Abstract
Background:
Transthyretin cardiomyopathy (ATTR-CM) is a debilitating disease that has received much attention since the emergence of novel treatments. The Transthyretin Cardiomyopathy Clinical Trial showed that tafamidis, a transthyretin tetramer stabilizer, effectively reduced the declines in functional capacity and quality of life. However, Ala97Ser (A97S) hereditary ATTR-CM is underrepresented in major ATTR-CM tafamidis trials.
Objectives:
We aim to investigate the change in global longitudinal strain (GLS) of A97S ATTR-CM patients after 12 months of tafamidis treatment.
Methods:
We retrospectively analysed a prospective cohort of patients with A97S ATTR-CM who received tafamidis meglumine (61 mg/day) at the National Taiwan University Hospital. Echocardiography with speckle tracking strain analysis was performed at baseline and 12 months after treatment.
Results:
In all, 20 patients were included in the cohort. The baseline left ventricular ejection fraction (LVEF) and interventricular septum (IVS) thickness were 59.20 ± 13.23% and 15.10 ± 3.43 mm, respectively. After 12 months of tafamidis treatment, the LVEF and IVS were 61.83 ± 15.60% (p = 0.244) and 14.59 ± 3.03 mm (p = 0.623), respectively. GLS significantly improved from −12.70 ± 3.31% to −13.72 ± 3.17% (p = 0.048), and longitudinal strain (LS) in apical and middle segments significantly improved from −16.05 ± 4.82% to −17.95 ± 3.48% (p = 0.039) and −11.89 ± 4.38% to −13.58 ± 3.12% (p = 0.039), respectively. Subgroup analysis showed that patients with LVEF < 50% had a better treatment response and improvement in GLS. The patients with an IVS ⩾ 13 mm had an improvement in two-chamber LS from −10.92 ± 4.25% to −13.15 ± 3.87% (p = 0.042) and an improvement in apical left ventricular LS from −15.30 ± 5.35% to −17.82 ± 3.99% (p = 0.031).
Conclusion:
Tafamidis significantly improved GLS, and particularly apical and middle LS in A97S ATTR-CM patients.
Plain language summary
Transthyretin cardiomyopathy (ATTR-CM) is a severe heart condition that has gained attention due to recent advancements in treatments. One of these treatments, called tafamidis, has been shown to be effective in maintaining heart function and quality of life. However, there has been limited research on a specific genetic variation of ATTR-CM: A97S. Our aim was to determine whether A97S ATTR-CM patients experienced improved heart function after one year of tafamidis treatment. We conducted this study at the National Taiwan University Hospital, where we enrolled 20 A97S ATTR-CM patients. We used echocardiography to evaluate their heart function, focusing on a parameter called global longitudinal strain. The results showed that after one year of tafamidis treatment, these patients experienced a significant improvement in their global longitudinal strain, particularly in the apical and middle regions of the heart. In conclusion, tafamidis appears to be beneficial for A97S ATTR-CM patients by enhancing their heart’s global longitudinal strain, which is a positive sign for their cardiac health.
