Abstract
Osteonecrosis of the femoral head (ONFH) is a prevalent orthopedic disorder characterized primarily by compromised blood supply. This vascular deficit results in cell apoptosis, trabecular bone loss, and structural collapse of the femoral head at late stage, significantly impairing joint function. While MRI is a highly effective tool for diagnosing ONFH in its early stages, challenges remain due to the limited availability and high cost of MRI, as well as the absence of routine MRI screening in asymptomatic patients. . In addition, current therapeutic strategies predominantly only relieve symptoms while disease-modifying ONFH drugs are still under investigation/development. Considering that blood supply of the femoral head plays a key role in the pathology of ONFH, angiogenic therapies have been put forward as promising treatment options. Emerging bioengineering interventions targeting angiogenesis hold promising potential for ONFH treatment. In this review, we introduce the advances in research into the pathology of ONFH and summarize novel bioengineering interventions targeting angiogenesis. This review sheds light upon new directions for future research into ONFH.

Introduction
Osteonecrosis of the femoral head (ONFH), also known as avascular necrosis of the femoral head, is a common orthopedic disease caused by the compromised blood supply to the femoral head. Interruption of blood supply is the initiation step, followed by the death of bone cells and subsequent structural collapse of the femoral head, which eventually results in hip joint dysfunction. 1 Based on different pathologies, ONFH can be divided into two main categories: traumatic and non-traumatic. The former is mainly caused by femoral neck fracture, hip dislocation, and other hip injuries, while the latter most results from the use of corticosteroids and alcoholism. 2 As a debilitating condition, ONFH is commonly seen in patients aged between 30 and 65, and often results in the deterioration of the hip joint, causing pain, limited range of motion, and functional impairment. 3 The etiologies of ONFH vary depending on the patients’ race, lifestyle, and culture. Among all the risk factors, corticosteroid use and alcohol abuse account for more than 80% of all cases. 4 While the precise incidence of osteonecrosis is unknown, it is estimated that 20,000–30,000 new patients are diagnosed with osteonecrosis annually in the United States, accounting for approximately 10% of the 250,000 total hip arthroplasties performed annually. 5 In Asia, 100,000–200,000 new cases are reported in China every year. 6 Nationwide surveys from Japan and Korea report more than 10,000 new cases annually. 7
ONFH are often asymptomatic in the early stages. 8 In the later-stage, pain in the groin, thigh and buttock is a commonly reported symptom. 9 Findings of physical checks for ONFH are generally nonspecific. Positive signs include reduced range of joint motion, painful ambulation, Trendelenburg sign, and/or crepitus. 10 Imaging techniques are important methods for the diagnosis of ONFH. 11 In addition to imaging results, clinical features, physical examinations, bone biopsy, and pathological manifestations have been used for the ONF diagnosis. Multiple classification systems of ONFH have been developed to assist in the diagnosis and treatment of this potentially debilitating disorder. Among them, Ficat and Arlet, 12 University of Pennsylvania, 13 Association Research Circulation Osseous (ARCO), 14 and Japanese Orthopedic Association 15 are the most commonly used systems. An analysis of patients can be made with any of the four major systems based on the findings of MRI and various radiographic tests. 16 While MRI is a highly effective tool for diagnosing ONFH at an early stage, early diagnosis remains challenging in clinical practice due to the subtle nature of symptoms, limited accessibility to advanced imaging modalities in some settings, and the lack of definitive clinical or biochemical markers to guide early screening. Besides, the unclear pathological mechanisms add to the challenges facing ONFH treatment.
Compromised blood flow is considered a core feature of ONFH, which can be triggered by various factors such as trauma, corticosteroid therapy, alcohol use, HIV, lymphoma/leukemia, blood dyscrasias, chemotherapy, radiation therapy, Gaucher disease, and Caisson disease. 17 The mechanisms by which these factors diminish femoral perfusion remain unclear. Possible mechanisms include vascular endothelial damage, intramedullary adipogenesis, and microvascular thrombosis formation, which increase the intraosseous pressure and lead to venous stasis and arterial obstruction.18–20 The limited understanding of the precise pathophysiological mechanisms underlying ONFH presents a significant challenge in the development of targeted therapeutic strategies. Current treatments are mostly palliative, providing temporary alleviation of joint symptoms but failing to prevent ONFH or halt/reverse its progression. Researchers are working on unraveling the intricate factors contributing to ONFH and its treatment, with a particular focus on vascular abnormalities.
ONFH management presents significant challenges due to the singular mode of blood supply to the femoral head and the structural damage caused by vascular injury. 21 Certain areas of the body are characterized by no significant capillary network and depend exclusively on terminal arteries for blood supply. The femoral head is a notable example. 22 Because of this unique vascular arrangement, regions like the femoral head are especially susceptible to ischemia and injury. Any interruption in their blood flow can rapidly lead to irreversible pathological changes. The femoral head receives its primary blood supply from the medial and lateral circumflex arteries, especially the retinacular arteries, which traverse the femoral neck and supply blood to the superior portion of the femoral head. 21 Consequently, any disruption to the circumflex arteries, such as in femoral neck fractures or hip dislocations, can severely compromise blood flow to the femoral head and lead to ischemia and subsequent complications like ONFH. 23 Clinical strategies such as core decompression and intraosseous drilling aim to address this challenge by by reducing venous congestion and bone marrow pressure and creating channels that enhance blood supply to the epiphysis. 24 Bioengineering approaches, including scaffold implantation and growth factor delivery, have been developed to complement these techniques by promoting tissue regeneration. 25 For example, bioengineered scaffolds seeded with mesenchymal stem cells (MSCs) or coated with osteoinductive growth factors like bone morphogenetic proteins (BMPs) have shown promise in stimulating bone repair and angiogenesis in preclinical and clinical studies. These scaffolds provide a supportive structure for new tissue growth while promoting the recruitment of progenitor cells and the formation of new blood vessels.26,27 These advanced therapies, aiming to achieve long-lasting restoration of joint functionality, promise to improve the prognosis for patients with ONFH.
In recent years, significant advancements have been achieved in the treatment of ONFH via using biomaterials and tissue engineering techniques. Several reviews summarized the research progress of this field, providing an overview of past and current strategies.25,28–31 However, most previous reviews lacked an in-depth discussion of the critical role of angiogenesis in ONFH, and few comprehensively summarized ongoing and completed relevant clinical trials. In this review, we examine the pathology and therapies for ONFH by highlighting the critical role of angiogenesis. In addition, we summarize recent advances in regenerative medicine and bioengineering techniques that target angiogenesis in the prevention and treatment of ONFH. Besides, completed, ongoing, and suspended clinical trials aiming at improving angiogenesis for ONFH therapy are screened. Uncompleted registered clinical trials are also analyzed to guide future research. The goal of this review is to reveal ONFH pathology and explore potential therapies, paving the way for future clinical management with improved efficacy.
The pathology of ONFH
The classification of ONFH is crucial for understanding its pathology and choosing appropriate therapeutic strategies. Based on different classification methods, ONFH can be categorized into various types. Traumatic ONFH results from direct hip joint injury, while non-traumatic ONFH is associated with factors that affect blood flow, such as corticosteroid use, excessive alcohol consumption, systemic diseases, and vascular diseases. Although the etiology of ONFH varies, with traumatic and non-traumatic causes, the pathologies converge on a common final pathway: disruption of blood flow leading to massive bone and marrow cell death. This review will primarily focus on non-traumatic ONFH, as it represents the majority of cases and involves distinct etiological factors frrom traumatic ONFH.
