Abstract
Short sequences of amino acids called peptides have a wide range of biological functions and the potential to treat a number of diseases. Bioactive peptides can be derived from different sources, including marine organisms, and synthetic design, making them versatile candidates for production of therapeutic agents. Their therapeutic effects span across areas such as antimicrobial activity, cells proliferation and migration, synthesis of collagen, and more. This current review explores the fascinating realm of bioactive peptides as promising therapeutic agents for skin wound healing. This review focuses on the multifaceted biological effects of specific peptides, shedding light on their potential to revolutionize the field of dermatology and regenerative medicine. It delves into how these peptides stimulate collagen synthesis, inhibit inflammation, and accelerate tissue regeneration, ultimately contributing to the effective repair of skin wounds. The findings underscore the significant role several types of bioactive peptides can play in enhancing wound healing processes and offer promising insights for improving the quality of life for individuals with skin injuries and dermatological conditions. The versatility of peptides allows for the development of tailored treatments catering to specific wound types and patient needs. As continuing to delve deeper into the realm of bioactive peptides, there is immense potential for further exploration and innovation. Future endeavors may involve the optimization of peptide formulations, elucidation of underlying molecular and cellular mechanisms.
Introduction
One of the major issues facing the world today is delayed wound healing, which has substantial therapeutic and financial implications. Numerous forms of epidermal skin injury are regularly caused by burns, hot water or oil, flames, diseases, accidents, and amounting to hundreds of patients each year. Around 30,000 people die annually from burns and scalds, according to the World Health Organization (WHO). The process of wound healing can be either acute or chronic, and it may suggest that certain treatments have substantial limits on how effectively they work to cure the underlying conditions. 1 In addition, it is estimated that 2.6 million Malaysians are diabetic, where 15.2% of our total population and these numbers have been projected to increase sharply over the years. The statistics of diabetes mellitus disease are of great concern because Malaysia is the country with the highest number of people with diabetes in Southeast Asia and ranked 10th in the world. The diabetic foot ulcer could affect 15%–25% of diabetic patients and becoming one of the major problems for those chronic wounds.2,3 It shows an important need to identify new and more effective therapeutic agents as drug candidates to become realistic options for wound treatment.
Up till now, the utilization of therapeutics has been developed over time and continues to evolve with changes in drug development. A wide range of therapeutics have been studied extensively and approaches to support wound healing such as metal nanoparticles,4,5 scaffolds,6,7 bioactive dressings,8,9 hydrogels, 10 polymeric nanostructures, growth factors, 11 secretomes, 12 and membranes. 13 However, the efficacy of these therapeutics are limited by constrains of side effects, besides being inefficient and costly. 14 The new therapeutic agent that is applied to the wound, in combination with support or carrier or alone, have become interesting strategies and necessary to improve their efficacies toward wound treatment. 15 With the discovery of peptide, the bioactive peptide with good stability, high activity and specificity have shown promise in the field of medicinal chemistry especially for wound treatment. Peptides represent a unique class of pharmaceutical compounds in which it is molecularly poised between small molecules and proteins. It has several benefits over other medicinal medications, including several biological activities that seem to be health-promoting, the ability to synthesis collagen because it is a building block of proteins, and affordability.16–18 Peptides are emerging as promising candidates for wound healing due to their diverse biological activities and ability to influence various aspects of the healing process. These short chains of amino acids can mimic natural biological processes, promoting cell proliferation, migration, and tissue regeneration. Peptides play crucial roles in modulating inflammation, enhancing collagen production, and preventing infections. Additionally, peptides can be engineered to have specific functions, making them versatile tools for targeted therapy. Their biocompatibility, low toxicity, and potential for reducing scarring further highlight their therapeutic potential in improving wound healing outcomes. Towards this end, several intrinsic peptides have shown promise in promoting wound healing due to their various biological activities. For instance, transforming growth factor-Beta (TGF-β) stimulates collagen production and modulates inflammation; epidermal growth factor (EGF) is known to promote cell proliferation and migration, accelerating re-epithelialization; fibroblast growth factor (FGF) enhances angiogenesis and granulation tissue formation; platelet-derived growth factor (PDGF) attracts cells to the wound site and stimulates cell growth and division; defensins play a role in killing pathogens and regulating the immune response, aiding in maintaining a sterile wound environment; while collagen peptides derived from collagen, can enhance collagen synthesis, improve skin elasticity, and support the structural integrity at the wound site.19,20
Improper wound healing occurs when the body’s natural process of repairing tissue is disrupted, often due to factors such as infection, poor blood circulation, chronic diseases (like diabetes), nutritional deficiencies, certain medications, or lifestyle choices such as smoking and alcohol consumption. This can lead to complications including chronic, non-healing wounds, increased risk of infection, abnormal scar formation, and potential loss of function in the affected area. Proper wound care, infection control, nutritional support, lifestyle modifications, and management of underlying health conditions are essential for promoting effective wound healing and preventing complications. Therefore, to avoid the wound condition from becoming worse, cells that have evolved bioactive peptides were lead to distribute treatments to the wound site in a precise manner and to improve therapeutic outcomes of the wound’s response to drug.21,22 In particular, the therapeutic activity of antimicrobial peptides (AMPs) is noteworthy for tissue and wound healing since they are attractive candidates that can prevent the progression of microbial infections on wounds, a process inevitably associated with delayed wound healing. AMPs are mostly produced by epithelial surfaces and have been described as mediators of the body’s innate defense response that directly kill or inhibit the growth of microorganisms. They are the short peptides usually comprising a stretch of 12–50 amino acids that have broad-spectrum microbial activity.
In the case of skin injury, AMPs expression is highly up-regulated at the wound edge due to increased synthesis by keratinocytes and deposition from degranulation of recruited neutrophils, which indicates that AMPs are potential wound-healing stimulator.23,24 The rapid bactericidal activity of AMPs makes them promising candidates for therapeutic action which is broad range in certain areas such as complicated skin and soft tissue infections, where a rapidly increasing incidence of poly-microbial infections involving both Gram-positive and Gram-negative organisms has been reported over the last decade. 25 AMPs generally target microbial membranes, leading to their disruption and subsequent cell death. Their main mechanisms include membrane disruption, wherein they interact with microbial membranes, integrating into the lipid bilayer and causing physical disruption, pore formation, or complete membrane disintegration. Alternatively, certain AMPs can penetrate microbial cells and interfere with intracellular functions such as protein synthesis, nucleic acid synthesis, or enzyme activity. Furthermore, AMPs can modulate the host immune response, enhancing the clearance of infections and promoting healing. 26 Studies have demonstrated that arginine and lysine amino acid substitution in peptides favorably facilitate insertion into bacteria membrane due to their positive charge interacting with bacterial membrane, while the presence of hydrophobic amino acids such as tryptophan and phenylalanine within peptide sequences are associated intensified antimicrobial activity against bacteria.27,28 Alternatively, existing literature strongly indicates that most classes of AMPs possess certain characteristic amino acids, most notably proline, tryptophan, histidine, glycine and arginine; with certain classes of AMPs described to possess disulfide linkages due to presence of cysteine. 29 For example, defensins and cathelicidins are among the most well studied classes of AMPs known to possess immunomodulatory properties beneficial for promoting wound healing.
Beyond the scope of AMPs, there exists other categories of peptides that are extremely relevant in the context of wound healing therapeutics. Among them, collagen peptides are gaining significant attention in the field of wound healing due to their remarkable ability to support and enhance the body’s natural repair processes. Collagen, the most abundant protein in the human body, provides structural integrity to various tissues. 30 When broken down into smaller chains, known as collagen peptides, this protein becomes highly bioavailable and effective in promoting wound healing. Notably, Type-I collagen peptides predominantly present in skin provides tensile strength and structural support, forming the primary framework for new tissue growth; while Type-III collagen Peptides found alongside Type-I collagen in skin is essential for the formation of granulation tissue during the early stages of wound healing, which helps maintain the structural integrity of newly formed tissues. Additionally, Type-IV collagen peptides located in the basal lamina, plays a pivotal role in cell adhesion due to abundance of RGD-motifs, thereby supporting the re-epithelialization process by providing a scaffold for new cell growth. 31 Yet another class of short peptides functioning as matrix metalloproteinase inhibitors (MMPI’s), modulate MMP activity, and prevent excessive tissue breakdown and promote proper tissue remodeling during healing. 32 Alternatively, angiogenic peptides play a critical role in wound healing by promoting the formation of new blood vessels through angiogenesis. This is essential for providing the necessary oxygen and nutrients to the regenerating tissues, facilitating rapid and effective healing. Angiogenic peptides, such as vascular endothelial growth factor (VEGF) and FGF, stimulate endothelial cells to proliferate, migrate, and form capillary structures within the wound site; while thymosin β-4 promotes angiogenesis, and reduces inflammation. By enhancing blood flow to the affected area, these peptides ensure that immune cells and growth factors are efficiently delivered to the wound, supporting autolytic debridement and the formation of granulation tissue. 33
Consequently, researchers are increasingly interested in using peptides for skin wound healing due to their potent bioactivity, specificity, and minimal side effects. Peptides can effectively promote various aspects of the healing process, including inflammation modulation, antimicrobial action, and tissue regeneration. They can be designed to target specific cellular pathways, enhancing fibroblast proliferation, collagen synthesis, and angiogenesis, which are crucial for rapid and efficient wound closure. Additionally, peptides can be engineered for stability and targeted delivery, making them versatile tools in developing advanced wound care therapies. Their ability to integrate seamlessly with the body’s natural healing mechanisms positions peptides as promising candidates for improving wound healing outcomes. Nevertheless, the enthusiasm for peptide therapeutics was subsequently tempered by certain limitations such as short plasma half-life and another obstacle for peptide drug development is oral bioavailability. Digestive enzymes designed to break down amide bonds of ingested proteins are effective at cleaving the same bonds in peptide hormones, and the high polarity and molecular weight of peptides severely limits intestinal permeability. As oral delivery is often viewed as attractive for supporting patient compliance, the need for injection made peptides a less appealing option for indications that required chronic, outpatient therapy. 34 Researchers began to use medicinal chemistry techniques to make therapeutic agents more drug-like by improving half-life, stability under physiological conditions, and receptor selectivity. Peptide analogs of native hormones with improved pharmaceutical properties thus entered the clinic. Furthermore, natural and synthetic peptides have attracted great attention in research and are widely used in medical and pharmaceutical fields for producing drugs.35–39 This is because peptides have advantages such as high biological activity, high specificity (greater efficacy and selectivity), low cost (synthetic vs recombinant production), greater stability (lengthy storage at room temperature and no changes of secondary structures) and high penetration ability. 40
Even though bioactive peptides have been identified from several sources and their activities investigated in many disciplines, this paper aims at reviewing noteworthy several bioactive active ingredients in wound healing embedded with therapeutic peptides which can prevent and/or treat infected wound. At the same time, we have collected and included various recent data that underlined the matchless potential bioactive peptides that optimize the therapeutic outcomes of wound healing. This review represents a significant advancement in the field of wound healing and regenerative medicine. The points highlighting the novelty of this discovery focus on the derivation of bioactive peptides from natural sources and synthetic design with their regenerative or biological properties, stimulating various stages of the wound healing process including inflammation, angiogenesis, collagen synthesis, and tissue remodeling. This review is a promising approach with the objective to articulate the idea of revolutionizing wound care with different peptide combinations that can be topically applied. Continued research and development in this field holds promise for the advancement of regenerative medicine and personalized wound healing strategies.
Data search management
A comprehensive literature search was conducted within 7 years of publications (2017–2023) through platforms including PubMed, Web of Science (WoS), Scopus, and Google Scholar. The search strategy used the terms “peptide,” “therapeutic agent,” ’biological effect’, “skin,” “wound healing,” and “tissue repair.” The exclusion criteria for this review would be all secondary literature or any original articles written and submitted in languages other than English.
