Abstract
Background:
Numerous first-line immune checkpoint inhibitors (ICI) were developed for patients with advanced non-small cell lung cancer (NSCLC) lacking driver gene mutations. However, this group consists of a heterogeneous patient population, for whom the optimal therapeutic choice is yet to be confirmed.
Objective:
To identify the best first-line immunotherapy regimen for overall advanced NSCLC patients and different subgroups.
Design:
Systematic review and Bayesian network meta-analysis (NMA).
Methods:
We searched several databases to retrieve relevant literature. We performed Bayesian NMA for the overall survival (OS), progression-free survival (PFS), objective response rate (ORR), and treatment-related adverse events (tr-AEs) with a grade equal or more than 3 (grade ⩾ 3 tr-AEs). Subgroup analysis was conducted according to programed death ligand 1 (PD-L1) levels, histologic type, central nervous system (CNS) metastases and tobacco use history.
Results:
For the PD-L1 non-selective patients, sintilimab plus chemotherapy (sinti-chemo) provided the best OS [hazard ratio (HR) = 0.59, 95% confidence interval (CI):0.42–0.83]. Nivolumab plus bevacizumab plus chemotherapy (nivo-bev-chemo) was comparable to atezolizumab plus bevacizumab plus chemotherapy (atezo-bev-chemo) in prolonging PFS (HR = 0.99, 95% CI: 0.51–1.91). Atezo-bev-chemo remarkably elevated the ORR than chemotherapy (OR = 3.13, 95% CI: 1.51–6.59). Subgroup analysis showed pembrolizumab plus chemotherapy (pembro-chemo) ranked first in OS in subgroups of PD-L1 < 1%, non-squamous, no CNS metastases, with or without smoking history, and ranked second in OS in subgroups of PD-L1 ⩾ 1% and PD-L1 1–49%. Cemiplimab and sugemalimab plus chemotherapy ranked first in OS and PFS for squamous subgroup, respectively. For patients with CNS metastases, nivolumab plus ipilimumab plus chemotherapy (nivo-ipili-chemo) and camrelizumab plus chemotherapy provided the best OS and PFS, respectively.
Conclusions:
Sinti-chemo and nivo-bev-chemo were two effective first-line regimens ranked first in OS and PFS for overall patients, respectively. Pembro-chemo was favorable for patients in subgroups of PD-L1 < 1%, PD-L1 ⩾ 1%, PD-L1 1–49%, non-squamous, no CNS metastases, with or without smoking history. Addition of bevacizumab consistently provided with favorable PFS results in patients of all PD-L1 levels. Cemiplimab was the best option in squamous subgroup and nivo-ipili-chemo in CNS metastases subgroup due to their advantages in OS.
Plain language summary
Patients with advance non-small cell lung cancer (NSCLC) lacking driver gene mutations are a group of heterogeneous people. Although numerous therapeutic regimens were developed, the optimal choice for advanced NSCLC patients and specific subgroups is yet to be identified. We conducted a Bayesian network meta-analysis with the currently available data, and performed subgroup analyses according to programed death ligand 1 (PD-L1) levels, histologic type, CNS metastases and tobacco use history. Our key findings were as follows: (1) in non-selective PD-L1 groups, sinti-chemo and pembro-chemo provided the best OS outcome; nivo-bev-chemo and atezo-bev-chemo resulted in the most prolonged PFS; atezo-bev-chemo and pembro-chemo yielded significantly improved ORR; (2) pembro-chemo was favorable for patients in subgroups of PD-L1 < 1%, PD-L1 ⩾ 1%, PD-L1 1–49%, non-squamous, no CNS metastases, with or without smoking history; (3) immunochemotherapies involving anti-PD-1 agents generally exhibited potential advantages over those with anti-PD-L1 drugs; (4) addition of anti-VEGF drugs to immunochemotherapies consistently provided with favorable PFS results in advanced NSCLC patients with or without PD-L1 selection; (5) in patients with squamous NSCLC, cemiplimab and suge-chemo were the optimal drugs for improving OS and PFS, respectively; in patients with non-squamous NSCLC, pembro-chemo provided the best OS, while nivo-bev-chemo, atezo-bev-chemo, sinti-chemo, and pembro-chemo showed comparable advantages in improving PFS; (6) for patients with CNS metastases, nivo-ipili-chemo and camre-chemo provided the best OS and PFS, respectively. Our findings provide evidence for a more precise selection of first-line immunotherapy regimen for advanced NSCLC patients.