Glucocorticoid-induced ONFH (GONFH)
Glucocorticoids are prescribed to suppress inflammation or the immune system for various medical conditions. 32 At present, approximately 2%–3% of the population is receiving systemic or local glucocorticoid treatment. 33 High doses and/or long-term use of glucocorticoids are associated with an increased risk of ONFH, which is known as GONFH or steroid-induced osteonecrosis of the femoral head (SONFH). 34 It is estimated that 100,000 Chinese and more than 20,000 Americans are disabled due to GONFH annually.5,35 The effect of corticosteroid doses on the incidence of hip osteonecrosis remains unclear. A meta-analysis conducted by Mont et al. revealed that the incidence rate of hip osteonecrosis is positively correlated with the dosage of corticosteroid used. Osteonecrosis incidence was 6.7% with corticosteroid treatment of >2 g (prednisone-equivalent). Each 10 mg/day increase was associated with a 3.6% increase in osteonecrosis rate, and >20 mg/day resulted in a higher osteonecrosis incidence. 36
The exact pathogenesis of GONFH remains largely unknown. Nonokawa et al. 37 proposed that the pathogenesis of GONFH involves a cascade initiated by glucocorticoid-induced platelet activation and neutrophil extracellular trap formation, which results in small vessel occlusion, ischemia, and subsequent osteocyte death in the femoral head. Zhu et al. revealed that pathological exosomes (ONFH-exos) from necrotic bone tissues disrupt key pathways required for osteogenic differentiation and osteoblast?/MSC? migration. Restoring CD41 levels via exosome engineering or targeting its signaling pathway may provide a promising approach to the diagnosis and treatment of ONFH. 38 Inflammation is also involved in the pathogenesis of ONFH. Hyperactivation of TLR4/NF-κB pathway induced by glucocorticoid was found to trigger the overproduction of inflammatory cytokines, which exacerbated tissue damage, impaired bone remodeling, and contributed to osteonecrosis in rats. 39 Sympathetic nerves are known to regulate bone homeostasis. A recent study by Shao et al. revealed that glucocorticoids lower sympathetic tone by disrupting the balance of glucocorticoid receptors and mineralocorticoid receptors in neurons of the hypothalamic paraventricular nucleus. The decreased sympathetic tone triggers endothelial cell apoptosis and reduces H-type vessels in the femoral head, inhibiting angiogenesis and osteogenesis in the femoral head. 40 Besides, the recruitment and hyperactivation of osteoclasts are believed to contribute to the loss of bone structural integrity and subchondral fracture in GONFH. 34 Glucocorticoid administration was found to increase serum levels of sclerostin, a key regulator of bone homeostasis and skeletal integrity. Histological analysis of GA-ONFH patient samples revealed high concentrations of sclerostin in the necrotic regions of the femoral head, which disrupted the balance of bone remodeling and vascular repair, accelerating osteonecrosis progression via suppressing Wnt signaling pathway. 41 Abnormal lipid metabolism, increased cell apoptosis, and disturbed BMSC differentiation caused by β-catenin inhibition have been reported to underlie GONFH pathology. 42 Recently, Chen et al. 43 found that gut bacteria loss also participated in GONFH. Glucocorticoid-induced loss of beneficial gut bacteria, Lactobacillus animalis, and extracellular vesicles from L. animalis (L. animalis-EVs) were associated with the pathogenesis and development of GONFH. Oral supplementation of L. animalis mitigates GONFH by increasing angiogenesis, augmenting osteogenesis, and reducing cell apoptosis. 43 In addition, LncRNAs have been found to regulate the nuclear location of β-catenin that influences the differentiation of BMSCs and the progression of GONFH. 44
Treatments targeting these potential pathogenic mechanisms have received promising results in tackling GONFH. For example, vitamin K2 effectively promotes angiogenesis and protects vascular integrity in the femoral head via enhancing endothelial cell migration and tube formation. 45 Calycosin ameliorates GC-induced ONFH by inhibiting the TLR4/NF-κB pathway, reducing inflammation, and promoting osteogenic differentiation and bone formation. 39 The activation of Keap1/Nrf2 pathway by inhibiting monoacylglycerol lipase attenuated GC-induced ONFH in rats. 46 Exosomes derived from synovial-derived MSCs and human CD34+ stem cells have been shown to protect against GC-induced ONFH in animal models by enhancing cell proliferation and reducing cell apoptosis in rats.47,48 Extracellular vesicles secreted by human urine-derived stem cells protected against GC-induced ONFH by enhancing angiogenesis and reducing apoptosis of MSCs apoptosis via the delivery of bioactive molecules like DMBT1 and TIMP1. 49 Restoring sympathetic tone and targeting downstream endothelial glycolytic pathways, such as PFKFB3 activation via Adrb2 signaling, represent promising therapeutic strategies for mitigating GC-induced ONFH. 40 Targeting sclerostin, through strategies such as SOST inhibitor use or genetic knockout, represents a potential therapeutic approach to mitigating GA-ONFH and improving bone and vascular outcomes in rats. 41 The proliferation and differentiation of MSC can be enhanced by lithium chloride to protect rats from GC-induced ONFH. 50 Although these studies have demonstrated promising effects, challenges remain in their clinical applications. For instance, it is difficult to effectively deliver drugs to targeted sites and maintain their therapeutic efficacy in the human body. Innovative drug delivery systems, such as nanoparticles, hydrogels, and other controlled-release platforms, provide new directions for overcoming these hurdles.
Alcohol-associated ONFH (AONFH)
AONFH is closely related to alcohol consumption. Alcohol intake, especially in daily drinkers, significantly increases the risk of ONFH. A meta-analysis study of the Japanese population revealed that the alcohol dose-dependent risk of ONFH was increased by 35.3% for every 100 g/week (95% confidence interval [CI], 1.24–1.47; p < 0.001) and by 44.1% for every 500 g drink-years (95% CI, 1.295–1.601; p < 0.001). 20 The microarchitectural disruptions and histopathological features in the necrotic region of AONFH are similar to those of GONFH. 51 The exact mechanism by which alcohol induces ONFH remains unclear. However, the detrimental effects of alcohol on blood vessels are believed to contribute to the pathology of AONFH. 52 Chronic alcohol intake is known to cause vascular constriction and endothelial dysfunction, resulting in reduced blood flow to the femoral head. This compromised blood supply deprives bone tissue of essential nutrients and oxygen, resulting in cellular damage and ultimately necrosis. 52 Additionally, alcohol metabolism generates toxic cellular byproducts that can disrupt the function of mitochondrion and lysosome, further exacerbating tissue injury and inflammation within the hip joint.53,54 Pre-clinical studies revealed several possible regulating pathways. For example, disturbed osteogenic-adipogenic differentiation of stromal cells and altered regulons such as myocyte enhancer factor 2C (MEF2C) and Jun protein D are related to AONFH. 55 Additionally, a subset of endothelial cells expressing atypical chemokine receptor 1 (ACKR1) exhibited enhanced chemotaxis but weakened angiogenic ability. These endothelial cells potentially regulate the differentiation of stromal cells via nicotinamide phosphoribosyl transferase (NAMPT)- and E-Selectin-related pathways. 55 Genetic factors also contribute to the onset of AONFH. For example, rs62030917 in RAB40C gene, PFKP rs10903966, GPC6 rs9523981, L3MBTL3/PTPN9 gene polymorphism, CARMEN (cardiac mesoderm enhancer-associated non-coding RNA) polymorphism, RETN genetic polymorphisms, OPG and RANKL polymorphisms are found to increase the susceptibility to AONFH in the Chinese Han ethnic group.56–61 Unique long non-coding RNA (lncRNA) expression profiles were found to distinguish AONFH from other types of ONFH, suggesting their potential as biomarkers for AONFH diagnosis and targeted treatment. 62 In addition, alcohol consumption can contribute to the progression of ONFH by influencing gut microbiota composition by, for example, increasing Lactobacillus and Roseburia and shifting fecal metabolites. 63
Considerable research efforts are underway to elucidate the underlying mechanisms for and improve the management of AONFH. Researchers found that betaine played a protective role and suppressed the progression of AONFH via the mammalian target of rapamycin (mTOR) pathway. 64 Furthermore, inhibiting Dickkopf-1 (DKK1) activates Wnt/β-catenin signaling and the nuclear translocation of β-catenin, thereby promoting osteogenesis and inhibiting adipogenesis of bone marrow-derived mesenchymal stem cells (BMSCs). 65 For the treatment, Fu and co-workers developed a heat-sensitive nanocomposite hydrogel system to provide long-term gene regulation in lesion cells in ONFH. The hydrogel, injectable at room temperature, solidifies in the body under body heat, releasing secondary nanostructures carrying gene-regulating plasmids and siRNA. 66 This system restored physiological activity in stem cells in the ONFH area, promoting repair and bone reconstruction via upregulating the expression of B-cell lymphoma 2 and inhibiting the secretion of peroxisome proliferators-activated receptors γ. In vivo experiments demonstrated its long-term efficacy, offering a promising treatment option for AONFH and potentially other gene therapy applications. 66
COVID-related ONFH
COVID-19 induces a hypercoagulable state and increases the risk of thrombosis and ONFH, which are known as symptoms of “Long COVID.” 67 Vascular damage caused by COVID-19 is believed to be achieved through a process known as immunothrombosis. 68 In this process, activated neutrophils and monocytes interact with platelets and activate the coagulation cascade, leading to the formation of intravascular clots in both small and larger vessels. These microthrombotic complications contribute to the reduction of blood flow within the vessels, which can exacerbate the progression of ONFH. 69 In addition, COVID-19 vaccine-related thrombosis and high-dose steroid therapy used in the treatment of COVID-19 also increase the risk of ONFH. 70 Several studies have reported ONFH diagnosis in patients who recovered from COVID-19 disease (Table 1). Agarwala et al. first reported three such cases. The mean dose of prednisolone used in these cases was 758 mg (400–1250 mg). The mean time to diagnose ONFH was 58 days after COVID-19 diagnosis. Their study emphasized that COVID-19 infection lowers the threshold of glucocorticoid use for the development of ONFH and greatly accelerates the progression of this disease. 71 Dhanasekararaja et al. reported 22 patients (39 hips) who had an aggressive presentation and rapid progression of ONFH after COVID-19. In their study, the average cumulative dose of methylprednisolone equivalent was 811 mg (range 200–2100 mg) and the average duration of steroid intake was 2.8 weeks. This low cumulative dose of steroids suggested that COVID-19-associated vasculitis may play a role in the pathogenesis of ONFH. The mean time to diagnose ONFH was 39.3 days (in the range of 10–90 days). The average duration from COVID-19 infection to the onset of hip symptoms was 7.5 months (in the range of 3–11 months). 72 Particularly, patients resented with elevated serological markers and extensive periarticular bone and soft tissue edema exhibited an acute and aggressive onset and rapid femoral head destruction. 72 Several single case reports also support the finding that after COVID-19 infection, glucocorticoid use is highly related to the increased risk of ONFH.73–77 In addition to ONFH, septic arthritis with avascular necrosis due to COVID-19 infection should also be taken into serious consideration. 78 In patients with a history of COVID-19 infection, especially those treated with corticosteroids, any joint symptom should draw our attention to the diagnosis of ONFH. Low therapeutic doses of corticosteroids with minimal effective duration remain the key to halting its occurrence. A retrospective evaluation conducted by Agarwala et al. evaluated the effects of bisphosphonates for the treatment of post-COVID ONFH. Forty-eight patients (88 hips) who were diagnosed with ONFH after COVID-19 infection received intravenous zoledronic acid (5 mg) at the initiation of therapy and oral alendronate (35 mg) twice weekly. At a mean follow-up of 10 months, 84 (95.5%) of the hips showed good clinical outcomes, and only 4 (4.5%) of the hips required surgical intervention. Their results highlighted the effectiveness of bisphosphonates in ONFH therapy. 79 For patients with end-stage ONFH, timely diagnosis and surgery are important for preserving their hip joints. A prospective study conducted by Veizi et al. 80 evaluated the incidence of osteonecrosis in COVID-19 patients who received or did not receive corticosteroid treatment. Their results showed that corticosteroid use significantly increased the number of painful joints. At 2 years, 11.9% (corticosteroid group) and 5.1% (non-corticosteroid group) of patients complained of at least one painful joint, respectively. Eight patients who received corticosteroid treatment developed osteonecrosis. This study emphasized the importance of paying attention to ONFH in COVID-19 patients who received corticosteroid treatment. With the the increased incidence of COVID-related ONFH, the number of patients seeking medical support are expected to increase in the following years. Accompanying this growing medical demand are additional challenges facing orthopedic surgeons, such as increased proportion of elderly patients and patients with complex disease histories and/or poor physical conditions.