Wound healing peptides
Natural-originated peptides
Understandably, there are several peptides of natural origin either from animals, plants, or microorganisms which address all relevant aspects in the topic of wound healing effects. Microbial fermentation offers a sustainable and scalable method for producing reparative peptides. Microorganisms, such as bacteria and fungi, can be genetically engineered to produce peptides with high efficiency and purity. This approach not only ensures a consistent supply of peptides but also allows for the customization of peptide sequences to enhance their reparative properties. They can also be synthesized in the laboratory to mimic naturally occurring peptides. In nature, peptides play critical roles in various biological processes. They can act as hormones, neurotransmitters, growth factors, and immune modulators. For example, insulin is a well-known peptide hormone that regulates glucose metabolism, while AMPs are part of the innate immune system and protect organisms from infections. On the other hand, many known peptides are of amphibians’ origin, and most of them have revealed wound healing properties. These properties are believed to contribute to the accelerated wound re-epithelialization commonly observed in many species within
In addition to the above, Liu et al. successfully isolated a new short AMP processing 11 residues from the skin of
Although peptides, especially ones derived from amphibians, hold immense potential for therapeutic applications, there are no completed or ongoing clinical trials specifically focused on amphibian-derived wound-healing peptides. While there are over 20 peptide-based clinical trials underway and more than 400 peptide drugs developed annually, the exploration of amphibian wound repair-promoting peptides still remains in the early stages.
51
Amphibian-derived wound healing peptides have consequently made it to the clinical trials for assessing their potency in promoting healing. For instance, Ot-WHP, a peptide derived from the Chinese concave-eared frog, has shown significant potential in promoting wound healing in preclinical studies. This peptide enhances the inflammatory phase of wound healing by recruiting neutrophils and macrophages, promoting keratinocyte migration, and facilitating the crosstalk between macrophages, keratinocytes, and fibroblasts. This results in improved healing outcomes, making it a promising candidate for acute wound management.
52
Notable examples include pexiganan, a synthetic analog of magainins from the African clawed frog (
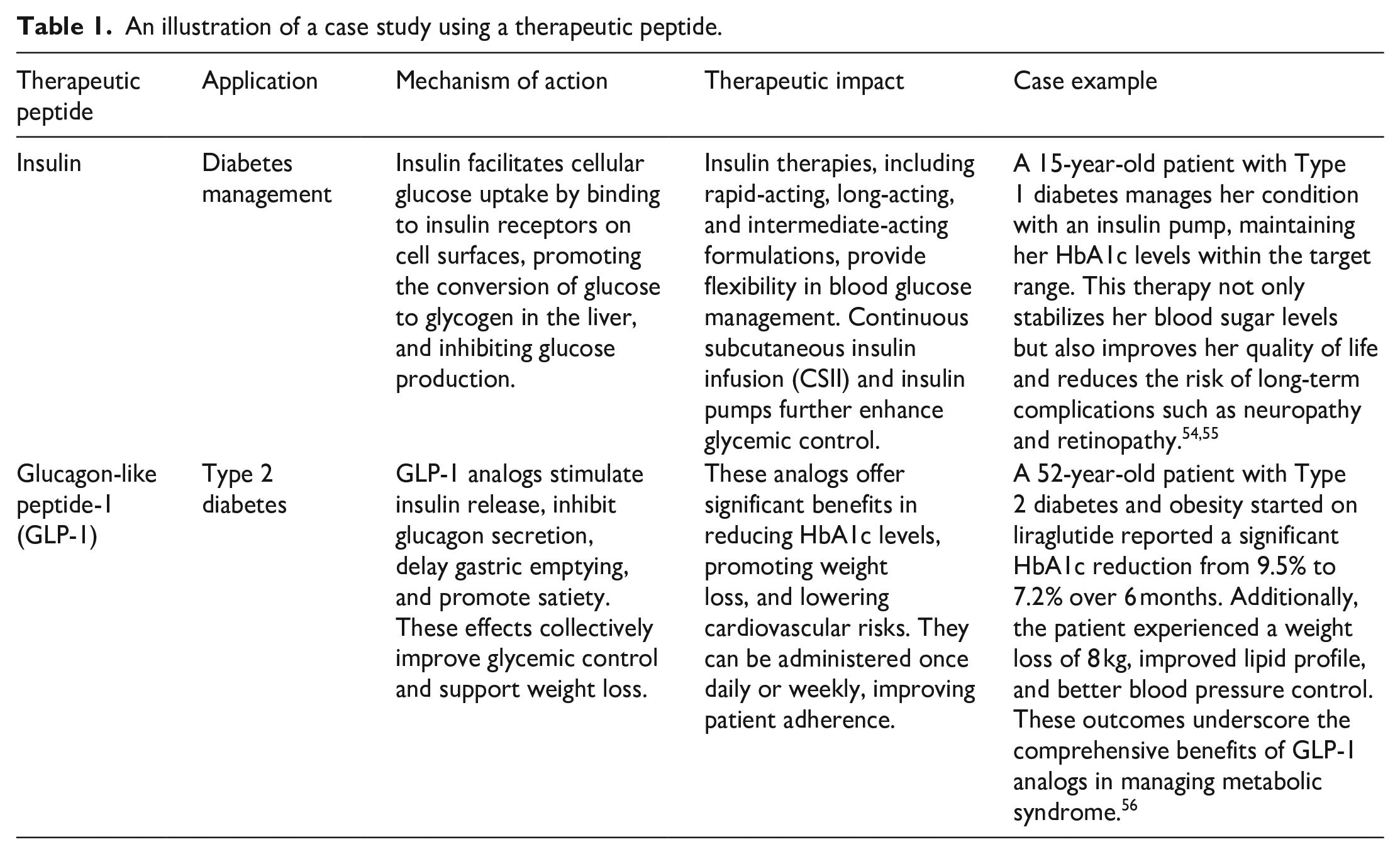
An illustration of a case study using a therapeutic peptide.
Moreover, some marine organisms can serve as safe collagen source since it is abundant in nature which can be broken down into small different proteins with various biological activities. Agreeably, the bioactive peptides derived from collagen have been proven in promoting cell migration and proliferation which is important process in wound healing.
57
Felician et al. have reported that collagen peptides (predominantly type-I) derived from the jellyfish
In relation to the growing number of new peptides being discovered, the therapeutic peptide field has opened a new horizon in the development of plant-based peptides as a drug candidate for the management of various diseases. In plants, bioactive peptides are produced ubiquitously in roots, seeds, flowers, stems, and leaves in which highlight their physiological importance. Most of bioactive peptides produced in plants possess microbicide properties and also involved in cellular signaling. 60 Encouragingly, research findings demonstrated that AMP from a potato gene, referred to as snakin-2 (StSN2) was locally induced by wounding. Furthermore, it was found to be active against fungal and bacterial plant pathogens particularly caused a rapid aggregation of both Gram-positive and Gram-negative bacteria. 61 Other notable examples include the plant-derived bioactive peptides which have opportunities to treat and cure diabetes diseases.62–64 Indeed, Nakatsuji and Gallo 65 found that more than 1200 types of AMPs have been discovered in living organism including animals and plants. In this regard, there is a diverse collection of naturally origin that have been demonstrated to possess excellent wound healing ability and has been listed in Table 2.
Examples of naturally based peptides with wound healing properties. Peptide sequences are displayed using the one-letter amino acid code, as per the IUPAC-IUBMB Joint Commission on Biochemical Nomenclature rules.

Synthetic-b ased p eptides
The use of synthetic peptides has been approved by the health authorities for vaccine development besides being favorable as drug candidates in drug delivery systems. Therapeutic peptides have been derived synthetically as bioactive peptides where there are specific protein fragments that give a positive effect and influence health. Until 2018, over 4800 peptides (4256 natural peptides and 593 synthetic peptides) have been reported as AMPs by DRAMP (data repository of AMPs) database.
91
The designed synthetic AMPs for increased structural stability and high antimicrobial activity under physiological conditions, was also found to indicate wound healing activity.
92
The potential role of synthetic-based peptides in extracellular matrix (ECM) synthesis and wound contraction has also been elucidated. Bioactive peptides can promote contraction capacity of fibroblast by inducing fibroblast to myofibroblast differentiation as well as enhancing wound healing.
93
Another example of a small synthetic peptide was named SHAP1 containing 19 amino acid residues which have showed and displayed stronger wound closure activity. Invivo analysis revealed that SHAP1 treatment accelerated closure and healing of full-thickness excisional wounds in mice. Moreover, SHAP1 effectively countered
Definitely, impaired wound healing is a problem especially for the elderly and diabetic patients. Previous research has found that the actin-binding domain of thymosin β4 duplicated in a seven amino acid residues synthetic peptide with sequence of LKKTETQ was able to promote dermal and corneal repair in normal rats. It was very active in accelerating wound repair in full-thickness dermal wounds in both diabetic and aged mice. This peptide significantly increased epidermal keratinocyte migration, wound contracture and collagen deposition in the wounds in the entire animal studied. 95 In addition to the above, wound healing involves the initial inflammation phase followed by a period of tissue regeneration including epithelialization, angiogenesis, and collagen deposition and ECM remodeling.96,97 Rapid keratinocyte migration could determine the efficiency of the initial wound healing process.98–100 Lee et al. have successfully synthesized and developed a synthetic peptide (AES16-2M) comprised of five amino acids sequences (REGRT), in which significantly showed healing effects in animal wound models. The AES16-2M has proven to speed up and accelerates skin wound healing in vivo via increasing keratinocyte migration through extracellular signal-regulated kinase (ERK) phosphorylation. 35 According to Piipponen et al., 101 keratinocytes are the most dominant cell type in the skin and it plays critical roles in wound repair as structural cells.
An interesting and very recent example of a highly promising synthetic therapeutic peptide is Palmitoyl-GDPH, developed by Fadilah et al. 102 who derived a fatty acid conjugated tetrapeptide in alleviating wound healing. Palmitoyl-GDPH was found to be a novel small peptide with four amino acid residues and revealed remarkable wound healing properties due to promotion of migration of normal human fibroblast (NHDF) cells in vitro; according to the authors, based on the data from scratch wound closure assays. The in vivo wound treatment topically with Palmitoyl-GDPH on the rats for 18 days, histologically demonstrated an almost perfect healing exhibited by increased re-epithelialization, enhanced collagen deposition, and diminished scar formation. Meanwhile, from hematological and biochemical results, the topical application of Palmitoyl-GDPH on the wound did not result in significant toxicity and no systemic side effect in the liver, kidney, and pancreas of the experimental animals. 103 Furthermore, the seven amino acid residues with sequence SVVYGLR, is a multifunctional synthetic peptide and has beneficial effects on oral wound healing through enhancement of the earlier phase consisting of angiogenesis and remodeling with granulation tissue. The synthetic SVVYGLR peptide promoted an increase in the cell motility of oral fibroblasts and keratinocytes. Additionally, the in vivo experiment and immunohistological staining of wound tissue revealed that SVVYGLR peptide accelerated the healing process of oral mucosal wounds by facilitating angiogenesis, increase the capillary growth and greater number production of fibroblasts and myofibroblasts. With the beneficial effects on oral wound healing including earlier phase of angiogenesis and remodeling with granulation tissue, it can be a valuable agent for treatment particularly for intractable mucosal wounds. 104 Altogether, these findings as above prompt other approaches to therapeutic peptides that seek inspiration in wound healing.
Identification of wound healing peptides and their pathway interactions
The bioactive peptides which accelerate wound healing are necessary to be identified to enhance the efficiency of studying the peptide sequence and their possible applications as therapeutics in cutaneous wounds. It is sometimes difficult to identify these peptides correctly because of the structural complexities in numerous native proteins reported as per literature. Since the composition and sequence of amino acids can affect the peptide’s ability to interact with cells and biomolecules present at the wound site, the effectiveness of screening the peptides can be increased by integrating state-of-the-art protein sequencing strategies and various bioinformatics tools. The Edman sequencer can be used to identify the primary structure of the desired peptide. Following that, the cDNA encoding the peptide precursor can be further screened using the cDNA library to identify the genes it is expected to express. NCBI BLAST can be then used to find the similarity of the novel peptide by comparing it with an existing peptide of the same family. 71 Once the sequence identification has been done, several in silico-based approaches can be performed further to predict the structural and functional interaction of the obtained peptide with the target molecule which promotes the healing process.