Keywords
Introduction
With an estimated 2.2 million new cases and 1.8 million deaths in 2020, lung cancer ranked second in the most commonly diagnosed cancer and first in the cause of cancer death. 1 Comprising a heterogeneous set of tumors, lung cancer can be generally divided into two categories, that is, non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC), which accounts for approximately 85 and 15% of patients, respectively. 2 However, nearly half of NSCLC patients are diagnosed at advanced stage, with limited therapeutic choices and poor prognosis. Although with limited benefit and non-neglectable side-effects, cytotoxic therapies like platinum-based doublet chemotherapy have been the backbone for the management of advanced NSCLC for a long time. Moreover, as driver gene mutations are further identified, individualized targeted therapy of advanced NSCLC has shown promising results. However, there is a considerable number of patients who do not present any driver mutation and require alternative treatments.3–5
It has been 30 years since the cDNA of programed death ligand 1 (PD-1) was identified and extracted from a murine T cell hybridoma and murine haematopoietic progenitor cell line which underwent a classic type of programmed cell death in 1992. 6 Piling researches have looked into the immune-modulating role of PD-1, which was confirmed to interact with its ligands on PD-L1 and programed death ligand 2 (PD-L2). Binding of PD-1 on the surface of T cells to PD-L1 on tumor cells effectively inhibits T cell activation, thus inducing immune escape. 7 Therefore, high level of PD-L1 results in immune avoidance and PD-L1 is expressed in 35–95% of NSCLC patients. 8 Nevertheless, it also provides a novel and promising therapeutic strategy for the treatment of NSCLC.
In recent 10 years, a series of randomized controlled trials (RCTs) have demonstrated significant clinical benefits in front-line treatment for NSCLC with PD-1/L1 inhibitors, either applied in monotherapy or immunochemotherapy. 9 As the new era unfolds, more therapeutic strategies have been investigated in multi-center clinical trials, such as dual immune checkpoint inhibitors (ICI) treatment involving both anti-PD-1/anti-PD-L1 agents and anti-CTLA-4 antibodies as well as combined therapy of anti-PD-1/anti-PD-L1 agents and anti-angiogenesis drugs.10,11 As the novel treatment strategies continued to spring up, there is an urgent need to establish the optimal therapeutic strategy for patients with NSCLC.
To address this issue, we performed a Bayesian network meta-analysis (NMA) of RCTs to evaluate the efficacy and safety of the currently available therapeutic regimens involving first-line PD-1/PD-L1 inhibitors, for NSCLC patients who do not present with driver gene mutations, in an attempt to identify the optimal treatment.
Materials and methods
Search strategy
Relevant RCTs that had evaluated the clinical efficacy of immunotherapy in the treatment of advanced NSCLC were thoroughly searched using the following databases: PUBMED, EMBASE, and Cochrane. The last retrieval was performed on 14th December 2021. The keywords adopted were as follows: all terms related to ‘NSCLC’, atezolizumab, durvalumab, avelumab, cemiplimab, camrelizumab, nivolumab, pembrolizumab, ipilimumab, tislelizumab, sintilimab, toripalimab, all terms related to clinical trial.
Eligibility criteria
Studies fulfilling all the following criteria were included: (1) RCTs; (2) immunotherapies including PD-1/L1 inhibitors as first-line therapy for advanced NSCLC; (3) studies included patients with no epidermal growth factor receptor (EGFR), anapastic lymphoma kinase (ALK), or other sensitive mutations; (4) with at least one of the following reported outcomes: overall survival (OS), progression-free survival (PFS), objective response rate (ORR) and treatment-related adverse events (tr-AEs); (5) studies published in English. For the overlapping reported data obtained from the same group of patients, the latest and most complete reports were included. Duplicate publications, unrelated review articles, editorials, case reports, letters, conference abstracts, animal or cell experiments and studies with incomplete data were excluded.
Inclusion criteria for patients enrolled in the immunotherapy group
As for the included trials in our work, the inclusion criteria for patients enrollment in the immunotherapy group are mostly as follows: (1) patients aged at least 18 years old; (2) Patients who have stage IIIB–IV squamous or non-squamous NSCLC and did not receive any prior systemic treatment; (3) patients without sensitizing EGFR mutation or ALK fusion oncogene; (4) patients should have measurable disease by imaging examination like computed tomography or magnetic resonance imaging; (5) patients should have an Eastern Cooperative Oncology Group (ECOG) performance status of ⩽ 1; (6) patients should have a life expectancy of at least 3 months; (7) patients voluntarily agreed to participate by giving written informed consent.
Data extraction
The references selection was performed independently by two investigators according to the predefined criteria. The information extracted from each study was as follows: the name of study, year of publication, trial number and design, ethnicity involved, sample size (female%), age (years), ECOG score, PD-L1 levels, clinical efficacy indicators including PFS, OS, ORR, and tr-AEs. A third investigator was consulted when there were any disagreements during the research process, and the discrepancies were resolved by discussion.
Quality assessment
The quality assessment of included studies was conducted independently by two investigators. The quality of any selected RCT was evaluated according to the Cochrane Collaboration tool, with a total of six items included: selection bias, performance bias, detection bias, attrition bias, reporting bias, and other bias (Supplemental Figure 1). There are three levels for each item, namely a high, low, or unclear risk of bias. A third investigator was consulted when there were any disagreements during the research process, and the discrepancies were resolved by discussion.
