Abstract
The rising prevalence of diabetes has underscored concerns surrounding diabetic wounds and their potential to induce disability. The intricate healing mechanisms of diabetic wounds are multifaceted, influenced by ambient microenvironment, including prolonged hyperglycemia, severe infection, inflammation, elevated levels of reactive oxygen species (ROS), ischemia, impaired vascularization, and altered wound physicochemical properties. In recent years, hydrogels have emerged as promising candidates for diabetic wound treatment owing to their exceptional biocompatibility and resemblance to the extracellular matrix (ECM) through a three-dimensional (3D) porous network. This review will first summarize the microenvironment alterations occurring in the diabetic wounds, aiming to provide a comprehensive understanding of its pathogenesis, then a comprehensive classification of recently developed hydrogels will be presented, encompassing properties such as hypoglycemic effects, anti-inflammatory capabilities, antibacterial attributes, ROS scavenging abilities, promotion of angiogenesis, pH responsiveness, and more. The primary objective is to offer a valuable reference for repairing diabetic wounds based on their unique microenvironment. Moreover, this paper outlines potential avenues for future advancements in hydrogel dressings to facilitate and expedite the healing process of diabetic wounds.
Introduction
Diabetes mellitus (DM) denotes a cluster of sophisticated metabolic disorders delineated by persistent elevation of blood glucose levels caused by the impaired pancreatic β-cell function and diminished responsiveness to insulin, known as insulin resistance (IR).1,2 According to the latest data, diabetes mellitus impacts an estimated 529 million individuals worldwide in 2021 with a prevalence rate of 6.1%. It also is projected to a notable escalation, estimating over 1.31 billion individuals affliction by the year 2050, 3 therefore lead to a substantial global health threat, presenting escalating challenges and imposing an immense burden on both patients and public healthcare systems.4,5 Moreover, persistent hyperglycemia significantly harms numerous organs and triggers consequential complications associated with diabetes, including nephropathy, diabetic ulcers or wounds, cardiovascular issues, peripheral neuropathy, and retinopathy.6,7 Among the diabetics, approximately 19%–34% patients are influenced by diabetic wounds or ulcers, 8 which places a significant financial burden on global medical expenditure and utilizes substantial medical resources. Furthermore, in the presence of hyperglycemia, peripheral blood vessel damage, immunosuppression, and motor neuropathy contribute to the gradual deterioration of diabetic ulcers or wounds, leading to tissue necrosis and bacterial infection. Consequently, this sequence of events can culminate in deformity and tissue atrophy, potentially necessitating amputation. Therefore, it is imperative to comprehensively understand the complicated pathogenesis and seek effective strategies for managing diabetic wounds.
Skin wound healing represents a finely orchestrated physiological process that occurs in response to injuries. 9 Throughout the overlapping phases of wound healing—encompassing hemostasis, inflammation, proliferation, and remodeling—complex and interrelated mechanisms orchestrate entangled interactions. These mechanisms involve a sophisticated network of complementary actions, operating through multifaceted hierarchical frameworks and the microenvironment, where multiple dynamic cytokines, various cell types (particularly macrophages), and intricate signaling pathways play pivotal roles. The preservation of homeostasis within the wound microenvironment significantly influences the progression of these interlacing stages of healing. Disruption or alteration of any stage may induce changes in the microenvironment, subsequently impacting the healing process. Typically, the resolution of most skin lesions may take approximately 1–2 weeks. In delayed healing wounds, persistent hyperglycemia leads to a diverse range of localized pathological alterations evident within the wound microenvironment, characterized by abnormal inflammation, 10 poor angiogenesis, 11 varied cell death and signaling pathways,12–14 advanced glycation end products (AGEs),15,16 ROS, 17 oxidative stress, 18 peripheral neuropathy, 19 impaired neuropeptide,20,21 variation of pH, dysfunction of macrophage polarization, 22 and high protease activity. 23 Simultaneously, additional bacterial infection exacerbates the already delayed healing process and complicates the microenvironment. Considering the complex pathogenesis of diabetic wounds, current clinical management strategies involve a spectrum of approaches encompassing debridement, autologous skin grafting, off-loading techniques, negative pressure wound therapy (NPWT), administering antibiotics, applying wound dressings, utilizing topical medications and oxygen therapy, employing shock wave therapy, intervening with growth factor delivery, and in severe cases, resorting to amputation. Unfortunately, prolonged and repeated debridement procedures lead to severe pain and considerable economic burdens for afflicted patients. Furthermore, conventional dressings, while serving a singular function, necessitate frequent replacements, thereby extending hospital stays or mandating repeated clinic visits for dressing changes. Additionally, the emergence of multi-drug resistant (MDR) bacteria can undermine the efficacy of antibiotic treatments. Hence, it is imperative to explore cost-effective and efficient clinical interventions capable of accommodating the dynamic healing process of diabetic wounds and augmenting therapeutic outcomes.
In recent years, considerable progress in materials engineering, polymer science, biomedical science, and chemistry has fostered the development and continuous enhancement of multifunctional biomaterials. Clinicians eagerly anticipate these advancements, hoping they can effectively tackle clinical challenges, particularly chronic and refractory diabetic wound healing. To confront these challenges, functionalized biomaterials such as hydrogels, foams, hydrocolloids, nanofibers, sponges, and semipermeable membranes have been engineered to expedite wound healing and mitigate infections. Among these biomaterials, hydrogels stand out as promising wound dressings for diabetic patients owing to their outstanding biocompatibility, favorable swelling capacity, remarkable permeability, complex 3D network structure, and excellent drug-loading capabilities. Crucially, hydrogels exhibit a spectrum of properties encompassing antibacterial, anti-inflammatory, antioxidant attributes, controlled drug release mechanisms, and responsiveness to varying stage-specific microenvironments, thereby enhancing the healing of diabetic wounds.
In this comprehensive discussion, first we delve extensively into the multifaceted microenvironment of diabetic wounds and explore the diverse factors influencing the trajectory of healing. Then the therapeutic potential of hydrogels in fostering the healing of diabetic wounds is underscored and recent advancements in hydrogel-based wound dressings is detailed meticulously specifically to target the distinctive characteristics of the microenvironment associated with diabetic wounds (Figure 1).
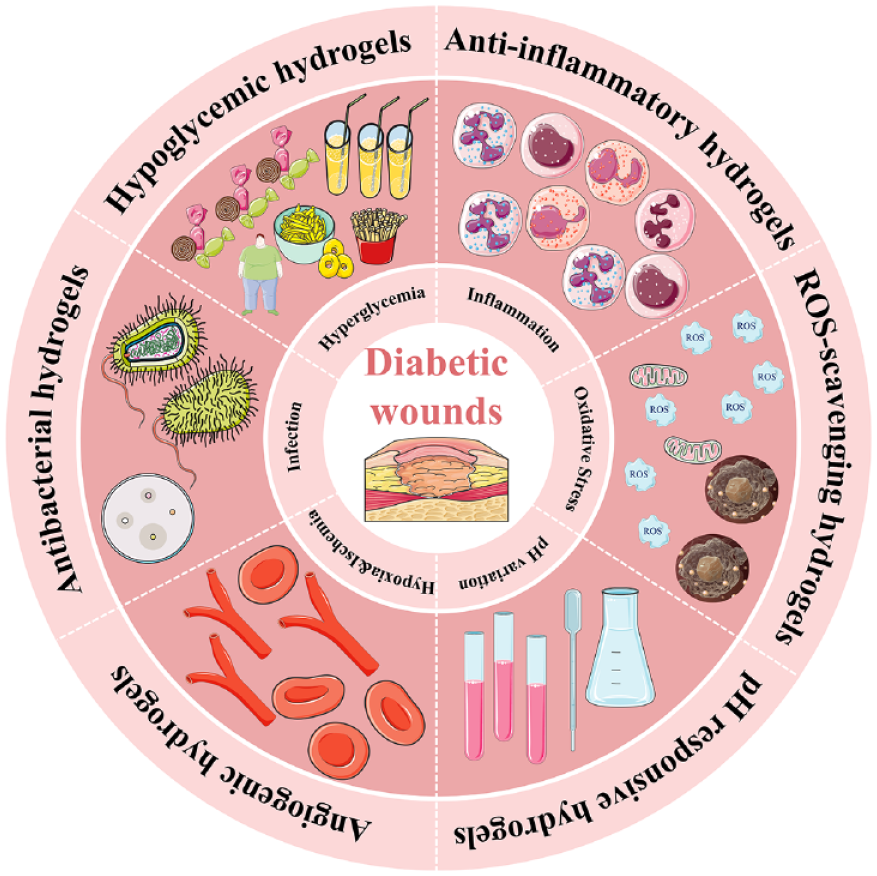
Illustration of recent hydrogels in accelerating diabetic wound healing.
The wound healing process
The skin, serving as a multi-layered external defense system of the human body, plays a pivotal role in preventing pathogenic infections, initiating vitamin D synthesis, facilitating excretion, and regulating body temperature. 24 However, being the largest organ on the body’s surface, the skin is prone to external damage, leading individuals to inevitably encounter cutaneous injuries at various points in their lives.