Previous studies on the relationship between corticoid use for COVID-19 treatment and the onset of ONFH.

Legg-Calvé-Perthes disease (LCPD)-associated ONFH
LCPD is a pediatric orthopedic disorder characterized by a temporary disruption of blood supply to the femoral head. Impaired blood supply further leads to chronic hip synovitis, irreversible deformity of the femoral head, and premature osteoarthritis. 81 LCPD is considered a self-limiting, self-healing, and non-systemic disease that occurs typically in children aged between 4 and 10. It primarily affects boys and often presents unilaterally. 82 The prevalence of LCPD varies geographically, with incidence rates ranging from 0.4 to 29.0 cases per 100,000 children. 82 Despite its relative rarity, LCPD poses a significant orthopedic challenge because of severe long-term outcomes such as persistent hip pain, limping, leg length discrepancy, and the early onset of osteoarthritis. 83 Therefore, early detection and intervention are important to mitigate the above adverse effects and preserve hip function. This highlights the need for an efficient and effective approach to the diagnosis and treatment of the complex condition of LCPD.
Although the precise etiology of LCPD is not fully understood, it is generally accepted that factors such as genetic predisposition, vascular abnormalities, and mechanical stress on the hip joint play integral roles in its progression. 84 These factors contribute to the temporary disruption of blood supply to the femoral head, leading to the characteristic manifestations of the disease. 84 Treatment strategies for LCPD aim to preserve the shape of the femoral head, maintain hip function, and prevent long-term complications. Non-surgical approaches may include activity modification, physical therapy, bracing, and anti-inflammatory medications to alleviate symptoms and promote healing. In cases where conservative measures fail to halt disease progression, 85 surgical interventions such as osteotomy, femoral or pelvic realignment procedures, and hip arthroplasty may be considered to restore hip joint integrity and function. 86
Regulatory mechanism for angiogenesis
Angiogenesis is defined as a process in which new blood vessels form from pre-existing vessels (Figure 1). This process is critical in maintaining normal body functions such as growth and wound healing. It is also involved in pathological conditions like cancer, diabetic retinopathy, and LCPD.87,88 Generally, angiogenesis starts with the vasodilation of existing vessels. Then the basement membrane degrades to allow the migration of endothelial cells toward the angiogenic stimulus. 88 Tip cells are specialized endothelial cells located at the leading edge of a growing blood vessel during the process of angiogenesis. They extend numerous filopodia, which are thin, actin-rich projections that explore the extracellular environment. These filopodia sense and respond to guidance cues, directing the sprouting vessel toward areas of high angiogenic factor (such as vascular endothelial growth factor, VEGF) concentration. 89 Tip cells direct the proliferation and migration of endothelial cells toward the leading front of migrating cells and form new capillary loops. Finally, pericytes are recruited and new basement membranes are deposited to help the maturation and stabilization of capillaries. 87 While significant progress has been made in understanding and modulating angiogenesis for therapeutic purposes, challenges remain such as resistance to anti-angiogenic therapies and the side effects associated with systemic inhibition of angiogenesis. 90 Therefore, a deeper understanding of the regulatory processes of angiogenesis is crucial for developing selective angiogenic therapies.
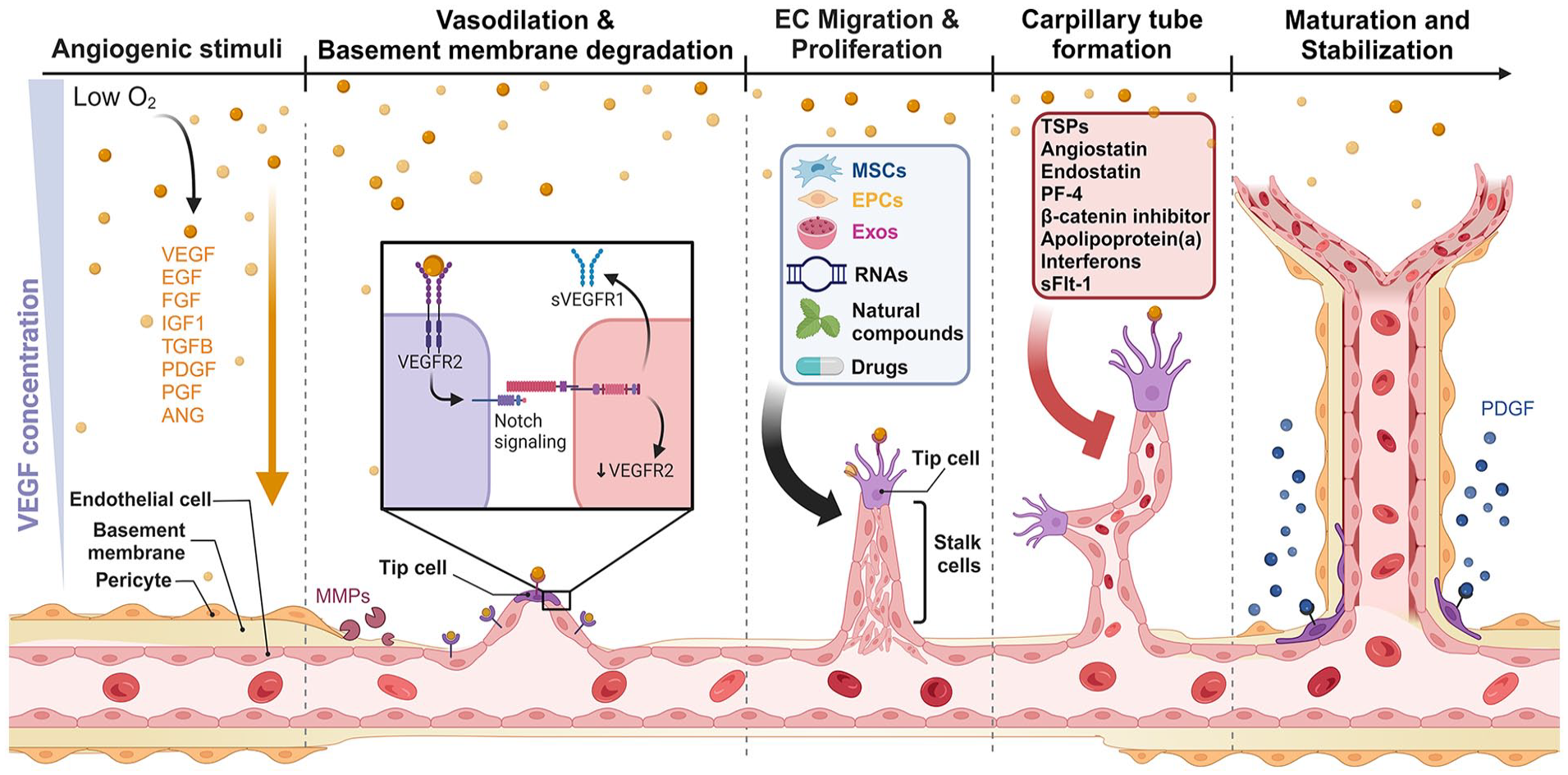
Angiogenic process and key molecular regulators involved in the formation and maturation of new blood vessels. Angiogenesis triggered by internal and external stresses such as low oxygen levels (hypoxia) lead to the release of angiogenic factors. Tip cells sense the stimuli and transmit stimuli signals to neighboring endothelial cells (ECs) through cell-to-cell communication. ECs respond to angiogenic stimuli, resulting in vasodilation and degradation of the basement membrane facilitated by matrix metalloproteinases (MMPs). ECs then migrate and proliferate to form new capillary tubes. The newly formed vessels undergo maturation and stabilization, aided by pericytes and smooth muscle cells. Several factors influence the angiogenesis process via targeting different stages. Created with biorender.com.