Statistical analysis
All data were included to compare the therapeutic efficacy of different regimens. OS and PFS were considered as primary outcome, while ORR, any-grade tr-AEs and tr-AEs of grade equal to or greater than 3 (grade ⩾ 3 tr-AEs) were secondary outcomes. Given that grade ⩾ 3 tr-AEs were more often reported in the included trials than tr-AEs, only grade ⩾3 tr-AEs were included for safety analysis in our work. Adverse events reported in the included trials were graded by National Cancer Institute Common Terminology Criteria for Adverse Events. Hazard ratios (HR) with 95% confidence intervals (CIs) for OS and PFS, odds ratios (OR) with 95% CIs for ORR and grade ⩾ 3 tr-AEs were either extracted from the original report or calculated with the method provided by Tierney et al. 12
R software (version 4.1.0) with package gemtc was adopted to perform Bayesian NMA in random-effect model. For each outcome, a random-effect consistency model was used and four chains were generated for running 20,000 burn-ins and 50,000 sample iterations per chain simultaneously with one step-size iteration. Moreover, the overall ranking probabilities could be calculated based on surface under the cumulative ranking curves for each endpoint. And the network maps were plotted with STATA software (version 15.1).
Subgroup analysis was conducted according to the PD-L1 expression levels. Statistical inconsistency was evaluated with global inconsistency model and local inconsistency model. For global inconsistency, the DIC values and the statistic inconsistency index (I 2 ) were calculated and compared. An I 2 value less than 25% indicates low heterogeneity. For local inconsistency, node-splitting method was adopted. The R software (version 4.1.0) with package anohe was adopted for heterogeneity assessment.
Results
Study selection
We identified 6076 records from online databases. After excluding 1208 duplicates and 4868 reports for unfit titles and abstracts, a total of 42 studies were reviewed for full-text assessment. Finally, 28 studies were included in our work (Figure 1).

Flowchart of study selection.
Overall, a total of 17,702 patients from 28 studies were enrolled in the following 24 different therapeutic regimens13–40: chemotherapy (chemo), atezolizumab (atezo), cemiplimab (cemi), durvalumab (durva), nivolumab (nivo), pembrolizumab (pembro), atezolizumab plus chemotherapy (atezo-chemo), atezolizumab plus bevacizumab plus chemotherapy (atezo-bev-chemo), bevacizumab plus chemotherapy (bev-chemo), camrelizumab plus chemotherapy (camre-chemo), cemiplimab plus chemotherapy (cemi-chemo), durvalumab plus chemotherapy (durva-chemo), durvalumab plus tremelimumab (durva-treme), durvalumab plus tremelimumab plus chemotherapy (durva-treme-chemo), nivolumab plus chemotherapy (nivo-chemo), nivolumab plus bevacizumab plus chemotherapy (nivo-bev-chemo), nivolumab plus ipilimumab (nivo-ipili), nivolumab plus ipilimumab plus chemotherapy(nivo-ipili-chemo), pembrolizumab plus chemotherapy (pembro-chemo), pembrolizumab plus ipilimumab (pembro-ipili), tislelizumab plus chemotherapy (tisle-chemo), toripalimab plus chemotherapy (tori-chemo), sintilimab plus chemotherapy (sinti-chemo), sugemalimab plus chemotherapy (suge-chemo).
All studies were multi-center phase III RCTs except for Canadian Cancer Trials Group (CCTG) BR-34 and Keynote 021G (both phase II). Of the 28 studies included in our research, there were 1 four-arm trial (Checkmate-227), 4 three-arm trials (Impower 150, MYSTIC, POSEIDON, and RATIONALE-307), and 23 double-arm trials. Only the wild-type groups from Impower 130 and Impower 150 studies were analyzed. The detailed information of included studies was listed in Tables 1 and 2.
Baseline characteristics of selected studies in this meta-analysis.

NSCLC, non-small cell lung cancer; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; tr-AE, treatment-related adverse event.
Baseline characteristics of patients included in this meta-analysis.

BM, brain metastases; ECOG, Eastern Cooperative Oncology Group; NG, not given; PD-L1, programed death ligand 1.
Tumor response in PD-L1 non-selective NSCLC patients
To evaluate the treatment efficacy, OS and PFS were considered as primary outcomes while ORR was of secondary interest. In the advanced NSCLC population without PD-L1 selection, the relevant data was obtained from all 24 regimens [Figure 2(a)].

Comparative network plots on OS, PFS, ORR, tr-AEs, and grade ⩾ 3 tr-AEs of reported treatment regimens in advanced NSCLC patients according to PD-L1 levels. (a) OS, PFS, and ORR in PD-L1 non-selective patients; (b) tr-AEs and grade ⩾ 3 tr-AEs in PD-L1 non-selective patients; (c) OS, PFS, and ORR in PD-L1 < 1% patients; (d) OS, PFS, and ORR in PD-L1 ⩾ 1% patients; (e) OS, PFS, and ORR in PD-L1 1–49% patients; (f) OS, PFS, and ORR in PD-L1 ⩾ 50% patients. The size of the node and the width of the line are proportional to the number of RCTs and comparisons, respectively.