The healing process for most wounds typically involves reparative healing, which unfolds through a series of coordinated and overlapping stages within a complex biological framework. These stages can be simplified into four primary phases: hemostasis, inflammation, proliferation, and remodeling. These stages work in concert to restore the structural integrity of the skin, thereby facilitating the expected healing outcome. Following cutaneous injury, the hemostasis stage promptly commences, involving the rapid contraction of damaged blood vessels to facilitate the formation of blood clots, thereby preventing hemorrhage. This stage can be broken down into vasoconstriction, primary hemostasis, and secondary hemostasis for simplified understanding. 25 Fibrin, fibronectin, vitronectin, and thrombospondin collaborate to create an insoluble clot. This clot not only halts bleeding but also serves as a provisional scaffold. This scaffold facilitates the infiltration of immune cells and acts as a reservoir for growth factors (including platelet-derived growth factor (PDGF), epidermal growth factor (EGF), and vascular endothelial growth factor (VEGF)), as well as cytokines, for instance, platelet-derived chemokine (C-X-C motif) ligand 4 (CXCL4), in the subsequent phases of the healing process.26,27 The initiation of the inflammatory phase is distinguished by the arrival of inflammatory cells, which is instigated during hemostasis and corresponds to injury signals such as pathogen-associated molecular patterns (PAMPs) and danger-associated molecular patterns (DAMPs). Neutrophils, as the initial circulating immune cells, are drawn into the wound bed due to chemokines like interleukin 1 (IL-1), tumor necrosis factor-alpha (TNF-α), and lipopolysaccharide (LPS). They are recruited from damaged vessels and primarily dominate during the early stages of the inflammatory phase. 28 Neutrophils play critical roles in eliminating invasive pathogens and necrotic tissue by responding to the activation of inflammatory signaling pathways. They produce various bioactive substances, including ROS, matrix metalloproteinases (MMPs), antimicrobial peptides, and serine proteases. Upon activation, neutrophils degrade virulence factors, capture bacteria, and eliminate pathogens through the formation of prominent extracellular structures known as neutrophil extracellular traps (NETs). 29 Ultimately, neutrophils undergo apoptosis or necrosis and are subsequently cleared by macrophages through processes such as efferocytosis or re-entry into the circulatory system, marking the resolution of inflammation. Macrophages, characterized by their high heterogeneity and plasticity, play indispensable roles in wound healing dynamics. Their pivotal contribution lies in the extraordinary capacity for macrophage polarization, allowing them to modulate their phenotype in accordance with spatial and temporal signals encountered at the injury site. 30 Embryonically, skin macrophages originate from the yolk sac and fetal liver, while postnatally, they stem from the bone marrow. Following differentiation, they give rise to resident macrophages and perform hematopoietic functions. The skin’s resident macrophage population comprises various subtypes, including vessel-associated macrophages marked by Lyve1highMHCIIlow expression (indicative of lymphatic endothelium hyaluronan receptor-1 and major histocompatibility complex class II), sensory nerve-associated macrophages characterized by Lyve1lowMHCIIhigh expression, as well as dendritic cells and Langerhans cells.31,32 Upon tissue damage, a restricted subset of resident macrophages rapidly detects signals of injury and instigates an inflammatory response. 33 Simultaneously, circulating blood monocytes differentiate into macrophages, collaborating with tissue-resident macrophages to facilitate the restoration of tissue integrity. 34 The process of phagocytosing senescent neutrophils by macrophages, combined with microenvironmental cues, triggers a shift in macrophage polarization from pro-inflammatory M1 macrophages to anti-inflammatory M2 macrophages. This transition sets the stage for the proliferation phase of wound healing, characterized by substantial activation of keratinocytes, fibroblasts, macrophages, and endothelial cells. Their concerted efforts aim to orchestrate wound closure, deposit new matrix, and stimulate angiogenesis. Subsequently, the remodeling stage ensues, focusing on the restoration of tissue structure and the enhancement of tensile strength. This phase can persist for 2 years or longer as it gradually refines the healed tissue to achieve optimal functionality and resilience.
The complex microenvironment of diabetic wounds and the customized hydrogel dressings
The concept of the wound microenvironment was first introduced by Carla R. Kruse and colleagues in 2015. This theory encompasses both the external and internal components of the wound microenvironment, providing a comprehensive understanding of the factors influencing the healing process. 35 The wound healing process in diabetic wound deviates from the conventional steps and fails to generate the appropriate anatomical structure or restore physiological function due to their intricate microenvironment which remains in persistent chronic inflammation, recurrent infections, and vascular impairment associated with hyperglycemia, thus the healing period generally sustain more than a month clinically. Nowadays, hydrogels have demonstrated significant potential in the field of wound healing, especially in the context of diabetic wounds. Their porous structure, high water absorption, and biocompatibility make them excellent candidates for wound dressings. By being classified into natural and synthetic categories, hydrogels offer diverse options for biomedical applications. In the case of diabetic wounds, where the microenvironment poses unique challenges, hydrogels present an array of properties that can be customized to address these specific concerns. The ability to regulate hyperglycemia, monitor wound status, respond to pH changes, combat bacteria, reduce ROS levels, and promote angiogenesis can significantly contribute to improving the healing process in diabetic wounds. Herein, we are committed to meticulously classify and succinctly summarize the alterations occurring in the microenvironment of diabetic wounds, as well as recent advancements in hydrogel-based wound dressings, aiming to provide a comprehensive understanding of diabetic wounds’ pathogenesis, with a specific focus on the application and customization of hydrogels to better suit their complex microenvironment (detailed in Table 1). It will also cover the modification techniques, incorporation of bioactive agents, and innovative strategies implemented in hydrogel formulations to cater to the specific needs of diabetic wound healing.
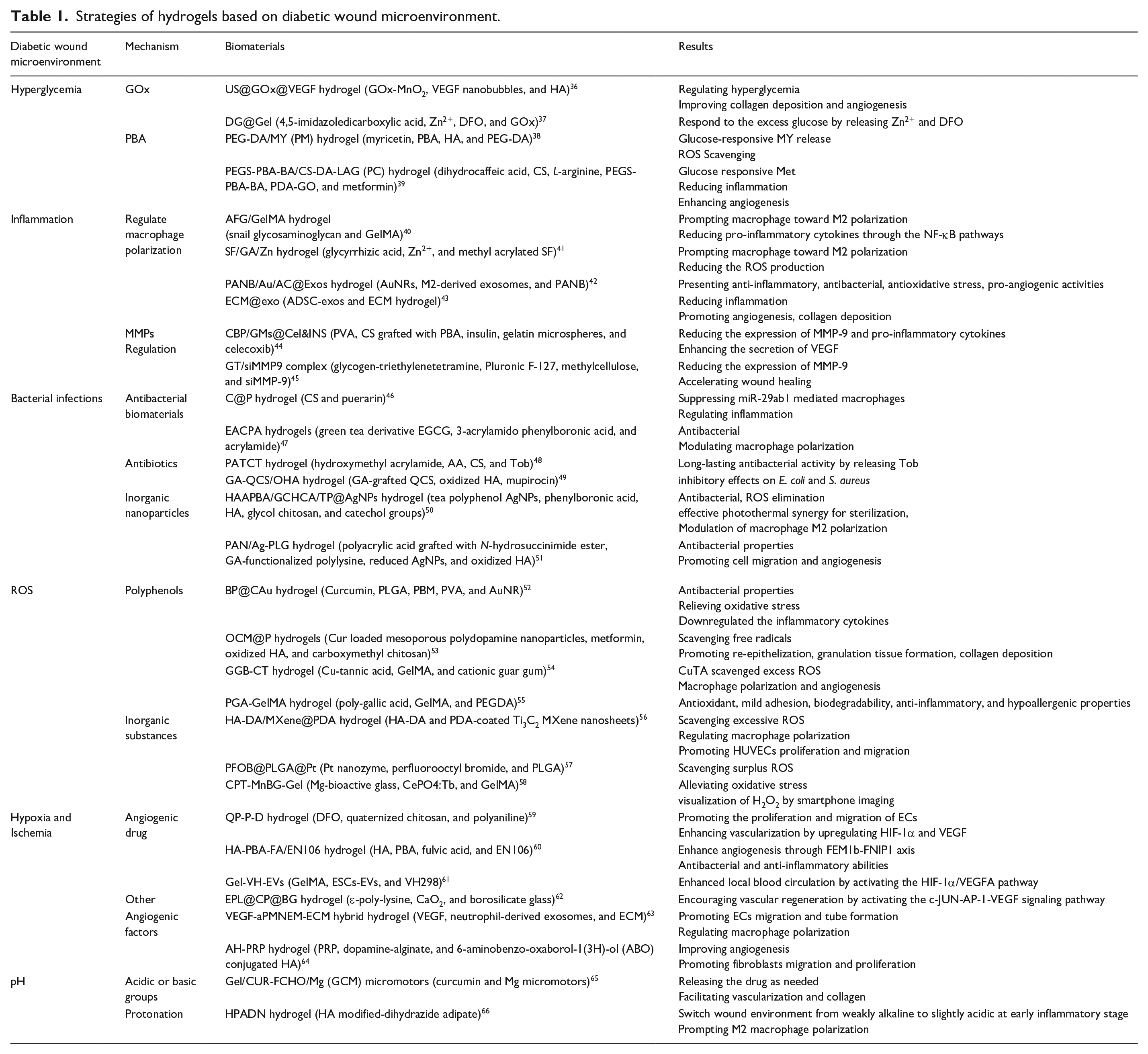
Strategies of hydrogels based on diabetic wound microenvironment.
Hyperglycemia and hyperglycemic hydrogels
Diabetes imposes constraints on the body’s ability to maintain normal physiological blood glucose levels due to IR or dysfunction of β-cells. Factors such as glucolipotoxicity and inflammation associated with obesity culminate in persistently elevated blood glucose levels, leading to a phenomenon known as hyperglycemia or even creating a hyperglycemic memory under certain circumstances. Simultaneously, insulin resistance exacerbates hyperglycemia by increasing glucose production while reducing glucose uptake in the liver, muscles, and adipose tissue. Additionally, hyperglycemia contributes to the dysfunction of β-cells by triggering endoplasmic reticulum stress, a process regulated by both apoptosis and the unfolded protein response (UPR). 67 The persistent hyperglycemic microenvironment disrupts the normal functioning of keratinocytes, thereby hindering the tissue repair process in diabetic wounds. Huang et al. 68 have suggested that the hyperglycemic environment diminishes the expression of Galectin-7 (Gal-7), a molecule crucial for keratinocyte migration. This reduction in Gal-7 levels leads to delayed wound healing in individuals with diabetes. The hyperglycemic environment has been observed to decrease the expression of human β-defensin-2 (hBD2) in keratinocytes. This reduction in hBD2 levels can result in insufficient migration of keratinocytes and impaired neovascular development due to the downregulation of the STAT-1 signaling pathway. Ultimately, these effects compromise the healing process of diabetic wounds. 69 Furthermore, the hyperglycemic microenvironment has been shown to cause increased neutrophil infiltration, thus impeding the wound healing process in individuals with diabetes. This effect is mediated through the epidermal growth factor receptor (EGFR)–extracellular regulated kinase (ERK) signaling pathway. Prolonged hyperglycemia induces non-enzymatic glycation reactions involving proteins, lipids, and nucleic acids, leading to the formation of AGEs. These AGEs are believed to contribute to dysfunctional cellular microenvironments triggered by hyperglycemia, presenting a significant obstacle to the healing of diabetic wounds.70–72 The interaction between AGEs and their principal cellular receptor, RAGE (receptor for AGEs), which belongs to the immunoglobulin superfamily, initiates numerous signaling pathways. This interaction triggers elevated oxidative stress and inflammatory response. Consequently, these cascading events ultimately lead to compromised insulin signaling, 73 disruption of metabolic homeostasis, 74 epigenetic modifications, 75 and RAGE-induced toxicity in pancreatic β cells. 76 Furthermore, hyperglycemia microenvironment promotes progressive accumulation of AGEs and ROS, 77 thereby exacerbating oxidative stress and facilitating activation of inflammatory responses via AGE-specific receptors and inhibiting Ca2+ mobilization.78,79 Continual oxidative stress and the accumulation of excessive AGEs can impair the function of two receptors: AGE-R1 and Sirtuin1 (SIRT-1). These receptors not only serve crucial roles in identifying and counteracting AGEs but have also been verified to collaboratively regulate adipocyte insulin signaling in vitro. 80 Meanwhile, investigations have revealed that endothelial cells demonstrate compromised structural integrity and increased vulnerability to apoptosis in an in vitro high-sugar environment. This condition leads to cell shedding and their subsequent entry into the bloodstream, thereby contributing to complications associated with angiogenesis. 81 This also makes hyperglycemic environment become one of the reasons that hinder the healing of diabetic wounds. Therefore, effective management of both systemic and wound-specific blood glucose levels is advantageous in improving the microenvironment of diabetic wounds and facilitating the process of wound healing.