For instance, PeptideMine webserver is a sequence-based approach used to identify the conserved functional motifs which are often found by aligning several sequences. 105 Following that, these sequences are altered to produce a list of potential candidates which might be able to interact with a certain target protein, thereby blocking non-specific interaction with other proteins. Contrastingly, the conformation-based methods are focused on creating peptide structures and conformational ensembles that are further optimized through the study of structure-activity connections. PEP-FOLD is one such example which uses a Hidden Markov Model to create a structural alphabet and create segments of “letters” that are put together to create three-dimensional structures that are subsequently improved by Monte-Carlo computations. 106 Furthermore, de novo design techniques and knowledge-based strategies are examples of target-based approaches. For example, PiPred evaluates databases of fragments for the most suitable peptides matching the target surface by identifying anchor residues in protein complexes. 107 PepComposer distributes a set of backbone scaffolds that can target a variety of protein surfaces after examining a pool of them. The newly designed peptides seen in the final peptide-protein complex have their conformation refined by a further Monte Carlo simulation. Analyzing protein complexes, PepCrawler and its homolog PinaColada identify candidate peptides that are then randomly altered to improve their affinity for the target. 108
Overall, these wound-healing peptides serve as excellent therapeutic agents to improve healing outcomes in cutaneous injuries. Peptides are generally more specific in their action and are safe as well as more efficient than conventional chemical drugs. However, their bioavailability within physiological systems demands extensive pharmacokinetic studies to determine peptide concentrations in vivo that can exert a beneficial effect on wound healing in vitro. When studying the wound healing properties of novel peptides or any other bioactive molecules, it is imperative to thoroughly validate their wound healing potential. Several strategies can be used to examine progressive changes in wounds during the healing process, each focusing on a distinct aspect. Appropriate wound assessment is reliant on knowing the healing pathophysiology, variables that delay the process, and the optimal wound bed conditions required to enhance healing and therapeutic success. 109 Wound healing measurements often include wound size, wound bed features, scarring extent, and a variety of underlying vascular and pathophysiological problems that may be impairing healing. Qualitative and quantitative techniques are used to assess wound progression and should be followed until tissue regeneration occurs. There exist a few standard experimental protocols that are routinely performed to ascertain the possible beneficial effects of such wound-healing peptides in terms of promoting cell migration, improving collagen deposition, enabling wound contraction, or warding off microorganisms from wound site. These methods inevitably allow the inference of different aspects of wound healing and monitor through its stages. An overview of these methods is provided in Table 3.
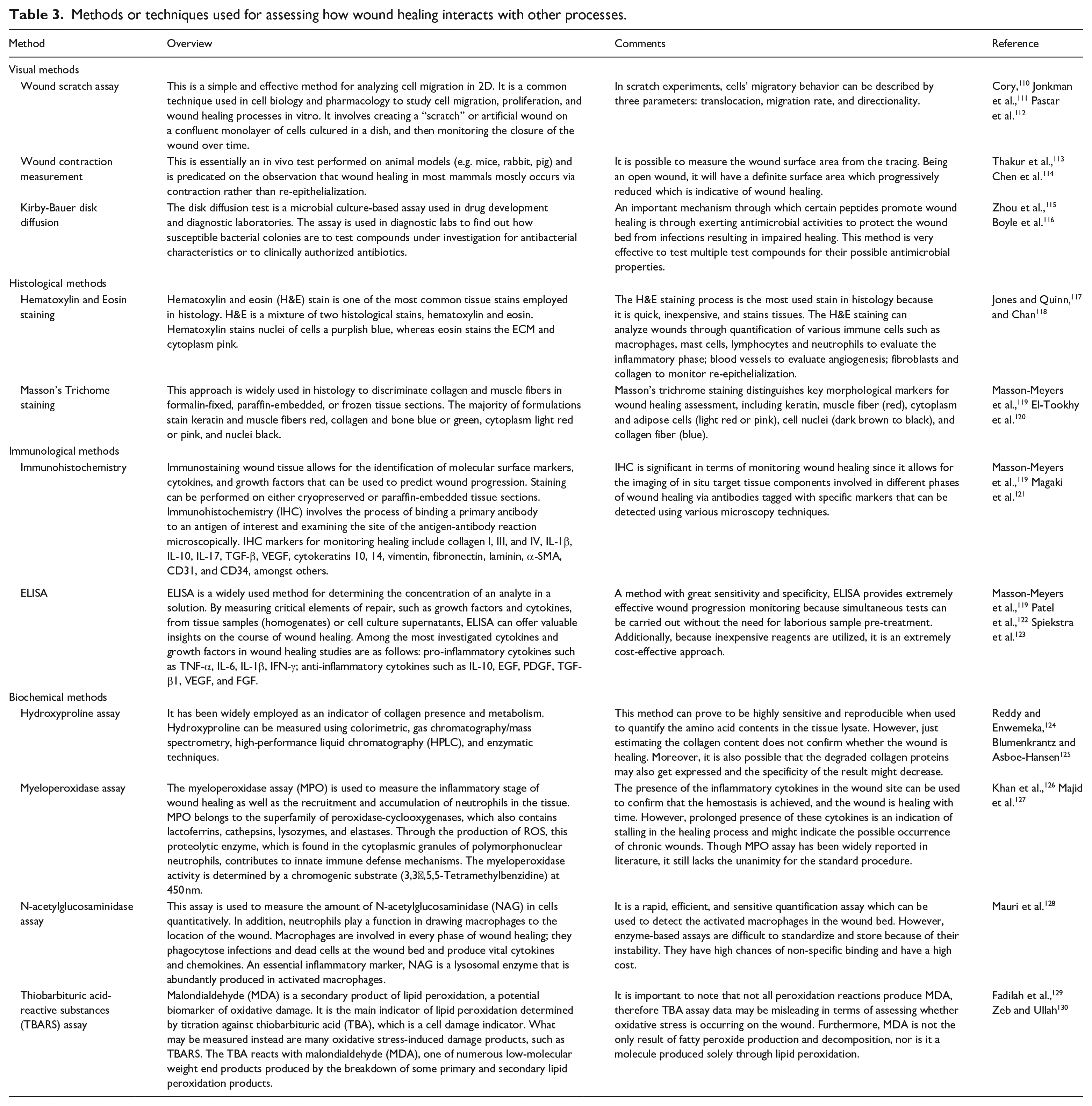
Methods or techniques used for assessing how wound healing interacts with other processes.
Understanding the mechanism of interaction of wound healing peptides with various proteins and growth factors on the wound bed is necessary to elucidate the downstream metabolic pathways which are activated by subjecting the wound to these peptides. An observed metabolic response even in terms of wound healing is the result of the coordinated activation and interaction between multiple genetic pathways. 131 A general rule of thumb that is usually performed is to thoroughly perform a transcriptomic analysis in order to detect differential gene expression at the wound site, followed by gene ontology correlation studies in order to map the main metabolic pathways of genes related wound healing. 132 Appropriate validation of these differentially expressed genes related to wound healing metabolic pathways using quantitative real-time PCR and correlating them with of RNA-sequencing can ensure that the transcriptome data are reliable. Wound healing is a very complex process, where there is an involvement of more than one pathway getting activated, depending on the phase of healing. Therefore, novel approaches based on detailed knowledge of signaling pathways that are activated during acute or chronic cutaneous wound healing enables effective elucidation of interaction mechanisms of such peptides in wound healing.
One of the most known databases to compile all the anticipated interactions of proteins both physically and functionally is the STRING database. 133 STRING provides a list of automated pathways such as MAPK, TGF-β1/SMAD, and NF-κB signaling pathways which might be followed by the peptides to accelerate wound healing.66,134,135 Hence, the enrichment and network tools in STRING provide a thorough evaluation of gene lists and functional genomics datasets to aid the user in comprehending the underlying mechanism easily. Alternatively, the KEGG pathway database is yet another collection of manually verified maps of biological interactions, including the entire set of signal transduction pathways and other cellular activities. Rapid mapping of differentially expressed genes to KEGG pathways can provide insight into the functional importance of gene lists corresponding to high-throughput expression data and hence the underlying pathways governing the wound healing response. 136
Potential biological effects of peptides on wound healing
The previous sections have explained about the bioactive peptides to stimulate both antimicrobial and wound healing properties including granulation tissue formation, re-epithelialization, angiogenesis and collagen synthesis. Therefore, the biological effects of peptides on wound healing-related cells are of interest in which could also provide a theoretical basis for the application of the peptides in promoting wound healing (Figure 1). However, the activity of peptides is dependent on different factors such as their size (peptide length or number of amino acids),
137
conformation (e.g.
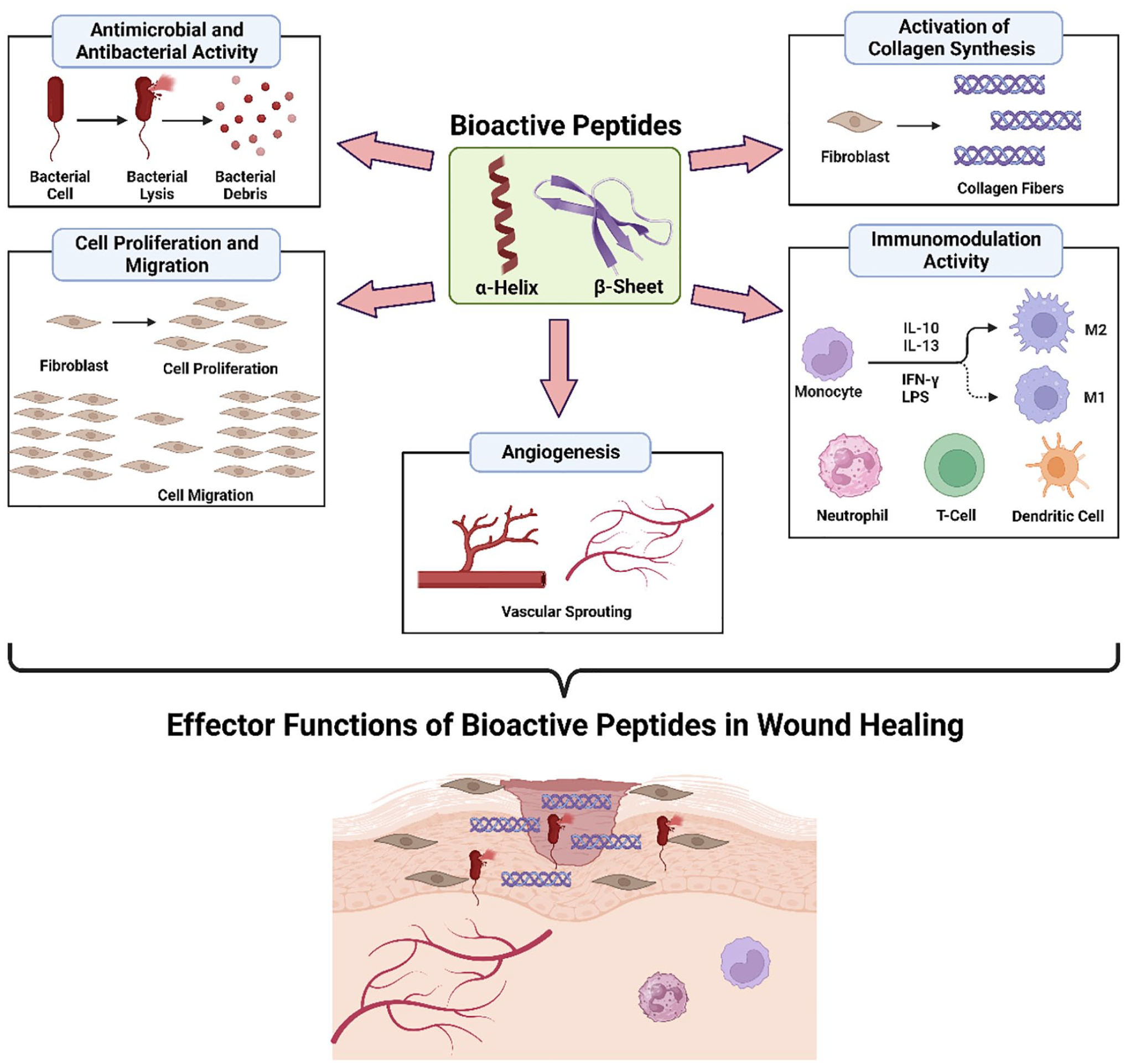
Schematic representation of the potential biological effects of AMPs on wound healing.