Central illustration: After 12 months of tafamidis treatment, ATTR-CM patients had significantly improved GLS
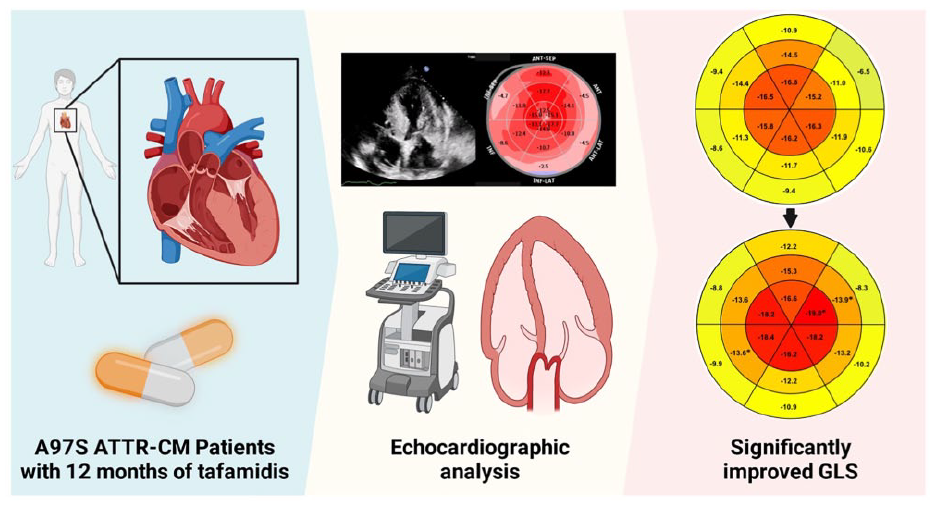
Introduction
Systemic amyloidosis is a progressive disease caused by the deposition of insoluble homomeric amyloid fibrils in vital organs. Systemic amyloidosis is classified according to the type of amyloid fibril deposited. Hereditary transthyretin amyloidosis (hATTR) is the deposition of amyloid fibrils due to inherited gene mutations. hATTR can manifest with multiple organ involvement, such as the cardiovascular, neurological and gastrointestinal systems. 1 Transthyretin cardiomyopathy (ATTR-CM) is the myocardial involvement of hATTR, and it has received increasing attention due to the availability of treatment options. The natural history of ATTR-CM includes progressive heart failure, arrhythmias and conduction system disease. 1 The Ala97Ser (A97S) genotype, the most common variant in Taiwan, has not been well investigated in major ATTR-CM studies.2–4
Since the tafamidis in Transthyretin Cardiomyopathy Clinical Trial (ATTR-ACT), the outcomes and response to tafamidis treatment for ATTR-CM have received increasing attention. Most reports on treatment groups have demonstrated slower deterioration or stable clinical course compared to non-treated groups, while some have demonstrated improvements in myocardial biomarkers.5–8 However, the treatment response and effect on various genotypes have yet to be extensively studied, particularly the A97S genotype.
Echocardiography remains the preferred imaging study for the assessment of cardiac amyloidosis. 9 However, typical findings of ATTR-CM, such as increased left ventricular (LV) wall thickness and impaired systolic and diastolic function, may remain normal until the late stages of the disease. Analysis of cardiac deformation with two-dimensional speckle tracking echocardiography (STE) is a sensitive measure of systolic and diastolic function. LV global longitudinal strain (GLS) assessed by STE plays an important role in the diagnosis and risk stratification of ATTR-CM. In addition, a relative ‘apical sparing’ regional longitudinal strain (LS) pattern in which the LV apical region shows relatively normal strain compared with progressively worse strain in the mid and basal regions is consistently observed in ATTR-CM. 10
STE is used for longitudinal surveillance of ATTR-CM. 11 Emerging studies have reported that tafamidis reduced the deterioration in GLS after 12 months of treatment in wild-type ATTR-based patients.5,12 Tafamidis was also shown to reduce the deterioration in GLS over 30 months in ATTR-CM patients enrolled in the ATTR-ACT trial. 13 These echocardiographic data provide additional insight into the efficacy of tafamidis; however, studies evaluating the efficacy of tafamidis in patients with the A97S genotype are still needed. In this study, we aimed to use comprehensive echocardiographic evaluations to investigate the effects of tafamidis on A97S ATTR-CM.
Methods
Study population
We retrospectively analysed a prospective cohort of ATTR-CM patients who received tafamidis (61 mg/day) treatment at the National Taiwan University Hospital. The diagnosis of A97S ATTR-CM was based on the presence of heart failure symptoms in conjunction with (1) positive technetium-99m-pyrophosphate (PYP) scintigraphy with the exclusion of light chain disease 14 and (2) A97S mutation and genetic tests were performed using either Sanger sequencing or restriction enzyme specific for the A97S mutation. The exclusion criteria were as follows: (1) patients lost to follow-up or who died within the 12-month follow-up period; (2) those who were intolerant to tafamidis or had poor prescription adherence and (3) those with non-A97S ATTR-CM.
