Abstract
Anaplastic thyroid carcinoma (ATC) is one rare type of thyroid carcinoma without standard systemic treatment for advanced disease. Recent evidence has demonstrated promising efficacy of immune checkpoint inhibitors, particularly those targeting programmed death-1 (PD-1)/programmed death ligand 1 (PD-L1), in a variety of solid tumors. However, there have been no research of immune checkpoint inhibitors plus chemotherapy in ATC. Here, we present the case of a 37-year-old man with metastatic ATC with positive PD-L1 expression, who achieved long-term remission of 34 months after later-line treatment with zimberelimab (a PD-1 inhibitor) and nab-paclitaxel, followed by single-agent zimberelimab maintenance therapy. After three cycles of the combination treatment, the thyroid lesion and the liver metastases shrank dramatically, leading to the best overall response of partial remission. PD-L1 expression may serve as a potential biomarker for tumor response to immune checkpoint inhibitors in ATC. Our review highlights the need for further studies investigating the role of PD-L1 status as biomarker to predict the prognosis of immunotherapy in the treatment of ATC.
Keywords
Introduction
Anaplastic thyroid carcinoma (ATC) is a relatively rare cancer with a morbidity of 1%–2% in patients with primary thyroid carcinoma. 1 As its diagnostic criteria is un-uniform, the evidence for treatment is limited to case series reports and single-institutional retrospective studies. There has been a lack of a standard treatment regimen until now. Furthermore, the prognosis of advanced ATC is significantly poorer than that of early disease. 2
Currently, there are limited treatment options for ATC, and achieving satisfactory outcomes with a single treatment approach is challenging.3,4 Therefore, an urgent need is to develop effective therapy for advanced ATC. In recent years, the combination therapy of programmed death-1 (PD-1)/programmed death ligand 1 (PD-L1) inhibitors immune checkpoint inhibitors (ICIs) with various kinase inhibitors has emerged as a more effective approach in the treatment of ATC.5,6 While, clinical trial results demonstrated that the efficacy of ICIs as a single-agent therapy remained to be enhanced and cannot completely replace the current treatment approaches.7.8 Combination of chemotherapy and ICIs like PD-1/PD-L1 inhibitors,9–11 has demonstrated marked efficacy in various solid tumors; however, to the best of our knowledge, there have been no reports of ICIs plus chemotherapy in ATC.
Here, we report the first case of metastatic ATC with positive PD-L1 expression, which was presented to our hospital and achieved long-term remission for 34 months after later-line treatment with zimberelimab, a novel PD-1 inhibitor and nab-paclitaxel, followed by single-agent zimberelimab maintenance therapy.
Case presentation
A 37-year-old male patient was admitted to a local hospital in November 2019 (Figure 1), complaining of shortness of breath and poor sleep quality. Computed tomography (CT) revealed a thyroid mass with a maximum cross-section of 5.2 × 7.1 cm, which was considered malignant. Multiple nodules were noted in the left lung, with the most dominant measuring 1.3 × 1.0 cm, and distant metastases were confirmed. The patient underwent endotracheal stent implantation in December 2019, and a coarse needle bioptic histopathology revealed a poorly differentiated squamous cell carcinoma of the thyroid. According to the World Health Organization’s 2022 classification of thyroid tumors, squamous cell carcinoma of thyroid was grouped as ATC. 12

Entire treatment process of the patient with poorly differentiated thyroid cancer.
The study was conducted in accordance with the Declaration of Helsinki, but ethical review and approval were waived for this retrospective case report, which did not impact the management of the patient. Informed Consent Statement: Written informed consent was obtained from the subject involved in the case report.
Immunohistochemical staining results were positive for AE1/AE3, pan cytokeratin, Epithelial Membrane Antigen (EMA), Cytokeratin5&6, and Cytokeratin5&14; focal positivity for P63; partial positive for P40; negative for TTF-1, as shown in Figure 2. The tumor proportion score (TPS) of PD-L1 staining (CST E1L3N) was 30% (Figure 3). Positron emission tomography-CT further indicated thyroid cancer (6.7 × 6.1 × 4.4 cm in size) with multiple metastases in lymph nodes, lung, liver, and bone (Supplemental Figure S1).

Pathologic diagnosis of poorly differentiated thyroid squamous cell carcinoma. (a, b) Hematoxylin and eosin staining (magnifications 200× and 400×); (c–h) immunohistochemistry staining: CK5/6 (magnifications 200× and 400×); P40 (magnifications 200× and 400×); P63 (magnifications 200× and 400×).

The TPS of PD-L1 immunohistochemical staining (200×) from the patient.
