Abstract
Millions of people worldwide suffer from skin injuries, which create significant problems in their lives and are costly to cure. Tissue engineering is a promising approach that aims to fabricate functional organs using biocompatible scaffolds. We designed ultrashort tetrameric peptides with promising properties required for skin tissue engineering. Our work aimed to test the efficacy of these scaffolds for the fabrication of dermal grafts and 3D vascularized skin tissue models. We found that the direct contact of keratinocytes and fibroblasts enhanced the proliferation of the keratinocytes. Moreover, the expression levels of TGF-β1, b-FGF, IL-6, and IL-1α is correlated with the growth of the fibroblasts and keratinocytes in the co-culture. Furthermore, we successfully produced a 3D vascularized skin co-culture model using these peptide scaffolds. We believe that the described results represent an advancement in the fabrication of skin tissue equivalent, thereby providing the opportunity to rebuild missing, failing, or damaged parts.
Graphical abstract

Introduction
Every year worldwide, hundreds of thousands of patients require hospitalization for organ failure, which affects the quality of life and is costly to treat. Tissue engineering is an alternative approach for creating tissue constructs and fabricating functional organs using biological scaffolds. The scaffold is a major component of a tissue-engineered construct made from a biomaterial, with the 3D architecture to accommodate more cells and promote cell adhesion and proliferation. Recently, the fabrication of a biomimetic scaffold has attracted the interest of researchers. 1 Among these biomaterials are self-assembling peptide hydrogels, which mimic the morphology and mechanical properties of the native extracellular matrix. 2 These biocompatible peptide hydrogels have been shown to have promising applications in various biomedical applications such as tissue engineering, drug delivery, regenerative medicine, microbiology, and biosensing.3 –9
From this viewpoint, we have designed self-assembly ultrashort peptide hydrogels, Ac-IVFK-NH2 (Ac-Ile-Val-Phe-Lys-NH2) and Ac-IVZK-NH2 (Ac-Ile-Val-Cha-Lys-NH2), with promising properties for wound healing applications. 10 These peptides have an innate tendency to self-assemble in aqueous conditions without needing gelators, cross-linkers, or mechanical stimulators into helical nanofibers. These helical nanofibers organize themselves into supramolecular 3D meshed nanofibrous networks, resembling the extracellular matrix in their architecture. The self-assembly of these peptides occurs through the antiparallel pairing interaction of two peptide monomers, forming helical intermediate structures. The condensation of these fibers leads to hydrogel scaffold formation.3 –5
We evaluated the effect of these materials on skin cell proliferation and graft in treating full-thickness wounds in minipigs for skin tissue engineering.10,11 From this promising observation, the present study aimed to use the abovementioned peptide scaffolds to fabricate bilayer 3D skin tissue models. Bell et al. and his group were the first to use dermal fibroblasts and epidermal keratinocytes to treat chronic burn wounds. 12 Moreover, they developed Apligraft®, consisting of dermal fibroblasts and collagen, to treat full-thickness chronic wounds. Nevertheless, the results were unsatisfactory as the wound was not covered and needed too much time to cover the wound entirely.13,14
Thus, the development of a new scaffold for fabricating a functional bilayer skin substitute to treat full-thickness chronic wounds for optimal wound healing has to be implemented to achieve a complete cure for chronic wounds within a short period. To successfully achieve a bilayer skin replacement, the scaffold should be biocompatible to maintain cellular viability for extended periods while being biodegradable in a controlled manner. The encapsulated cells can then secrete their own ECM, replace the degrading scaffold material, and eventually achieve dermal regeneration. Furthermore, the scaffold should be an animal-free source to avoid disease transmission and adverse immune responses.
Therefore, we previously developed two compounds of rationally designed ultrashort peptide materials. To fulfill the criteria of a tissue-engineered scaffold in maintaining the in vitro proliferation of human dermal fibroblasts, enhancing collagen deposition, and supporting the pre-revascularization of the dermal construct. 11 Based on these findings, in this study, we fabricated a non-vascularized skin substitute by co-culturing human dermal fibroblasts within the abovementioned novel scaffolds and seeding epidermal keratinocytes on top of the surface of both scaffolds. We found that the direct contact of keratinocytes and fibroblasts enhanced keratinocyte proliferation. Moreover, the significant expression of different cytokines and growth factors confirmed the functionality of our skin co-culture model. We assessed the levels of pro-inflammatory cytokines IL-1 alpha and IL-6, and also the levels of growth factors TGF-β1 and b-FGF, known to boost keratinocyte proliferation and differentiation.15 –18
Another consideration of the in vitro fabrication of these 3D models is the common problem of cell viability loss in long-term cultures, 19 resulting from a lack of neovascularization from the peripheral vessels of the wound edge. 20 This leads to poor nutrition and oxygen deficiency inside tissues. 21 Several studies have confirmed that insufficient vascularization leads to cellular necrosis or the complete loosening of implanted skin replacements.22,23 Furthermore, the successful survival of any engineered tissue upon implantation depends on the efficient diffusion of nutrients and oxygen. 24 Thus, vascularization of engineered tissue is necessary for the fabrication of three-dimensional tissue constructs. This study successfully produced a 3D vascularized bilayer skin model by seeding keratinocytes on top of the vascularized dermal construct using the tetrameric peptide scaffolds. Considering our results, we believe that both peptide hydrogels are promising materials for fabricating dermal grafts and 3D vascularized skin equivalents, particularly for wound healing.
