Abstract
Cardiac amyloidosis usually presents with diastolic dysfunction, but sometimes systolic dysfunction develops, particularly at its advanced stage. However, the therapeutic strategy for patients with cardiac amyloidosis and systolic dysfunction remains unknown. We report a 77-year-old man who was diagnosed with wild-type cardiac amyloidosis and systolic dysfunction with a left ventricular ejection fraction of 27%. Following 6-month medical therapy of tafamidis 80 mg and neurohormonal blockers (carvedilol 5.0 mg, enalapril 2.5 mg, and spironolactone 25 mg), the left ventricular ejection fraction improved to 55%. Tafamidis-incorporated neurohormonal blocker therapy might be a promising strategy to facilitate cardiac reverse remodeling in patients with cardiac amyloidosis and systolic dysfunction.
Keywords
Introduction
Cardiac amyloidosis is a predominant and fatal comorbidity of wild-type transthyretin amyloidosis (ATTR), which is a systemic and progressive disease caused by the deposition of wild-type transthyretin amyloid in multiple organs. 1 The clinical presentation of cardiac amyloidosis is usually diastolic dysfunction with a preserved left ventricular ejection fraction (LVEF).
Tafamidis, which binds to the thyroxine-binding site of transthyretin and stabilizes transthyretin tetramers, 2 has recently been introduced in Japan for patients with wild-type ATTR. The ATTR-ACT trial showed an advantage of tafamidis in preventing all-cause death and cardiovascular hospitalization over placebo. 3 However, tafamidis suppresses the progression of amyloidosis, but does not regress amyloid that is already deposited in the myocardium. 2 Additionally, the effect of neurohormonal blockers, which are a guideline-directed medical therapy for heart failure with reduced ejection fraction (HFrEF) to facilitate cardiac reverse remodeling, 4 on patients with HFrEF due to cardiac amyloidosis remains unknown. Therefore, a therapeutic strategy for those with HFrEF due to cardiac amyloidosis has not been established.
We present a patient with HFrEF due to cardiac amyloidosis whose LVEF considerably improved by 6-month medical therapy of tafamidis and neurohormonal blockers.
Case report
Before referral
Two years before referral to our institute, a 74-year-old man was admitted to another institute to investigate suspected cardiomegaly in a chest X-ray. This possible condition was pointed out in a health examination, although he was asymptomatic. Transthoracic echocardiography showed that the LVEF was 40% with cardiac hypertrophy. An endomyocardial biopsy from the right ventricular septum showed deposition of transthyretin-positive amyloidosis. 99mTechnetium-pyrophosphate scintigraphy showed a positive stain in the heart. On the basis of these findings, he was diagnosed with cardiac amyloidosis.
Two months before the referral, he complained of dyspnea on effort and was classified as New York Heart Association functional class III. He had gained 7 kg of body weight over the past month. Transthoracic echocardiography showed a reduction in the LVEF to 27% (Figure 1a). Spironolactone 25 mg and azosemide 60 mg were administered to treat his systemic congestion. He was referred to our outpatient clinic for further treatment.