Indeed, the management of hyperglycemia in individuals with diabetic wounds poses significant challenges, especially when it comes to achieving optimal blood glucose control. The use of insulin and oral hypoglycemic drugs is a standard approach in clinical settings to regulate blood glucose levels for diabetic wounds, such as foot ulcers. However, patient compliance and adherence to these treatments can often be suboptimal, leading to difficulties in maintaining consistent blood glucose levels. In this context, local management of blood glucose levels within the wound microenvironment has garnered attention. Hydrogels engineered to mitigate glucose levels within wounds present a promising avenue for facilitating diabetic wound healing. Among these, glucose-responsive hydrogels stand out due to their ability to act as self-monitoring systems for individuals with diabetes. Glucose-responsive hydrogels are equipped with glucose sensors capable of detecting glucose levels and fluctuations. These sensors, such as glucose oxidase (GOx), concanavalin A (Con A), and phenylboronic acid (PBA), play a critical role in detecting changes in glucose concentration. This information is then used to trigger specific responses within the hydrogel, enabling the release of insulin or other therapeutic agents to manage local glucose levels effectively which offers the potential for more targeted and precise management of glucose levels within the wound, thereby promoting an optimal healing environment for diabetic wounds. 82
The integration of GOx within tissue engineering has proven to be an effective catalyst in controlling glucose levels. GOx facilitates the conversion of glucose to hydrogen peroxide (H2O2) and glucuronide, exhibiting hypoglycemic efficacy. Additionally, this conversion contributes to the decrease in pH levels and demonstrates antibacterial properties. For instance, a novel hydrogel formulation, utilizing multiple components including 3′,5,5′-tetramethylbenzidine (TMB), Fe2+, Pluronic F127 (PF127), and GOx, has been developed to promote the healing of diabetic wounds (Figure 2(a)). The included GOx enzymatically reduces blood glucose levels, generating H2O2 and gluconic acid. Meanwhile, these components support the Fe2+-based Fenton reaction, leading to the production of hydroxyl radicals, which possess bactericidal effects, combining with the ability to regulate glucose levels, this specific platform presents promising prospects for diabetic wound healing applications.
83
However, GOx-based hydrogels face challenges such as enzyme degeneration, limited oxygen supply for catalysis, and insufficient insulin loading capacity which necessitate the modifications to the carrier and drug delivery systems. Furthermore, the pH alterations in the wound bed due to the application of GOx-based hydrogels need to be monitored regularly, and preemptive measures should be taken to mitigate any potential adverse effects caused by pH changes. Another approach involves Con A, a naturally derived protein known for its glucose selectivity. Yin et al.
84
developed microgels combining glycidyl methacrylate modified dextran (DexG) and Con A (diameter of 5.126 ± 1.546 μm, Figure 2(b)). The loaded insulin resulted in sustained and consistent insulin release in response to varying glucose concentrations, ensuring the maintained bioactivity of the released insulin. However, Con A based glucose response biomaterials, are almost employed to experiments in vitro, rather than diabetic wound model.85–87 Further investigation is warranted to ascertain the efficacy of its potential in facilitating the healing process of diabetic wounds. Both GOx and Con A are naturally sourced proteins that not only exhibit exceptional selectivity toward glucose, but also exist adverse immunogenicity and protein stability. Compared with them, PBA derivatives make the chemical modification for achieving optimal glucose sensitivity more easily, hence exhibit immense potential in various applications on glucose-responsive hydrogels. PBA exhibits reversible binding to 1,2/1,3-
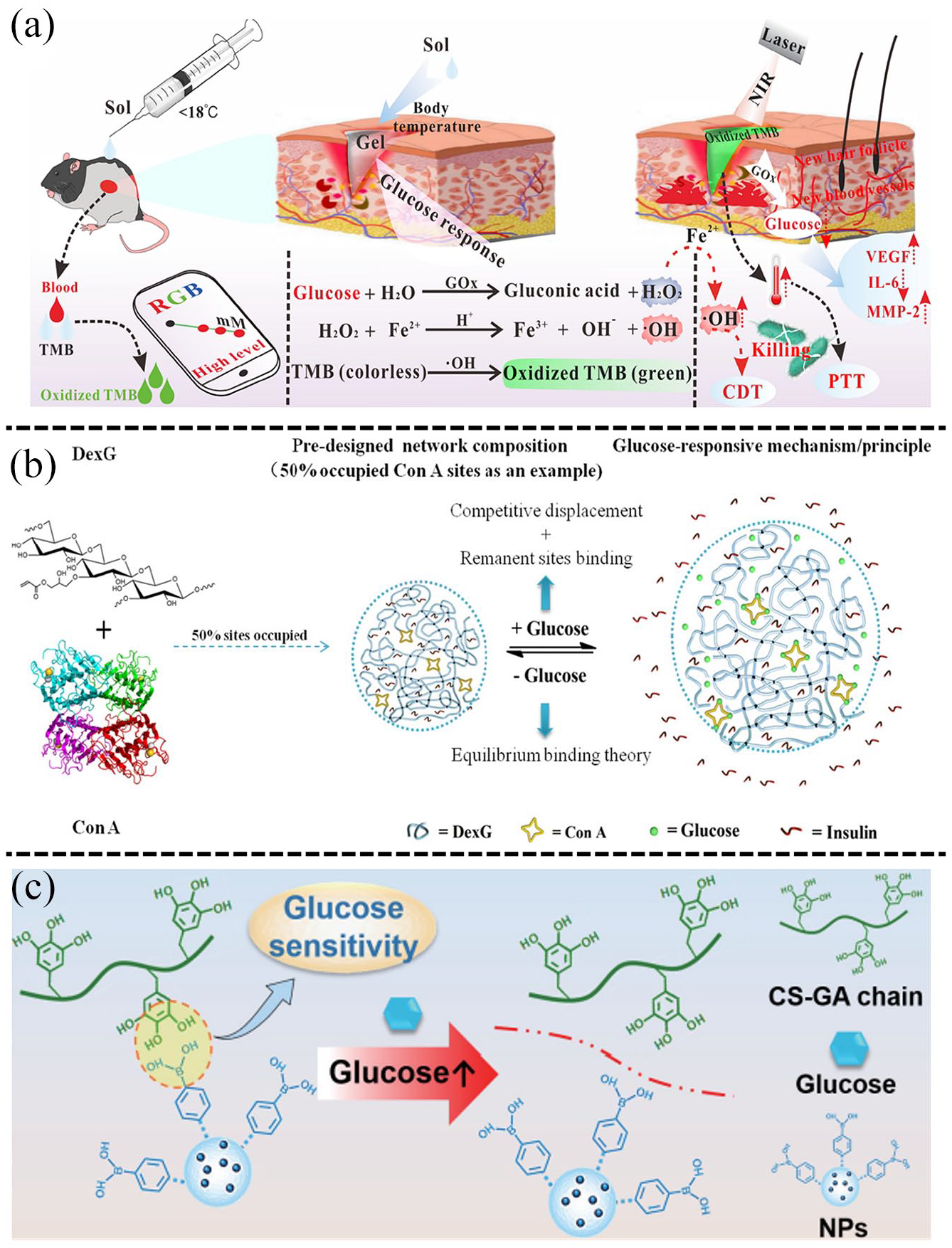
The glucose-responsive hypoglycemic hydrogels: (a) the glucose-responsive TMB/Fe2+/PF127 hydrogel based on GOx and its application in promoting diabetic wound healing. Reproduced with permission. 83 Copyright 2023, American Chemical Society, (b) con A-DexG based system and its glucose-responsive and hypoglycemic effect mechanism in vitro. Reproduced with permission. 84 Copyright 2018, Elsevier B.V, and (c) the glucose-responsive ODEX-DA/HTCC hydrogels based on PBA for releasing insulin NPs to exert hypoglycemic effect. Reproduced with permission. 88 Copyright 2023, Springer Nature.
Chronic inflammation and anti-inflammatory hydrogels
Prolonged inflammation represents a notable hallmark of the healing process in diabetic wounds, intricately linked with various pathological factors, particularly hyperglycemia as previously discussed. In contrast to acute wounds, the initial phase of diabetic wounds displays a restrained inflammatory response, which then transitions into the chronic inflammatory state which absolutely impedes repair progression. Macrophage plasticity plays a crucial role in transitioning from the inflammatory to the proliferative phase of tissue repair. This flexibility allows macrophages to perform various functions in response to the dynamic wound microenvironment, ensuring efficient healing. Diabetic wounds commonly manifest an intensified inflammatory response, contributing to chronic inflammation that impedes the macrophage shift from the pro-inflammatory M1 to the anti-inflammatory M2 phenotype. This imbalance results in an increased M1/M2 ratio, observed consistently in both diabetic patients and animal models, and subsequently prolongs the shift from the inflammatory phase to tissue proliferation during wound healing. 90 Moreover, the hyperglycemic environment in diabetic wounds leads to increased levels of ROS which hinder chemokine expression and impair macrophage stimulation to produce pro-inflammatory cytokines like TNF-α, IL-1β, and IL-6. This exacerbates a detrimental cycle, perpetuating sustained M1 macrophage polarization and chronic inflammation, further impeding the healing process. 91 Studies in diabetic mice have indicated that the activation of the cGAS-STING pathway prompts the polarization of macrophages toward the pro-inflammatory M1 phenotype. This polarization leads to the release of pro-inflammatory cytokines, exacerbates endothelial cell dysfunction, and consequently hampers the healing of diabetic wounds. Additionally, compelling evidence suggests that individuals with diabetes exhibit an increased expression of Nod-like receptor protein 3 (NLRP3) inflammasome in macrophages. This heightened expression not only sustains chronic inflammation and oxidative stress but also triggers excessive infiltration of innate immune cells into diabetic wounds which adversely affects the proliferation and migration of keratinocytes.92–94 Therefore, controlling macrophage polarization represents a critical approach in diabetic wound recovery.