The OS data of each and every kind of therapeutic regimen was obtained and compared [Figure 3(a)]. Patients treated with sinti-chemo experienced the best OS outcome as compared with chemo (HR = 0.59, 95% CI: 0.42–0.83) and pembro-chemo was demonstrated to provide comparable OS advantage with sinti-chemo (HR = 0.93, 95% CI: 0.62–1.36). Generally speaking, immunotherapy strategies involving anti-PD-1 potentially provided better OS results than those containing anti-PD-L1, anti-CTLA-4, and anti-vascular endothelial growth factor (VEGF) drugs. Noteworthy, suge-chemo, a protocol including anti-PD-L1 treatment reported recently, was found to yield significantly favorable OS results as compared with chemo (HR = 0.67, 95% CI: 0.47–0.96). What’s more, cemi (HR = 0.68, 95% CI: 0.49–0.94), nivo-chemo (HR = 0.73, 95% CI: 0.6–0.89), nivo-ipili-chemo (HR = 0.73, 95% CI: 0.57–0.94), and pembro (HR = 0.75, 95% CI: 0.6–0.9) were all found to provide marked OS benefits over chemo alone.

Efficacy and safety profiles for OS, PFS, ORR, and grade ⩾ 3 tr-AEs in PD-L1 non-selective patients based on network consistency model. (a) HRs and 95% CIs for OS (lower triangle in blue) and PFS (upper triangle in orange). HR < 1 indicates better efficacy. (b) ORs and 95% CIs for ORR (lower triangle in blue) and grade ⩾ 3 tr-AEs (upper triangle in orange). OR > 1 indicates better efficacy, while OR < 1 implies better safety.
As for PFS [Figure 3(a)], nivo-bev-chemo and atezo-bev-chemo revealed notably longer PFS as compared with chemotherapy (HR = 0.43, 95% CI: 0.21–0.88; HR = 0.44, 95% CI: 0.26–0.74, respectively), followed by suge-chemo and sinti-chemo (HR = 0.48, 95% CI: 0.3–0.77; HR = 0.51, 95% CI: 0.36–0.72, respectively). Additionally, no significant advantage was noticed on PFS in treatment strategies involving anti-CTLA-4 therapy (ipilimumab and tremelimumab) and durvalumab when compared with chemo alone. A total of 12 regimes were found to provide marked PFS benefits over chemo, durva-trem, and durva.
Regarding ORR [Figure 3(b)], a total of 22 therapeutic regimens provided with ORR data were included for analysis. Atezo-bev-chemo and pembro-chemo yielded significantly improved ORR over chemo (OR = 3.13, 95% CI: 1.51–6.59; OR = 3.09, 95% CI: 2.04–4.69, respectively), followed by cemi-chemo and suge-chemo (OR = 2.62, 95% CI: 1.29–5.3; OR = 2.58, 95% CI: 1.31–5.11, respectively). Similar to OS, potential superiority was observed in immunotherapy strategies involving anti-PD-1 on ORR over those containing anti-PD-L1 and anti-CTLA-4 drugs, with an exception of suge-chemo protocol.
Bayesian ranking profiles (Supplemental Figure 2) suggested that sinti-chemo was most likely to be ranked first in providing the best OS outcome (cumulative probability of 32%), while nivo-bev-chemo had the highest possibility to offer best PFS (cumulative probability of 33%) and atezo-bev-chemo was the best possible treatment for ORR (cumulative probability of 24%). These results were consistent with the pooled results of HRs and ORs, suggesting stability and reliability.
Safety and toxicity
A total of 21 therapeutic regimens provided with tr-AEs data were included for analysis [Figure 2(b)]. The safety profile was determined based on tr-AEs that were equal or greater than grade 3 (grade ⩾ 3 tr-AEs). Pooled results [Figure 3(b)] showed that all mono-immunotherapies resulted in significantly lower grade ⩾ 3 tr-AEs than standard chemotherapy as well as their immunochemotherapy counterparts. No significant difference was noticed among all mono-immunotherapies (atezo, cemi, nivo, pembro, and durva). In addition, the grade ⩾3 tr-AEs of atezo-chemo, tisle-chemo nivo-chemo, pembro-Ipili, camre-chemo were markedly higher than chemotherapy alone. Noteworthy, addition of chemotherapy to dual ICI regimens significantly increased the chance of grade ⩾ 3 tr-AEs as compared with its dual ICI counterparts (nivo-ipili-chemo versus nivo-ipili; durva-trem-chemo versus durva-trem), while no obvious difference was noticed between those two groups: nivo-ipili-chemo versus nivo-chemo, durva-trem-chemo versus durva-chemo, suggesting the potential safety of supplementing anti-cytotoxicT-lymphocyte-associated protein 4 (CTLA-4) antibodies.
Bayesian ranking profiles (Supplemental Figure 2) showed that camre-chemo had the highest probabilities of ranking first in inducing grade ⩾ 3 tr-AEs (cumulative probability of 65%), while ranked last (cumulative probability of 67%).
