regeneration. Spheroid-based therapies enhance cell-cell interactions and more effectively mimic the native tissue environment, thereby improving cartilage repair. However, several challenges remain, including the need for protocol standardization, the optimization of cell-material interactions, and the development of more complex tissue models. To address these challenges, integration of biomimetic materials and advanced biofabrication techniques will be necessary to create a more physiologically relevant environment. These advancements will promote cell survival, ECM production, and integration into host tissues. Future studies should focus on optimizing composite spheroid culture protocols, exploring novel biomaterial formulations, and integrating mechanical stimulation to enhance their structural and functional properties. By overcoming these challenges, composite spheroid technology holds significant potential for advancing cartilage regeneration and improving treatments for osteoarthritis and other cartilage-related disorders.
Footnotes
Acknowledgements
The authors sincerely thank Phatcharapon Udomluck, MD (Department of Orthopaedics, School of Medicine, University of Phayao, Thailand), for his valuable academic guidance on articular cartilage regeneration and cartilage repair evaluation.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
NU collected the information, organized the review, and wrote the manuscript. JYL and HP revised the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government Ministry of Science and ICT (RS-2024-00449435) and AI-based GIST Research Scientist Project grant funded by the GIST in 2024.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
No data was used for the research described in the article.