Angiogenesis is a complex and dynamic process regulated through a balance between pro-angiogenic and anti-angiogenic factors 87 (Figure 1). Among them, VEGF is the most potent and critical pro-angiogenic factor. It binds to the receptors on endothelial cells, stimulating their proliferation, migration, and new vessel formation.91,92 Strategies targeting VEGF and related pathways, such as the use of anti-VEGF antibodies (e.g. Bevacizumab), have been applied for cancer treatment as an adjunct therapy of chemotherapy or immunotherapy. 92 Other pro-angiogenic factors, like fibroblast growth factors (FGFs), promote the proliferation and differentiation of endothelial cells and induce their physical organization into tube-like structures. 93 Integrins are critical for the interaction of endothelial cells with the extracellular matrix, facilitating cell adhesion and migration. 94 Angiopoietins (Ang-1 and Ang-2) have dual roles in angiogenesis. Ang-1 promotes vascular maturation and stability. Ang-2, unlike Ang-1, facilitates vascular regression in the absence of VEGF but promotes angiogenesis when VEGF is present. 95 Sphingosine 1-phosphate is an important signaling sphingolipid that regulates angiogenesis and endothelial barrier integrity. 96
Anti-angiogenic factors are natural or synthetic molecules that inhibit the angiogenesis process. Although anti-angiogenic factors are not the primary focus of this review, their role in impairing vascularization and exacerbating ischemia in ONFH warrants a brief discussion. For example, elevated levels of anti-angiogenic molecules such as thrombospondin-1 (TSP-1) have been associated with reduced neovascularization, a critical barrier to effective bone regeneration in ONFH. 97 TSP-1 is a multifunctional glycoprotein that suppresses angiogenesis by inducing apoptosis and inhibiting the proliferation of endothelial cells. Besides, TSP-1 influences the bioavailability and activity of other growth factors involved in angiogenesis, such as VEGF and FGFs, functioning as a key modulator within the vascular environment and impacting various stages of angiogenesis. 98 Angiostatin is an endogenous protein fragment derived from the proteolytic cleavage of plasminogen. Its anti-angiogenic role is achieved by inhibiting the proliferation of endothelial cells. 99 Apolipoprotein(a), when bound to oxidized phospholipids, leads to the apoptosis of endothelial cells. 100 In addition, apolipoprotein(a) inhibits the functionality of endothelial progenitor cells and upregulates nuclear factors within endothelial cells, resulting in the disruption of the integrity of the vascular lining and hampering the natural repair and regeneration of vascular endothelium. 100 Endostatin is a fragment of collagen XVIII. It functions as a potent inhibitor of angiogenesis via suppressing vascular cell proliferation and migration. This suppression thereby inhibits new blood vessel formation.99, 101 Interferons (IFNs), known for their immune-modulating effects, also have strong anti-angiogenic properties via inhibiting the proliferation of endothelial cells and disrupting epithelial repair.102,103 Platelet factor-4 (PF-4) is a chemokine released by platelets that inhibits endothelial cell migration and proliferation. 104 Soluble Fms-like tyrosine kinase-1 (sFlt-1) is a soluble receptor that binds to VEGF, preventing it from activating its endothelial receptors. 105
Targeting angiogenesis for ONFH treatment
ONFH is characterized by a critical reduction in vascular supply that is essential for bone healing and remodeling. The resultant hypoxic environment within the femoral head further impairs angiogenic responses. Therapeutic strategies targeting angiogenesis have shown promising progress in alleviating symptoms of ONFH. 45 For example, periarticular injection of VEGF receptor 2 antibody in rats suppressed intraosseous vessels and resulted in the development of ONFH. 106 On the contrary, co-transplantation of angiotensin II-pretreated MSCs and endothelial cells induce both osteogenesis and angiogenesis in a rabbit model of SONFH. 107 Utilizing angiogenic factors in tissue engineering techniques by loading these factors in biomaterial scaffolds, has been explored extensively to support the formation of new blood vessels over time. 108 However, it is noteworthy that excessively high levels of VEGF exhibit detrimental effects on vascular permeability and bone repair. For example, Cao et al. tested the effects of different concentrations of VEGF on human umbilical vein endothelial cells (HUVECs) and bone marrow cell (BMC)-derived osteoclast precursors. Their results revealed that treatment with high concentrations of VEGF (50 ng/ml) induced the disruption of the junctional integrity of endothelial cells. Furthermore, VEGF promoted the formation and activity of osteoclast from BMC-derived osteoclast precursors in vitro. 109 Therefore, while VEGF has therapeutic potential in ONFH treatment, its levels must be carefully regulated to avoid its adverse effects on blood vessels and bone cells. Targeting angiogenesis in the treatment of ONFH represents a cutting-edge area of study that has gained significant attention in recent years. Clinical trials targeting angiogenesis for the treatment of ONFH are summarized in Table 2. Current clinical trials are primarily concentrated on evaluating the effects of autologous cell-based therapy and bone marrow concentrates on the early-stage ONFH. Gene editing and bioengineering biomaterials have not been explored in ONFH treatment in clinics, probably due to the complexity and security concerns of these technologies. In this section, we summarize the research progress in this area and present the latest advances in angiogenesis promotion and tissue-engineered delivery platforms for angiogenesis enhancement.
Registered clinical studies targeting angiogenesis for ONFH.

BMMNCs: bone marrow mononuclear cells; MSCs: mesenchymal stem cells; NA: not applicable; XCEL-MT-OSTEO-ALPHA: “Ex vivo” expanded autologous bone marrow mesenchymal stem cells fixed in allogenic human bone tissue.
Current animal models for studying ONFH
Preclinical models of ONFH vary in their ability to mimic the human condition and provide insights into different aspects of the disease. Currently, there is no consensus on a standardized ONFH animal model. Each ONFH model has unique characteristics that make it suitable for certain types of studies but may also present limitations that should not be neglected. 110
To date, a variety of animal models of ONFH have been created, with relevant research efforts being focused on quadrupeds, such as mice, rats, rabbits, dogs, pigs, sheep, goats, and horses. 111 However, these quadrupeds do not possess comparable load-bearing capacities to humans. Studies on bipedal animals, such as geese, chickens, ostriches, and emus, reveal that while their weight-bearing characteristics are similar to those of humans, their anatomical structures and physiological features are not. 112 Large animal models, such as canines, allow for surgical procedures and implant testing, while small animal models, such as rats and mice, are particularly useful for genetic and molecular studies due to the availability of genetically modified strains. 113 Emu models of ONFH provide a unique advantage due to their anatomical similarity in femoral heads to humans. 114 Common methods for establishing ONFH models include traumatic, non-traumatic and combined approaches. Overall, the success rates of surgical vascular deprivation-induced ONFH models ranged from 60% to 100%. 114
Regardless of induction methods, impaired angiogenesis is consistently observed across different types of models. This impairment contributes significantly to the progression of ONFH, emphasizing the importance of enhancing angiogenesis to effectively manage and treat ONFH.115–117
Cell-based therapies and molecular innovations
The concept of promoting angiogenesis for the treatment of ONFH is based on the widely recognized critical role of adequate blood supply in bone health and repair. Enhancing new blood vessel formation helps to deliver nutrients and oxygen to the affected areas and facilitating the repair processes and bone regeneration of ONFH.