Subgroup analysis
Based on PD-L1 expression levels
PD-L1 < 1%
In patients with PD-L1 expression levels less than 1%, 16 different treatment strategies were included for subgroup analysis [Figure 2(c)]. As for OS [Figure 4(a)], pembro-chemo showed marked superiority over chemo (HR = 0.62, 95% CI: 0.43–0.89), while other treatments were equivalent. For PFS [Figure 4(a)], advantages over chemo alone were confirmed in nivo-bev-chemo (HR = 0.38, 95% CI: 0.16–0.87), atezo-bev-chemo (HR = 0.49, 95% CI: 0.26–0.88), suge-chemo (HR = 0.56, 95% CI: 0.32–0.99), sinti-chemo (HR = 0.59, 95% CI: 0.38–0.94), pembro-chemo (HR = 0.61, 95% CI: 0.41–0.85), and atezo-chemo (HR = 0.68, 95% CI: 0.48–0.9) groups. Similar to OS, only pembro-chemo yielded higher ORR than chemotherapy (OR = 3.67, 95% CI: 1.69–9.93) [Figure 4(b)].

Efficacy and safety profiles for OS, PFS, and ORR in PD-L1<1% patients based on network consistency model. (a) HRs and 95% CIs for OS (lower triangle in blue) and PFS (upper triangle in orange). HR < 1 indicates better efficacy. (b) ORs and 95% CIs for ORR (lower triangle in blue). OR > 1 indicates better efficacy.
PD-L1 ⩾ 1%
In patients with PD-L1 expression levels equal or more than 1%, 17 different treatment strategies were included for subgroup analysis [Figure 2(d)]. When compared with chemo, only pembro-chemo was associated with significantly improved OS (HR = 0.67, 95% CI: 0.51–0.92), while atezo-bev-chemo (HR = 0.43, 95% CI: 0.23–0.81), tisle-chemo (HR = 0.45, 95% CI: 0.32–0.64), sinti-chemo (HR = 0.46, 95% CI: 0.32–0.65), suge-chemo (HR = 0.46, 95% CI: 0.29–0.74), pembro-chemo (HR = 0.48, 95% CI: 0.37–0.67), camre-chemo (HR = 0.56, 95% CI: 0.33–0.95), and atezo-chemo (HR = 0.61, 95% CI: 0.39–0.96) resulted in remarkedly prolonged PFS [Figure 5(a)]. Among those, atezo-bev-chemo (HR = 0.43, 95% CI: 0.23–0.81) ranked first. As for ORR [Figure 5(b)], pembro-chemo (OR = 2.76, 95% CI: 1.06–6.44) was superior to chemo alone.
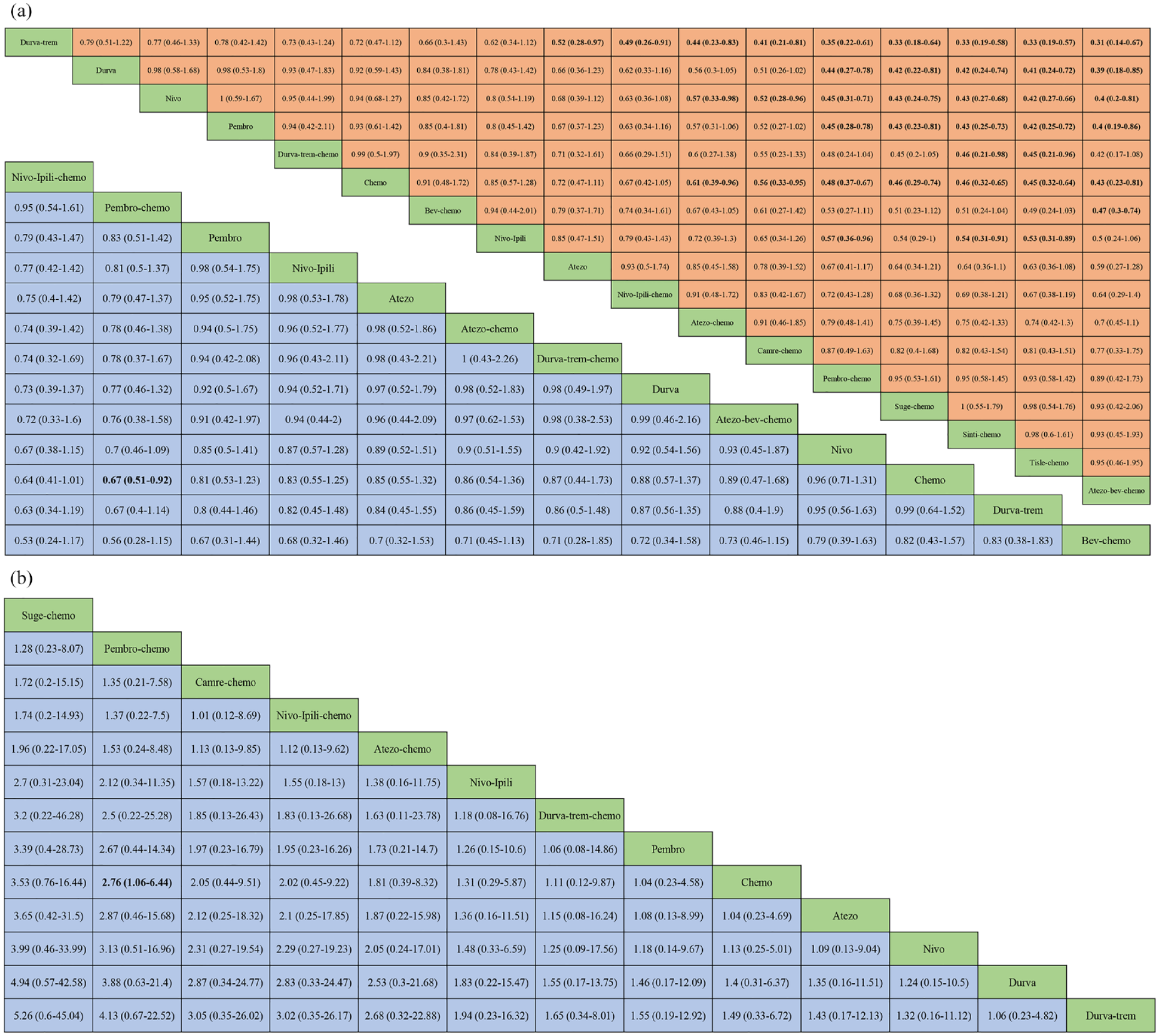
Efficacy and safety profiles for OS, PFS, and ORR in PD-L1 ⩾ 1% patients based on network consistency model. (a) HRs and 95% CIs for OS (lower triangle in blue) and PFS (upper triangle in orange). HR < 1 indicates better efficacy. (b) ORs and 95% CIs for ORR (lower triangle in blue). OR > 1 indicates better efficacy.
PD-L1 1–49%
In patients with PD-L1 levels at 1–49%, 11 different treatment strategies were included for subgroup analysis [Figure 2(e)]. Pembro-chemo provided the best OS (HR = 0.62, 95% CI: 0.41–0.96) and ORR (OR = 3.94, 95% CI: 1.02–15.53) results when compared with chemotherapy [Figure 6(a) and (b)]. Additionally, tisle-chemo yielded the best benefit regarding PFS (HR = 0.52, 95% CI: 0.3–0.91) [Figure 6(a)].