Antimicrobial and antibacterial activity
Skin infections and wounds represent one of the most common indications for the use of antimicrobial therapy. This is why bacterial wound infections are a major cause of delayed wound healing and even make the wound worse. Almost all open wounds are colonized with microorganisms, bacteria, and spores where some wounds are clearly infected thus maybe toxic to the tissues. Therefore, it is not surprising that the development of new effective antimicrobial agents is highly desired and frequently introduced. Another newly popular topical remedy for wound infections is bioactive peptides which are referred to as AMPs. In recent times, AMPs have received more attention from researchers due to their good properties which is having antibacterial and antimicrobial efficacy.143,144 It has shown a significant promise as novel therapeutic agents to treat infections caused by multidrug-resistant pathogens for example
The mechanism of antibacterial and antimicrobial activity is generally due to the electrostatic interactions with microbial cell membranes of anionic phospholipids. 152 This interface relationship will lead to the disruption or disunity of the microbial cell membrane that result in cell death153,154 through different mechanism as presented in Figure 2. Interestingly, many antimicrobial compounds or agents have multiple positive charges, and they are polycationic where may alter the stability of cell membrane which affected bacterial function. 155 The polycationic compounds can isolate anionic lipids to induce lipid phase separation. According to the molecular interactions among lipid and their lateral packing density, the phase boundary will defected and thus lowering the permeability barrier between the cell and its surrounding. 152 Subsequently, the AMPs will access the phospholipid bilayer membrane, interact with intracellular components, start to aggregate and form of different complex structures which in turns leads to cell lysis and apoptosis. 156 The AMPs basically do not have specific microbial cell targets instead of bacterial membrane which is considered to be the main target of AMPs. Another prominent example includes mechanism in the integrity of bacterial membranes was by Ma et al. The author found that a novel engineered amphipathic peptide, WRL3 can treat burn wound infections by destroying the cell cytoplasmic membrane leading to cell death. The cell membrane surfaces treated with WRL3 become atrophy, creping, breakage, roughened and corrugated. 157 Agreeably, an efficacious antibacterial and antimicrobial agent should be able to selectively inhibit and kill bacteria for the main determinant of therapeutic outcome. Their bactericidal activities are generally observed at µM or µg/ml concentrations. 158 A designed antimicrobial peptide (RP557) was displayed superior and broad-spectrum antimicrobial activity (relatively low doses, 2 µg/ml) against both Gram-positive and Gram-negative bacterium including drug resistant phenotypes and fungi. 159
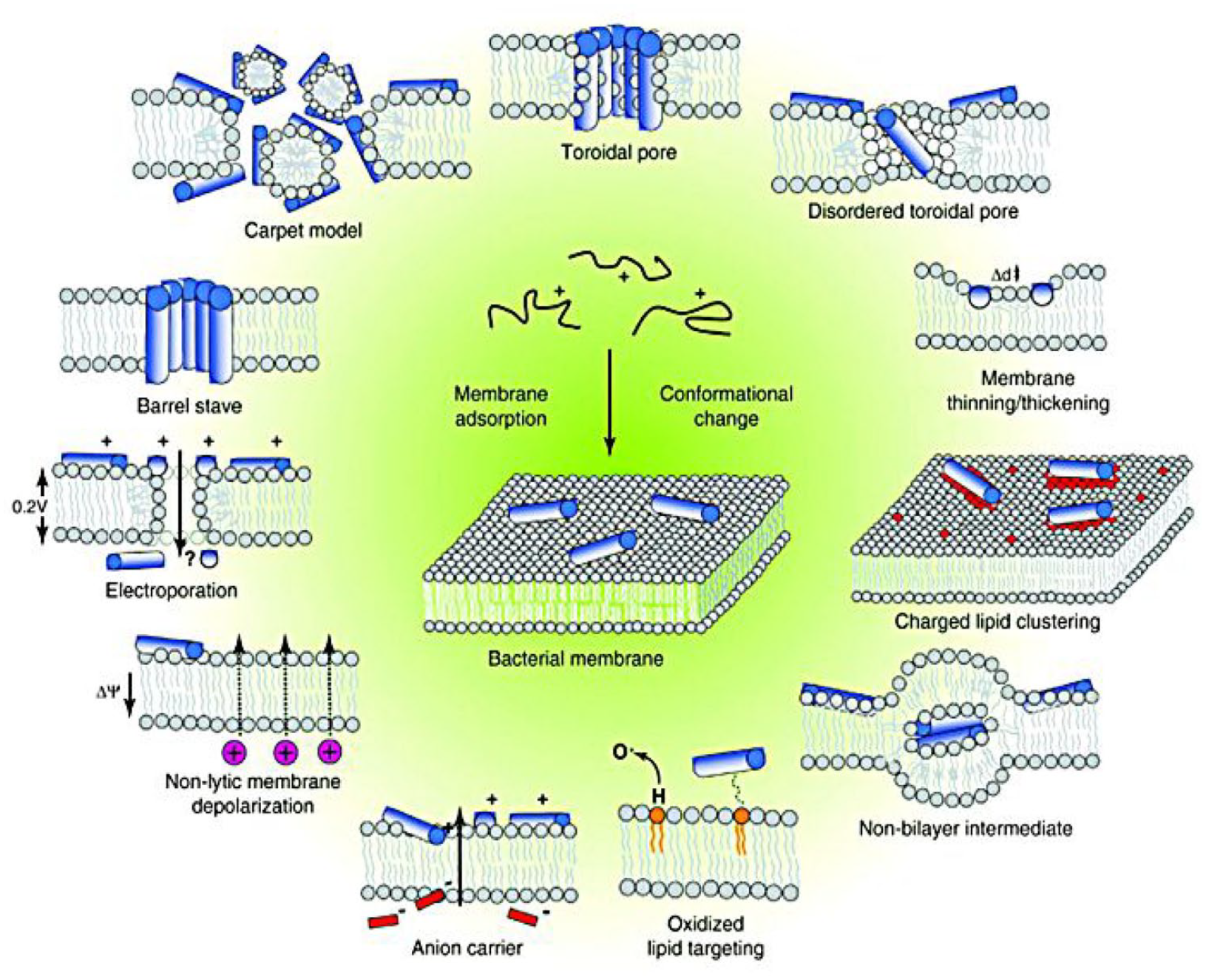
Events occurring at the bacterial cytoplasmic membrane following initial antimicrobial peptides (AMPs) adsorption. The figure is adapted from Nguyen et al. 160 under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Many AMPs with their antimicrobial and antibacterial activity could promote wound healing both in vitro and in vivo. For instance, Stigmurin which is a linear peptide composed of 17 amino acid residues was shown to induce resistant pathogens in the treatment of epithelial injuries. The topical application of Stigmurin in a wound infection model induced by
Cell proliferation and migration
During wound healing process, excessive inflammation or long-term inflammatory reactions delay healing and sometimes will become an ulcer where it is difficult to heal. Thus, inhibition of inflammation also has a very important effect on wound healing, especially in the period from inflammation to proliferation.165,166 Despite that, the closure of wound site is primarily achieved by re-epithelialization, proliferation, and migration of epithelial cells. When the skin is damaged, fibroblasts are attracted by cytokines and growth factors which are secreted by platelets and macrophages. They are starting to migrate from the edge of the wound to the injured area. All epithelial cells continue to divide, proliferate and migrate to the wound surface in order to reform a new layer of thin epidermis. 167 Following these various wound healing processes, each of steps play a key role to contribute to accelerating wound healing.
More importantly, most of the studies with bioactive peptides have been performed on wounds both in acute and chronic that affected the overall therapeutic activity observed. It was later found that a small peptide isolated from wheat germ (YDWPGGRN) enhanced the proliferation and migration of keratinocytes and fibroblasts in skin wound therapeutics. Specifically, the wound area was significantly narrower, and the gap distance was significantly reduced after the 24 h of peptide treatment. 168 The epithelial cells as the main keratinocytes migrate to the wound surface and first cover the wound area are thus beneficial for wound repair. Meanwhile, fibroblasts contribute to the formation of granulation tissue after migration and facilitate timely and wound repair. 169 Besides, Iosageanu et al. reported that the bioactive peptides also presented high capability and capacity to influence the human keratinocytes migration process. As the keratinocytes is the major cell type of the epithelial barrier, it supports the progress of wound healing together with fibroblast. In the study, they found that fish bone peptides successfully involved in treatment of cutaneous wound healing, in particularly stimulation of keratinocytes metabolism and migration. 170 The proliferative activity of human skin fibroblasts was probably related to the increases in ECM because wound closure depends entirely on the migration of cells irrespective of cell growth. Moreover, further mechanistic investigations revealed that bioactive peptide from rice bran proteins (RBPs) induced cell proliferation and migration in endothelial cells most efficiently at low concentrations between 0.1 and 1.0 µM. That cationic peptide significantly dependent on the proliferation-promoting effects. 171
Due to the special contributions of bioactive peptides in normal human dermal fibroblast cells, other studies have demonstrated that synthetic bioactive peptide is able to promote wound healing by increasing fibroblast cell migration and extension. Following that, Fadilah et al. found out a therapeutic agent, Palmitoyl-GDPH which promoted growth of normal human fibroblast cells after 24, 48, and 72 h as shown in Figure 3(a). The cells showed proliferation in time-dependent manner at low concentrations of 1.56–100 µg/ml. From the data in Figure 3(b), the cells treated with different concentration presented different migration rates and healing progression. Furthermore, by analyzing cell migration paths in the scratch area, it was found that the migration path was significantly shortened meanwhile the migration rate was increases after the application of bioactive peptide. The cells reached full gap closure faster with Palmitoyl-GDPH concentration level of 100 µg/ml as displayed in Figure 3(c). Though, Palmitoyl-GDPH with a low concentration of 12.5 µg/ml is exhibited a positive relationship between the cell migration speed and the distance from the wound scratch. 76 Encouragingly, the active compounds will stimulate the cellular activity even treated with low concentration doses and this enhanced migration may explain the promoted cell proliferation. 172
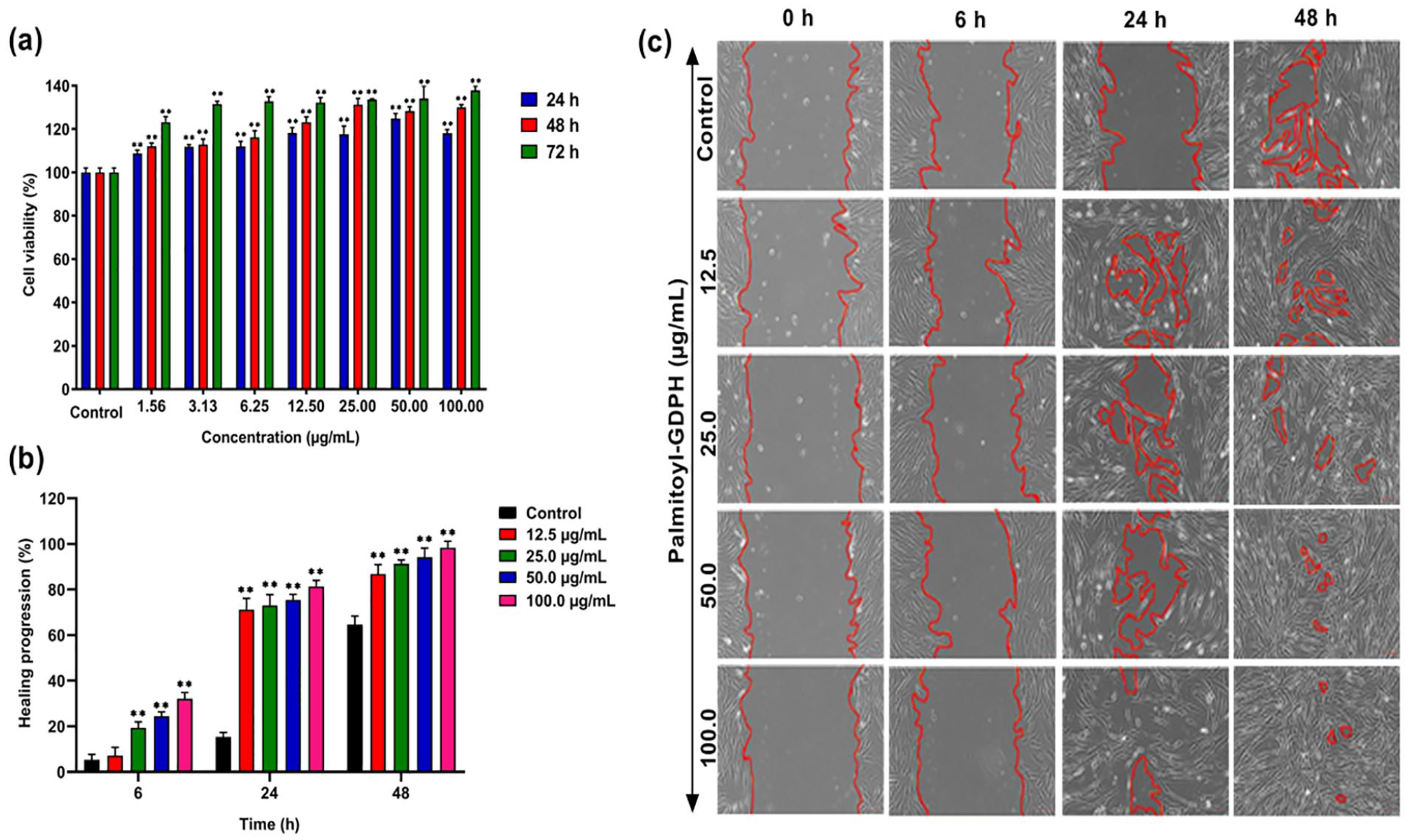