Recent advances in cell-based therapy, particularly those based on mesenchymal stem cells (MSCs), have emerged as potent strategies for enhancing angiogenesis in ONFH. Localized cell-based therapy relies on core decompression to create a bone tunnel, which serves as a pathway for implanting materials containing various cell types into the femoral head. Cells contained in these inplants, like MSCs, are capable of differentiating into various cell types, including vascular endothelial cells, and secrete a range of angiogenic factors that promote new blood vessel formation. 118 For example, Xu et al. developed a composite implant using carboxymethyl chitosan/alginate (CMC/ALG) scaffolds loaded with BMSCs and endothelial progenitor cells (EPCs). In vitro, this implant enhanced osteogenic and angiogenic differentiation while reducing adipogenic differentiation of BMSCs. 119 In a rabbit model of SONFH, a 27 mm bone tunnel from the greater trochanter to the femoral head was created via core decompression. Different implants were transplanted into the femoral head through this bone tunnel. Compared with other groups, the CMC/ALG/BMSC/EPC implant showed significantly faster bone tissue repair via promoting osteogenesis and angiogenesis and reducing adipogenesis. 119 Vascular cell adhesion molecule 1 (VCAM-1) is significantly downregulated in BMSCs from patients with trauma-induced ONFH. Motivated by this phenomenon, Shao et al. utilized a lentiviral vector to overexpress VCAM-1 in BMSCs, which enhanced the migration and improved the angiogenic potential of BMSCs in vitro. mRNA transcriptome sequencing revealed that VCAM-1 promoted angiogenesis via suppressing the cellular communication network factor 2 (CCN2)/Apelin signaling pathway. 120 However, MSC therapy for ONFH faces challenges such as limited cell self-renewal and donor availability. To address these limitations, Zhou et al. explored the use of MSCs derived from induced pluripotent stem cells (iPSCs). BMSCs of ONFH patients (ONFH-BMSCs) were reprogramed into iPSCs, following which the iPSCs were differentiated into MSCs (iPSC-MSCs). They found that these iPSC-MSCs had similar properties to normal MSCs, but showed higher proliferation ability and no tumorigenic potential. These iPSC-MSCs were then transplanted into an ONFH rat model through intrabone marrow injection. Specifically, a needle was inserted through the intercondylar region into the femur’s marrow cavity to deliver 0.2 ml of iPSC-MSCs (10⁷ cells/ml). The injection site was sealed with sterilized bone wax after the procedure. Micro-CT imaging and staining results indicated that this method effectively promoted bone repair and angiogenesis in a rat model of SONFH. 121 In the study by Zhao et al., 107 enhanced ossification and revascularization in the femoral head in a rabbit model of SONFH were obserfved following co-transplantation of angiotensin II-modified mesenchymal stem cells (Ang II-MSCs) and endothelial cells (ECs) at a 5:1 ratio through the core decompression tunnel under C-arm fluoroscopy of an X-ray machine. Lee et al. injected VEGF- and bone morphogenetic protein 2 (BMP2)-transfected ASCs into critical-size calvarial defects (4 mm) and long-bone segmental defects (4 mm) in a rat model. Their findings demonstrated that the transfected ASCs promoted rapid angiogenesis and osteogenesis, significantly enhancing bone regeneration while remaining localized at the defect site without migration by day 56. This approach holds potential for clinical application in the treatment of ONFH. 122
Exosomes are small extracellular vesicles secreted by various cell types. They function as cargos that can transfer proteins, lipids, and nucleic acids between cells. 123 Unlike cell transplantation, which typically requires core decompression and drilling into the femoral head to deliver the therapeutic cells, exosomes can be applied with higher flexibly for ONFH treatment. Exosomes can be administered either through direct injection into the femoral head via core decompression or by systemic delivery through the bloodstream or intra-articular injection. The minimally invasive systemic or articular delivery method represents a promising alternative for ONFH treatment. 124 For example, an engineered exosome-functionalized ECM-mimicking hydrogel (Lightgel-Li-Exo) was injected into the femoral head via core decompression tunnel in a rat model of GONFH. These exosomes enhanced macrophage M2 polarization, osteogenesis, and angiogenesis, ultimately promoting bone repair. 125 Intra-articular injection of exosomes from miR-1a-3p deprived, glucocorticoid-stimulated M1 macrophages suppressed adipogenic differentiation and promote osteogenic differentiation of BMSCs in a mouse model of GONFH. 126 In a GC-induced ONFH rat model, intra-articular injection of miR-26a-CD34+-Exos increased vessel density and improved trabecular bone integrity, thereby inhibiting the progression of ONFH. 48 Platelet-rich plasma-derived exosomes (PRP-Exos) encapsulating platelet-derived growth factor-BB (PDGFBB), transforming growth factor beta (TGF-β), FGF, and VEGF were identified to promote angiogenesis and inhibit apoptosis in ONFH. These exosomes, injected via the tail vein, activated the Akt pathway, increasing the expression of anti-apoptotic proteins such as Bcl-2 and countering the apoptotic effects of glucocorticoid-associated endoplasmic reticulum (ER) stress. 127 Another systematic delivery example is the microRNA (miR)-21-5p delivered by human umbilical MSC-derived exosomes (hucMSC-Exos). These exosomes, injected daily via the tail vein, were found to reduce the number of empty cavities or abscesses with poor necrotic trabecular bone structure and increased the volume and number of blood vessels via suppressing SOX5 and EZH2 transcription in ONFH rat models. 128 Other in vitro studies showed that exosomes derived from MSCs and endothelial cells have been shown to enhance angiogenesis by delivering pro-angiogenic factors directly to the affected area. 129 siRNAs encapsulated in BMSC-derived exosomes were found to be effective in promoting angiogenesis and osteogenesis in a cell model of ONFH.129,130 miR-26a overexpressed in CD34+ stem cell-derived exosomes (miR-26a-CD34+-Exos) enhanced endothelial cell migration and tube formation, as well as increased the osteogenic differentiation of BMSCs in vitro.48,127
Tissue engineering and delivery techniques
Tissue engineering involves the combination of scaffolds, cells, and regulatory signals to create functional tissues capable of repairing or replacing damaged structures. Several tissue engineering approaches have been synergistically combined with angiogenic therapy to enhance their therapeutic effects for ONFH treatment. 25 Emerging delivery systems are designed for different therapeutic needs based on their unique advantages (Figure 2). In Table 3, we summarized the key features, strengths, and limitations of different delivery systems, including nanoparticles (e.g. lipid nanoparticles), nanosuspensions, nanomicelles, nanofibers, liposomes, ethosomes, phytosomes, liquid crystals, microspheres, exosomes, dendrimers, hydrogels, AAV system, transdermal drug delivery systems, and nano and micro emulsions. The primary goals of these systems are to overcome biological barriers, improve drug bioavailability, enhance cell/tissue targeting ability, and achieve controlled and sustained drug release. 131 Take hydrogels as an example, which is a 3D biphasic material composed of porous, permeable solids and at least 10% (an approximate lower limit by weight or volume) of interstitial fluid. 132 This water content is essential for their characteristic ability to swell and retain moisture, which makes hydrogels particularly suitable for applications in tissue repair. Hydrogels’ high water content, biocompatibility, and ability to mimic the physical properties of biological tissues enable them to repair tissues with complex shapes and provide a platform for controlled drug release. 133 For example, Fu et al. developed a heat-sensitive mesoporous silica nanoparticle composite hydrogel that could be injected and undergo solidification under body temperature. With the gradual degradation of the hydrogel, the contained miR-21-5p was released, which promoted bone regeneration in a rat model of AONFH. 134 Moreover, hydrogels can be engineered to be highly sensitive to environmental stimuli, such as changes in pH, temperature, or ionic strength. For instance, thermosensitive hydrogels gelate at body temperature, facilitating minimally invasive delivery and localized therapeutic effects, and pH-sensitive hydrogels can release bioactive factors in response to the acidic microenvironment of necrotic bone. 135 However, the limited mechanical strength of hydrogels and concern for their long-term stability and potential toxicity remain as obstacles for their extended applications. 136 It should be noted that a lengthy exploration is often required before the translation of drug delivery techniques from basic research to clinical applications due to their biological complexities and safety concerns. 137 In this section, we introduce typical novel drug delivery systems as examples and illustrate their mechanisms of function.

Illustration of delivery systems for angiogenesis. There are diverse strategies employed to deliver angiogenic factors, each with unique advantages and applications. These advanced delivery systems are pivotal in the development of therapeutic approaches for enhancing angiogenesis, with significant implications for regenerative medicine and the treatment of various vascular diseases. Created with biorender.com.
Strengths and limitations of different delivery systems for modulating angiogenesis.
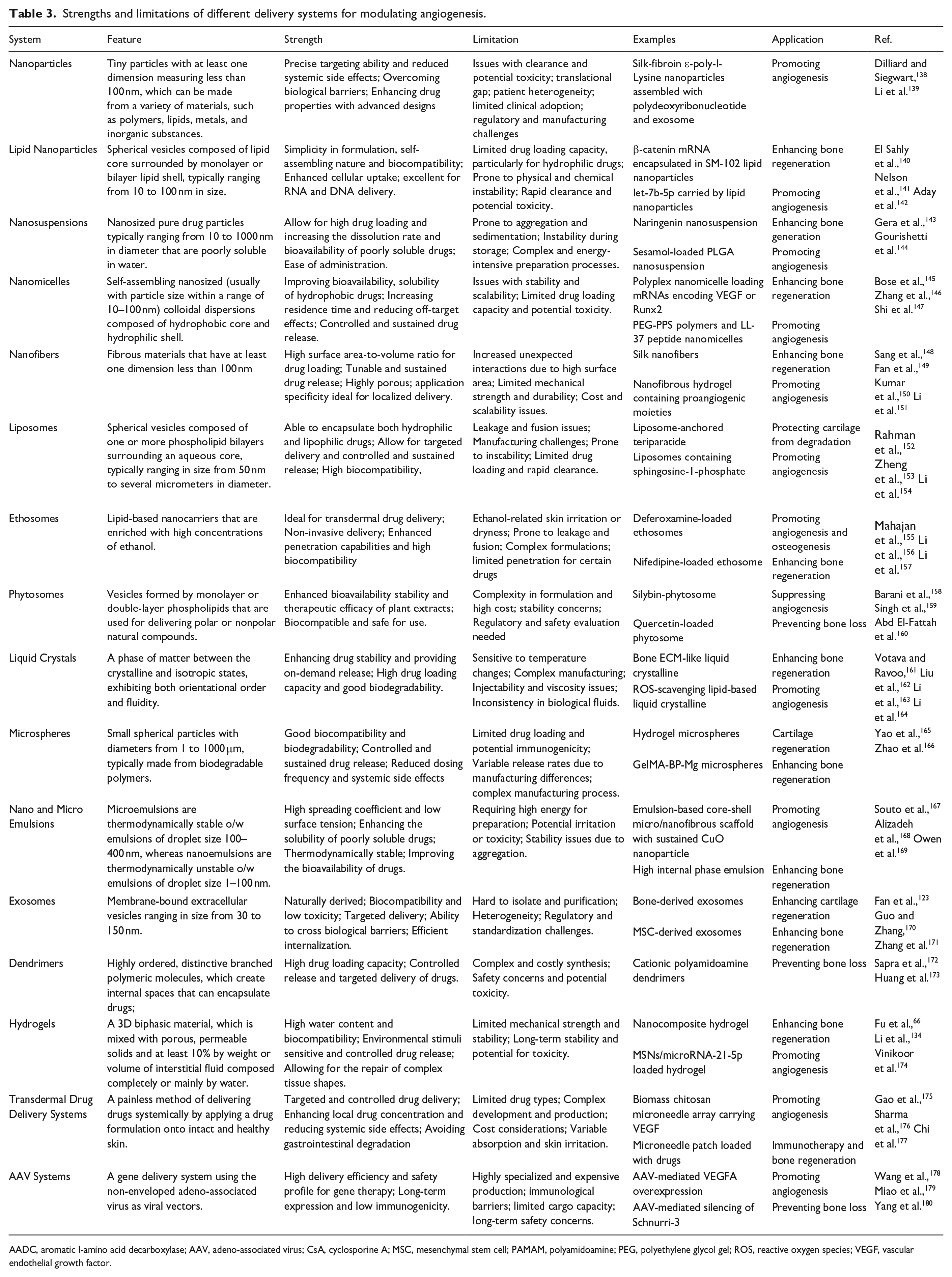
AADC, aromatic l-amino acid decarboxylase; AAV, adeno-associated virus; CsA, cyclosporine A; MSC, mesenchymal stem cell; PAMAM, polyamidoamine; PEG, polyethylene glycol gel; ROS, reactive oxygen species; VEGF, vascular endothelial growth factor.