Recently, scientists have focused on developing hydrogels to regulate macrophage polarization, targeting the inflammatory response linked to diabetic wounds to accelerate recovery. Tu et al.
95
developed the GDFE hydrogel to modulate immune cells and facilitate the healing of diabetic wounds (Figure 3(a)). The hydrogel was created by integrating polydopamine-modified graphene oxide (GD) and F127-EPL (FE) into a F127 solution, using hydrophilic-hydrophobic interactions along with hydrogen bonding. Results showed that the GDFE hydrogel effectively promoted macrophage polarization toward the M2 phenotype, reducing inflammation during the later stages of diabetic wound healing. Additionally, the hydrogel demonstrated thermal responsiveness, self-healing capabilities, and increased antibacterial and antioxidant activity, presenting a promising approach for diabetic wound treatment. Xu et al.
96
utilized GelMA along with bletilla striata polysaccharide (BSP) and the tannic acid/ferric iron complex (TA/Fe3+) to create the GBTF hydrogel. Their research demonstrated that the TA/Fe3+ complex within the GBTF hydrogel effectively triggered the polarization of anti-inflammatory M2 macrophages, promoting the healing of diabetic wounds and tissue regeneration via modulation of the PI3K/Akt1/mTOR signaling pathway (Figure 3(c)). Additionally, the GBTF hydrogel exhibited an enhanced antibacterial effect, with a notable antibacterial rate of over 90% against
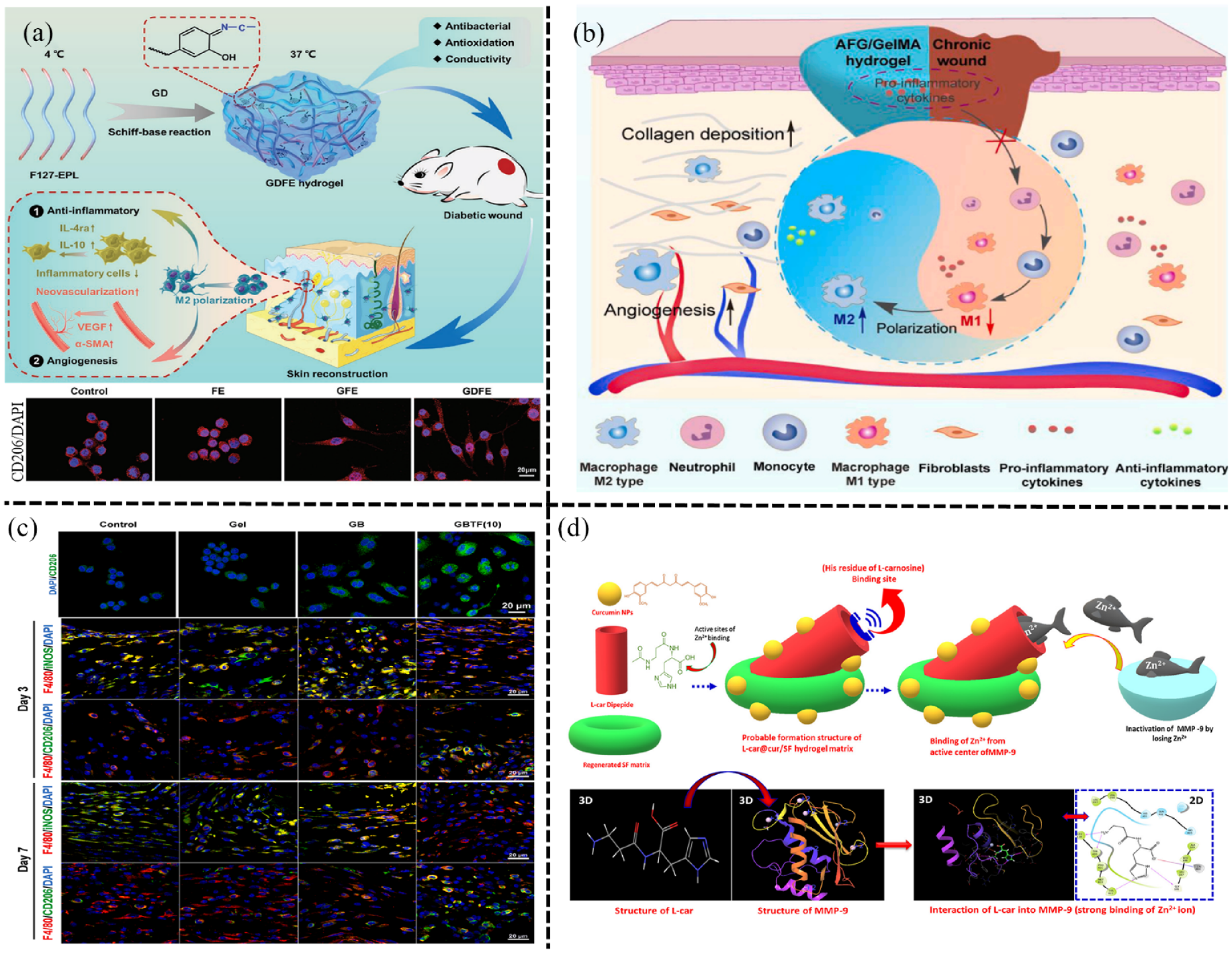
The anti-inflammatory hydrogels based on modulation of macrophage polarization and decreasing the expression of MMP-9 in diabetic wound: (a) the GDFE hydrogel could effectively activate macrophages polarization to M2 phenotype, and remarkably accelerated wound healing. Reproduced with permission.
95
Copyright 2021, Wiley-VCH GmbH, (b) the AFG/GelMA hydrogel by natural adhesive from snail mucus improved the healing of diabetic wound via promoting macrophage polarization to M2 phenotype. Reproduced with permission.
40
Copyright 2023, Elsevier Ltd, (c) evaluation of the GBTF hydrogel on macrophage polarization in diabetic wound. Reproduced with permission.
96
Copyright 2023, Elsevier B.V, and (d) the
Persistent chronic inflammation can induce the upregulation of matrix metalloproteinases (MMPs), which constitute a family of zinc endopeptidases capable of degrading components within ECM. This upregulation influences the diabetic wound healing process by participating in various biological processes relevant to ECM secretion, granulation tissue formation, angiogenesis, collagen synthesis, re-epithelialization, and the inflammatory response. For instance, MMP-1 aids in wound re-epithelialization, while MMP-2 is significant during the process of angiogenesis and the extended remodeling of the matrix. Additionally, MMP-3 plays a crucial role in normal wound contraction and the remodeling of the basement membrane. These distinct functions highlight the multifaceted involvement of MMPs in various aspects of the wound healing process. Among these MMPs, MMP-9 plays a pivotal role in the diabetic wound healing process. In 2009, Liu et al.
102
initially observed elevated levels of MMP-9 in wound fluid, marking the first identification of this phenomenon. Subsequent comprehensive and rigorous research has demonstrated the association between the dysregulation of MMP-9 and various axis, including Notch1/NF-κB/MMP-9,
103
Nrf-2/MMP-9,
104
and MMP-9/FasL/Fas.
105
These findings serve to further validate the detrimental impact of MMP-9 on impeding the healing process of diabetic wounds, underscoring its significance in contributing to this inflammatory state. Additionally, researchers have identified that (
Infection and antibacterial hydrogels
Diabetic wounds exhibit heightened susceptibility to bacterial infections and the formation of biofilms whihc impedes the natural healing process and contributes to recurring along with prolonged local inflammation leading to severe outcomes such as osteomyelitis, the necessity for amputation, and even sepsis. Additionally, the hyperglycemic microenvironment characteristic of diabetic wounds exacerbates the challenge of wound healing. In most instances, diabetic wounds are commonly contaminated by Gram-positive bacteria, including species such as
Consequently, current biomedical research predominantly emphasizes the development of hydrogels with antibacterial properties for managing diabetic wounds. These antibacterial hydrogels encompass various types, including those based on antibacterial biomaterials, antibiotics loading, inorganic nanoparticles integration, and those with synergistic effects in combating bacteria. Hydrogels that incorporate antibacterial biomaterials such as polysaccharides, polyphenols, and antibacterial peptides (AMPs) utilize direct interactions to bacteria for inherent antibacterial effects. Chitosan (CS), derived from chitin, is a polysaccharide known for its inherent antibacterial properties by disrupting bacterial cell membranes or biomolecules, causing a disruption in membrane permeability and metabolic processes. CS-based hydrogels are promising for improving the healing of vulnerable diabetic wounds. For instance, Lin et al.