(a) Cell viability of normal human dermal fibroblast cell upon treatment with Palmitoyl-GDPH for 24, 48, and 72 h, as determined by MTT assay, (b) healing progression of Palmitoyl-GDPH at dose dependent, and (c) migration wound scratch assay using normal human fibroblast cell treated with Palmitoyl-GDPH. All the figures (a)–(c) adapted and reprinted from Fadilah et al. 102 under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
The ability of a new non-toxic skin amphibian peptide, OA-RD17 to alter the cellular and molecular processes involved in skin wound healing was investigated. As the major cellular component of the epidermis, keratinocytes not only exhibit immune functions but are also the executors of the re-epithelialization, which is essential for wound healing. 101 Consequently, the molecular mechanism of OA-RD17 on keratinocytes and wound repair was revealed by RNA-seq, which may aid in the identification of new therapeutic targets and the creation of innovative agents for wound repair. At the mRNA level, OA-RD17 regulated differentially expressed genes (DEGs), associated with ribosomal and extracellular components, and involved in nucleic acid binding, intranuclear trafficking of proteins and phosphorylation biological processes. Regarding the molecular mechanism include in the wound healing, the MAPK signaling pathway is tightly associated with cell proliferation and migration. 173 It was promoted macrophage proliferation and migration by activating MAPK and suppressed inflammation by inhibiting NF-κB. The OA-RD17 upregulated miR-632 by activating MAPK pathway via Toll-like receptor 4 (TLR4), and miR-632 promoted keratinocyte proliferation and migration by targeting GSK3β to activate Wnt/β-catenin signaling pathway. Notably, OA-RD17 accelerated granulation and epidermal regeneration, facilitated the shift from the inflammatory to the proliferative stage, and shown therapeutic effects on ex vivo skin wounds from diabetic patients and mice. MiR-632 stimulated Wnt/β-catenin to facilitate the healing of full-thickness skin wounds in rats. 174
According to a different study, an amphibian-derived peptide homodimer OA-GL17d showed potential ability to promote skin wound regeneration through the miR-663a/TGF-β1/Smad axis. 175 The OA-GL17d exhibited remarkable repair-promoting abilities in HaCaT cells. They carried out miRNA sequencing to investigate the role of miRNA in OA-GL17d in skin wound repair, as miRNA is involved in several biological processes, including as proliferation, angiogenesis, apoptosis, and inflammation. According to the findings, there was a significant up-regulation of 12 miRNAs and a down-regulation of 72 miRNAs. Notably, miR-663a was among the most severely down-regulated genes. The miRNA sequencing analysis and verification revealed that OA-GL17d markedly reduced the expression of miR-663a. Further investigation revealed that both OA-GL17d therapy and miR-663a suppression markedly increased TGF-β1 expression. They also found that OA-GL17d treatment and miR-663a inhibition activated the Smad signaling pathway and significantly reversed the effect of miR-663a mimic on the Smad pathway, thereby promoting skin wound healing.
Angiogenesis
During wound healing, angiogenesis in the vascular system becomes an essential part and plays a critical role in the healing process, whereby it can provide oxygen and nutrients to the wound area. When tissue is damaged, the microvascular endothelial cells in the surface layer of the vessels are activated by pro-angiogenic factors such as hypoxia/hypoxia tension and vascular endothelial growth factor (VEGF). Simultaneously, when the ECM is degenerate or degraded, endothelial cells begin migrating and proliferate after that re-establish cell-cell connections to produce new capillaries. This way would increase the perfusion of the blood around wound bed and provide essential metabolites and nutrients for wound healing. 176 Moreover, previous studies have focused on the effect of AMPs on epithelial cell migration and re-epithelialization toward wound healing nevertheless, angiogenesis role also has a vital response in wound treatment. For that reason, it needs to be explored whether AMPs also play a role in other stages of rapid wound healing.
Recent studies have demonstrated the angiogenic abilities of peptides in the wound area. A study of peptide extract derived from natural source which is
Cutaneous wound healing is a well-organized reaction to injury that calls for exact coordination of epithelialization, dermal repair, and angiogenesis and involves numerous different growth factors. 180 Takahashi et al. looked at how antimicrobial peptide human β-defension-3 (hBD-3) affected human fibroblasts’ cell migration, proliferation, and generation of angiogenic growth factors. They also assessed how hBD-3 affected angiogenesis and wound healing in vivo. 181 Furthermore, the expression of various angiogenic growth factors was increased in the hBD-3-treated wounds. From the in vitro research, results showed that hBD-3 increased the secretion of angiogenic growth factors like FGF, PDGF, VEGF, and encouraged human fibroblast migration and proliferation as well. The hBD-3-mediated activation of fibroblasts involves the fibroblast growth factor receptor 1 (FGFR1)/Janus kinase 2 (JAK2)/signal transducer and activator of transcription 3 (STAT3) pathways, as evidenced by the inhibitory effects of pathway-specific inhibitors as illustrated in Figure 4.
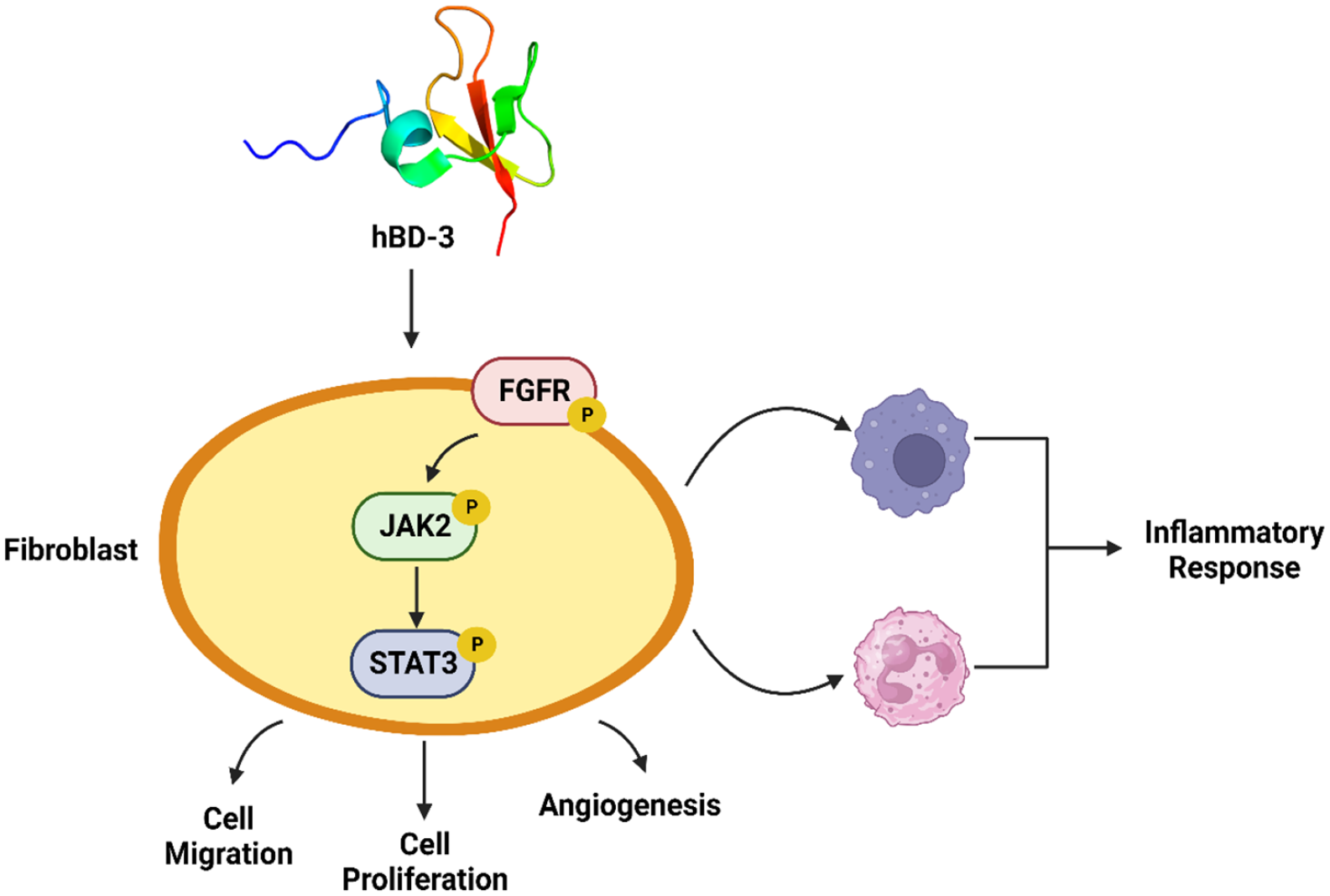
Schematic illustrating how hBD-3 is involved in wound healing. hBD-3 induces accumulation of neutrophils and macrophages in the early phase of wound healing and reduction of these phagocytes in the latter phase, leading to regulation of inflammatory responses during the wound healing process. hBD-3 also stimulates fibroblasts to migrate, proliferate and promote angiogenesis via activation FGFR/JAK2/STAT3 pathways. The idea of the figure has been adapted from Takahashi et al., 181 under the terms and conditions of the Creative Commons Attribution License (CC BY).
Immunomodulatory activity
After a wound has developed, our immune system draws in inflammatory cells that release inflammatory peptides known as cytokines. Depending on how quickly the wound is healing, these cytokines may be pro-inflammatory, anti-inflammatory, or both. The primary function of the pro-inflammatory responders is to trigger an inflammatory phase to fight any possible bacterial invasion that may have taken place. 182 Under normal healing conditions, immunomodulation controls these inflammatory cascades, enabling following regeneration cascades led by anti-inflammatory cytokines to advance the wound toward complete healing. The sequence of inflammatory and regenerative cascades is, however, dysregulated in cases of sluggish or defective wound healing, affecting the phenotypic and functional states of local and systemic immune cells. 183 Usually, dysregulated wound healing is exacerbated by existing comorbidities, multiple tissue injuries or wound infection due to microbial invasion. Such occurrences affect the usual course of wound healing, making it necessary to develop efficient immunomodulation strategies for reducing inflammation during wound healing. 184 Immunomodulation, in its most basic form, refers to the modification of an immune response or the activity of the body’s immune system through the physiological effects of an immunomodulator molecule. The immune system is critical in wound healing with immune cells promoting wound healing by secreting immunomodulatory molecules mainly cytokines, chemokines, and growth factors, facilitating cellular crosstalk and guiding tissue repair besides tackling recurring infections culminating into chronic wounds.