Materials and methods
Materials
The two tetrameric self-assembling peptides IVFK (Ac-Ile-Val- Phe-Lys-NH2) and IVZK (Ac-Ile-Val-Cha-Lys-NH2) are rationally designed peptides. The peptides are amphiphilic with a dominant hydrophobic part and a hydrophilic positively charged residue lysine head group at the C-terminus. All peptides are acetylated at the N-terminus and amidated at the C-terminus. Both peptide compounds were synthesized by solid-phase peptide synthesis (SPPS) using a CS136X CS Biopeptide synthesizer. The following materials were purchased from (Gibco, USA): human dermal fibroblasts-neonatal (HDFn, C0045C), human epidermal keratinocytes (HEKn, C-001-5C), Epilife keratinocyte medium and its growth supplements (HKGS, S-001-5), Dulbecco’s modified eagle medium (DMEM), fetal bovine serum (FBS), Dulbecco’s phosphate-buffered saline (PBS) solution, and penicillin-streptomycin antibiotics (P/S). In addition, primary human keratinocytes isolated from foreskin samples were provided as a kind gift from Professor Abdalla Awidi at The University of Jordan/Cell Therapy Center in Jordan and used throughout this study. Human umbilical vein cells (HUVEC, CC-2517A) and their growth-supplemented medium (EGM-2) were purchased from Lonza. A CellTiter-Glo® luminescent 3D cell viability assay kit, an MTT cell proliferation assay kit, a cytotoxicity assay kit (CytoTox96), and a LIVE/DEAD Viability/Cytotoxicity kit were purchased from Promega, USA. CD31 and anti-vimentin antibodies were procured from Abcam. Cell culture flasks and 96-well plates were ordered from Corning, USA. ELISA kits for IL-1α, IL-6, TGF-β1, and b-FGF were purchased from Sigma-Aldrich.
The study including its experimental protocols was approved by the Institutional Biosafety and Bioethics Committee (IBEC) of King Abdullah University of Science and Technology (KAUST) (21IBEC023).
Hydrogel preparation
The peptide hydrogels were prepared by dissolving the peptide powders in Milli-Q water. Then, the peptide solution was mixed with 10x phosphate-buffered saline to enhance the gelation to reach a final concentration of 1xPBS. The critical gelation concentration (CGC) of IVFK (4 mg/mL) and IVZK (3 mg/mL) was identified using the vial inversion assay and used throughout the study. 6 At CGC for IVFK and IVZK peptides, the gelation occurred within 5 min at room temperature.
Scanning electron microscopy (SEM)
The fiber structures were evaluated with a field emission scanning electron microscopy system (FEI Nova Nano 630 SEM, Oregon, USA). The samples were prepared as previously described. 10 Briefly, the peptide nanogels were dried out by gradually increasing concentrations of 30%, 50%, 70%, 90%, and 100% (v/v) ethanol solutions for 15 min in each solution. Then, 100% ethanol solution was used twice for further dehydration for 15 min each. After that, the dehydrated samples were kept in a 1:2 ratio of HMDS and ethanol for 20 min. Next, the samples were incubated in a fresh solution of a 2:1 ratio of HMDS and ethanol for 20 min, then 100% HMDS. This last step was done twice for 20 min, and then the samples were incubated overnight inside a fume hood to evaporate the HMDS. The samples were sputter-coated with a 5 nm thick coating of Iridium and a 3 nm thick coating of gold/palladium. The coated samples were then imaged with a field emission scanning electron microscopy system (FEI Nova Nano630 SEM, Oregon, USA).
Transmission electron microscopy (TEM)
The fiber diameter was analyzed using transmission electron microscopy (FEI Titan G2 80-300 CT). The TEM samples were prepared by dropping diluted peptide hydrogels on carbon-coated copper grids. The grid was then stained with 2% uranyl acetate.
Cell culture of human dermal fibroblasts (HDFn)
The HDFn cells were cultured in a T75 culture flask in complete DMEM media (10% fetal bovine serum and 1% penicillin/streptomycin). Cells were kept in a 37°C incubator with 95% air and 5% CO2. The medium was replenished every 2 days. The cells were subcultured using trypsin/EDTA at approximately 80% confluence. Passage 4–7 fibroblasts were used.