The A97S cohort comprised 26 patients, of whom two died within the 12-month follow-up period due to non-cardiovascular aetiologies. Another four patients were excluded due to underlying major cardiovascular conditions that may have altered the interpretation of STE: one patient had type A dissection, one patient had received coronary bypass surgery due to coronary artery disease and two patients were implanted with permanent pacemakers. Finally, 20 patients were included in this study (Figure 1). Of these 20 patients, 10 were enrolled in an ongoing clinical trial (NCT02791230) and the other 10 received compassionate tafamidis. The study was approved by the National Taiwan University Hospital Institutional Review Board and conducted in accordance with institutional guidelines.
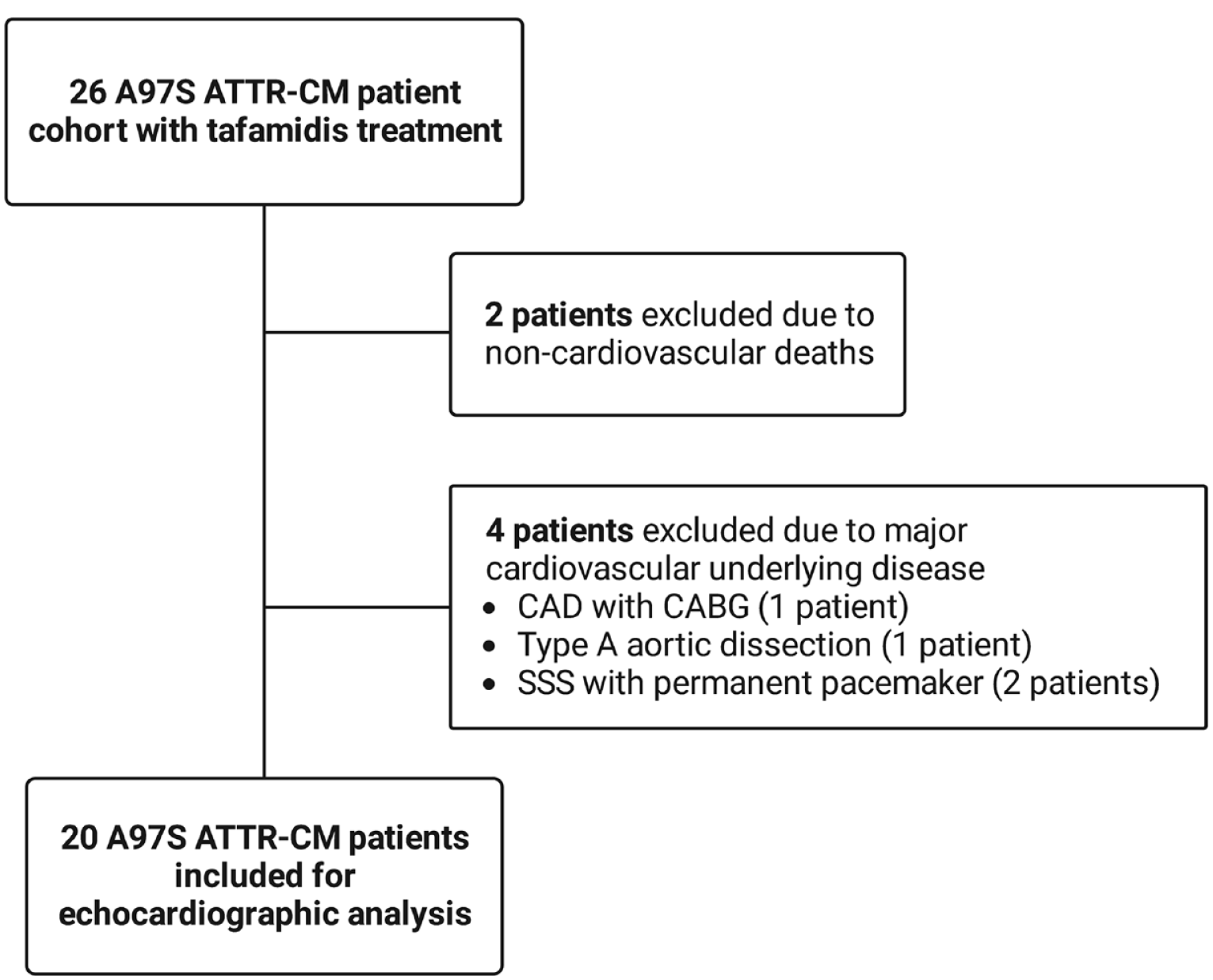
Patient selection.