At this juncture it is worth highlighting that besides the immunomodulatory mediators secreted by polarized macrophages, other immune cells of our body are also associated with immunomodulatory activities toward progression of wound healing. A typical example includes defensins which are short (28–42 amino acids length) amphipathic peptides produced and secreted particularly by leukocytes. 185 They are cysteine rich cationic peptides known to exhibit therapeutic immunomodulatory activities in that they are capable of inducing inflammation and suppressing inflammatory responses by acting on specific cells through distinct mechanisms. Defensins can also influence immune responses by forming complexes with cellular molecules such as proteins, nucleic acids, and carbohydrates. 185 Defensin-mediated immunomodulation appears to be cell-type and context dependent. Nevertheless, human defensins are typically synthesized as a pre-pro-peptide and are then processed intracellularly or extracellularly depending on the defensin and cell type. In humans, there are reportedly six α-defensins including four human neutrophil peptides (HNP1, HNP2, HNP3, and HNP4), and two additional human α-defensins (HD5 and HD6). Furthermore, six human β-defensins (hBD1, hBD2, hBD3, hBD4, hBD5, and hBD6) have also been reported and previously characterized to be associated with wound healing. Immunomodulation effect of a-defensins, and β-defensin is pro-inflammatory and anti-inflammatory, respectively. 186
Furthermore, multiple studies have indicated that defensins promote leukocyte recruitment and accumulation at inflammatory sites while additionally releasing a few chemokines associated with immunomodulation functions resulting in abating inflammation and promoting wound closure toward a healing cause.187,188 HNPs are primarily released as effector molecules from neutrophils which are among the first cells to migrate to the wound bed. Although HNPs have direct and potent antimicrobial activities, 185 these peptides also control infection during acute infection by modulating various immune activities such as chemotaxis, phagocytosis, and cytokine induction. Additionally, HNPs function majorly as chemoattractant for multiple cell types monocytes, immature DCs, and naïve CD4+ T cells but not for memory CD4+ T cells or mature DCs. 189 Among these HNP1, 2 and 4, especially HNP1 has been reported to be highly expressed at wound center as compared to wound edges, suggesting massive infiltration of neutrophils at wound center as observed during normal circumstances. 187 HNP1 under homeostatic physiological circumstances have not been reported to be involved in any immunomodulatory activity, however in an inflammatory state HNP1 recruits leukocytes in a positive feedback-loop, 188 stimulates the production of IL-6 and IL-8 via the p38 and ERK1/2 MAPK-dependent signaling pathways.190,191 Inspired by the immunomodulatory properties of defensins, Luo et al. 192 developed Nanodefensin, a wound healing therapeutic inspired by HD5, which following local administration at wound site via a hydrogel encasing system promoted wound regeneration and increased myofibroblast expression. One of the most important indicators of chronic wounds is failure to re-epithelialize. Keratinocyte migration, proliferation, and differentiation all contribute to the re-epithelialization process. Notably, members of the defensin family such as HNP1, hBD2, hBD3, and hBD4 have been reported to induce keratinocyte proliferation and migration, which can then secrete hBDs, promoting cellular barrier reconstruction and accelerating wound healing. 193
On the other hand, Cathelicidins represent another group of endogenous peptides which are abundantly stored as inactive precursors within the granules of neutrophils and mast cells. Cathelicidins, unlike defensins, are predominantly α-helical, amphipathic, cationic, and short length comprising 23–37 amino acids. 194 They are essentially protein precursors with a conserved amino-terminal (N-terminal) signal peptide domain, an antimicrobial C-terminal domain mature peptide, and a cathelin-like domain. 195 When neutrophils degranulate and secrete peptides, the cathelin domain is cleaved by serine proteases, resulting in the active form of the peptide. 196 Furthermore, the potential human peptide known as LL-37, regulates the production of chemokines as well as pro- and anti-inflammatory cytokines to maintain the delicate balance between both responses. The capacity to maintain equilibrium is critical in pathogen clearance at the wound site while maintaining immune system stability to promote tissue regeneration. 197 Through activation of the p38 and ERK1/2 MAPK pathways, LL-37, in conjunction with hBD-2, hBD-3, and hBD-4, induce IL-18 secretion from keratinocytes which acts as a pleiotropic factor engaged in immunomodulation cascades associated with host defense and immunomodulation. 198 Consequently, LL-37 is considered one of the most preferred peptides-based therapeutics for wounds with poor healing and infections. Previously, nanoparticle lipid carriers encapsulating LL-37 administered topically have been shown to expedite wound closure, re-epithelisation, and reduce inflammation in vitro and in vivo. 199 Another study showed that topical application of LL-37 encapsulated within PLGA nanoparticles accelerated wound closure, and re-epithelialization, besides enhanced granulation tissue formation at the wound bed. 200 Overall, cathelicidins are key regulators of the inflammatory response associated with wound healing, with multiple studies supporting their immunomodulatory actions over their antimicrobial ones.201,202
Activation of collagen synthesis
Collagen, the most abundant protein in the body, becomes a crucial part of the ECM, and the biological adhesive that holds cells together. When there is an injury, the affected site’s tissue integrity is lost, thus destroying the ECM. Collagen will performs crucial functions in the regulation of wound healing stages, either in its original state or as little peptide fragments in the wound environment. 203 During the inflammatory phase, soluble collagen fragments attract immune cells such as macrophages, which patrol the wound for the elimination of microorganisms and devitalized tissue. 204 This facilitates the shift to the proliferative phase, which is characterized by the creation and activation of collagen. Numerous investigations have verified the importance of macrophages’ role in wound healing. Proinflammatory M1 macrophages eliminate cellular debris, degraded matrix, microorganisms, and neutrophils following initial infiltration. They also release pro-inflammatory cytokines and growth factors, which mobilize additional immune cells and enhance keratinocyte, fibroblast, and epithelial cell proliferation observed during the proliferative and remodeling phases. 205
Arguably, PDGF is a more potent chemoattractant for wound macrophages and fibroblasts and can induce these cells to express endogenous growth factors such as TGF-β, a process that in turn directly stimulates new collagen synthesis and sustained enhancement of wound healing over a longer time. The findings suggest that PDGF indirectly stimulates collagen type I biosynthesis by stimulating proliferation and migration of activated fibroblasts through TGF-β signaling where TGF-β1 attracts fibroblasts into the wound and may directly stimulate collagen synthesis. They start secreting anti-inflammatory cytokines including TGF-β1, which promotes ECM production and wound contraction, during this phase. TGF-β1 acts as a growth-inhibitory cytokine in unwounded epidermis to help maintain tissue homeostasis through a SMAD-mediated transcriptional regulation of multiple cell-cycle regulators. The TGF pathway directly affects and arrests the cell cycle at the early G1 phase. 206
The ability of collagen peptides to exerts a physiological impact on the cutaneous wound healing is greatly determined by their absorption and distribution into the skin. Mistry et al. 207 carried out the potential of bovine collagen peptides to promote cutaneous wound healing of fibroblast and keratinocytes derived from both young and aged individuals. According to their research, collagen peptides promote wound healing through increased cellular proliferation, as demonstrated by an increase in cell viability and peptide induced Ki67 expression. As an essential amino acid that speeds up the healing of diabetic wounds, L-arginine has been demonstrated to improve wound strength and collagen deposition in both people and animals. Lin et al. 208 also found that a compound that rich of Arginine might contribute to the potential of wound healing effect. Their ability to stimulate collagen production, inhibit degradation, and promote tissue repair makes them valuable tools in various medical, cosmetic, and wellness contexts. Further research and development in this field may reveal even more ways in which peptides can be utilized to support collagen synthesis and benefit human health.
Peptide combination types for topical applications
To heal a wound, a series of overlapping processes occur sequentially which includes phagocytosis, chemotaxis, mitogenesis, collagen synthesis, and the production of other matrix components. These processes do not happen randomly; instead, they follow a precisely timed and organized cascade that corresponds with the development of distinct cell types in the wound at various stages of the healing process. There is a hypothesis that suggests insufficient growth factor stimulation could lead to poor wound healing. According to Schultz et al., fibroblast cultures were induced to synthesis DNA at high levels and the fluid recovered from the chest drains of healed mastectomy wounds had significant concentrations of growth factors. On the other hand, there were insufficient concentrations of growth factors and no ability to promote DNA synthesis in the fluids taken from chronic, nonhealing wounds. When it comes to animals suffering from diabetes, infections, malnourishment, or impairments from steroids, chemotherapy, or radiation, growth factors have been demonstrated to enhance wound healing. 209
Some human-based defense peptides such as human β-defencins (hBDs), dermcidins and cathelicidin LL-37are well known for promoting wound healing in chronic conditions.76,210,211 Brown et al. demonstrated that PDGF (BB homodimer) in combination with TGF- α has shown enhanced wound healing in genetically diabetic mice. It was inferred that PDGF itself promoted faster closure of wounds and TGF-α when combined further accelerated the process. 212 Another study by Lynch et al. experimentally showed that doses up to 1500 ng of TGF-β when singly administered, promoted collagen maturation, connective tissue formation and angiogenesis. However, this caused enhanced inflammatory responses and abnormal differentiation of epithelial cells. This side effect was found to be reversed by further administration of PDGF-2 homodimer combined with IGF-I. It was concluded that PDGF-2 with IGF-I not only promoted wound healing but also reduced unnecessary inflammations caused during the healing process. 213 Furthermore, a study carried out by Gope gave a contrasting comparison among the three combinations of peptides which were EGF with PDGF-AA, PDGF-BB and PDGF-AB in several combinations. The best results were observed in the EGF and PDGF-BB combination in the concentrations 10 and 15 ng, respectively. 214 Other than that, an experiment by Qu et al. 215 reported that chemically altered collagen membranes were released by the addition of bFGF and keratinocyte growth factor (KGF) which facilitated cellular proliferation, faster cell migration and tissue regeneration.