The main goal of scaffold-based approaches is to provide a 3D structure that supports cell attachment, proliferation, and differentiation. Besides, 3D scaffolds, such as hydrogels, can encapsulate cells and biomolecules, such as growth factors, and be engineered to release the encapsulated molecules gradually to promote angiogenesis and bone repair for a relatively long period. 181 Currently engineered scaffolds often combine different types of materials to generate a complex network, leveraging the strengths of each to achieve better results. For example, Lai et al. integrated poly (lactide-co-glycolide) (PLGA), β-tricalcium phosphate (TCP) and magnesium (Mg) powder to formulate a porous PLGA/TCP/Mg scaffold using a low-temperature rapid prototyping technology. This scaffold exhibited osteogenic and angiogenic properties in an GONF rabbit model. 181 Zhu et al. designed a platelet-coated micro-scaffold composed of cross-linked gelatin within a 3D-printed porous titanium framework. This scaffold has a stable, degradable structure that promoted osteogenesis and vascularization in a rabbit ONFH model through the slow release of growth factors, specifically TGF-β1 and VEGF, over 21 days. 182 Wang et al. 26 developed a novel tissue engineering scaffold by compositing CaO2/gelatin microspheres with a porous scaffold, gelatin/alginate hydrogel, and BMSCs. In their research, medical-grade gelatin and calcium peroxide were cross-linked with glutaraldehyde to create microspheres (Figure 3(A)). A 3D multi-layer porous tubular structure was printed using a mixture of nano-hydroxyapatite and polycaprolactone. Gelatin-coated microspheres were molded into rod-shaped complexes and inserted into the tubular scaffold. BMSCs were encapsulated within a gelatin and sodium alginate matrix. This mixture was injected into the scaffold containing the microspheres. 26 This scaffold was capable of providing necessary stiffness and releasing oxygen via the decomposition of calcium peroxide within the microspheres over 19 days (Figure 3(A)). In vivo and in vitro tests demonstrated its high biocompatibility and ability to promote cell proliferation in hypoxic conditions and reduce local cell apoptosis. Additionally, the composite 3D scaffold significantly enhanced osteogenesis and angiogenesis in a rabbit ONFH model demonstrating improved efficiency of a tissue engineering treatment for ONFH. 26 In addition to combining materials of different properties, coating is another effective method to add new characteristics to the scaffold. For example, the presence of a Matrigel coating on the CMC/ALG scaffold provided a friendly environment for cell adhesion and proliferation in a rabbit model of SONFH. 119 In this scaffold, the loaded BMSCs and EPCs promoted bone regeneration and angiogenesis while reducing fat accumulation. 119 Certain biomaterials can stimulate desired cellular behaviors by creating specific microenvironments or releasing metal ions. For example, Quinlan et al. developed hypoxia-mimicking bioactive glass/collagen glycosaminoglycan composite scaffolds. These scaffolds significantly enhanced the production and expression of VEGF in endothelial cells and promoted the formation of new blood vessels and bone tissue, representing a promising option for ONFH treatment (Figure 3(B)). 183 Zamani et al. incorporated zinc and magnesium ions (SiO2-P2O5-CaO-ZnO-MgO, 60% SiO2, 26% CaO, 4% P2O5, 5% ZnO, 5% MgO, in mol.%) into bioactive glass-ceramic particles. Their results showed that incorporating iron-containing bioactive glasses into alginate networks improved the antibacterial efficacy, biological activity, and mechanical properties, including toughness, elastic modulus, and compressive strength, of the bone composite scaffolds (Figure 3(C)). 184 Eriksson et al. developed S53P4 bioactive glass scaffolds with a composition of 53% SiO2, 23% Na2O, 20% CaO, and 4% P2O5. This scaffold, after undergoing pressureless sintering, exhibited a significantly higher porosity of approximately 50% and greater compressive strength of 4.80 MPa than previously reported bioactive glass scaffolds. These characters make the scaffold suitable for load-bearing applications in long bone defects. In vivo studies found that this scaffold induced BMP expression and osseointegration in a rabbit critical-sized bone defect model. 185 Bian et al. developed a bifunctional scaffold made by integrating MgCuFe-Layered Double Hydroxide (LDH)-derived polymetallic sulfide (MCFS) nanosheets into a 3D-printed bioactive glass scaffold (BGS). The MCFS nanosheets incorporate Mg²⁺, Cu²⁺, and Fe³⁺ ions and enhance bone regeneration and vascularization. In a rabbit cranial defect model, BGS/MCFS resulted in an 8.5-fold increase in bone mass and a 2.3-fold increase in neovascularization compared to pristine BGS (Figure 3(C)). 186 Gómez-Cerezo et al. 187 developed a type of mesoporous scaffold by mixing ε-polycaprolactone (PCL) with mesoporous bioactive glass (MBG-58S) powder. Drugs can be incorporated and released controllably over 30 days with the degradation of the scaffold. This complex scaffold effectively supported angiogenesis and bone regeneration in an osteoporotic sheep model (Figure 3(D)). 187 However, the role of these bioactive glass scaffolds on angiogenesis and ONFH has not been investigated, which warrants further research in the future.

Scaffold-based bioengineering approaches to ONFH management. (A) The CaO2/gelatin-based, oxygen-releasing microspheres and the 3D printed PCL/nHA tubular scaffold were molded into rod-shaped complexes. Micro-CT confirmed bone regeneration and immunohistochemical staining of CD 31 indicated angiogenesis in the femoral heads in rabbit ONFH model 4 weeks after implantation. Sc, PCL/nHA Scaffold; Hy, hydrogel; Cc, calcium carbonate; CPO, calcium peroxide. Each value is the mean ± standard error of mean (n = 3); Figures reproduced with permission from Wang et al., 26 Copyright 2021, RSC. (B) Images of toluidine blue-stained cobalt bioactive glass/collagen-glycosaminoglycan scaffolds. These two scaffolds, with different structural integrity and porosity, are prepared at a controlled freezing rate of 1 and 4℃/min, respectively. Scanning electron microscope images revealed that these scaffolds, loaded with small- and large-diameter bioactive glass particles, elute cobalt effectively. Figures reproduced witi perrmission from Quinlan et al., 183 Copyright 2015, Elsevier Ltd. (C) Schematic illustration of the fabrication of a versatile MCFS nanosheet-functionalized 3D-printed BGS for periprosthetic infection preven tion/treatment and vascularized osteogenesis confirmed by CLSM and digital images. Figures reproduced witi permission from Bian et al. 186 (D) Scanning electron micrographs illustrating the structural changes in MBG-PCL-zol scaffolds before and after 21 and 30 days of drug release tests, respectively. Confocal images of the morphology of osteoclast-like cells 1 and 6 days after immersing the MBG-PCL-zol scaffolds in the medium of osteoclast cultures. Actin was stained with rhodamine-phalloidin (red) and cell nuclei with DAPI (blue). Figures reproduced with permission from Gómez-Cerezo et al., 187 Copyright 2019, Elsevier Ltd.