In January 2020, the baseline CT imaging of thyroid and liver metastases were shown in Figure 4(a) and (f), respectively. Then the patient received first-line treatment with six cycles of paclitaxel liposomes 300 mg (day 1) plus cisplatin 50 mg (day 2–3), 40 mg (day 4), intravenously (IV) every 21 days, and ibandronate for bone metastases. Subsequently, paclitaxel 300 mg (day 1) was administered every 21 days for a total of five cycles (Figure 1). The best tumor response to chemotherapy was achieving stable disease (SD) condition with tumor reduction.

CT imaging of thyroid (a–e) and liver metastases (f–j) before and after zimberelimab plus nab-paclitaxel in the patient with ATC; arrows: the thyroid lesion and liver metastases. (a) Thyroid lesion before the first-line chemotherapy (tumor size: 5.2 × 7.1 cm). (b) Thyroid lesion after three cycles of gemcitabine+S-1 and one cycle of S-1 (tumor size: 3.4 × 1.9 cm). (c) Thyroid lesion before zimberelimab plus nab-paclitaxel (tumor size: 3.3 × 1.9 cm). (d) Thyroid lesion after three cycles of zimberelimab plus nab-paclitaxel (tumor size: 2.3 × 2.2 cm). (e) Thyroid lesion after nine cycles of zimberelimab plus nab-paclitaxel (tumor size: 3.0 × 1.6 cm). (f) Liver metastases before the first-line chemotherapy (tumor size: 4.8 × 5.8 cm). (g) Liver metastases after three cycles of gemcitabine+S-1 and one cycle of S-1 (tumor size: 1.3 × 1.1 cm). (h) Liver metastases after three cycles of gemcitabine+S-1 and one cycle of S-1 (tumor size: 2.8 × 1.8 cm). (i) Liver metastases after three cycles of zimberelimab plus nab-paclitaxel (tumor size: 1.5 × 1.4 cm). (j) Liver metastases after nine cycles of zimberelimab plus nab-paclitaxel (tumor size: 1.5 × 1.4 cm).
Since November 2020, the patient has been treated with gemcitabine 1.6 g plus toripalimab 240 mg (a PD-1 inhibitor) IV (Figure 1). In response to the first dose, the patient developed grade 3 immune-related rash and grade 1 immune-related hepatotoxicity (elevated alanine aminotransferase and aspartate aminotransferase with levels of less than two times the upper limit of normal). Further, these immune-related adverse events (irAE) were resolved with symptomatic therapy, and toripalimab was discontinued.
From March 2021, gemcitabine 1.6 g, IV (day 1) plus S-1 60 mg oral twice daily was administered for three cycles. Subsequent re-examination of the tumor showed SD condition, and S-1 was administered as a maintenance treatment (Figure 1).
In November 2021, the imaging revealed that thyroid lesions remained stable (Figure 4(b) and (c)), while the liver lesions were markedly enlarged from 1.3 × 1.1 to 2.8 × 1.8 cm (Figure 4(g) and (h)), indicating disease progression to the third-line chemotherapy. Therefore, the patient received fourth-line therapy with another PD-1 inhibitor, zimberelimab 240 mg, plus nab-paclitaxel 200 mg IV (day 1), every 2 weeks. After three cycles of the treatment, the thyroid lesion shrank from 3.3 × 1.9 to 2.3 × 2.2 cm (Figure 4(d)), and the liver metastases shrank from 2.8 × 1.8 to 1.5 × 1.4 cm (Figure 4(i)), leading to the best overall response (BOR) of partial remission confirmed by subsequent imaging per RECIST v1.1 (Figure 4(c), (d), (h), and (i)). The patient was kept on this combination regimen for a total of nine cycles, which remained stable (Figure 4(e) and (j)). The patient followed by a single zimberelimab (240 mg, Q2W) maintenance therapy, remained in remission for 34 months until September 2024 (the last follow-up).
During the combined treatment, the patient experienced immune-related endocrine toxicity. In April 2022, he was found to develop hypothyroidism (grade 2 irAE) along with an elevated thyroid-stimulating hormone level of 50.10 μIU/ml (normal range: 0.27–4.20) and decreased free thyroxine level of 6.46 pmol/l (normal range: 12.00–22.00) (Figure 5). Levothyroxine sodium titrated to 150 μg daily was administered from May 2022, and antitumor treatment was withheld. In August 2022, thyroid function improved (Figure 5); therefore, zimberelimab monotherapy was resumed every 3 weeks.