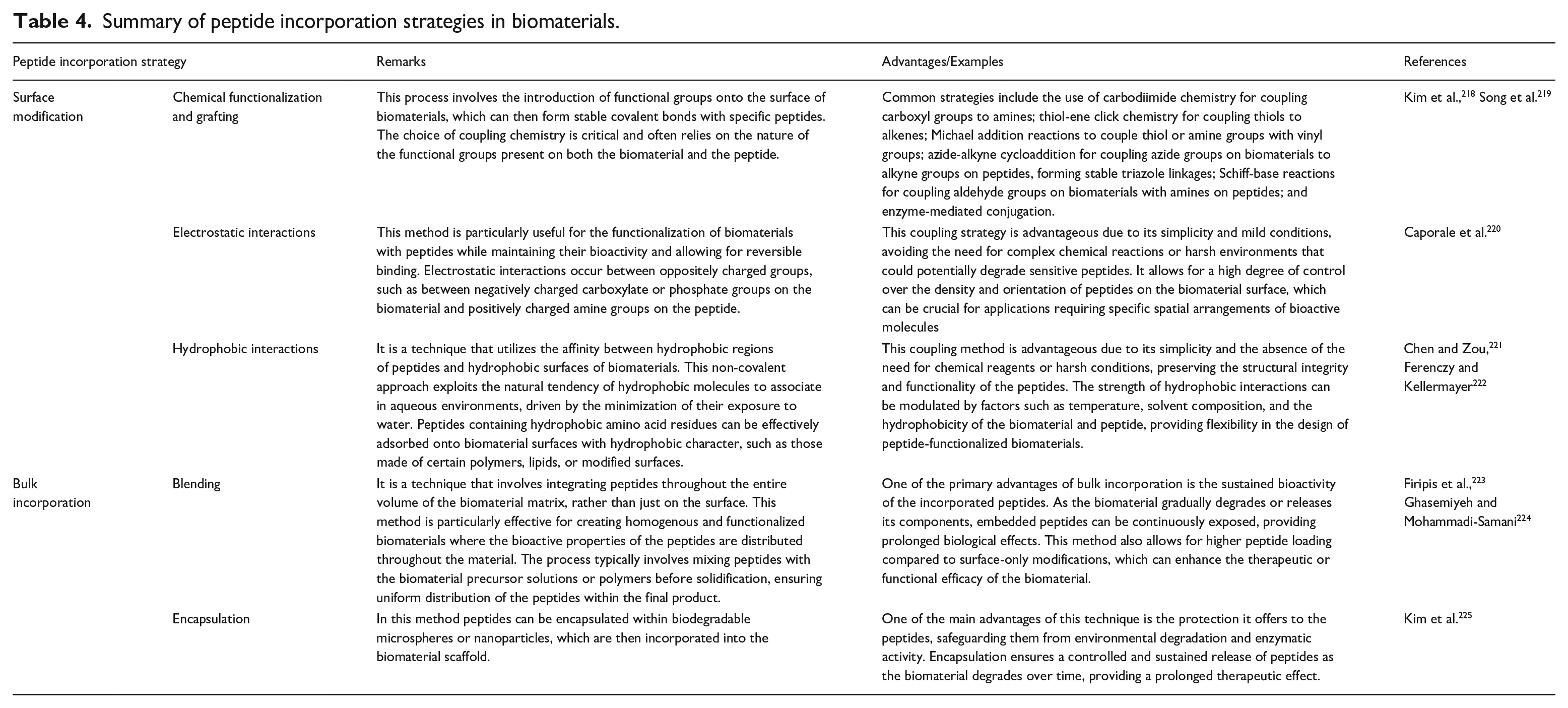
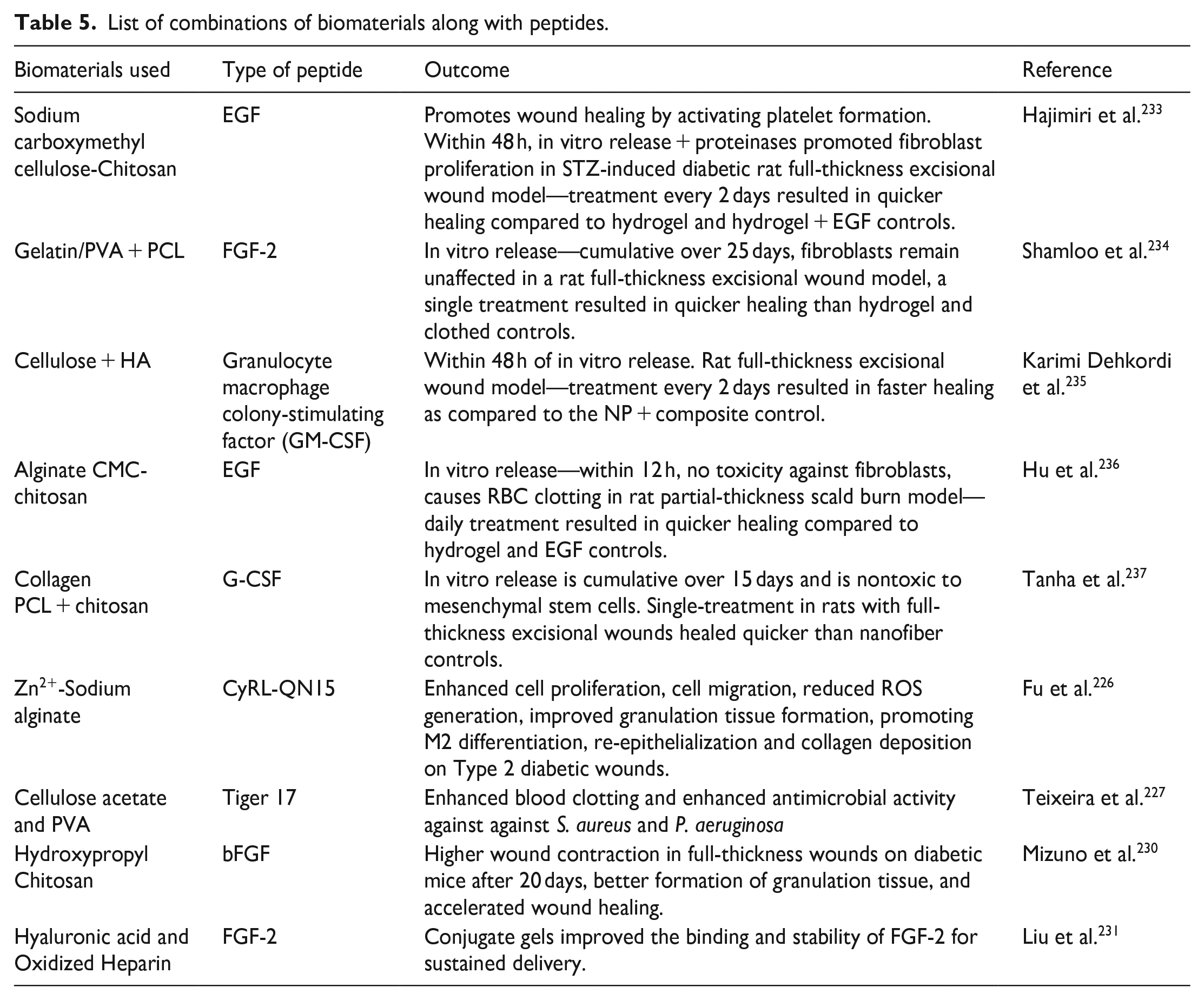
Active peptides have emerged as a potent tool in material science, offering innovative intervention strategies for various biomedical applications including wound healing. These short chains of amino acids, known for their bioactivity, can be engineered to interact specifically with biomaterials, thereby enhancing or imparting desired properties. One prominent strategy involves incorporating active peptides into biomaterials to promote cell adhesion, differentiation, and proliferation, making them invaluable in tissue engineering and regenerative medicine. For instance, the incorporation of RGD (arginine-glycine-aspartic acid) peptides into scaffolds has been shown to significantly improve cell attachment and growth. 216 Another approach is the utilization of AMPs in material coatings to prevent biofilm formation and microbial infection. These peptides can be integrated into surfaces such as medical devices, implants, and wound dressings, offering a bio-friendly alternative to traditional antibiotics and chemical agents. The peptide-coated materials demonstrate high efficacy in reducing bacterial colonization while being biocompatible and non-toxic to human cells. 217 In the realm of bio-nanotechnology, active peptides are employed to functionalize nanoparticles, enhancing their stability, targeting ability, and therapeutic potential. Incorporating peptides into biomaterials involves several strategies to leverage their bioactivity for enhancing the material’s properties and functionality. These strategies can be broadly categorized into surface incorporation, and bulk incorporation systems as summarized in Table 4.
Summary of peptide incorporation strategies in biomaterials.
Over the years, various polymeric delivery systems (e.g. hydrogels, films, microspheres, nanofibers) for incorporating these peptides have become increasingly important in pharmaceutical and biomedical applications due to their ability to enhance the stability, bioavailability, and controlled release of therapeutic peptides. Notably, several collagen-based drug delivery systems and wound healing dressings are already available in the market such as Silvakollagen® Gel, Synerheal® Collagen Hydrogel Wound Dressing, Collatek® Collagen Gel, Coloplast triad hydrophilic wound dressing, Collogel, Stimulen™ collagen gel, Coloplast wounds collagen hydrogel dressing, and others. Polymer delivery systems offer a versatile platform for enhancing the delivery and efficacy of peptide-based therapeutics. For example, Fu et al.
226
showed that a cyclic heptapeptide (CyRL-QN15) possessed efficient potency to heal skin wounds. The efficiency of CyRL-QN15 increased when loaded with sodium alginate crosslinked with Zn2+ comprising hollow polydopamine nanoparticles. This hydrogel showed accelerated cell migration, cell proliferation, regulated cytokines secretion from macrophages and reduction of ROS at the wound site thus enhancing wound healing. Moreover, it also promoted the formation of M2 macrophages thereby reducing inflammation and improving re-epithelialization, collagen deposition, granule tissue formation and enhanced angiogenesis in full-depth type 2 diabetic skin wounds. Alternatively, Teixeira et al.
227
further investigated an antimicrobial peptide Tiger 17 combined with electrospun cellulose acetate and PVA crosslinked with glutaraldehyde vapor. The incorporation of Tiger 17 into the hydrogel enhanced the clotting of re-calcified plasma. Additionally, the hydrogel showed efficient antimicrobial activity against
Since the peptides have a short half-life because of their small size, they can easily get degraded by the enzymes present inside the human body and filtered out by the kidneys. To combat this issue, several biocompatible biomaterial combinations including collagen, alginate, chitosan, κ-carrageenan, gelatin, polylactic acid (PLA), and polycaprolactone (PCL) along with the peptide can be used to fabricate novel wound healing dressings which will degrade within a stipulated time period and hence enables a sustained release of the peptides. 229 According to a study by Mizuno et al., integration of bFGF to chitosan films as a delivery vehicle improved the peptide’s stability and permitted a prolonged release at the wound site. The findings revealed that in genetically diabetic mice, the combination action of bFGF and chitosan hastened wound closure and enhanced wound healing. 230 Research has demonstrated the antibacterial effects of hyaluronic acid, a naturally occurring polysaccharide. According to Liu et al., 231 conjugating recombinant FGF-2 with amine-modified hyaluronic acid conjugated with oxidized heparin improved the peptide’s binding kinetics and stability characteristics. Interestingly, FGF-2 was released in vitro from imine-bonded (HAHPi) gels in the form of FGF-2-heparin complexes by imine bond hydrolysis. In contrast, the more stable amine-bonded (HAHPa) gels required free heparin treatment or enzymatic breakdown of the hyaluronate portion to release growth factor. Overall, functional examination of the released FGF-2 revealed that HAHP conjugate gels improved the stability and activity of FGF-2. Moreover, alginate is a biomaterial derived from algae that has gelling qualities that allow it to form a stable hydrogel. When VEGF-A was added to the alginate hydrogel, the peptide’s half-life was longer, and it aided in wound healing. 232
Recently, Hajimiri et al. 233 fabricated sodium carboxymethyl chitosan (NaCMCh) hydrogel nanoparticle carriers for tethering recombinant human epidermal growth factor conjugate (rhEGF) to improve the peptide’s stability and sustained release through the enzymatic action of α-amylase. Importantly, in vitro data showed that the conjugated NaCMCh-rhEGF improved stability of rhEGF and protected it against proteolysis while still retaining biological activity. Additionally, after 15 days, examination of excised wounds in diabetic mice revealed that the wound area in the NaCMCh-rhEGF-hydrogel dressing group was much less than the other groups, with histological finding strongly corroborating with the wound contraction findings as showed in Figure 5(a)–(c). In another study, Shamloo et al. 234 reported a hybrid PVA-gelatin-chitosan hydrogel system incorporating PCL microsphere for accelerating wound healing by sustained release of basic fibroblast growth factor (bFGF). The addition of microspheres to the hydrogel resulted in zero-order release kinetics and lowered degradation time. Moreover, in vivo data also revealed sustained release of bFGF from PCL microspheres contained in the hydrogel framework, thereby significantly promoting wound regeneration, particularly during the early phases of healing.