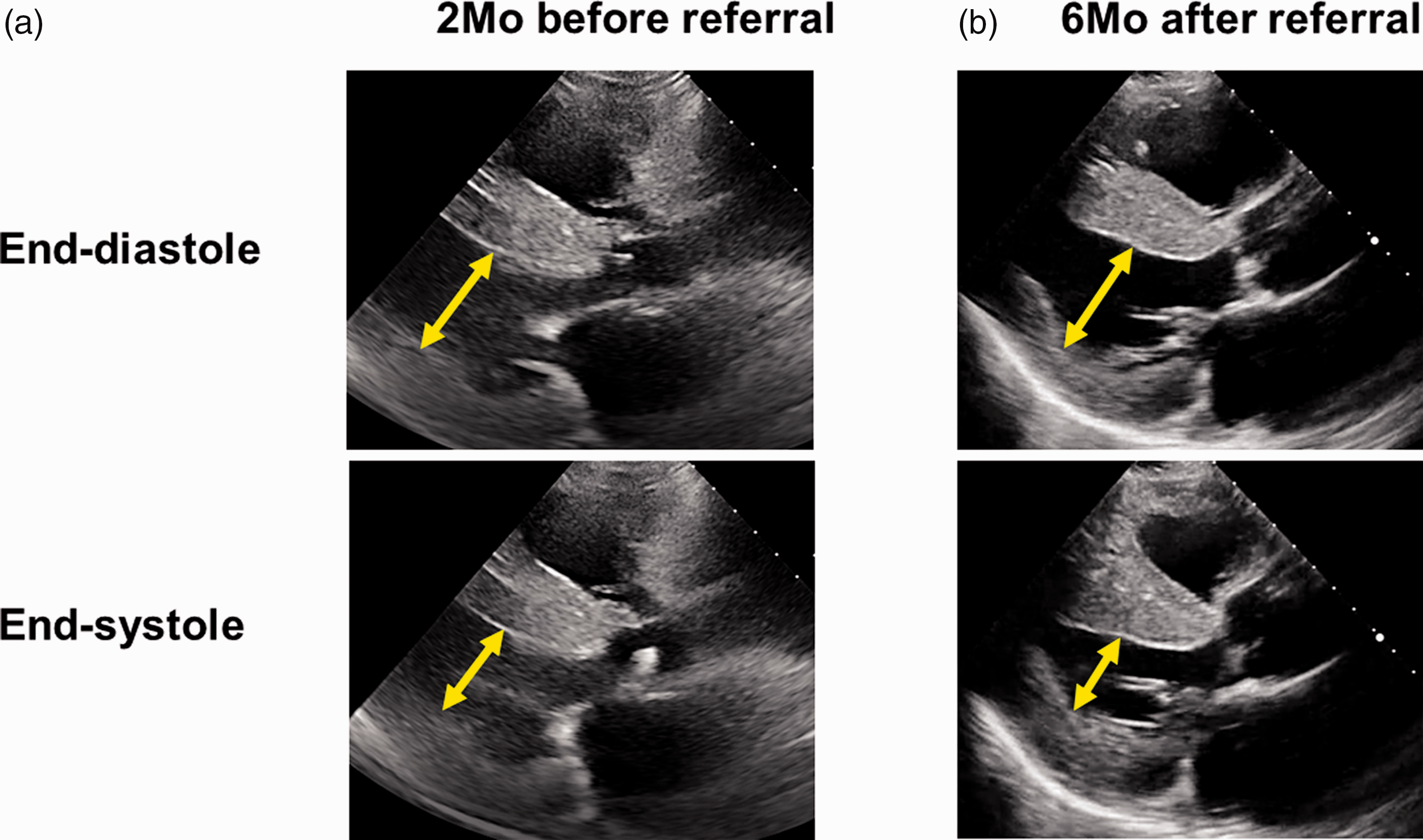
Transthoracic echocardiography (long-axis view) obtained at 2 months before referral (a) and 6 months after referral (b). Yellow arrows indicate the left ventricular diameter.
On referral
On referral to our out-patient clinic, the patient’s blood pressure was 114/68 mmHg and his heart rate was 84 beats/minute (Table 1). Transthoracic echocardiography showed a left ventricular end-diastolic diameter of 52 mm and LVEF of 40% with diffuse mild hypokinesis. The left ventricular mass index was 151.1 g/m2. The plasma B-type natriuretic peptide concentration was 160 pg/mL, and the serum troponin I concentration was 94.1 pg/mL. The QRS duration in an electrocardiogram was 142 ms. A genetic test showed wild-type ATTR.
Time course of clinical parameters.

BNP, B-type natriuretic peptide; eGFR, estimated glomerular filtration rate; bpm, beats/minute; LVDd, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; LVMI, left ventricular mass index.