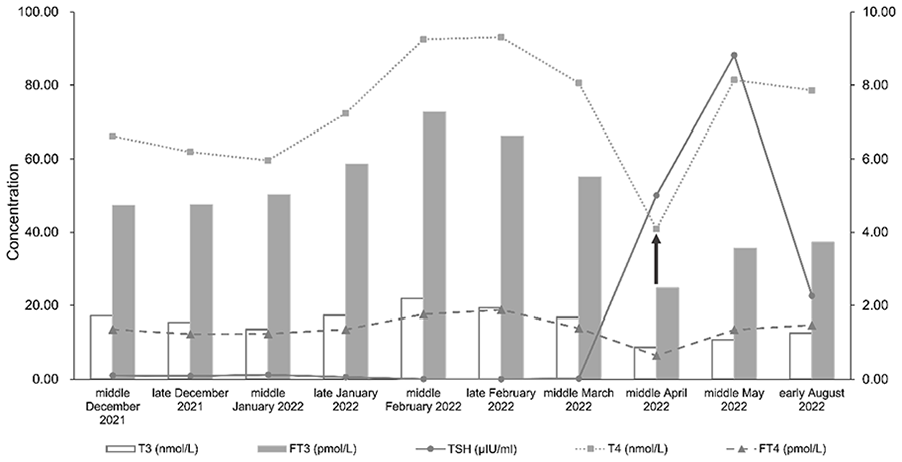
Results of five thyroid function tests of the patient from December 2021 to August 2022. The left vertical axis reflects TSH, T4, and FT4 values, whereas the right vertical axis shows the values for T3 and FT3. T3, triiodothyronine (normal range: 1.30–3.10); T4, thyroxine (normal range: 66.00–181.00); FT3, serum-free triiodothyronine (normal range: 3.10–6.80); FT4, free thyroxine (normal range: 12.00–22.00); TSH, thyroid-stimulating hormone (normal range: 0.27–4.20).
The patient described in this report provided written informed consent for collecting and publishing clinical and molecular data and de-identified images.
Discussion
Histological confirmation of poorly differentiated thyroid cancer (PDTC) and ATC is highly complex and largely dependent on rarity of the disease, evolving diagnostic criteria, experience, and subjectivity of pathologists. Therefore, true boundaries between these two subtypes remain unclear. This case was initially diagnosed as a poorly differentiated squamous cell carcinoma of the thyroid in 2019. However, according to the World Health Organization’s 2022 classification of thyroid tumors, 12 squamous cell carcinoma of thyroid is now considered a morphologic pattern/subtype of ATC.
Owing to the rarity and heterogeneity of ATC, no relevant guidelines or consensus on its treatment exists. ATC is highly aggressive and a median survival duration of approximately 5 months and a 1-year overall survival rate of 20%.13,14 The present patient was at an advanced stage with multiple distant metastases at diagnosis and was ineligible for surgery and radiotherapy with a poor life expectancy.
The patient received six cycles of paclitaxel plus cisplatin as the first-line regimen, followed by five cycles of paclitaxel alone to minimize cisplatin toxicity, and remained in SD with a final PFS of 10 months in the first-line setting.
In recent years, led by anti-PD-1/PD-L1 antibodies, ICIs with or without chemotherapy have demonstrated promising efficacy in a variety of solid squamous cell carcinomas.15–18 Currently, PD-L1 is widely recognized as a validated biomarker for selecting patients who may benefit from anti-PD-1 or anti-PD-L1 antibodies in various solid tumors. 19 Studies have shown that lymphocyte infiltration in ATC was significantly higher than that in differentiated thyroid carcinoma, 20 the expression of PD-L1 in ATC was higher than that in other subtypes.21–23 A recent study involving 15 ATC patients and 13 PDTC patients discovered that PD-L1 expression is prevalent in ATC, being present in 60% of cases, 22 and its expression is higher compared to PDTC, suggesting that ICI may emerge as a novel treatment modality for selected patients with PDTC.
One retrospective study has reported the antitumor activities of pembrolizumab and lenvatinib in patients with ATC (n = 6) and PDTC (n = 2). The results showed that four patients with ATC achieved complete response (CR), while two achieved PR among the patients with PDTC. Notably, all patients with long-term (>2 years) or CRs had either increased tumor mutational burden or PD-L1 TPS >50%. 24 In a phase II trial involving 42 predominantly metastatic ATC patients, PD-1 inhibitor spartalizumab was administered. The findings revealed that patients with PD-L1 positive tumors exhibited significantly higher ORR and longer median OS compared to those with PD-L1 less than 1%. 25 Similarly, in a phase II KEYNOTE-158 study that enrolled patients with papillary or follicular thyroid carcinoma, the ORR was slightly higher in PD-L1 positive disease (n = 46) than in those with negative disease (n = 53). 26 These results suggested that PD-L1 status may serve as a potential predictive biomarker for thyroid carcinoma. In addition, in vivo studies showed that the PD-L1 antibody effectively suppresses tumor growth in cases of ATC with PD-L1 expression.27,28
A study conducted immune scoring on 505 patients with thyroid cancer, and the results showed that the degree of differentiation of thyroid cancer was significantly negatively correlated with immunosuppressive markers CTLA-4 and PD-L1. Compared with wild-type tumors, these immunosuppressive markers were more frequently found in BRAF V600E-mutated tumors. 29 The elevated expression level of PD-L1 has been confirmed in some solid tumors and is associated with poor prognosis.30–33 Tumor cells expressing PD-L1 can inhibit the activation of T cells in the tumor microenvironment, thereby protecting tumor cells from the influence of immune responses. Massari et al. 34 analyzed the prognostic potential of PD-L1 in 185 PTCs and 66 benign nodules, and the results showed that the increased expression level of PD-L1 in thyroid cancer was associated with a higher risk of recurrence and shorter disease-free survival. Especially for ATC, amplification of immune escape genes usually occurs. 35 These results suggest that targeting PD-L1 may be an effective treatment method for advanced metastatic thyroid cancer.