Cell culture of human epidermal keratinocytes, neonatal (HEKn)
The HEKn cells were cultured in a T75 culture flask in the Epilife medium supplemented with growth factors (HKGS, S-001-5). Cells were kept in a 37°C incubator with 95% air and 5% CO2. The medium was changed every 2 days. The cells were subcultured using trypsin/EDTA before reaching 80% confluence. Passage 4–7 keratinocytes were used.
Cell culture of human umbilical vein cells (HUVEC)
The HUVEC cells were cultured in a T75 culture flask in epidermal supplemented medium EGM-2. The cells were incubated at 37°C incubator with 95% air and 5% CO2 until reaching 80% confluency, then subcultured using trypsin/EDTA.
Coculture of HDFn and HEKn
Fibroblast and keratinocyte cells were encapsulated separately in different peptide hydrogels, IVFK (4 mg/mL) and IVZK (3 mg/mL), at a density of 2 × 104 cells/well and 4 × 104 cells/well, respectively. A co-culture was performed by seeding keratinocytes on top of a 3D cultured fibroblast encapsulated within our peptide hydrogels cultured for 2 days. The effect of fibroblasts on keratinocytes growing after 5 days was observed using an inverted confocal microscope (Zeiss LSM 710, Germany).
Tracking the co-culture of keratinocytes and fibroblasts within the 3D scaffold
Dual fluorescent stains were used to distinguish between keratinocytes and fibroblasts in a 3D co-culture. Cell tracker red was used to label keratinocytes, and cell tracker green for labeling fibroblast cells. Keratinocytes were seeded after 2 days at a density of 4 × 104 cells/well on the upper surface of dermal fibroblasts (2 × 104 cells/well) embedded in different hydrogels on the lower surface. The increase in cell density for both cell types was observed by confocal microscopy (Zeiss LSM 710, Germany) at different time points. Moreover, the co-cultured constructs were stained with different markers: vimentin against fibroblasts and cytokeratin 14 against basal keratinocytes to confirm that both cells were mitotically active.
Volume scope scanning electron microscopy
Teneo volume scope scanning electron microscopy was carried out after 18 days of co-cultured keratinocytes and fibroblasts to visualize the interaction between epidermal keratinocytes and dermal fibroblasts at higher magnification. This test was conducted by constructing the dermis layer (fibroblasts) for 14 days; then, epidermal keratinocytes were applied on top of the dermis layer. Finally, the cryosections were done 4 days after seeding keratinocytes.
Quantitative expression of IL-1α, IL-6, TGF-β1, and b-FGF
Different cytokines and growth factors are involved in the wound healing process and skin reepithelization; among those are pro-inflammatory cytokines, including IL-1 and IL-6, and growth factors, such as TGF-β1 and b-FGF. IL-1 and IL-6 are produced by keratinocytes and fibroblasts and are involved in keratinocytes chemotaxis and fibroblasts activation. TGF-β1 is important for fibroblast activation and ECM deposition, and b-FGF plays an essential role in angiogenesis and keratinocyte proliferation. To evaluate whether our 3D co-culture conditions induced a better production of different cytokines and are better suited for generating dermal/ epidermal skin substitutes that would also enhance wound healing. We measured the expression level of IL-1, IL-6, TGF-β1, and b-FGF under different culture conditions by enzyme-linked immunosorbent assay (ELISA). The supernatant of 3D monoculture and co-culture human dermal fibroblast and human epidermal keratinocytes were collected after 72 h, and ELISA kits detected the respective cytokines according to the manufacturers’ instructions. Briefly, 100 μL of each standard and sample were added to each well of a pre-coated plate with the cytokines (IL-1α or IL-6) or the growth factors (TGF-β1 or b-FGF) to be measured. The plate was incubated for 2.5 h at room temperature with gentle shaking, then the supernatant was discarded, and the wells were rinsed four times.
Next, a 100 μL biotinylated detection antibody was added to each well and incubated for 1 h at room temperature with gentle shaking. After four times washing of the detection antibody, 100 μL HRP-streptavidin solution was added to each well and incubated for 45 min at room temperature with gentle shaking. Following the incubation and washing steps, 100 μL TMB ELISA reagent was added to each well for 30 min in the dark at room temperature. The color developed in proportion to the amount of target protein bound. After adding a stop solution, the color changed from blue to yellow. Finally, a plate reader (PHERAstar FS, Germany) was used to record the intensity of the color at 450 nm.
Statistical analysis
All results are presented as the mean ± standard deviation. Three independent experiments were conducted for each test type. One-way analysis of variance (ANOVA) with a Tukey post-test was used to determine the statistical differences between the experimental groups. The results were considered to be statistically significant when p < 0.05.