116
developed a CS/hyaluronic acid (HA) hydrogel to aid in healing
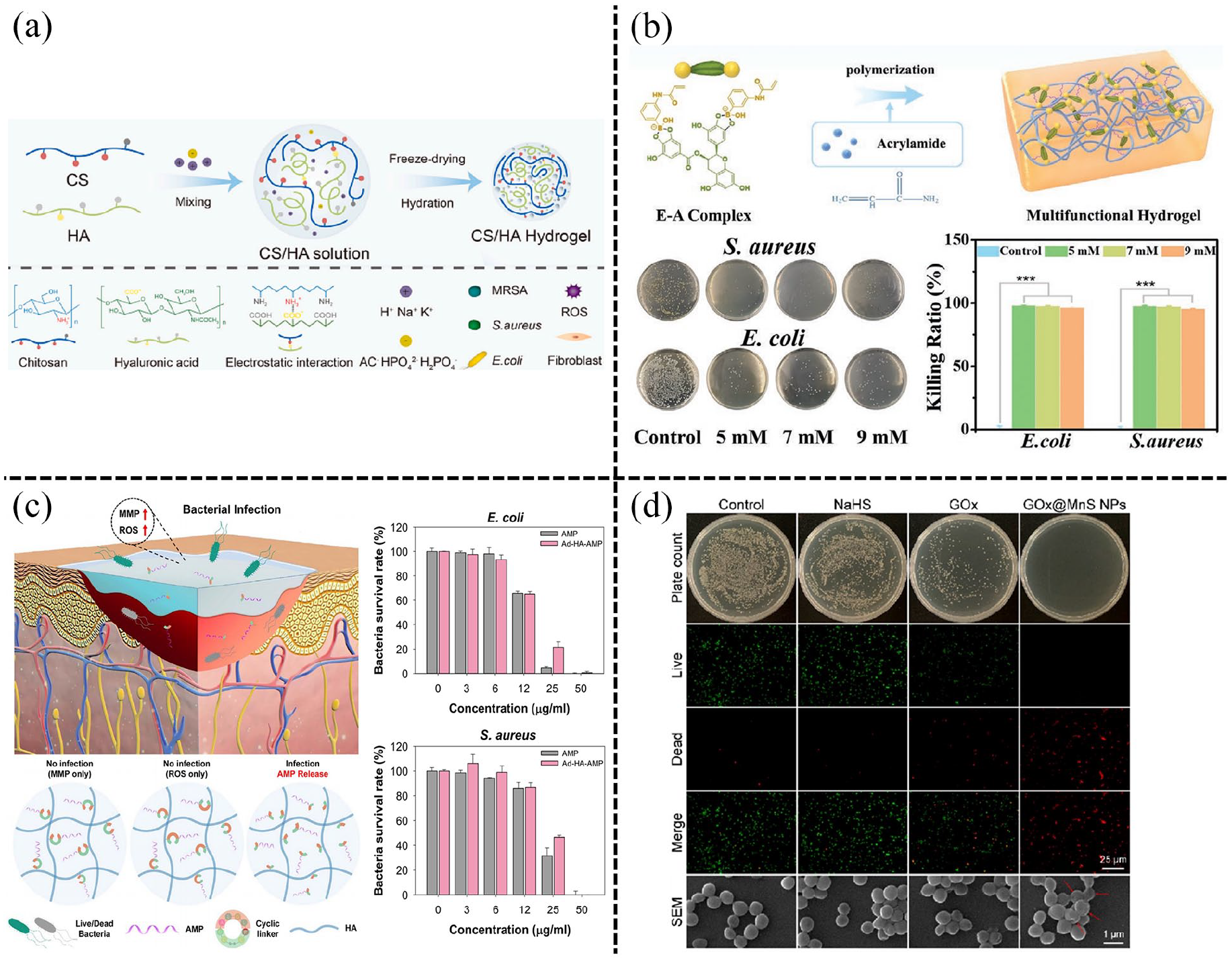
The antibacterial hydrogels in diabetic wounds: (a) the CS/HA hybrid hydrogels. Reproduced with permission.
116
Copyright 2023, Elsevier Ltd, (b) the bacteria-killing efficiency of EACPA hydrogel against
In current clinical practices, antibiotics play a central role in managing infections within diabetic wounds. However, their prolonged use can lead to antibiotic resistance and disturb the natural bacterial balance. Hydrogels loaded with antibiotics aim to locally address inflammation and infection promptly, speeding up wound healing and reducing the need for systemic antibiotic administration. Zhang et al.
120
developed the OCMC-Tob/PEI hydrogel, a positively charged hydrogel with injectable, self-healing, and biocompatible properties. This hydrogel was created by linking tobramycin (Tob) and polyethylenimine (PEI) to oxidized carboxymethyl cellulose (OCMC) backbone through Schiff base reaction. Through its “Pull-Push” strategy, this hydrogel effectively improved the healing of
Oxidative stress and ROS-scavenging hydrogels
ROS play a pivotal role in governing various stages of the wound healing process. Maintaining a low level of ROS is crucial for mounting a robust defense against external damage. However, excessive ROS production can lead to oxidative stress, stemming from an imbalance between heightened ROS levels and inadequate antioxidant defense mechanisms. This oxidative stress impedes the healing process of diabetic wounds by inducing lipid peroxidation, DNA damage, and mitochondrial dysfunction. Moreover, oxidative stress triggers the upregulation of specific biochemical pathways, including the formation of AGEs, increased expression of the receptor for AGEs, elevated polyol flux, activation of protein kinase C (PKC), and augmentation of the hexosamine pathway. These upregulated pathways further contribute to the complexities and challenges encountered during the healing of diabetic wounds. 123 The escalation of oxidative stress during the diabetic wound healing process is primarily influenced by elevated glucose levels, the presence of AGEs, and hypoxic conditions. Lan et al. 124 discovered that hyperglycemia induced heightened levels of ROS by activating the EGFR–ERK signaling pathway. This elevated ROS production subsequently led to increased levels of IL-8, impairing wound healing in both diabetic rats and human keratinocytes. Hyperglycemia triggers the disruption of the mitochondrial membrane, resulting in a decline in mitochondrial membrane potential. This disturbance leads to excess of mitochondrial ROS (mtROS) due to mitochondrial respiratory leakage observed in diabetic wounds.125,126 In diabetes mellitus, there is an observed increase in mitochondrial fission and a compromised autophagy process. These alterations result in the disruption of mitochondrial networks, the accumulation of small dysfunctional mitochondria, and the augmented production of mtROS. 127 Mitochondrial fission, also known as the process of dividing mitochondria, is typically regulated by proteins such as mitofusins (Mfn), optic atrophy 1 (Opa1), and dynamin-related protein 1 (Drp1). Shi et al. 128 discovered that downregulation of Drp1 facilitated angiogenesis and suppressed the generation of mtROS in diabetic wounds. Indeed, mitigating the excessive generation of ROS and mtROS is crucial for intervening in the diabetic wound microenvironment.
To counteract this, ongoing research focuses on ROS-scavenging hydrogels aimed at efficiently eliminating ROS, regulating the wound microenvironment, curbing inflammation, and fostering wound healing. These hydrogels often utilize ROS-scavenging agents and adopt various strategies, including organic-based and inorganic-based substances, to manage ROS levels effectively in diabetic wounds. 129 The organic functional groups like phenols, sulfur, and boronic acid effectively counteract ROS through mechanisms involving electron transfer or proton donation. Polyphenols like curcumin, tannin acid (TA), and gallic acid (GA) are commonly utilized in ROS-scavenging hydrogels, while sulfur or boronic acid groups are integrated into ROS-responsive hydrogels, as their respective bonds can be cleaved by oxidants following oxidation. Curcumin, a naturally occurring polyphenol characterized by its low molecular weight, exhibits antioxidant, anti-inflammatory, and antibacterial properties. Hu et al. 130 engineered a hydrogel based on hyaluronic acid (HA) and chitosan (CS) to deliver nanotechnologically-modified curcumin (CNP) and epidermal growth factor (EGF). In this formulation, curcumin was swiftly and continuously released, effectively mitigating inflammation and oxidative stress during the initial stages of wound healing, ultimately accelerating the process of diabetic wound recovery. Shi et al. 131 developed an innovative ROS-responsive injectable hydrogel called HA@Cur@Ag through Michael addition chemistry which involved modifying poly (ethylene glycol) diacrylate (HB-PEGDA) to create hyperbranched poly (β-hydrazide esters; HB-PBHEs) for ROS-responsive antioxidant properties (Figure 5(a)). By encapsulating curcumin within liposomes, the sustained presence of curcumin in the wound bed was ensured. Additionally, both liposomes and AgNPs exhibited responsiveness to ROS, enabling sustained release and thereby effectively delivering antioxidant and antibacterial effects, ultimately promoting the healing of diabetic wounds. Tannic acid (TA), a natural polyphenolic compound, possesses notable capabilities in scavenging free radicals and exhibiting antioxidant effects. It possesses the ability to create complexes with various substances like polysaccharides, proteins, alkaloids, and metal ions. Researchers integrated TA into CS-grafted-dihydrocaffeic acid (CS-DA), benzaldehyde-terminated PF127 (PF127-CHO), and 3D adipose-derived mesenchymal stem cells-derived exosomes to produce the dynamic CS-DA/PF/TA/3D-Exo hydrogel. This hydrogel demonstrated impressive antioxidant properties by effectively scavenging ROS, thereby promoting the healing of diabetic wounds. 132 Gallic acid (GA), found in various plant sources, possesses the phenolic hydroxyl group with significant ROS elimination capabilities and the ability to interrupt the cycle of new free radical generation. This property makes GA an effective antioxidant by impeding the oxidation process of enzymes involved in lipid, DNA, and protein synthesis, consequently reducing free radical formation. Wang et al. 133 utilized GA to produce nanoparticles with catalase-like properties, which were then encapsulated within a thermosensitive hydrogel (Figure 5(b)). The controlled release of GA from the outer layer of these nanoparticles demonstrated anti-inflammatory and ROS-scavenging properties facilitated effective healing of diabetic wounds. Protocatechuic aldehyde (PA) is a natural compound sourced from Salvia miltiorrhiza, known for its potent ROS scavenging capabilities. Its unique phenolic aldehyde structure distinguishes it from other natural polyphenols possessed the phenol structure like TA and GA. Incorporating PA into hydrogels own exceptional antioxidant properties, making it a promising strategy for developing ROS-scavenging hydrogels in diabetic wound healing.97,134 Hydrogels integrating inorganic substances with ROS scavenging properties are anticipated to facilitate diabetic wound healing. Chen et al. 135 introduced an injectable thermosensitive hydrogel (Nb2C@Gel) incorporating niobium carbide (Nb2C), effectively targeting ROS within diabetic wound sites and providing protection to wound cells against oxidative stress damage. Qi et al. 136 utilized melanin-coated Aurum (Au) and platinum (Pt) nanoparticles to develop AuPt@melanin nanoparticles. These nanoparticles were then integrated into a hydrogel composed of gelatin modified with phenylboronic acid and hyaluronic acid modified with thiol, resulting in the formation of the AuPt@melanin-incorporated (GHM3) hydrogel (Figure 5(c)). The inclusion of AuPt@melanin nanoparticles notably augmented the antioxidant activity of the GHM3 hydrogel compared to the GHM1 hydrogel lacking AuPt@melanin, indicating the enhancement in scavenging capabilities for ROS. Zhao et al. 137 incorporated polydopamine-modified MnCoO nanoparticles into hydrogel to harness their exceptional ROS-scavenging capabilities (Figure 5(d)), expediting the healing process of diabetic wounds. To summarize, ROS-scavenging hydrogels exhibit the potential to alleviate oxidative stress, accelerate regeneration, and synergize with additional therapeutic agents or molecules. Consequently, these hydrogels show substantial promise as therapeutic biomaterials in the treatment of diabetic wounds.