Efficacy and safety profiles for OS, PFS, and ORR in PD-L1 1–49% patients based on network consistency model. (a) HRs and 95% CIs for OS (lower triangle in blue) and PFS (upper triangle in orange). HR < 1 indicates better efficacy. (b) ORs and 95% CIs for ORR (lower triangle in blue). OR > 1 indicates better efficacy.
PD-L1 ⩾ 50%
In patients with PD-L1 expression levels equal or greater than 50%, 20 different treatment strategies were included for subgroup analysis [Figure 2(f)]. The results suggested that patients obtained significantly higher OS benefit from cemi (HR = 0.57, 95% CI: 0.35–0.94), atezo-chemo (HR = 0.65, 95% CI: 0.43–0.98), and pembro (HR = 0.66, 95% CI: 0.47–0.92), as compared with chemo alone [Figure 7(a)]. For PFS, all included therapeutic regimens consistently revealed remarkable improvements in PFS than chemo, except for nivo and bev-chemo. Of those, camre-chemo was associated with the best PFS outcome (HR = 0.14, 95% CI: 0.04–0.44) [Figure 7(a)]. For ORR, the efficacies of pembro-chemo (OR = 4.84, 95% CI: 1.87–12.79), tisle-chemo (OR = 4.1, 95% CI: 1.92–9), sinti-chemo (OR = 3.32, 95% CI: 1.29–8.77), atezo-chemo (OR = 2.98, 95% CI: 1.24–5.76), and cemi (OR = 2.53, 95% CI: 1.13–5.76) were superior to that of chemo [Figure 7(b)]. However, no significant objective response difference was found between these five regimens.

Efficacy and safety profiles for OS, PFS, and ORR in PD-L1 ⩾ 50% patients based on network consistency model. (a) HRs and 95% CIs for OS (lower triangle in blue) and PFS (upper triangle in orange). HR < 1 indicates better efficacy. (b) ORs and 95% CIs for ORR (lower triangle in blue). OR > 1 indicates better efficacy.
Based on histologic type
The efficacies of different treatment regimens were further explored based on histological type. In patients with squamous NSCLC, cemiplimab was most likely to be the optimal choice in improving OS (HR = 0.53, 95% CI: 0.23–1.25), while suge-chemo had the highest possibility to rank first in terms of providing PFS benefit (HR = 0.34, 95% CI: 0.11–1.02), though no significant difference was noticed [Supplemental Figure 3(a)]. In patients with non-squamous NSCLC, however, pembro-chemo provided the best OS outcome as compared with chemotherapy (HR = 0.6, 95% CI: 0.41–0.93), while nivo-bev-chemo, atezo-bev-chemo, sinti-chemo, and pembro-chemo demonstrated similar effects in improving PFS [Supplemental Figure 3(b)].