Recent studies have also explored the use of nanoparticles as carriers of angiogenic growth factors, aiming to improve the precision of delivery and the duration of therapeutic effect. Their affinity for specific tissues makes these particles a versatile platform for releasing growth factors, genes, and other bioactive molecules at the targeted site. 66 Previous studies have shown that growth factor-loaded nanoparticles and gene delivery nanoparticles enhanced angiogenesis and bone regeneration in animal models of ONFH.66,188 For example, Xu et al. 111 developed a porous nano-hydroxyapatite/polyamide 66 scaffold, which demonstrated significant potential in supporting bone regeneration in dog ONFH model. Microcapsules and microspheres are another class of delivery vehicles that can encapsulate cells, growth factors, and drugs. These systems offer controlled release properties and can be engineered to target specific tissues. For example, CaO2/gelatin microspheres developed by Wang et al. 26 constantly released oxygen, which facilitated the treatment of ONFH via enhancing angiogenesis and survival of grafted stem cells. Currently, research into nanotechnology-based therapies of ONFH, especially those for promoting angiogenesis, is limited. Studies in related areas can provide valuable insights. 189 For example, researchers used functionalized mesoporous silica nanoparticles in a porcine model of myocardial infarction to achieve anti-inflammatory and proangiogenic effects. 134 A biomimetic, hierarchical scaffold composed of nanoparticles (DFO@PCL NPs) facilitated bone regeneration and angiogenesis via activating the hypoxia-inducible factor-1α pathway. 190 Oxygen-releasing nanoparticles loaded with MSC-Exos promoted angiogenesis and muscle regeneration without causing significant inflammation or overproduction of ROS. 191 A dual delivery system developed by O’Brien et al. 192 incorporated a miR-210 mimic and a miR-16 inhibitor within a collagen-nanohydroxyapatite scaffold, which could enhance bone repair by enhancing angiogenic-osteogenic coupling. However, despite significant advancements in tissue engineering and biomolecule delivery techniques, several challenges remain, including the limited availability of donor cells, potential immune reactions, and the need for precise control over the delivery and release of therapeutic agents.
Other angiogenesis-promoting methods
Non-invasive therapies have been actively pursued in the treatment of ONFH. These therapies offer significant benefits by minimizing the risks and complications associated with invasive surgical procedures. For example, extracorporeal shockwave treatment (ESWT) is a non-invasive treatment option for various diseases, including musculoskeletal and vascular disorders. High-dosage ESWT (6000 impulses of ESWT at 24 kV) for 1 month was found to be associated with improved angiogenesis and osteogenesis, anti-inflammatory effects, higher pain threshold, and tissue regeneration. 193 Wu et al. 194 found that ESWT promoted the proliferation, migration, and angiogenesis of endothelial cells, relieving endothelial injury and dysfunction in steroid-induced rat ONFH via downregulating miR-135b and FOXO1 modulation. Another type of therapy, hyperbaric oxygen therapy (HBOT), involves breathing pure oxygen in a pressurized environment, which increases oxygen delivery to tissues. 195 This therapy has been reported to enhance angiogenesis and improve tissue healing under various conditions, like accelerating skin wound healing in diabetic mice and reducing fibrosis in patients with breast cancer who received adjuvant radiotherapy for breast cancer.196,197 HBOT may be particularly useful in early-stage ONFH to enhance the oxygenation of the necrotic area and support new blood vessel formation. 198 Low-intensity pulsed ultrasound (LIPUS) is another non-invasive method that uses ultrasound waves to stimulate tissue repair and angiogenesis. LIPUS has been shown to enhance the expression of angiogenic factors and promote blood vessel formation in ONFH. LIPUS therapy is considered safe and can be applied as an adjunct to other treatments. 199 The efficacy of exercise and mechanical loading in treating ONFH is under debate. Weight-bearing activities and specific exercise regimens can promote blood flow and enhance the delivery of nutrients and oxygen to the necrotic area, thereby supporting the repair process. Though physical activities can stimulate angiogenesis through the physical stress they impose on bones and tissues, the optimal intensity and frequency of such activities have not yet been standardized.
Natural compounds and nature-derived molecules represent an emerging category of therapeutics for ONFH treatment, with several preclinical studies demonstrating their potential to modulate inflammation, enhance vascularization, regulate metabolism, combat oxidative stress, and promote bone regeneration., For example, icariin, a bioactive compound derived from the Epimedium plant, promoted microvascular endothelial cell migration, tube formation, and expression of angiogenesis-related cytokines via activating protein kinase B (Akt), increasing B-cell lymphoma 2 (Bcl-2) expression and reducing Bcl-2-associated X protein (Bax) expression. In vivo, icariin treatment resulted in a lower ratio of empty lacunae, higher blood vessel volume, and more CD31-positive cells in a rat model of GONFH. 200 Resveratrol and urolithins, found in grapes and pomegranates, have been noted for their protective effects on bone health through their anti-inflammatory, antioxidant, and anti-apoptotic properties.201,202 Vitamin B2, also known as riboflavin, is a vital nutrient that plays a crucial role in energy production, and cellular antioxidant defense system and aids in the metabolism of fats, drugs, and steroids.203,204 Recent research has highlighted the potential therapeutic benefits of vitamin B2 in the treatment of ONFH. 170 Guo and Zhang 205 found that vitamin B2 can inhibit ONFH-like changes by suppressing cell apoptosis, promoting blood vessel regeneration, and increasing bone mass in rat models of GONFH. In vitro studies using HUVECs have further supported these findings, showing that vitamin B2 enhances cell migration, boosts the expression of angiogenesis-related factors, and inhibits cellular apoptosis. These effects are believed to be mediated through the activation of the phosphoinositide 3-kinase (PI3K)/Akt signaling pathway. 205 Osthole is a bioactive derivative of coumarin. It has an extensive range of pharmacotherapeutic effects, including anti-inflammatory, antioxidant, anti-tumor, angiogenetic and neuroprotective properties. A study revealed that osthole reversed the detrimental effects of ethanol on BMSCs and HUVECs by modulating the Wnt/β-catenin pathway. In vivo study further confirmed that osthole enhanced bone formation, increased angiogenesis, and reduced adipogenesis in a rat model of AONFH. 52 Aldehyde dehydrogenase 2 (ALDH2) is a crucial enzyme in the metabolism of ethanol. It plays a pivotal role in the detoxification process by converting acetaldehyde, a toxic byproduct of alcohol metabolism, into less harmful acetic acid. 206 A recent study investigated the protective role of ALDH2 in AONFH. In a rat model, Alda-1, an ALDH2 activator, protected against ethanol-induced ONFH by enhancing bone formation, reducing adipogenesis, and promoting angiogenesis. The protective effects were achieved via activating PI3K/AKT and AMPK signaling pathways. 206
Various pharmacological agents and bioactive molecules play significant roles in the treatment of ONFH. Deferoxamine, for instance, is an iron chelator that can remove excess iron, reduce oxidative stress, and promote angiogenesis in a rabbit SONFH model. 207 Traditional Chinese medicine (TCM) represents a promising source of therapeutic agents for ONFH treatment, which may have multiple pharmacological actions. For example, polydatin, a natural bioactive ingredient extracted from the roots of the Reynoutria japonica Houtt, was found to promote the proliferation and osteogenic differentiation of BMSCs. 208 Lai et al. developed a porous PLGA/TCP scaffold containing icariin, a bioactive component derived from Herba Epimedii. This scaffold was precisely printed to provide mechanical support and stable icariin release, which improved angiogenesis and bone regeneration in rabbit model of SONFH. 209 Icariin was also found to activate autophagy and rejuvenate osteogenesis of senescent BMSCs, thereby alleviating inflammaging and bone loss in osteoporotic mice. 210 Other typical TCM that possess osteogenic function and promote bone regeneration include but are not limited to Drynariae Rhizoma, Psoralea corylifolia, Astragalus radix, Eucommiae Cortex, Deer Antler, curcumin and resveratrol.211,212 Xu et al. developed a novel 3D-printed bone repair scaffold (SGC) by incorporating xonotlite nanofibers into a silk fibroin/gelatin scaffold. This SGC scaffold promoted osteogenic and angiogenic differentiation of bone mesenchymal stem cells and reprogramed macrophages to create a favorable osteoimmune microenvironment. 213 Synthetic growth factors, such as recombinant BMP-2 and VEGF, directly stimulate bone growth and vascularization. 214 Statins, commonly used for lowering cholesterol, have been shown to promote angiogenesis and bone healing. 215 Zinc, magnesium, and iron ions are important bioactive agents in bone tissue. 216 The combined supplementation of Mg and vitamin C significantly inhibited osteoclast differentiation of BMCs and promoted angiogenesis in a rat model of GONFH. 217 A randomized controlled clinical trial conducted by Zhao et al. confirmed that biodegradable Mg screws were reliable options for the fixation of vascularized bone graft in ONFH patients. Patients in the Mg screw group showed improved functional recovery and increased Harris hip score at 12-month post-operative follow-up without increased risk of side effects. 218 A self-adaptive biomimetic periosteum composed of a novel interpenetrating double network hydrogel developed by Zhou et al. 219 continuously releases NO, activating the NO-cGMP signaling pathway, thereby enhancing angiogenesis and osteogenesis. Other pharmacological agents, such as bisphosphonates and prostaglandin E1, hold the potential to enhance blood flow and support bone regeneration in ONFH.220,221
In addition, external shape and internal architecture of scaffolds and tissue engineering constructs can dramatically impact tissue regeneration outcomes beyond the addition of growth factors, as different geometric designs can modulate the cellular environment and vascularization. For instance, scaffolds with smaller fiber diameters and higher porosity, such as those fabricated using melt electrowriting (MEW), have been shown to promote better bone regeneration by fostering dense microvascular networks, while scaffolds with larger fiber diameters, like those made with fused deposition modeling (FDM), support the formation of larger blood vessels. 222 Yan et al. 223 used FDM to produce scaffolds with PCL, 6-hexanediamine, and angiogenic agents, which showed enhanced vascular regeneration and improved bone formation in a rat large bone defect model. The type H vessel, which is a specific capillary subtyte, has been found to couple angiogenesis with osteogenesis via regulating various signal patways, such as Notch, PDGF-BB, Slit3, HIF-1α, and VEGF. 224 Chitosan (CS)-based microporous scaffolds have gained attention for its role in promoting and angiogenesis during bone regeneration. 225
Discussion
Overview of ONFH and current treatment strategies
ONFH remains a challenging orthopedic condition with a significant impact on the patients’ quality of life as it progresses toward joint dysfunction. Current recommendations for ONFH treatment are listed in Table 4.4,5 Nonsurgical treatment modalities, such as weight-bearing restriction and pharmacological therapies may improve outcomes when an early diagnosis is made. 226 Once the disease progresses to late stages characterized by the presence of a crescent sign, femoral head flattening, and acetabular involvement, total hip arthroplasty becomes the only viable treatment option. 227 Innovative approaches that target the underlying pathology of the disease, particularly vascular insufficiency, are currently under extensive investigation. Researchers are relentlessly exploring a diverse array of potential treatments, including antioxidants like Resveratrol, extracellular vesicles like exosomes, cell-based therapy, cutting-edge biotechnologies such as gene editing, metabolic reprograming, and non-operative physical therapy such as high-energy focused extracorporeal shock wave.228–232
Recommended treatments for ONFH.