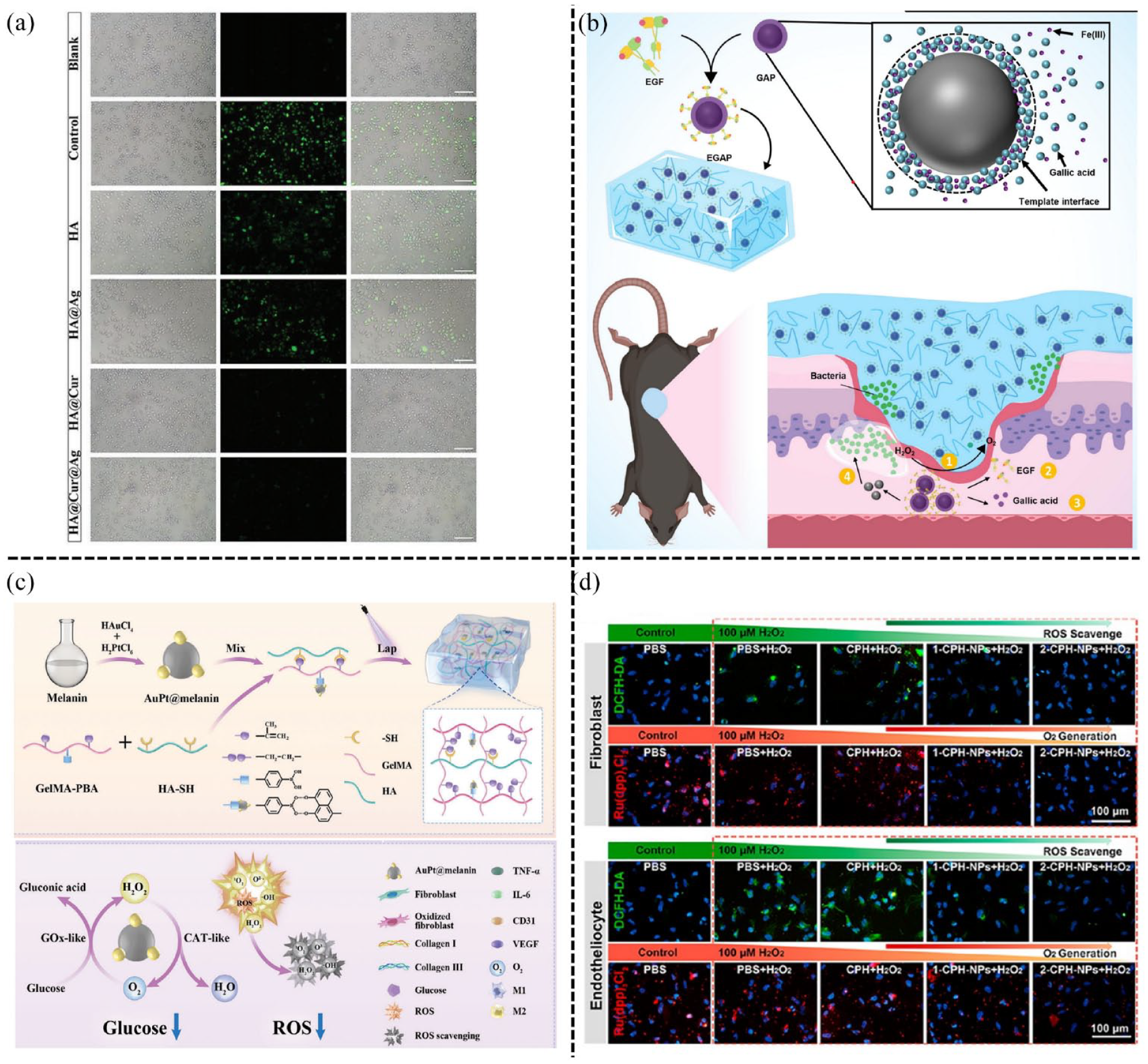
ROS-scavenging hydrogels in diabetic wounds: (a
Hypoxia and ischemia and angiogenic hydrogels
Oxygen plays a critical role in regulating multiple wound healing processes, encompassing fibroblast proliferation, granulation, re-epithelialization, angiogenesis, and tissue regeneration. However, in diabetes, hypoxia is commonly associated and has been observed to impede neovascularization induced by ischemia, along with other adaptive cellular and tissue responses to low oxygen levels. Consequently, the presence of hypoxia within the diabetic wound microenvironment can significantly hinder the healing process. Presently, studies suggest that hypoxia-inducible factor 1 (HIF-1), especially HIF-1α, functions as a hypoxia sensor and plays a pivotal role in translating hyperglycemia into impaired cellular responses to hypoxia in diabetic wounds.138,139 Typically, HIF-1α demonstrates substantial upregulation in response to hypoxic conditions. Hariharan et al. 140 discovered that the hypoxic and ischemic microenvironment decreased the expression of VEGF in cells and tissues obtained from diabetic patients and diabetic mice. This effect was attributed to the impairment of HIF-1α/p300 interaction, consequently hindering the angiogenetic process in diabetic wound healing. Moreover, in response to low oxygen levels or hypoxia, cells upregulate the generation of ROS as compensatory mechanism due to the insufficient oxygen supply. 141 This adaptive strategy aims to sustain energy production through anaerobic glycolysis, ensuring cellular survival in response to insufficient oxygen levels. Hypoxia brings about prolonged inflammation and impaired angiogenesis within the chronic hypoxic diabetic microenvironment due to persistence of pro-inflammatory M1 macrophages. 142 Additionally, hypoxia can compromise the bactericidal ability of neutrophils through respiratory burst metabolism, 143 and this compromise has the potential to exacerbate wound infections and impede the healing process. In the clinical management of diabetic wounds, apart from regulating blood glucose levels, promoting local angiogenesis is crucial to expedite wound healing. Consequently, there’s a notable drive in developing angiogenic hydrogels that aim to enhance the microenvironment of diabetic wounds by incorporating agents that facilitate blood vessel formation, improve oxygen delivery, or deliver growth factors promoting vascularization.
Deferoxamine (DFO) is an FDA-approved angiogenic drug attributed to promoting vascularization by upregulating the expression of HIF-1α that induces the secretion of VEGF. Shao et al.
144
engineered a self-adaptive multifunctional hydrogel named QCSFP with self-healing and injectable qualities. This hydrogel was created by merging the PBA group from 3-carboxy-4-fluorophenylboric acid grafted quaternary ammonium chitosan (QCSF) with the hydroxyl group of PVA, forming a hydrogel network that incorporated gelatin microspheres loaded with the angiogenic drug DFO (DFO@G). QCSFP displayed responsiveness to the elevated MMP-9 levels characteristic of diabetic wounds, allowing for controlled release of the Fe2+ chelating agent DFO. This action disrupted the degradation of HIF-1, consequently enhancing the expression of HIF-1, VEGF, α-smooth muscle actin (α-SMA), and CD31, thereby significantly promoting angiogenesis within diabetic wounds (Figure 6(a)). Additionally, vasotropic drugs have been incorporated into hydrogels in diverse forms to create angiogenic hydrogels. Li et al.
145
developed a cationic hybrid hydrogel inspired by the natural properties of hot springs. This hydrogel exhibits mild thermal effect specifically at the site of diabetic wounds with bacterial infections. It facilitates the controlled release of minute amounts of Fe2+, prompting the upregulation of VEGF, HIF-1α, basic fibroblast growth factor (bFGF), and bFGF receptor (bFGFR) to foster angiogenesis. Moreover, it responds to the infected microenvironment by releasing cationic antibacterial poly pentahydropyrimidine (PPHP), effectively eradicating bacteria, and synergistically accelerating the healing of STZ-induced infected diabetic wounds within a span of 10 days. Mesenchymal stem cell-derived exosomes (MSC-Exos) play a pivotal role in facilitating angiogenesis.
146
Therefore, Guo and co-workers incorporated exosomes derived from adipose-derived mesenchymal stem cells (hADSCs-Exos) into hydrogels aiming to enhance angiogenesis and facilitate the healing process of diabetic wounds (Figure 6(b)).
132
In addition, GelMA hydrogel and extracellular vesicles were employed as carriers for VH298, a small molecule drug known for its ability to inhibit the interaction between Von Hippel-Lindau (VHL) and HIF-1α, thereby increasing the expression of HIF-1α. The outcomes revealed that the sustained release of VH298 notably boosted angiogenesis in wounds of diabetic mice (Figure 6(c)). This effect substantially enhanced local blood circulation, playing a pivotal role in expediting wound healing by activating the HIF-1α/VEGFA pathway.