Based on CNS metastases
For patients presenting with central nervous system (CNS) metastases, nivo-ipili-chemo and pembro-chemo showed significant benefits in improving OS compared to chemotherapy, whereas camre-chemo, cemi, nivo-ipili-chemo, pembro-chemo, and suge-chemo demonstrated better efficacy than chemotherapy in terms of PFS, with camre-chemo yielding the best PFS benefit [Supplemental Figure 4(a) and (b)]. Nevertheless, for those with no CNS metastases, pembro-chemo and sinti-chemo provided the best OS and PFS benefits, respectively [Supplemental Figure 4(a) and (b)].
Based on tobacco use history
For patients who were current or former smokers, nivo-ipili-chemo and pembro-chemo showed significant benefits in improving OS compared to chemotherapy (HR = 0.62, 95% CI: 0.41–0.94; HR = 0.62, 95% CI: 0.41–0.94; respectively), while suge-chemo yielded the best PFS outcome as compared with others (HR = 0.48, 95% CI: 0.32–0.71) [Supplemental Figure 5(a) and (b)]. For patients who never smoked, pembro-chemo (HR = 0.3, 95% CI: 0.09–0.98) and tisle-chemo (HR = 0.33, 95% CI: 0.11–0.8) provided the best OS and PFS benefits, respectively [Supplemental Figure 5(a) and (b)].
Inconsistency and heterogeneity assessment
Evaluation of inconsistency was performed with global inconsistency model and local inconsistency model. Our data suggested that there is no inconsistency existing (Supplemental Tables 1 and 2). Therefore, we conducted this NMA in consistency model. The heterogeneity analysis demonstrated no obvious heterogeneity existed (Supplemental Table 3).
Discussion
With well-designed study protocol, we conducted a comprehensive review and systematic evaluation of the currently available therapeutic regimens for advanced NSCLC. In the hope of providing evidence for clinical practice, we present our key findings as follows: (1) in non-selective PD-L1 groups, sinti-chemo and pembro-chemo provided the best OS outcome; nivo-bev-chemo and atezo-bev-chemo resulted in the most prolonged PFS; atezo-bev-chemo and pembro-chemo yielded significantly improved ORR; (2) pembro-chemo was favorable for patients in subgroups of PD-L1 < 1%, PD-L1 ⩾ 1%, PD-L1 1–49%, non-squamous, no CNS metastases, with or without smoking history; (3) immunochemotherapies involving anti-PD-L1 agents generally exhibited potential advantages over those with anti-PD-L1 drugs. (4) addition of anti-VEGF drugs to immunochemotherapies consistently provided with favorable PFS results in advanced NSCLC patients with or without PD-L1 selection; (5) in patients with squamous NSCLC, cemiplimab, and suge-chemo were the optimal drugs for improving OS and PFS, respectively; in patients with non-squamous NSCLC, pembro-chemo provided the best OS, while nivo-bev-chemo, atezo-bev-chemo, sinti-chemo, and pembro-chemo showed comparable advantages in improving PFS; (6) for patients with CNS metastases, nivo-ipili-chemo and camre-chemo provided the best OS and PFS, respectively.
In our study, three anti-PD-L1 antibodies were adopted in the treatment for advanced NSCLC, namely atezolizumab, durvalumab, and sugemalimab. Based on 28 RCTs, our results indicated a favorable role of anti-PD-1 agents than anti-PD-L1 when combined with standard chemotherapy, which anchored with other previously published findings.41,42 One possible explanation would be the inherent differences between anti-PD-1 and anti-PD-L1. The PD-1 antibodies can simultaneously block the interaction between PD-1 and both PD-L1 and PD-L2, while the PD-L1 antibodies fail to inhibit the binding of PD-1 and PD-L2, thereby resulting in a chance for tumor escape. Nevertheless, sugemalimab (also known as CS1001), exhibits promising efficacy combo with standard chemotherapy in the GEMSTONE-302 trial. Though the OS data is not mature, its positive effects observed in PFS were maintained across all PD-L1 subgroups. Preclinical analysis suggested that sugemalimab, except for its affinity to PD-1, could effectively induce the CD4+ T cell proliferation, upregulate M1 macrophage population, and enhance interferon-γ and interleukin-2 production. 43
CTLA-4, another negative regulator of T cells, also serves as a key inhibitory target for treating a wide range of malignancies. And the anti-CTLA-4 antibodies mainly function via two mechanisms, that is, blockade of the CTLA-4 and B7 interaction and depletion of regulatory T cells through Fc-mediated effector functions. 44 In this work, two anti-CTLA-4 antibodies, namely ipilimumab and tremelimumab, were investigated in both dual ICI interventions and dual ICI plus chemo regimens. According to the pooled analysis, nivo-ipili-chemo provided statistically prolonged OS benefit as compared with standard chemotherapy. In a report by Hanna et al., the use of ICIs including CTLA-4, PD-1, and PDL-1 inhibitors yielded favorable results irrespective of age in patients with advanced cancers. However, the patients’ functional status and the risk of therapy-related toxicity should be carefully evaluated in clinical practice. 