Results
Nanofibrous morphology of self-assembling peptides
Scanning electron microscopy (SEM) was used to observe the nanofibrous morphology of self-assembling peptides (Figure 1). The nanofibrous structure of both peptides was compared to those observed in bovine collagen (Supplemental Figure S1), comprised of a unique triple-helical structure. 25 SEM results confirmed that both IVFK and IVZK peptide hydrogels formed a nanofibrous network, resembling the fibrous structure of collagen. 25 This nanofibrous structure was produced first by the antiparallel pairing of two peptide monomers, 4 followed by stacking the peptide pairs that generated the protofibrils. Condensation of these tiny fibers resulted in hydrogel formation. Furthermore, the diameter of a single fiber was observed using TEM. The TEM micrographs (Figure 1) revealed the formation of fiber bundles with the width of a single fiber of about 3 nm for both peptides.

The morphology of self-assembling IVFK and IVZK peptide hydrogels at a concentration of 4 mg/mL and 3 mg/mL, respectively. The images were taken using scanning electron microscopy (SEM) and transmission electron microscopy (TEM).
Interaction of fibroblasts and keratinocytes
We previously confirmed the biocompatibility of these scaffolds on 3D cultured HDFn cells. 10 Herein, the biocompatibility studies were repeated to ensure our materials’ suitability before fabricating the bilayer skin substitute. This test was conducted by constructing the dermis layer by encapsulating HDFn within both peptide scaffolds and then seeding the HEKn on top of the 3D cultured HDFn. Bright-field microscopy images revealed that in the early stage of the culture (after day 7), the proliferation of epidermal keratinocytes contacting dermal fibroblasts was significantly faster (Figure 2(g)–(i)) than those not in contact with fibroblasts (Figure 2(d)–(f)).
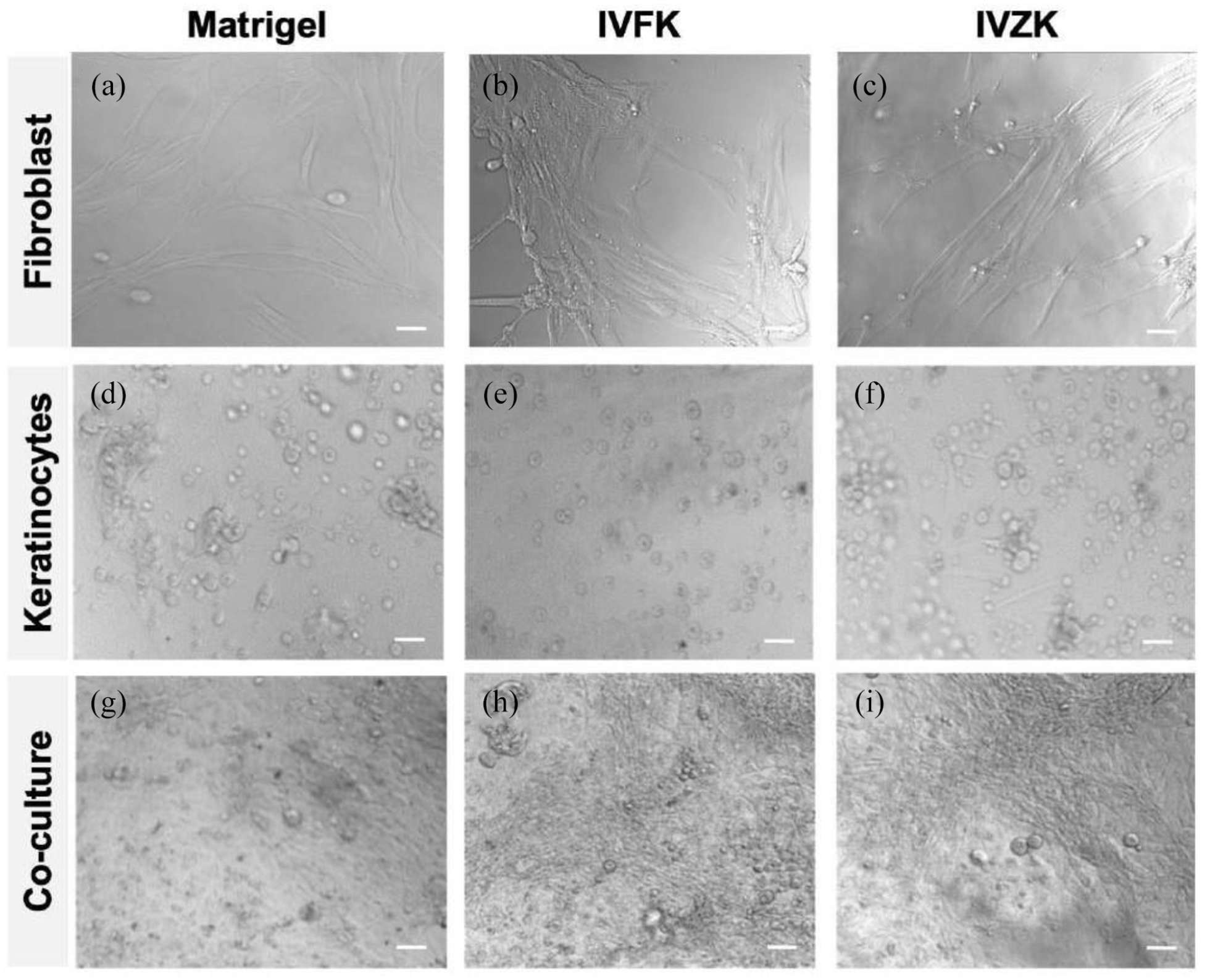
Bright-field images evidencing the enhancement of keratinocyte proliferation in co-culture with fibroblasts for different scaffolds: Matrigel (a, d, g), IVFK (b, e, h), and IVZK (c, f, i). Scale bars 50 μm.