Echocardiography
All subjects received standard transthoracic echocardiography using IE33 and EPIQ-CVx systems (Philips Medical Systems, Andover, MA, USA). All data were stored digitally for offline analysis. Conventional echocardiographic measurements were performed in accordance with the recommendations of the American Society of Echocardiography. 15 Interventricular septal (IVS) thickness, LV posterior wall (PW) thickness, LV end-diastolic and end-systolic diameters and LV ejection fraction (LVEF) were obtained. Doppler analysis included the E/e′ ratio (calculated with septal e′). GLS was calculated offline using QLAB cardiac analysis (Philips Medical Systems). LS was calculated for apical, middle and basal segments. Relative apical sparing and septal apical to base ratio were obtained according to a previously published study. 9
To ensure the reproducibility of STE analysis, GLS was remeasured in five random anonymized samples by a second operator. The inter-observer intra-class correlation coefficient (ICC) was 0.984 (95% confidence interval 0.855–0.998), with a mean GLS difference of 0.14 and a standard deviation of 0.52 (Supplemental Table S1).
Statistical analysis
Data were expressed as numbers (%) for categorical variables and as mean ± standard deviation for all continuous variables. Normality was determined using the Shapiro–Wilk test. Comparisons of data were performed using the paired sample t-test (normally distributed data) and Wilcoxon signed-rank test (non-normally distributed data). Interobserver ICCs were calculated with a two-way random model using an absolute agreement definition. All statistical tests were two-sided, and a p value of <0.05 was considered statistically significant. All statistical analyses were performed using SPSS for Windows, version 25.0 (SPSS, Inc., Chicago, IL, USA).
Results
Clinical and heart failure characteristics
The baseline clinical characteristics of all 20 tafamidis-treated patients are shown in Table 1. The majority of the patients in this cohort were male (80%), with a mean age of 62.4 ± 6.5 years. Notably, 20% of the patients had atrial fibrillation, and none were diagnosed with coronary artery disease. Heart failure characteristics are shown in Table 2. In all, 18 patients were New York Heart Association (NYHA) functional class II, and two patients improved to functional class I after 12 months of treatment (p = 0.625). The median N-terminal pro b-type natriuretic peptide (NT-pro-BNP) level was 558.60 pg/ml at baseline and 592.45 pg/ml at 12 months (p = 0.073). All patients were treated with tafamidis meglumine 61 mg daily. None of the patients discontinued treatment due to adverse effects, and adherence was confirmed at each clinical visit every 3 months.
Clinical, biomarker and heart failure characteristics.

AC, Ante Cibum (before meal); ATTR-CM, transthyretin cardiomyopathy.
Change in heart failure and echocardiographic parameters between baseline and 1 year of tafamidis treatment.

Fc, Funcational class; GLS, global longitudinal strain; IVSD, interventricular septum diameter; LS, longitudinal strain; LV, left ventricle; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; LVESD, left ventricular end-systolic diameter; LVPWD, left ventricular posterior wall diameter; RELAPS, relative apical sparing; SAB, septal apical to base ratio.
Baseline and 12-month echocardiographic characteristics
The baseline and 12-month echocardiographic values are shown in Table 2. None of the patients had a major valvular disease, and none of the patients received an implanted cardiac device or cardiac surgery or experienced any episodes of myocardial infarction during the study period (as the index date was different for every patient, the study spanned across 2019–2022).