61
Based on gas therapy, a novel hydrogel comprising
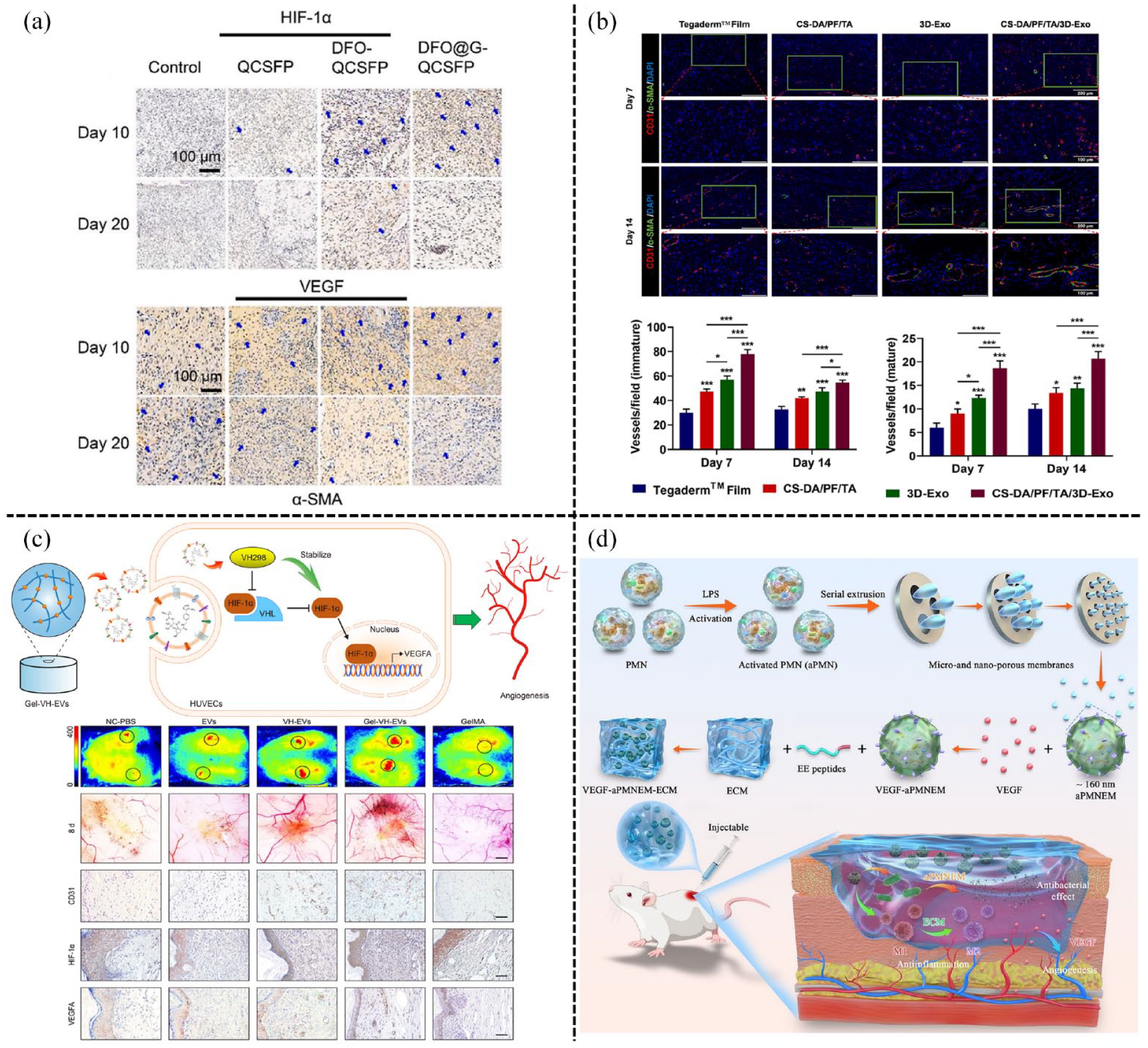
The angiogenic hydrogels in diabetic wounds: (a)the self-adaptive DFO@G-QCSFP hydrogel released DFO@G on demand to promote angiogenesis and accelerated the wound healing of diabetic rats. Reproduced with permission. 144 Copyright 2022, Elsevier B.V, (b) the CS-DA/PF/TA/3D-Exo hydrogel exhibited stronger vascular promoting ability. Reproduced with permission. 132 Copyright 2023, Elsevier B.V, (c) the Gel-VH-EVs released the VH-EVs to enhance angiogenesis in diabetic wound via HIF-1α mediated pathway. Reproduced with permission. 61 Copyright 2022, Elsevier Ltd, and (d) VEGF–aPMNEM–ECM hybrid hydrogel promoted diabetic wound repair through enhancing the long-distance reproduction of VEGF. Reproduced with permission. 63 Copyright 2023, Springer Nature.
pH variation and pH-responsive hydrogels
The physiological pH of the normal human epidermal layer is naturally maintained at a slightly acidic level. The skin surface typically ranges from 4.2 to 5.9, showing an increasing pH gradient across the horny layer until it reaches 6.8 in the lower stratum corneum. In contrast, the pH of diabetic wounds typically exists in the range from 7.2 to 8.9.150,151 Generally, the acidic environment supports wound healing by influencing macrophage and fibroblast activity, enhancing oxygenation levels, and facilitating enzymatic activity. Conversely, the alkaline environment promotes bacterial infection and contributes to oxygen depletion on the wound’s surface. Pivian et al. 152 demonstrated that pH significantly influences the modulation of wound healing rates. They found that acidic buffers formulated at pH 4 resulted in a more expedited recovery of injured tissues compared to pH 6 buffers by improving re-epithelialization. Indeed, the acidic microenvironment may not always be conducive to optimal wound repair. In infected wounds, bacterial growth and metabolism can lead to localized acidification through the production of lactic acid and acetic acid. Additionally, fibroblast proliferation and migration tend to occur in alkaline microenvironment. As a result, precise regulation of pH levels at different stages becomes essential for promoting efficient healing of diabetic wounds.
Researchers are currently investigating the creation of pH-responsive hydrogels tailored to the distinctive pH fluctuations in diabetic wounds. These hydrogels aim to regulate drug release in response to the wound’s pH variations, considering the lower pH levels during prolonged inflammation and higher initial pH levels prone to bacterial growth and increased oxygen utilization. These pH-responsive drug delivery hydrogels are structured with molecular components containing acidic or basic groups (such as carboxyl, sulfonic, and amino groups) that exhibit pH-sensitive behavior. Alternatively, dynamic covalent bonds are employed to create hydrogels that can respond to changes in pH levels. Li et al.
153
devised a self-healing and injectable hydrogel using polysaccharide components coupled with insulin glargine, created by integrating N-carboxyethyl chitosan (N-CS), HA-aldehyde (HA-ALD), and adipic acid dihydrazide (ADH) through reversible dynamic bonds like acylhydrazone and imine bonds (Figure 7(a)). This hydrogel exhibited pH-responsive characteristics owing to the inherent instability of the hydrazone bond in acidic conditions. The research findings highlighted that when the pH decreased from 7.4 to 6.5, the hydrogel degraded faster, resulting in the sustained release of insulin glargine over a 14-day period. Consequently, this property aided in diabetic wound healing by lowering glucose levels, promoting granulation tissue formation, enhancing collagen deposition, expediting re-epithelialization, and fostering neovascularization. Tang et al. developed a pH-responsive and thermosensitive injectable self-healing hydrogel by combining chitosan oligosaccharide (COS) with Pluronic F127 (F127-COS), hyaluronic acid treated with sodium periodate (NaIO4), COS, and boric acid (BA; Figure 7(b)). This formulation, termed FCAB hydrogel, was loaded with DFO to aid in diabetic wound healing and tissue regeneration. This enhanced and precise delivery of DFO under specific pH conditions showcased potential for promoting vascularization and aiding in wound healing.
154
Furthermore, pH-responsive hydrogels using protonation have been developed. Hu et al.
155
engineered a multifunctional hydrogel by incorporating antibacterial Ag NPs and the angiogenic drug DFO within the dual-crosslinked structure. This structure was created by combining chitosan quaternary ammonium salt (HTCC) and oxidized dextran-dopamine (OD-DA), forming double Schiff base bonds (DA and OD, HTCC, and OD-DA). In pH 7.4 phosphate-buffered saline (PBS), approximately 20% of Ag NPs were released. However, when exposed to acidic environment with PBS (pH 5.0), the release of Ag NPs dramatically increased to 83.9%. Simultaneously, over 90% of DFO was released within PBS (pH 5.0), whereas less than 30% was released at pH 7.4 (Figure 7(d)). This intelligent hydrogel exhibits pH responsiveness specifically at
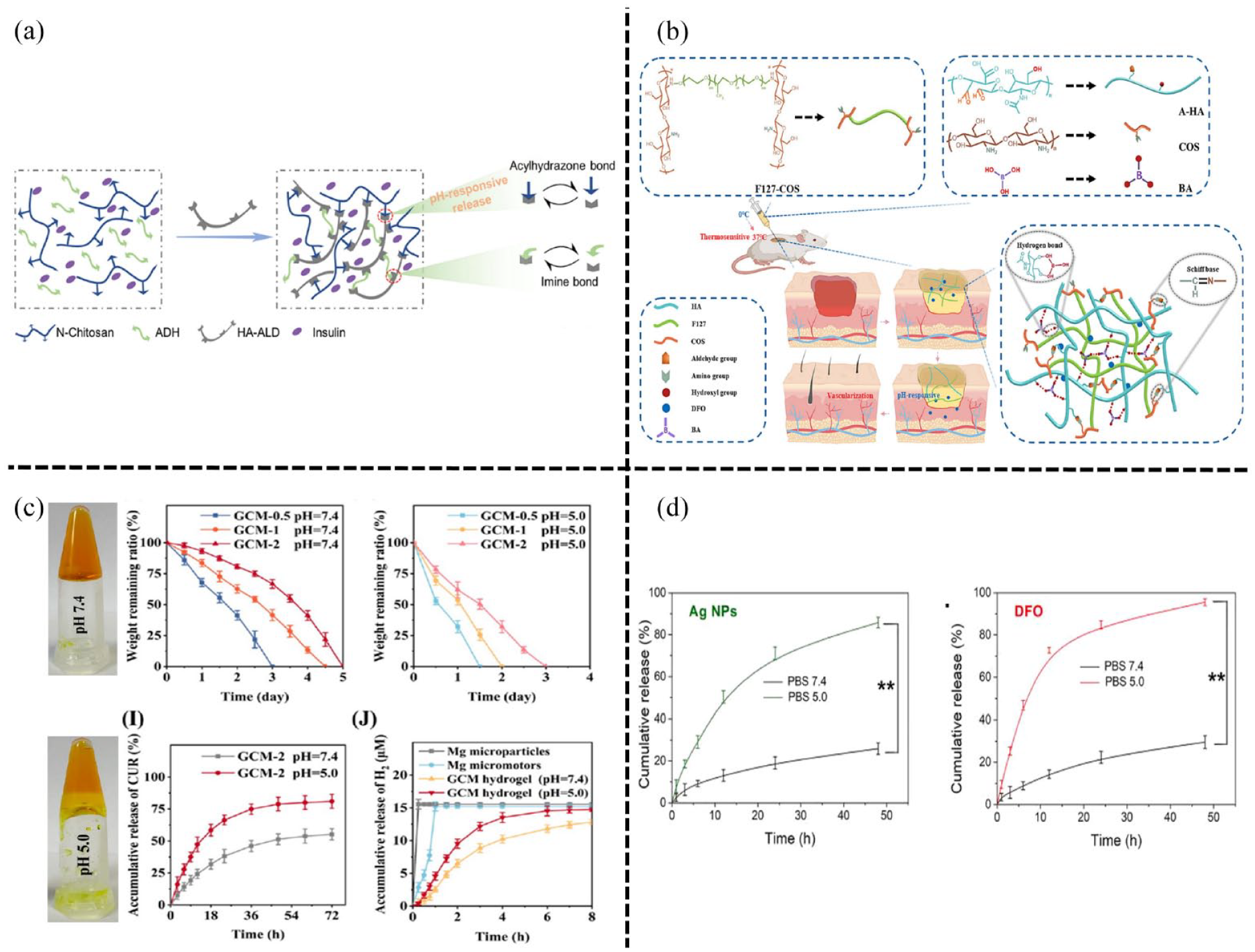
The pH responsive hydrogels in diabetic wounds: (a) the insulin-loaded hydrogels with pH-responsive property attributed to the inherent instability of the hydrazone bond under acidic conditions. Reproduced with permission. 153 Copyright 2021, Elsevier Ltd, (b) The FCAB/D hydrogel loaded with DFO possess pH response based on Schiff bone. Reproduced with permission. 154 Copyright 2023, Elsevier B.V, (c) the GCM hydrogel with pH-responsive ability to deliver drug on-demand and facilitated diabetic wound healing. Reproduced with permission. 65 Copyright 2023, American Chemical Society, and (d)the pH-responsive multifunctional hydrogel by loading antibacterial AgNPs and the angiogenic drug DFO. Reproduced with permission. 155 Copyright 2021, Elsevier B.V.