Grade A indicates good evidence (Level-I studies with consistent findings); grade B, fair evidence (Level-II or III studies with consistent findings); grade C, poor-quality evidence (Level-IV or V studies with consistent findings); and grade I, insufficient or conflicting evidence not allowing a recommendation.
Challenges facing angiogenic therapies
Recent advances in regenerative medicine and bioengineering have fueled the exploration of angiogenic therapies as potential treatments for ONFH. 233 In vitro and in vivo studies confirmed that the restoration of blood supply via enhancing blood vessel formation significantly prevented the apoptosis of osteocytes and the loss of trabecular bone.45,234 While therapeutic targeting of angiogenesis represents a promising direction in ONFH treatment, several challenges remain. First, the application prospect of these new treatments in clinical practice has not been clearly defined. Comprehensive multicenter clinical trials should be conducted to evaluate the long-term safety and efficacy of these approaches before their clinical application. Another concern is the heterogeneity of ONFH. Because of various pathologies, the response to angiogenic therapies may differ based on the underlying cause of the disease and in diverse patient populations. 2 Thus, personalized treatment approaches may be necessary to maximize the therapeutic outcomes. Additionally, real-time imaging and monitoring technologies should be applied to in vivo and clinical research, as useful insights can be obtained by monitoring the dynamics of angiogenesis within the femoral head. 235
Bioengineered scaffolds and bioprinting innovations
Bioengineered scaffolds are a highly active research field attracting increasing attention. Scaffolds implanted into the femoral head can provide a 3D structure that mimics the natural extracellular matrix, which facilitates cell adhesion, proliferation, and differentiation. 183 Angiogenic factors or cells incorporated in these scaffolds can enhance the therapeutic potential of these structures by gradually releasing the loaded or cell-secreted angiogenic factors (Figure 4). This sustained release mechanism creates a well-modulated environment and ensures a prolonged angiogenetic effect. 196 Advanced techniques like 3D bioprinting are being investigated to create more precise and functional tissue constructs for ONFH treatment.182,236 However, potential undesirable vascular growth in adjacent areas has to be avoided, which requires more precise regulatory and delivery systems.

4D bioprinted self-folding vascular structures. (A) 4D bioprinting of cell-laden tubular structures: (a) Schematic of the 4D bioprinting process for cell-laden AlgMA or HA-MA structures. Cells are mixed with AlgMA or HA-MA and printed on a substrate. Figures reproduced with permission from Kirillova et al., 240 Copyright 2017, Wiley-VCH. (b) The printed structures are crosslinked using green light, followed by drying. (c) The dried constructs are folded into tubular shapes upon immersion in water, PBS, or cell culture media. (d) Microscopic images of the cell-laden tubular structures after folding, demonstrating structural integrity and cell viability (scale bar: 400 µm). (B) Self-folding PEGDA bilayer structures: (d–f) The first and second layers are photocrosslinked sequentially, leading to a self-folding construct when immersed in water. (g and h) Microscopic images showing various self-folded micropatterns in the bilayer structures. (i) Fluorescent imaging of cell-laden scaffolds indicates successful cell seeding and cell distribution (scale bar: 200 µm). Figures reproduced with permission from Jamal et al., 241 Copyright 2013, Wiley-VCH. (C) 4D printing and transformation of vascular structures: 3D bioprinting or PDMS molding to create a grid-like scaffold, followed by transformation into complex vascular shapes in aqueous solutions. Magnetically driven shape transformation showing the 2D-to-3D transition to form branching vascular structures. Confocal microscopy images of the constructed vascular channels showing network formation by NHLFs and HUVECs within the matrix (scale bar: 100 µm). Figures reproduced with permission from Xie et al. 239
4D bioprinting incorporates time as the fourth dimension and uses smart materials and smart design to create dynamic 3D structures. 237 The key feature of 4D bioprinting is that the produced constructs can change their shape or functionality in response to external stimuli, such as water, heat, pH, light, electric current, and magnetic fields. 238 Unlike static 3D bioprinted products, 4D bioprinted products are programmable and capable of undergoing controlled dynamic changes, making them uniquely advantageous for certain biomedical applications. 239 For example, 4D bioprinted vascular scaffolds can not only mimic the geometry of native blood vessels, but also dynamically respond to the surrounding biological milieu. Figure 5 provides several examples of 4D bioprinted constructs. These constructs were designed to act as dynamic scaffolds that can support and guide the formation of new blood vessels.240–242 The creation of dynamic and functional vascular structures via 4D bioprinting presents transformative possibilities for ONFH treatment. As 4D bioprinting technology continues to evolve, its application in orthopedic regenerative medicine could revolutionize the treatment of ONFH. Future research will likely focus on optimizing the material properties and design of bioprinted vascular constructs to enhance their functionality and integration with host tissues.

Cell and cellular component-based therapy on angiogenesis and osteogenesis. (A) Schematic diagram illustrating the sources and types of stem cells and cell-derived component used as advanced therapeutics. Stem cells can be derived from bone marrow, adipose tissue, muscle, neonatal tissues, dental pulp, and skin. Different types of stem cells have been employed in relevant previous studies, including iPSCs, bone marrow stem cells, embryonic stem cells, and adipose-derived stem cells. The processes may involve cell culture, identification, intervention, and extraction of required components. (B) Effects of PRP-Exos on angiogenesis of cells or tissue treated with glucocorticoids. Osteogenesis and angiogenesis of the femoral head in each group were analyzed by immunohistochemical staining. The red arrows indicate vessels. Scale bars = 100 μm. 3D micro-CT images showing the subchondral region of the femoral head in each group. Figures reproduced with permission from Xu et al., 119 Copyright 2021, Wiley Periodicals LLC. (C) The angiogenesis of HMEC-1 cells treated with DEX alone or with Dex + PRP-Exos was evaluated using a tube formation assay. Angiographic images show the blood supply in the different groups. Scale bar: 100 μm. Figures reproduced with permission from Tao et al., 127 Copyright 2017, Ivyspring International Publisher.(D) Angiographic images showing the blood supply in a rat model of ONFH treated with different interventions. The density and structure of vessels in each group are visualized in red. Figures reproduced with permission from Zuo et al. 48
In addition, 3D/4D bioprinting holds great potential in not only providing regenerative constructs for ONFH management but also generating in vivo-like multi-tissue models for replicating the (patho)physiology of ONFH and predicting the safety and efficacy of potential drugs. 239 Besides, organs-on-chips (OoCs) are micro-engineered living systems that recapitulate the functions and interactions of human organs on a miniature, chip-like platform. These devices integrate human cells, 3D scaffolds, and physiologically relevant microenvironments to replicate the physiological and pathological processes of human organs. 243 There are currently no OoCs specifically developed for ONFH. Filling this gap with a dedicated ONFH-relevant OoC model could profoundly impact the understanding, diagnosis, and treatment of the disease, offering a more precise and comprehensive approach to tackling ONFH.
Conclusion
In this review, we comprehensively summarized the advancement and challenges in angiogenesis-targeted treatments of ONFH. As we have detailed, the use of angiogenic therapies is a promising strategy to enhance blood supply and support tissue regeneration in the femoral head. Emerging biochemical agents and advanced biotechnologies, such as gene editing, stem cell therapies, bioengineered scaffolds, and 4D bioprinting, provide exciting opportunities for future precision medicine. Several challenges remain regarding the translation of research findings related to ONFH mechanisms and intervention into clinical practice, including safety concerns, ethical issues, technical complexity, individual variability in response to treatments, and financial barriers. A deeper understanding of the molecular mechanisms underlying compromised angiogenesis in ONFH is expected to result in innovative, efficacious therapeutic interventions, which will greatly benefit from interdisciplinary efforts by clinicians, biologists, and tissue engineers.
Footnotes
Acknowledgements
We thank Prof. Hang Lin from the University of Pittsburgh for his helpful comments on the manuscript.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Science and Technology Innovation Program of Hunan Province (No. 2023JJ40823), Health Research Project of Hunan Provincial Health Commission (No. W20243122) and Scientific Research Launch Project for new employees of the Second Xiangya Hospital of Central South University (QH20230212). ZAL gratefully acknowledged the Vice-Chancellor Early Career Professorship Scheme of CUHK