The LVEF and E/e′ values were 59.20 ± 13.23% and 21.38 ± 11.19, respectively, at baseline, compared to 61.83 ± 15.60% and 20.53 ± 11.45, respectively, after 12 months of tafamidis treatment (LVEF: p = 0.244 and E/e′: p = 0.948). The IVS and PW diameters prior to tafamidis treatment were 15.10 ± 3.43 and 14.10 ± 2.69 mm, respectively, which decreased to 14.59 ± 3.03 and 13.53 ± 2.60 mm after 12 months of tafamidis treatment (IVS: p = 0.623 and PW: p = 0.194).
In speckle tracking analysis, the GLS significantly improved from −12.70 ± 3.31% to −13.72 ± 3.17% (p = 0.048). Most notably, the two-chamber LS improved from −11.98 ± 4.19% to −13.86 ± 3.80% (p = 0.042). Segment-wise, the GLS of apical and middle segments significantly improved from −16.05 ± 4.82% to −17.95 ± 3.48% (p = 0.039) and −11.89 ± 4.38% to −13.58 ± 3.12% (p = 0.039), respectively. The basal segment GLS did not significantly change (−9.46 ± 4.59% versus −10.08 ± 3.59%, p = 0.451) after 12 months of tafamidis treatment. The four-chamber (−13.24 ± 3.76% versus −13.01 ± 3.15%; p = 0.686) and three-chamber (−13.43 ± 4.25% versus −13.61 ± 3.41%; p = 0.824) LS did not significantly change after treatment. In addition, relative apical sparing (1.58 ± 0.29 versus 1.58 ± 0.33, p = 0.986) and septal apical to base ratio (3.92 ± 8.52 versus 2.92 ± 4.09, p = 0.645) did not significantly change after treatment.
Analysis of each LS segment was performed, and individual segments of the echocardiography were averaged [Figure 2]. In the apical segment, one of the six segments significantly improved (p < 0.05, Supplemental Table S2). While in the middle segments, two of the six segments significantly improved (p < 0.05, Supplemental Table S2). No basal segments statistically improved. Overall, the apical and middle LS improvement contributed to the improvement in overall GLS.

Eighteen-segment LV bull’s eye plots showing average segmental peak systolic longitudinal strain at baseline and after 12 months of tafamidis treatment. One out of six apical and two out of six middle segments significantly improved (*p < 0.05). The segments are colour coded according to the colour scale above.
Subgroup analysis: stratified by LVEF
Subgroup analyses were conducted to identify the subgroup with the most favourable outcome after treatment with tafamidis. We first performed a subgroup analysis of the patients with LVEF ⩾ 50% and LVEF < 50%. In all, 15 patients had LVEF ⩾ 50% and five had LVEF < 50%. The results revealed that the E/e′ ratio after 12 months of tafamidis treatment decreased from 26.76 ± 12.08 to 21.10 ± 9.93 in the patients with LVEF < 50% (p = 0.068), and from 19.31 ± 10.59 to 17.33 ± 11.09 in the patents with LVEF ⩾ 50% (p = 0.463). In addition, the GLS significantly improved from −8.46 ± 2.41% to −10.14 ± 2.50% (p = 0.043) in the LVEF < 50% subgroup, while the GLS remained stationary (−14.11 ± 2.15% versus −14.91 ± 2.40%; p = 0.177) in the LVEF ⩾ 50% group. In the LVEF < 50% subgroup, the four-chamber LS (−8.52 ± 1.96% versus −9.50 ± 2.01%; p = 0.043), apical LV LS (−10.36 ± 5.90% versus −14.75 ± 3.30%; p = 0.043) and middle LV LS (−6.61 ± 3.96% versus −10.54 ± 3.12%; p = 0.043) all significantly improved after 12 months of tafamidis treatment (Table 3).
Change in speckle tracking echocardiographic parameters stratified according to LVEF at baseline and 1 year of tafamidis treatment.

GLS, global longitudinal strain; LS, longitudinal strain; LV, left ventricle; LVEF, left ventricular ejection fraction.