Tracking the co-culture of keratinocytes and fibroblasts within the 3D scaffold
Different tracker fluorescent stainings were applied to the 3D co-cultured system to track and clearly distinguish the proliferation of co-cultured HDFn with HEKn at different time points. Dual fluorescent staining results confirmed the cellular viability and expansion in a 3D co-cultured environment (Figure 3). In addition, the homogenous distribution of fibroblasts (green) and keratinocytes (red) in the peptide hydrogels was detected through confocal microscopy (Figure 3). On day 7, comparable overgrowing of fibroblasts was observed in all tested groups. In contrast, on day 14, keratinocytes highly proliferated and covered most fibroblasts and tended to form skin-like structures on top of the fibroblasts.

The viability of co-cultured cells on different scaffolds (4 mg/mL IVFK and 3 mg/mL IVZK). Matrigel was used as a positive control (4 mg/mL). Fibroblasts are stained with tracker green fluorescence and keratinocytes with tracker red fluorescence. Significant fibroblast growth on day 7 in Matrigel and IVZK compared to IVFK. On Day 14, a layer of keratinocytes (red fluorescence) has been formed on top of fibroblast (green fluorescence) on IVZK 3D culture compared to IVFK and Matrigel. Scale bars 100 μm.
Furthermore, biomarker CK14 (keratinocytes surface marker, expressed in both keratinized and non-keratinized epithelial tissue and conserves its structural integrity) was significantly expressed in non-vascularized constructs, indicating that the basal keratinocytes in the epidermis layer were mitotically active. Moreover, skin-like structures formed on top of fibroblasts in all tested conditions, as shown in Figure 4.

The mitotic activity of fibroblast (green), keratinocytes (red), and nucleus (blue) in 3D co-cultured constructs. The cells were cultured on different scaffolds (4 mg/mL IVFK and 3 mg/mL IVZK), and Matrigel was used as a positive control (4 mg/mL). Scale bars 100 μm.
The morphological observation of fibroblasts and keratinocytes co-culture (skin co-culture model)
We carried out volume scope SEM to see the cell-cell interactions between Fibroblast/keratinocytes at higher magnifications. Cross-sections of peptide hydrogels showed the bilayer skin formation with the epidermal layer (Figure 5) on top of the spongy three-dimensional dermal matrix of fibroblast cells located between a high density of banded collagen fibrils (red arrows), confirming the construction of skin-like structures. 26 This result corroborated our previous findings on cellular distribution and collagen deposition by H/E and Masson trichrome. 11

Volume scope scanning electron microscopy images of skin co-culture model after 18 days in the submerged culture at different magnification. Cryosections indicated the formation of different keratinocyte layers (1–3), with cells showing keratohyalin granules (KG) and keratin filaments (KF). Red arrows denoted the collagen secreted from human dermal fibroblast cells (white arrows) within the middle dermis layer.
ELISA for IL-1α, IL-6, TGF-β1, and b-FGF
Many cytokines and growth factors play an essential role in the complicated wound healing process. We investigated the expression level of two pro-inflammatory cytokines (IL-1 α and IL-6) and two growth factors (TGF-β1 and b-FGF) that have an interconnected and substantial role in cell proliferation and re-epithelization.
Quantitative expression of cytokines and growth factors revealed a significantly higher level of IL-1α, IL-6, TGF-β1, and b-FGF in the 3D co-culture system than in monoculture (Figure 6). However, a slight reduction of IL-1α and b-FGF was observed in the 2D culture system (TCP), as shown in Figure 6(a) and (c). Moreover, IL-6, TGF-β1, and b-FGF were significantly expressed in 3D monoculture fibroblasts and co-culture, while IL-1α was significantly expressed in 3D monoculture keratinocytes and co-culture.

The in vitro release profile of cytokines IL-1α (a), IL-6 (b), and growth factors b-FGF (c) and TGF-β1 (d) in 3D monoculture and co-culture system after 72 h. Fibroblasts and keratinocytes were used for 3D monoculture and co-culture within IVFK and IVZK. Matrigel was used as 3D positive control and TCP (tissue culture plate) as negative 2D control.