We initiated tafamidis 80 mg because of the diagnosis of wild-type ATTR. Additionally, because of the patient’s reduced LVEF, we initiated carvedilol 2.5 mg (titrated up to 5.0 mg 1 month later) and enalapril 1.25 mg (titrated up to 2.5 mg later), as well as tolvaptan 15 mg to treat residual systemic congestion.
This report conforms to the CARE guidelines. 5
Follow-up
Following the initiation of these medications, the patient’s systolic blood pressure remained at approximately 100 mmHg and his heart rate was approximately 80 beats/minute. He did not complain of any tafamidis-related adverse events, including diarrhea, headache, urinary tract infection, and nausea.
At 6 months following the referral to our institute, transthoracic echocardiography showed improvement in the LVEF to 55%, with a left ventricular end-diastolic diameter of 52 mm (Figure 1b). His dyspnea on exertion was ameliorated, and the New York Heart Association functional class was improved to II. The plasma B-type natriuretic peptide concentration was decreased to 104 pg/mL, but the serum troponin I concentration remained at approximately 100 pg/mL. The left ventricular mass index remained at approximately 150 g/m2. The QRS duration was approximately 150 ms.
Discussion
Tafamidis and regression of cardiac hypertrophy
The effect of tafamidis on morphological change remains controversial. 6 Theoretically, tafamidis suppresses further deposition of amyloid in the myocardium and delays the progression of disease. 2 The ATTR-ACT trial consistently showed almost unchanged intraventricular thickness (−0.11 mm) during 30 months of tafamidis treatment compared with a 30-mm increase in the placebo arm. 3 In our patient, markers of disease progression, which comprised the left ventricular mass index, troponin I, and QRS duration, remained unchanged. Therefore, disease progression appeared to be suppressed by 6-month tafamidis therapy.
Neurohormonal blockers for HFrEF due to cardiac amyloidosis
Several studies recently showed the safety of neurohormonal blocker therapy in patients with cardiac amyloidosis, and these were administered to treat other comorbidities, such as arterial hypertension and tachycardia.7,8 In our case, hemodynamics and renal function, which sometimes deteriorate during neurohormonal blocker therapy, were maintained during the therapy. However, the effectiveness of neurohormonal blockers on cardiac amyloidosis has not yet been established. 9
We considered administering neurohormonal blockers to our patient, including beta-blockers and angiotensin-converting enzyme II inhibitors, in addition to mineralocorticoid receptor antagonists because these are guideline-recommended for general HFrEF. 4 However, their effect on secondary cardiomyopathy remains unclear and probably limited. We observed a drastic improvement in systolic function in our patient after administering neurohormonal blockers. Neurohormonal blockers might have facilitated cardiac reverse remodeling under suppression of disease progression by concomitantly administered tafamidis.
Future concerns
Further studies are warranted to validate our proposed strategy of combination therapy using tafamidis and neurohormonal blockers for patients with HFrEF and cardiac amyloidosis. The implications of novel anti-heart failure medications, including sacubitril/valsartan and sodium–glucose cotransporter 2 inhibitors, should also be investigated. In patients with diastolic dysfunction, the optimal heart rate might be relatively high, and aggressive beta-blocker therapy might reduce cardiac output. 10 The optimal medical therapy following the recovery of the LVEF, as found in our patient, also remains a future concern. Patisiran, which is currently used only for hereditary cardiac ATTR, might be more useful to achieve reverse remodeling than tafamidis. 11
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221078484 - Supplemental material for Combination therapy using tafamidis and neurohormonal blockers for cardiac amyloidosis and a reduced ejection fraction: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605221078484 for Combination therapy using tafamidis and neurohormonal blockers for cardiac amyloidosis and a reduced ejection fraction: a case report by Teruhiko Imamura, Toshihide Izumida, Masakazu Hori, Shuhei Tanaka and Koichiro Kinugawa in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
Ethical permission was waived because of the nature of the case report. Written informed consent was obtained from the patient on admission.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