Subgroup analysis: stratified by IVS thickness
We then performed a subgroup analysis of patients with IVS thickness ⩾13 mm and IVS <13 mm. In all, 15 subjects had an IVS ⩾13 mm and 5 had an IVS <13 mm. The subgroup analysis did not reveal the obvious changes in LVEF and E/e′ ratio after 12 months of tafamidis treatment, and GLS did not significantly improve in either group. In the IVS ⩾13 mm group, the two-chamber LS (−10.92 ± 4.25% versus −13.15 ± 3.87%; p = 0.042) and apical LV LS (−15.30 ± 5.35% versus −17.82 ± 3.99%; p = 0.031) both significantly improved after 12 months of tafamidis treatment (Table 4).
Change in speckle tracking echocardiographic parameters stratified according to intraventricular septum thickness at baseline and 1 year of tafamidis treatment.

GLS, global longitudinal strain; IVSD, interventricular septum diameter; LS, longitudinal strain; LV, left ventricle; LVEF, left ventricular ejection fraction.
Discussion
In this study, we demonstrated that 12 months of tafamidis treatment significantly improved GLS in patients with A97S ATTR-CM. The apical and middle LS contributed to the improvement, while the basal LS remained relatively unchanged. Subgroup analysis revealed that the patients with LVEF <50% seemed to benefit more compared to the LVEF ⩾50% subgroup. In patients with thicker IVS (⩾13 mm), the apical LS and two-chamber LS significantly improved compared to baseline.
A97S is the most common mutation in patients with hereditary ATTR in Taiwan, 16 and it has also been reported in China, 17 Malaysia 18 and Thailand. 19 A97S presents with equally severe polyneuropathy and restrictive cardiomyopathy. Refractory heart failure is the leading cause of death in these patients. Even though in vitro studies have shown that tafamidis can stabilize TTR tetramers with A97S mutations,20,21 this mutation was not investigated in the ATTR-ACT trial. 22
Previous studies have shown that amyloid infiltration causes a marked reduction in GLS, which often precedes significant changes in LVEF.10,23 Changes in LS after treatment have been reported in recent studies. In a Japanese cohort of wild-type ATTR-CM patients who continued tafamidis for 12–18 months, there was no significant worsening in LV GLS from baseline. 24 In another wild-type-predominant ATTR-CM cohort (91% wild type), tafamidis resulted in less deterioration in GLS over a 12-month period compared with a cohort (95% wild type) not treated with tafamidis. 5 Further analysis of changes in echocardiographic measures in the ATTR-ACT trial also demonstrated that tafamidis significantly reduced the deterioration in LV GLS in ATTR-CM patients. 13 In this study, we demonstrated the effects of tafamidis in patients with A97S hereditary ATTR-CM. Tafamidis treatment significantly improved GLS, especially in the apical and middle segments, which are believed to be less impacted during disease progression.
Apical sparing is a typical finding of cardiac amyloidosis, in which LS in the basal and mid-segments of LV is impaired more significantly compared with the apical segments.10,11,23 Ratios of apical to basal plus mid-ventricular strain have been shown to have good diagnostic accuracy for differentiating cardiac amyloidosis from other etiologies. 9 Segmental LV LS has also been investigated in recent studies. In a Japanese wild-type ATTR-CM cohort who were treated with tafamidis, an apical sparing pattern was consistently observed during follow-up and no significant changes in each segmental LS from baseline to 18 months were found. 24 In a post hoc analysis of the cardiac subpopulation from the phase III APOLLO study, a comparative improvement in LV GLS, driven primarily by attenuating disease progression in the basal region, over 18 months was observed in patients with hereditary ATTR-CM who were treated with patisiran, an RNA interference therapeutic that inhibits TTR synthesis. 25
We previously reported a case with A97S who was treated with tafamidis, and reverse cardiac remodelling was proven on multiple imaging modalities (echocardiography, magnetic resonance imaging and technetium-99m PYP scintigraphy). In that case report, the patient improved clinically, functionally and structurally. 26 Similar to our findings, a post hoc analysis of the ATTR-ACT cohort revealed clinical improvement after 2.5 years of follow-up in increased 6 min walk distance, NT-pro-BNP levels, improved Kansas City Cardiomyopathy Questionnaire Overall Summary Score (KCCQ-OS) and patient global assessment questionnaire scores and NYHA functional class. 27 However, current literature has reported conflicting results on functional and structural improvements, and several studies on tafamidis did not demonstrate significant changes in echocardiographic parameters before and after 12 months of treatment in patients with ATTR-CM.8,28 These studies further highlight the heterogeneous treatment response of ATTR-CM to tafamidis and the potential undiscovered benefits of tafamidis for different genotype mutations.