Pre-vascularization of the dermal construct (tube-like structure formation) and fabrication of the skin substitute
In a previous study, we successfully fabricated a 3D vascularized dermal layer without any supportive vascular growth factors by co-culturing HDFn with HUVEC using different nanogel scaffolds in a ratio of 1:1 and immunostained with different antibodies: CD31against HUVEC cells and vimentin against fibroblast as previously described. 11 In this study, a novel 3D vascularized skin substitute was fabricated by seeding the epidermal keratinocytes on top of the vascularized dermal construct. Confocal microscopy results revealed epidermal layer-like structure formation with both peptide hydrogels on top of the vascularized dermal construct (Figure 7). Notably, basal keratinocytes were mitotically active within all tested materials. Vascularization in IVZK nanogels was not as good as in Matrigel and IVFK nanogels. These results demonstrated that both peptides provided a favorable environment for the investigated cell types.
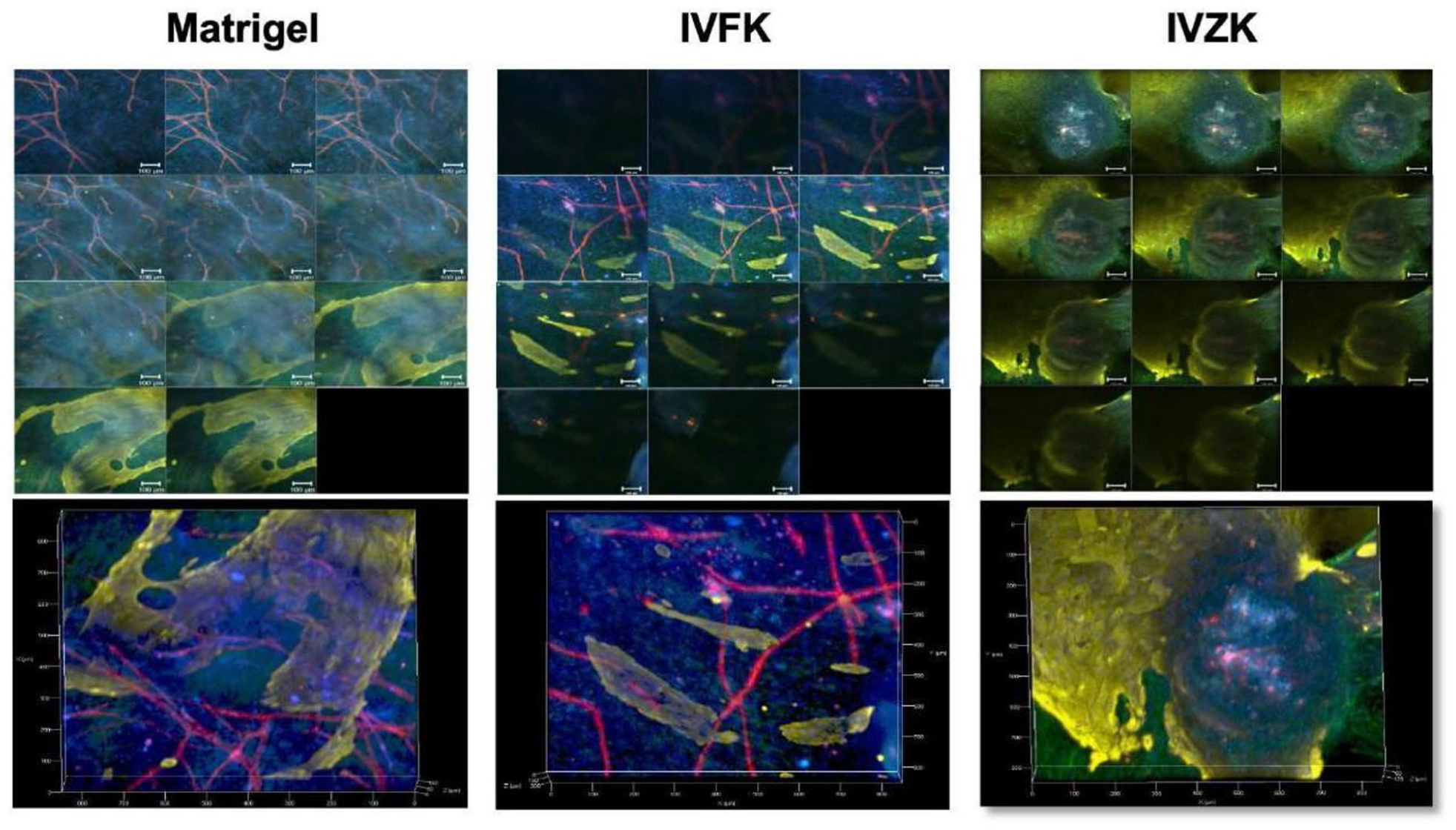
Fabrication of 3D co-culture vascularized skin constructs using (4 mg/mL IVFK and 3 mg/mL IVZK) Matrigel was used as a positive control (4 mg/mL). This figure shows Z-stack images across the entire depth of vascularized skin constructs. Vimentin, CD31, and CK14 antibodies were used to stain fibroblast, endothelial cells, and keratinocytes, respectively. Scale bars 100 μm.