45 Also, we observed a potential superiority of dual ICI plus chemo regimens over their dual ICI counterparts in regards to antitumor effects, irrespective of the PD-L1 levels. However, the accompanied increased adverse events should be taken into consideration as well. Moreover, we also noticed that both ICI–ICI and dual ICI plus chemo regimens involving ipilimumab (nivo-ipili, pembro-ipili, nivo-ipili-chemo) showed potential favorable efficacy results as compared with those involving tremelimumab (durva-treme, durva-treme-chemo). One possible explanation could be the broader blockade range of anti-PD-1 agents over anti-PD-L1, another could be attributed to the IgG2 nature of tremelimumab, which is less likely to induce Fc-mediated effector functions as compared with the IgG1 isotype of ipilimumab. 46
In our study, we demonstrated that both bevacizumab-containing regimens (atezo-bev-chemo and nivo-bev-chemo) provided the best PFS, and atezo-bev-chemo also brought about the best ORR for PD-L1 non-selective advanced NSCLC population. Bevacizumab, a humanized monoclonal antibody binds to VEGF and is the first available anti-angiogenic drug. The E4599 study proved that addition of bevacizumab to chemotherapy contributed to significantly prolonged median OS, thus bevacizumab was approved in the first-line treatment for NSCLC. 47 Further clinical researches confirmed the PFS, OS, and ORR benefits of bevacizumab in combination with chemotherapy or targeted therapy.48–51 Apart from the well-known angiogenesis effect, VEGF was demonstrated to play an immunomodulatory role in several steps of cancer immunity circle.52–54 First, by inhibiting the maturation from dendritic cells into antigen-presenting cells, tumor-derived VEGF interfered with the identification and capture of tumor neoantigens, which is the critical first step of cell immunity. Second, with decreased levels of selectins, integrins and adhesion molecules, the adhesion and migration of lymphocytes were hindered. In addition, VEGF also induces T cells apoptosis, leading to further immune tolerance. Third, VEGF is able to facilitate the proliferation of immune suppressive myeloid-derived suppressor cells, which then suppress the antigen-specific T cell production and promote the regulatory T cells development, contributing to immune suppressive microenvironment. The above-mentioned mechanisms might be the scientific rationales for the observed favorable synergistic effects of bevacizumab in combination with immunochemotherapy. In addition, together with ICIs treatment for cancer patients, concomitant medications including antibiotics, corticosteroid, non-steroidal anti-inflammatory drugs, or inactivated influenza vaccine yielded debatable results. 55 Therefore, further well-designed researches are warranted for this topic.
There are several potential limitations of the present meta-analysis which should be taken into consideration when interpreting our data. First, we did not include tumor mutation burden into the subgroup analysis, which might also affect the efficacy of PD-1/L1 inhibitors in advanced NSCLC patients. Second, to minimize the heterogeneity of our work, we only included the wild-type populations from Impower 130 and Impower 150 studies. However, the adverse events occurred in these specific populations without driver gene mutations were not obtained. Therefore, we failed to record and further analyze the tr-AEs induced by addition of bevacizumab. Third, the OS data of some trials including CameL, CHOICE-01, GEMSTONE-302, RATIONALE-304, ONO-4538-52, ORIENT-11, and ORIENT-12 were not mature, which awaits further reports.
Conclusion
Sintilimab plus chemotherapy and nivolumab plus bevacizumab plus chemotherapy were two effective first-line regimens ranked first in OS and PFS for overall patients, respectively. Pembrolizumab plus chemo was favorable for patients in subgroups of PD-L1 < 1%, PD-L1 ⩾ 1%, PD-L1 1–49%, non-squamous, no CNS metastases, with or without smoking history. Addition of bevacizumab consistently provided with favorable PFS results in patients of all PD-L1 levels. Cemiplimab was the best option in squamous subgroup and nivolumab plus ipilimumab plus chemotherapy in CNS metastases subgroup due to their advantages in OS.
Supplemental Material
sj-docx-1-taj-10.1177_20406223231189224 – Supplemental material for Comparison of the profiles of first-line PD-1/PD-L1 inhibitors for advanced NSCLC lacking driver gene mutations: a systematic review and Bayesian network meta-analysis
Supplemental material, sj-docx-1-taj-10.1177_20406223231189224 for Comparison of the profiles of first-line PD-1/PD-L1 inhibitors for advanced NSCLC lacking driver gene mutations: a systematic review and Bayesian network meta-analysis by Fu Wenfan, Xu Manman, Shi Xingyuan, Jiang Zeyong, Zhao Jian and Dai Lu in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