In this study, we found a significant improvement in GLS after 12 months of tafamidis treatment, indicating an improvement in myocardial function. Comparable with the post hoc analysis of the APOLLO study, we did not find a significant improvement in basal LS after tafamidis treatment. However, we found that the basal, apical and middle LS improvements were mainly attributed to the overall GLS improvement. Possible explanations for these findings include different treatment medications, patient populations and genetics. The APOLLO study compared the treatment group with a placebo group, and we compared individual patients who received tafamidis. 25 In our study, the LVEF was 59.20 ± 13.23%, while the LVEF of the treated cohort in the APOLLO study was 61.7 ± 10.0%. In addition, the IVS thickness was 15.10 ± 3.43 mm in our study and 15.8 ± 3.2 mm in the treated cohort in the APOLLO study. 3 Furthermore, all of the patients in our study had hereditary A97S ATTR-CM.
In subgroup analysis, we found that the significance of GLS improvement was mainly attributable to apical LS in the patients with a lower LVEF (Table 3). Those with a higher IVS thickness had more prominent apical LS improvement, but middle and basal LS did not improve significantly (Table 4). According to our results, we hypothesize that patients with more extensive disease such as low LVEF or thicker LV wall thickness may have a more significant treatment response compared to those with less cardiac involvement.
Finally, clinical phenotypes and presentations of ATTR-CM are diverse, 29 and even those with the same mutation may have various phenotype penetrance. 30 ATTR-CM A97S is an under-represented cohort in major tafamidis trials. This variant may have a different treatment response to tafamidis compared to wild-type or other variants. Therefore, findings for the A97S variant may not apply to other genotypes. Our findings require verification through future studies with larger sample sizes and extended follow-up periods.
Our study has several limitations. First, the limited number of cases. A larger treatment cohort and extended follow-up period are needed to support our findings. As ATTR-CM A97S is a rare disease, we could not calculate the study population size. Second, the absence of a placebo group in this study may have resulted in an underestimation of the clinical benefits of tafamidis treatment. Although the tafamidis-treated group showed limited improvement or stability, the lack of a placebo group means that the clinical deterioration expected in the placebo group cannot be taken into account. In our study, most clinical and echocardiographic parameters remained unchanged, but there was an improvement in GLS after treatment, which strongly suggested the potential benefits of tafamidis to improve cardiac function even without a placebo group for comparison.
Conclusion
Tafamidis significantly improved GLS, and particularly apical and middle LS in A97S ATTR-CM patients. Patients with more severe cardiac involvement, such as low LVEF or thicker LV wall, are likely to have a greater treatment response to tafamidis compared to those with less cardiac involvement.
Supplemental Material
sj-doc-1-taj-10.1177_20406223231222828 – Supplemental material for Tafamidis improves myocardial longitudinal strain in A97S transthyretin cardiac amyloidosis
Supplemental material, sj-doc-1-taj-10.1177_20406223231222828 for Tafamidis improves myocardial longitudinal strain in A97S transthyretin cardiac amyloidosis by Yuan-Kun (Aden) Wu, An-Li Yu, Mei-Fang Cheng, Lung-Chun Lin, Ming-Jen Lee, Chia-Hung Chou, Chia-Tung Shun, Hsueh-Wen Hsueh, Jimmy Jyh-Ming Juang, Ping-Huei Tseng, Siao-Ping Lin, Mao-Yuan Su, Chi-Chao Chao, Sung-Tsang Hsieh, Cheng-Hsuan Tsai and Yen-Hung Lin in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