Discussion
A considerable discrepancy between the number of patients on the waiting list for organ transplants and available donors has led to the search for alternative approaches to substitute compromised or missing tissues and organs. Hence, replacements for diseased and damaged skin tissues are in high demand.27 –29 In this study, we used tetrameric peptides and tested their ability as scaffolds for the fabrication of 3D vascularized skin grafts needed in wound healing.
The nanofiber network formed from the self-assembling of ultrashort peptides IVFK and IVZK was confirmed by SEM, with the average diameter of nanofibers in both the peptides of approximately 10–20 nm, where the fibers structurally resembled the collagen fibers in terms of their architecture. 2 Furthermore, the diameter of these nanofibers supported their use as scaffolds for skin regeneration,30 –33 where the natural ECM diametric range is between 5 and 300 nm, 34 in addition to the mechanical stability of both peptide nanogels.10,11
All the biocompatibility studies were repeated, which were extremely important for tissue repair and regeneration to ensure that both hydrogels were biocompatible and non-cytotoxic before constructing the bilayer skin equivalent. The results obtained correspond with the results obtained from our previous study (Supplemental Figures S2–S5). 10 Our previous findings demonstrated that the encapsulated cells were metabolically active.
Furthermore, fluorescent staining of the actin cytoskeleton, serving as direct evidence for cellular morphology and cytoskeleton structure, 35 demonstrated that dermal cells proliferated and extended within the scaffolds in their first days of encapsulation, and growth rates rapidly increased.10,11 Moreover, a network was created through cell-to-cell junctions, ultimately forming an extensive network saturating the nanogel matrix. The deposition of collagen in the histological section of the dermal fibroblast layer indicated that fibroblasts were mitotically and functionally active by producing their collagen matrix. 11
This study, we used the peptide hydrogels mentioned above to engineer a skin bilayer through the co-culture of fibroblasts and keratinocytes. The construction of this 3D skin model was performed by the 3D culturing of HDFn in both IVFK and IVZK hydrogels and then seeding HEKn on top of the 3D cultured HDFn. The outcome of co-culturing demonstrated that fibroblast cells tend to grow faster than keratinocytes, despite the higher cell density of keratinocytes. Furthermore, the proliferation of keratinocytes was enhanced when co-cultured with fibroblasts which might be attributed to the growth factors secreted by co-cultured fibroblasts that are essential for epidermal cell morphogenesis, 36 such as keratinocyte growth factor,37,38 insulin-like growth factor II, 39 and hepatocyte growth factor/scatter factor. 40 Others demonstrated the same observation, suggesting that the proliferation of keratinocytes during wound healing could be enhanced by direct contact of keratinocytes with fibroblast constructs.41 –48
Additionally, a skin-like layer was formed when keratinocytes were seeded on fibroblasts for 14 days, which showed similar results obtained earlier by others.49 –51 These results confirmed that the growth of keratinocytes does depend on fibroblast or fibroblast secretions. 41 Furthermore, it has been known that cross-talk between epidermis and dermis layers occurs through paracrine signaling that maintains homeostasis during tissue repair. 52 Moreover, fibroblasts secrete soluble angiogenic growth factors such as vascular endothelial growth factor (VEGF), platelet drive growth factor (PDGF), and transforming growth factor-beta (TGF-β1),53,54 which stimulate and promote the sprouting of endothelial cells, the formation of the lumen, and the maintenance of the long-term stability of new vessels. 55
We further demonstrated the importance of 3D co-culture conditions in wound healing applications by assessing the production level of inter-connected cytokines and growth factors. For example, IL-1α indirectly stimulates keratinocyte growth by stimulating dermal fibroblasts to express and release a series of growth factors essential for basal cell growth, including TGF-β1 and b-FGF. In addition, we also examined the level of b-FGF, besides its important role in re-epithelization and angiogenesis 15 ; it indirectly impacts wound healing, encouraging epithelialization by increasing TGF-β1. 16 Both IL-1α and b-FGF levels were higher in 3D co-culture conditions, further emphasizing the potential of our 3D co-culture model in wound healing applications.
Herein, we confirmed that direct contact of epidermal keratinocytes with dermal fibroblast construct on 3D scaffolds significantly enhanced epidermal cell proliferation, affirmed by the significant increase in the expression of IL-1α, an initiator of keratinocyte activation, in cultured keratinocytes alone as well as in 3D co-culture system, which, in turn, stimulated IL-6 that activates the proliferation of fibroblast and their production of extracellular matrix components as well as stimulate the secretion of TGFβ-1 (autocrine signal) and b-FGF (paracrine signal). As a result, TGF-β1 induced the expression of CK14 (early differentiation markers of basal keratinocytes) that returned the healthy phenotype of the basal keratinocytes in an autocrine manner.