Cell death and related signaling pathway
Cell death is a fundamental process in the life cycle of all organisms and occurs through various mechanisms, exerting influence on numerous aspects of human life. Recent years have seen significant advancements in understanding the pathogenesis of diabetic wounds through extensive research. Researchers have uncovered a strong correlation between diabetes-associated wounds and cell death, along with the associated signaling pathways. This progress has enhanced our knowledge in this field, shedding light on the intricate mechanisms underlying diabetic wound development.
Apoptosis, a genetically regulated process, refers to the programmed cell death that occurs in an organized manner to maintain internal homeostasis. The dysregulation of apoptosis in response to hyperglycemia in diabetic wounds is a common occurrence, leading to compromised wound healing. According to Awadhesh et al., 156 lymphocyte apoptosis is initiated by excessive ROS, resulting in increased expression of pro-apoptotic proteins such as Caspases-3, Bax, and FAS, while concurrently reducing the levels of anti-apoptotic proteins such as B-cell lymphoma 2 (Bcl-2) in the wound healing of diabetic patients. Liang et al. 105 demonstrated that the upregulation of MMP-9 induced by AGEs resulted in apoptosis in keratinocytes. This process hindered diabetic wound healing by activating the Fas Ligand/Fas pathway. 105
Pyroptosis is a form of programmed cell death triggered by inflammasome activation. During pyroptosis, the cell undergoes rapid expansion until its membrane ruptures, leading to the release of intracellular contents and eliciting potent inflammatory response. This process is initiated by two pathways: the classical signaling pathway mediated by caspase-1 and the non-classical signaling pathways induced by caspase-11/4/5. Both caspase-1 and caspase-11/4/5 cleave gasdermin D (GSDMD), a member of the gasdermin family, resulting in the liberation of its gasdermin-N domain. This liberated domain subsequently punctures the plasma membrane, causing cellular swelling and osmotic lysis.157,158 Emerging evidence suggests a noteworthy connection between pyroptosis and the onset of diabetic complications, particularly concerning diabetic wounds. Within inflammatory responses, the NLRP3 inflammasome has been implicated in the pathogenesis of diabetic wounds. The expression levels of NLRP3, pro-caspase-1, cleaved-caspase-1, pro-IL-1β, cleaved IL-1β, IL-18, and TNF-α are elevated within diabetic wounds.159,160
Researchers noted the increased expression of NLRP3 and NETs in diabetic wounds, observed both in human and rat models. Moreover, excessive production of NETs in diabetic wounds was found to elevate NLRP3 and pro-IL-1β levels through the activation of the TLR-4/TLR-9/NF-κB signaling pathway, while also triggering an excess generation of ROS. These processes facilitated the interaction between NLRP3 and thioredoxin interacting proteins, ultimately resulting in the activation of the NLRP3 inflammasome and the persistence of inflammatory responses.
92
Irena et al.
161
demonstrated that
Autophagy, categorized into macroautophagy, microautophagy, and chaperone-mediated autophagy, is pivotal for cellular homeostasis by dismantling aberrant proteins, damaged organelles, and recycling their components, especially during nutrient scarcity. It also aids in pathogen elimination. Dysregulated autophagy holds substantial implications in both normal and pathological states. Recent studies highlight the critical role of autophagy in diabetic wound healing. Liang et al.
162
observed a significant decrease in sequestosome 1 (SQSTM1/p62), an autophagy receptor, within keratinocytes in diabetic wounds. This decline impaired autophagic flux, contributing to delayed wound healing attributed to chronic hyperglycemia. Furthermore, the impaired migratory ability of keratinocytes in diabetic wounds impedes autophagy and hampers keratinocyte migration by suppressing the activation of the p38/MAPK signaling pathway. This effect is linked to the downregulation of autophagy-related proteins such as Atg5 and LC3-II.
163
Therefore, adjusting autophagy levels in diabetic wounds presents itself as a potentially promising therapeutic avenue. In an intriguing study by Ji et al.,
164
they enhanced autophagy in macrophages by elevating the expression levels of LC3 and Beclin-1. This intervention proved effective in alleviating
Presently, researchers are actively exploring not only the potential connections between diverse types of cell death and diabetic wounds but also the complex network of signaling pathways linked to these wounds. Recent studies have highlighted the involvement of the PI3K/Akt/mTOR signaling pathway in the regulation of processes such as proliferation, microvascular density, collagen deposition, and macrophage infiltration within diabetic wounds, aiming to promote and facilitate the healing process.168,169 In diabetes patients, growth factors and chemokines can trigger the activation of the JAK-STAT pathway. This activation has been observed to stimulate the proliferation and migration of human dermal fibroblasts, ultimately aiding the process of healing in diabetic wounds. 170 Suppression of the Wnt/β-catenin signaling pathway leads to reduced proliferation and cytokine expression in dermal fibroblasts. Additionally, it impairs endothelial cell functions and interferes with the development of granulation tissue, ultimately disrupting the re-epithelialization processes, thereby causing delays in wound healing.171–173 Dysfunction within the Notch pathway can hinder cell differentiation, disrupt normal cell proliferation, and impede angiogenesis, all of which contribute to interference in the process of diabetic wound healing.174,175
Despite significant research efforts, the exact relationship between diabetic wounds and interconnected signaling pathways remains elusive. Hence, there is currently a lack of hydrogels designed to enhance the healing of diabetic wounds through targeted modulation of relevant signaling pathways or cell death, further investigation is needed to thoroughly comprehend the etiology and pathogenesis of diabetic wounds so that researchers can develop new hydrogels with precise regulation to promote wound healing.
Conclusions and prospects
The exploration of the diabetic wound microenvironment has spurred the creation of targeted hydrogels, showcasing adaptive properties for treating these wounds. This review investigates the impact of recent diverse hydrogel formulations, designed to suit the specific conditions and facilitate of diabetic wounds healing. Given the unique physiological characteristics of diabetic wounds, hydrogels can be engineered to possess hypoglycemic, anti-inflammatory, anti-infective, ROS-scavenging, and angiogenic features. Additionally, responsive hydrogels answering to various stimuli like pH and glucose hold potential for tailored drug release, regulating the diabetic wound environment during different stages of healing. Concurrently, efforts are underway to develop real-time monitoring methods for wound biochemical markers and responsive drug delivery systems to enhance the healing. Despite the substantial therapeutic promise of hydrogels in treating diabetic wounds, ongoing research remains at the developmental stage and hasn’t progressed to clinical application. This indicates existing challenges in customizing hydrogels for diabetic wound healing, necessitating further exploration before clinical implementation can be realized.
Firstly, the prevalent models for studying diabetic wounds primarily rely on rats and mice due to their cost-effectiveness and ease of handling. While pigs offer a closer physiological resemblance to human skin, their high costs and operational complexities limit widespread use in wound research. Consequently, there’s been a focus on developing pre-experimental hydrogel models tailored specifically for diabetic wounds. However, these models often lack efficient clinical translation. Moreover, the healing timeline observed in these models doesn’t align well with the actual healing duration in diabetic patients, leading to limitations in applying experimental findings to clinical scenarios, resulting in suboptimal outcomes in clinical trials. Therefore, selecting animal models that closely mirror human skin structure and extending the duration of wound treatment can be crucial. These steps hold promise for accelerating the clinical translation of hydrogels to address real-world clinical challenges. Additionally, certain drugs and cytokines loaded into hydrogels often exhibit therapeutic effects specific to different healing stages. However, achieving precise control over drug release at these distinct stages remains a challenge, impacting the efficacy of diabetic wound healing. There is also no clear research regarding the optimal timing for application of hydrogel specifically designed to modulate the macrophages polarization in diabetic wounds for enhancing wound healing, which constitutes a critical issue warranting further investigation. Furthermore, some hydrogel-based visual monitoring systems designed for real-time assessment of biochemical indicators in diabetic wounds, coupled with sensing therapy, are complex and costly. As a result, these systems have mainly remained in the research phase and are yet to be practically implemented in clinics to address visualization challenges.
The prevalence of diabetic wounds presents a critical clinical challenge due to its impact on patients’ quality of life, causing discomfort, odor, and potential disability. Prolonged wounds can undermine patient confidence in treatments, fuel resistance to medical interventions, and worsen disease progression, imposing significant healthcare burdens and economic strain on society. This situation necessitates effective solutions to manage chronic wounds, especially those related to diabetes. The development of hydrogels tailored to the diabetic wound microenvironment offers promising prospects for addressing various healing stages. Through ongoing research advancements, hydrogels stand as potential solutions for the clinical treatment of chronic wounds.
Footnotes
Author contributions
Ying Zhao prepared the manuscript and designed figures. Yulan Zhao prepared the manuscript, designed partial figures, and helped with language editing. Bing Xu designed partial figures. Qiang Chang and Hongwei Liu designed and supervised manuscript. All authors approved this work for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the following foundation: National Natural Science Foundation of China (Grant number: 81772101, 82072196, and 82002066). We are also grateful for the support from the Science Fund for Distinguished Young Scholars of the Southern Medical University (Grant number: 2020J009).