In this case, the PD-1 inhibitor toripalimab combined with gemcitabine was attempted as the second-line regimen for PD-L1 positive disease, and toripalimab was discontinued due to severe irAEs after only one treatment cycle. Subsequently, third-line chemotherapy with gemcitabine plus S-1 was initiated, and the disease progressed 8 months later.
It was challenging to determine the fourth line of treatment. One reason was that the disease had progressed to two lines of chemotherapy (paclitaxel, gemcitabine, and cisplatin), and the other was that the patient experienced severe irAEs induced by a PD-1 inhibitor. It is noteworthy that first-line treatment with paclitaxel liposomes and cisplatin resulted in a long PFS of 10 months, which suggested that the tumor was sensitive to paclitaxel. Furthermore, we believe that the prior treatment duration of PD-1 inhibitors was too short to evaluate the tumor response to immunotherapy. Therefore, the combination of nab-paclitaxel and another PD-1 inhibitor, zimberelimab, was reconsidered in November 2021 for the patient.
Zimberelimab, a novel fully humanized anti-PD-1 antibody, was selected because it had a history of no subject experiencing grade 3 or 4 treatment-related rashes in the phase II study for relapsed/refractory classical Hodgkin lymphoma. 36 In addition, a phase II clinical trial evaluating zimberelimab monotherapy for cervical cancer with positive PD-L1 expression in the second or subsequent-line setting demonstrated a remarkable ORR of 27.6%, unparalleled within its therapeutic class. 37
The patient received nine cycles of this combination therapy, with the BOR of partial remission. This regimen was withheld because of grade 2 immune-related hypothyroidism, which was well managed with thyroxine. For hypothyroidism, the general approach is symptomatic supportive treatment. 38 Close monitoring of thyroid function and appropriate thyroid hormone replacement therapy are typically employed. 38 These measures aim to manage the symptoms and maintain normal thyroid function without necessarily requiring discontinuation of ICI treatment. Single zimberelimab was resumed as maintenance therapy. The combined therapy of zimberelimab and nab-paclitaxel achieved a PFS of 34 months by September 2024.
PD-L1 expression shows variability among different tumors, which can impact treatment outcomes.33,39,40 This heterogeneity poses challenges in accurately predicting response to immunotherapy based solely on PD-L1 status. Variability in PD-L1 expression within ATC tumors, along with tumor heterogeneity and the complex dynamics of the immune response, make it difficult to solely rely on PD-L1 for diagnosis or prognosis.21–23 Additionally, the lack of standardized PD-L1 testing methods contributes to inconsistent results. Its findings may not be generalizable to the broader population in this case report. To establish more robust and widely applicable conclusions, future research involving larger sample sizes and multiple cases is essential. Additionally, other emerging biomarkers or molecular factors might offer complementary information for ATC treatment strategies. Incorporating these into future studies could enhance our understanding and potentially improve patient outcomes.
Conclusions
To our knowledge, this is the first case in which zimberelimab plus chemotherapy successfully controlled PD-L1 positive ATC with a long response duration in a later-line setting. Our case suggests that PD-L1 may be a biomarker for ICIs therapy plus chemotherapy in ATC and can be integrated into decision-making for precision therapy in clinical practice.
Supplemental Material
sj-docx-1-sco-10.1177_2050313X241313084 – Supplemental material for Advanced anaplastic thyroid carcinoma with positive expression of PD-L1 response to immune checkpoint inhibitors: A case report
Supplemental material, sj-docx-1-sco-10.1177_2050313X241313084 for Advanced anaplastic thyroid carcinoma with positive expression of PD-L1 response to immune checkpoint inhibitors: A case report by Shanmin Fan, Yang Yuan, Yanfang Su and Die Sang in SAGE Open Medical Case Reports
Footnotes
Acknowledgements
None.
Author contributions
Conceptualization: D.S.; Methodology: S.M.F.; Investigation: Y.Y; Writing—original draft preparation and review and editing: Y.F.S. All authors have read and agreed to the published version of the manuscript.
Data availability statement
All of the data are available upon request to the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