Furthermore, IL-6 and b-FGF, secreted by dermal fibroblasts in response to the release of IL-1α, stimulated the proliferation and the sprouting of endothelial cells, consequently promoting angiogenesis and neovascularization in a paracrine manner.18,56,57 These outcomes suggested that the enhancement of keratinocyte proliferation was primarily due to the cytokines delivered by the two cells and that both cytokines (IL-1α and TGF-β1) played an important role in fibroblast-stimulated keratinocyte proliferation. Other investigators have reported a similar observation. 41 Indeed, the function of fibroblasts in skin tissue is stimulated by the presence of epidermal keratinocytes, 36 and the growth of keratinocytes cultured with fibroblasts occurs via a double paracrine manner. 58
The most recent studies have generated skin substitutes by co-culturing epidermal keratinocytes with dermal fibroblasts within a 3D scaffold. 59 However, cell viability was lost in long-term cultures, 19 resulting from poor nutrition and oxygen deficiency inside the construct, 21 which prevented using these scaffolds for clinical application. Thus, the vascularization of skin substitutes is in high demand. We have previously fabricated a pre-vascularized dermal construct by co-culturing HDFn with HUVEC using these nanogel scaffolds to overcome this hurdle. In this work, we fabricated a vascularized skin equivalent using our previously fabricated pre-vascularized dermal construct and seeding epidermal keratinocytes on top. The results showed that both peptide nanogels offered a favorable microenvironment for dermal fibroblasts but also provided suitable niches for epidermal keratinocytes’ growth, as they proliferated and formed a layer-like skin structure on top of the vascularized construct after 4 days.
Notably, the IVFK scaffold fulfilled the criteria of a tissue-engineered scaffold by maintaining the in vitro proliferation of dermal fibroblasts, enhancing collagen deposition, supporting the pre-revascularization of the dermal construct, and providing a favorable microenvironment for epidermal keratinocytes to form a layer like skin structure. Furthermore, the implantation of pre-vascularized 3D gel with keratinocytes has been shown to accelerate mice’s wound healing within 14 days. 60
Importantly, pre-vascularization of the scaffold using endothelial progenitor cells has been shown to accelerate skin wound recovery. 60 Moreover, expanded vessel arrangement inside the scaffold provides oxygen and nutrients necessary to maintain skin graft. 60 Consequently, our vascularized 3D skin substitute may have great potential for accelerating the wound healing process, thereby permitting prior anastomosis.
Conclusion
This study demonstrates that both IVFK and IVZK scaffolds support cellular attachment and substantially increased epidermal cell proliferation over time, eventually covering the surface of the entire construct to form skin-like 3D structures. The results represent an improvement in the fabrication of dermal grafts and 3D skin models and thus are safe for topical application. We suggest that both peptide nanogels are promising biomaterials for tissue engineering applications and could act as potential carriers for HDFn transplantation in vivo.
Notably, comparing both investigated peptide scaffolds IVFK and IVZK, the novel peptide material IVFK fulfilled best the criteria of a tissue-engineered scaffold and served as an efficient substrate for vascular tissue engineering, where it maintained the in vitro proliferation of HDFn, and HEKn, and enhanced collagen deposition, 10 in addition to its role in supporting the pre-revascularization of the dermal construct. Besides, IVFK hydrogel has offered a substantial increase in mouse myoblast cell viability and supported its expansion and differentiation to myotube muscle cells.61,62
Further in vivo studies should be conducted by applying vascularized skin construct, using autologous skin cells, on chronic diabetic full-thickness wounds. In addition, follow-up studies are needed to evaluate the fate of construct post-grafting.
Supplemental Material
sj-docx-1-tej-10.1177_20417314221111868 – Supplemental material for Peptide nanogels as a scaffold for fabricating dermal grafts and 3D vascularized skin models
Supplemental material, sj-docx-1-tej-10.1177_20417314221111868 for Peptide nanogels as a scaffold for fabricating dermal grafts and 3D vascularized skin models by Wafaa T Arab, Hepi H Susapto, Dana Alhattab and Charlotte A E Hauser in Journal of Tissue Engineering
Footnotes
Acknowledgements
The authors would like to acknowledge the support from Ohoud Alharbi, KAUST Core Laboratories, for SEM imaging. We also acknowledge Manola Moretti from King Abdullah University of Science and Technology for the valuable discussions and suggestions. Finally, we also acknowledge Prof. Abdalla Awidi from the University of Jordan/Cell Therapy Center for providing primary human keratinocytes.
Author contributions
Charlotte AE Hauser: Conceptualization, Funding acquisition, Supervision, Data curation, Writing – review, and editing. Wafaa T Arab: Conceptualization, Methodology, Investigation, Data curation, Writing – Original Draft, Formal analysis. Hepi H Susapto: Conceptualization, Methodology, Investigation, Writing – review, and editing. Dana M Alhattab: Conceptualization, Methodology, Validation, Writing – review, and editing. All authors contributed to the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by King Abdullah University of Science and Technology (KAUST).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