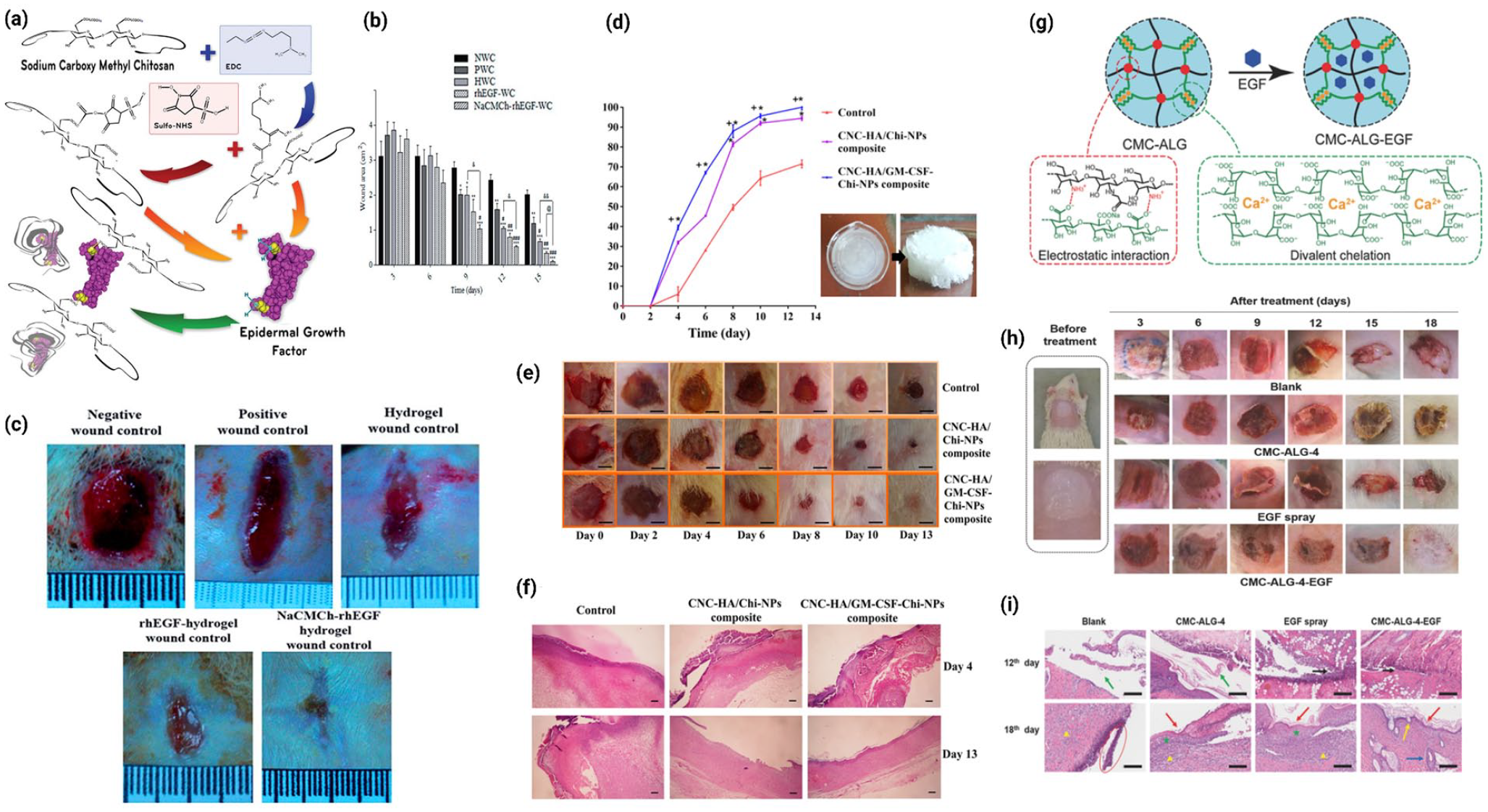
Schematic illustrating recent advances in polymer-based peptide delivery systems for wound healing: (a) schematic showing of NaCMCh–rhEGF conjugation reaction, (b) bar graph representing wound area (in cm2) during 15-day period, (c) typical wound contraction images, at post wounding day 15 in different treatment groups. Image reused with permission from Hajimiri et al. 233 (d) Effect of CNC-HA/GM-CSF-Chi-NPs composite, CNC-HA/Chi-NPs composite and normal saline on wound closure. Values are presented as a percentage of the wound area compared to day 2 (mean ± SD) (left). Images of CNC-HA/GM-CSF-Chi-NPs gels and CNC-HA/GM-CSF-Chi-NPs composites that prepared from these gels (right), (e) photographs of macroscopic appearance of wounds belonging to different treatment groups on different time points, (f) representative histological (H & E stained) images of skin wounds treated with CNC-HA/GM-CSF-Chi-NPs composite, CNC-HA/Chi-NPs composite and normal saline at 4th and 13th day after surgery. Image reused with permission from Karimi Dehkordi et al. 235 (g) schematic illustration of the CMC–ALG and CMC–ALG–EGF hydrogels, (h) representative photographs of wound healing from the 3rd day to the 18th day in different treatment groups, (i) H&E staining images of skin tissue samples at the 12th day and the 18th day for different treatment groups. Image reused with permission from Hu et al. 236
In line with the aforementioned studies, Karimi Dehkordi et al.
235
developed a novel nanocrystalline cellulose (CNC) reinforced HA-based composite containing chitosan nanoparticles loaded with GM-CSF (CNC-HA/GM-CSF-Chi-NPs composite) as an effective wound dressing as presented in Figure 5(d)–(f). The developed CNC-HA/GM-CSF-Chi-NPs composites exhibited excellent physicochemical qualities such as optimal mechanical properties, high swelling capacity, and controlled release of GM-CSF for up to 48 h, making it an ideal choice for wound dressing. In vivo analysis demonstrated that, after 13 days, the wounds treated with CNC-HA/GM-CSF-Chi-NPs composite could reach almost full wound closure and complete re-epithelialization compared to the normal saline treated wounds underscoring its superiority as an effective wound dressing. Toward the goal of developing advanced wound dressings, dual crosslinked amorphous polysaccharide hydrogels were prepared by Hu et al.
236
using
List of combinations of biomaterials along with peptides.
However, the major challenge encountered in using peptides for wound healing is their stability. Peptides are susceptible to enzymatic degradation which limits their efficacy. This can be overcome by chemical modification or encapsulation in biomaterials. Some peptides can also show immunogenicity and trigger potential allergic reactions in the wound site. Therefore, selecting a non-toxic peptide with potential wound-healing capabilities is a crucial process. Furthermore, complexities in isolating the peptides from their source is also a major challenge because of their low availability. Hence, the cost of isolation and production also increases significantly thereby making it less affordable for a lot of people. Moreover, screening the specificity of peptides is also necessary in order to avoid any non-specific binging or off-target delivery which might trigger potential side effects. While peptides possess potential wound healing properties, their efficacy varies depending on the severity of the wound. So, to fabricate a potential hydrogel with highly effective peptides, taking all the challenges into consideration is a must to avoid any sort of complications during clinical use.
Conclusions and future prospects of bioactive peptides used in wound treatment
Both the economy and the general public’s health are negatively impacted by skin damage. The effectiveness, safety, and cost of medications used to treat wounds are frequently concerns. Meanwhile, bioactive peptides have attracted interest as potential wound healing agents due to their high activity, selectivity, and stability. Although it is typically believed that ingesting bioactive peptides, whether in food or concentrated form, has no negative physiological effects, further research is required to substantiate this belief. This is because only a small number of research has been focused on potential toxicological effects in humans, and the great majority of toxicological studies are being carried out in vitro and on animals. These peptides have a great deal of potential for speeding up wound healing, but ensuring their safety is essential for their effective usage in clinical settings.
To further study on the use of bioactive peptides in wound healing, deeper studies are essential to learn more about the underlying principles of how peptides support wound healing procedures. For instance, it has been demonstrated that bioactive peptides in chicken egg whites (CEW) can reduce post-inflammatory hyperpigmentation by blocking the activity of the enzyme tyrosine, which is responsible for the aberrant generation of melanin during the healing process. To comprehend the mechanisms underlying bioactive peptides in wound healing, a variety of methods can be applied. Studying the molecular processes that regulate melanogenesis, confirming effects on human melanocytes, carrying out permeability and melanosome absorption assays, and utilizing Western blots to examine protein translation are all examples of this. Particularly, proteomic analysis through Western blots, ELISA, can be utilized to compare diabetic patients’ wound tissue protein expression levels. It can also be utilized to investigate the biochemical changes that occur in the wound microenvironment throughout the course of treatment. Moreover, a peptidomic approach, aided by mass spectrometry and bioinformatics, could provide novel means of identifying new peptide biomarkers for wound healing therapeutics. Parallelly, transcriptomic analysis through qPCR analysis can reveal how genes involved in cellular mobility are regulated during wound healing. Such investigation of gene expression during wound healing can potentially uncover the molecular pathways beneath wound healing and regeneration. Prior to conducting additional research using animal models or in human clinical trials, the effects on protein translation, degradation, melanosome maturation, and cell autophagy should be assessed. All these methods will contribute to a deeper comprehension of the mechanisms by which bioactive peptides derived from CEW affect melanogenesis.
In summary, bioactive peptides can effectively combat a wide range of pathogens, including antibiotic-resistant bacteria, which are common in chronic wounds. This antimicrobial property is essential for preventing infections and promoting a healthier wound environment. Moreover, many bioactive peptides can modulate the inflammatory response, reducing excessive inflammation that can delay wound healing. By controlling inflammation, these peptides help create a conducive environment for tissue repair. By stimulating angiogenesis, cell migration, and proliferation, bioactive peptides can quicken the healing process after wounds. Additionally, they improve the synthesis of elements of the ECM, which are essential for tissue regeneration. More research into and identification of novel bioactive peptides from plants, microbes, and marine species could result in the development of more potent medicines for healing wounds. The stability, bioavailability, and targeted administration of bioactive peptides can be improved by research into cutting-edge delivery technologies, such as nano-carriers and smart dressings, thereby optimizing their therapeutic potential. The synergistic effects of combining bioactive peptides with additional therapeutic agents, like growth factors, stem cells, or traditional antibiotics, may improve the overall results of wound healing. The development of more effective and targeted medicinal treatments will benefit from additional study to completely understand the mechanisms of action of bioactive peptides.
Due to the peptide compositions, AMPs are unfortunately limited in their potential to be used in vivo by a number of variables, including minimal tissue penetration, rapid removal from circulation, decreased bioavailability, limited stability, and low selectivity against bacterial cells relative to mammalian cells. Despite the immense potential of peptide therapeutic agents as next-generation treatments, some issues still remain to be resolved. For instance, many intracellular proteins have low cell membrane permeability due to their high molecular weight and charged amino acid residues. 238 Furthermore, their bioavailability is reduced by the vulnerability to denaturation and breakdown in the presence of proteolytic enzymes and low pH including dissociation of subunits and their rapid metabolism. 239 Hence, to minimize the challenges, significant advances have been made in preventing peptides and proteins from losing their bioactivity when subjected to adverse circumstances during processing, storage, and post-administration by incorporating them within polymeric materials. Polymeric systems using biodegradable synthetic polymers, like polyesters, polyorthoesters, and polyphosphoesters, are frequently utilized.240,241
However, the use of these polymeric systems may have disadvantages that could compromise the therapeutic outcomes of peptide and protein-based drugs. These disadvantages include the need for organic solvents, mechanical damage during mixing, water penetration during delivery and the production of inflammatory byproducts during degradation. 1 The design and formulation of polymeric-based peptides delivery systems utilizing optimum crosslinking densities, hydrophilicity of biopolymers used as deliver scaffold, polymer concentration and other factors play a crucial role in determining the drug release at the wound site. To illustrate, polymeric scaffolds with low crosslinking densities have high permeability which promotes the rapid release of the drug while high crosslinking density polymers lead to a sustained release. However, if the density is way much higher, it causes a more pronounced impermeability against water, thereby hindering the exchange of substances. Similarly, hydrophilic polymers tend to degrade at a much faster rate as compared to hydrophobic/amphiphilic polymers, thus effecting the release of incorporated peptides within them Hydrophobic polymers, however, might trigger some immunogenic reactions as side effects. 242 Nevertheless, despite numerous research and scientific findings, polymeric-based peptides are yet to achieve a significant position in the clinical market as a replacement for conventional wound healing dressings. Therefore, altering and modifying these potential parameters would aid in overcoming the existing barriers against the practically effective use of bioactive peptides as therapeutic agents for wound healing.
Footnotes
Acknowledgements
The authors would like to acknowledge the research team in the Center for Tissue Engineering and Regenerative Medicine (CTERM), Faculty of Medicine, Universiti Kebangsaan Malaysia for the technical support throughout this review.
Authors’ contributions
Conceptualization: N.I.M.F. and M.B.F.; methodology and software: N.I.M.F.; validation: N.I.M.F. and M.B.F.; formal analysis: N.I.M.F.; investigation and resources: N.I.M.F., N.A.S., R.A.M.R., A.S., A.G., H.A., M.M., A.M., and M.B.F; data curation: N.I.M.F.; writing—original draft preparation: N.I.M.F.; writing—review and editing: N.I.M.F., H.A., M.M. A.M., and M.B.F.; figure and visualization: N.I.M.F. and A.S.; supervision: M.B.F.; project administration and funding acquisition: H.A. and M.B.F.. All authors have read and agreed to the final version of this manuscript.
Availability of data and materials
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the
Informed consent statement
Not applicable.
