Abstract
Background:
Patients with psoriasis have a significant disease burden throughout the life course. Nevertheless, the lifetime risk and disease burden of psoriasis across the entire lifespan is rarely quantified in an easily understandable way.
Objective:
To estimate the cumulative incidence rate, life expectancy, loss-of-life expectancy, and lifetime healthcare expenditures for incident psoriasis.
Design and methods:
Using real-world nationwide data from the National Health Insurance Research Database of Taiwan for 2000–2017, along with the life tables of vital statistics, we estimated cumulative incidence rate, life expectancy, loss-of-life expectancy, and lifetime healthcare expenditures for those with psoriasis using a semi-parametric survival extrapolation method.
Results:
A total of 217,924 new psoriasis cases were identified. The lifetime risk of psoriasis in patients aged 18–80 for both sexes decreased in Taiwan with a cumulative incidence rate of 7.93% in 2000 to 3.25% in 2017. The mean (±standard error) life expectancy after diagnosis was 27.11 (± 1.15) and 27.14 (±1.17) years for patients with moderate-to-severe psoriasis and psoriatic arthritis, respectively. Patients with moderate-to-severe psoriasis and psoriatic arthritis had a mean (±standard error) loss-of-life expectancy of 6.41 (±1.16) and 6.48 (±1.17) due to psoriasis, respectively. Male patients have higher lifetime and annual lifetime healthcare expenditures than female. Mean life expectancy, loss-of-life expectancy, and lifetime cost were relatively higher for younger patients.
Conclusion:
Among psoriatic patients, patients with moderate-to-severe psoriasis and psoriatic arthritis had substantial years of life lost, particularly for younger patients. Our results provide a reliable estimation of lifetime disease burden, and these estimates will help health authorities in cost-effectiveness assessments of public health interventions and allocation of services resources to minimize loss-of-life expectancy, and lifetime healthcare expenditures in patients with psoriasis.
Introduction
Psoriasis entails extensive disease burdens throughout the life course. 1 Although previous studies have quantified the burden of psoriasis, they are mostly cross-sectional data rather than evaluation over a patient’s lifetime.2–9 Currently, there is no cure for psoriasis and as such the disease requires lifelong monitoring. In terms of physical burden, population-based studies have found an increased mortality risk in psoriatic patients, with an even higher risk for individuals with moderate-to-severe psoriasis (MSP).2–6 However, the majority of these studies used the mortality rate ratio (hazard ratio) to measure difference in mortality rates.2–6 Such measures are incomprehensible to lay people and some health authority staff, and incompletely represents life course disease burden of psoriasis.
Alternatively, cumulative incidence rate (CIR), life expectancy (LE), and loss-of-life expectancy (LoLE) and lifetime healthcare expenditures (HEs), can reflect the disease burden on life span10–12 and is more intuitive to the general public. 13 Nevertheless, data on the number of years of life lost among patients with psoriasis compared with the general population are limited.5,14 Furthermore, studies that have calculated years of life lost did so based on the difference in mean age at death between patients with psoriasis and either the provincial/national population or an inadequately matched reference group.5,14 Moreover, these studies measured the age at death from a prevalent cohort.5,14 In addition, potential confounders for estimates of LoLE, including differences in age, sex, subject identification period, or follow-up period between psoriasis and reference groups are not addressed in these prior studies.5,14
Psoriasis is not only a skin disease and associated with a variety of medical comorbidities, including psoriatic arthritis (PsA), cardiovascular diseases, metabolic syndrome, and so on. 4 Moreover, among dermatologic conditions, psoriasis has the highest association with psychiatric illness, including mood, anxiety, and personality disorders. 15 Due to comorbidities and the chronicity of psoriasis, long-term management of psoriasis incurs substantial cumulative expenses due to direct and indirect costs. Previous studies revealed that patients with psoriasis have significantly greater healthcare utilization and expenditures than those without psoriasis.16–18 However, as with LoLE, much of the available literature used a cross-sectional design and prevalence-based estimates to calculate the annual cost of HE over a relatively short follow-up period.16–19 Accrual of costs likely varies over a lifetime, and such cross-sectional estimates cannot fully capture lifetime HE after the diagnosis of psoriasis. Therefore, in this study, we applied the semi-parametric method by Hwang and Wang 20 in a real-world nationwide longitudinal cohort of incident cases of psoriasis with up to 18 years of follow-up to estimate the lifetime risk, LE, LoLE, and associated HE attributable to psoriasis. Our estimation of LE and LoLE from incidence patients provides a more reliable and easily understandable quantification of disease-specific impacts across the entire lifespan than those based on prevalent cohorts.
Materials and methods
Study population and data sets
This is a nationwide population-based cohort study from 2000 to 2017. We retrieved data from the National Health Insurance Research Database (NHIRD). 21 The NHIRD contains healthcare costs and detailed claim records of approximately 23 million beneficiaries (over 99% of the Taiwanese population) participating in the Taiwan National Health Insurance program. 21 Psoriasis patients were identified by a diagnosis of psoriasis [International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes 696.0, 696.1, 696.8 or ICD-10 codes L40.0, L40.1, L40.2, L40.3, L40.4, L40.8, L40.9, L40.50, L40.51, L40.52, L40.53, L40.54, L40.59, M07.0, M07.1, M07.2, M07.3, M09.0] during ambulatory visits or inpatient care at least twice between January 1, 1998 and December 31, 2017. To identify incident cases of psoriasis, we excluded those diagnosed with psoriasis in 2 years prior to 2000. In subgroup analysis, the psoriasis cohort was further stratified by sex, age, disease severity, and presence of PsA. For disease severity, we stratified patients with psoriasis into MSP and mild psoriasis. The criterion for MSP was requiring systemic antipsoriatic therapy, phototherapy, or both for more than 3 months in 6 months of follow-up; otherwise, patients were considered to have mild psoriasis.22–24 We also classified patients into groups with and without PsA. PsA was defined as at least two visits with a diagnosis of PsA based on ICD-9-CM 696.0 or ICD 10 code L40.50, L40.51, L40.52, L40.53, L40.54, L40.59, M07.0, M07.1, M07.2, M07.3, and M09.0.
Estimation of the CIR of psoriasis
We estimated age- and sex-specific incidence rates (IRs) using the age- and sex-specific number of new psoriasis cases from NHIRD for each calendar years as the numerator, and the corresponding populations at risk abstracted from vital statistics as the denominator. The CIR for psoriasis was calculated from ages 18 to 80 (CIR18–80) to estimate the lifetime risk of psoriasis according to the following formula:
where i = 18–19, 20–24, . . . , 75–80, IR i is the incidence rate for the ith age group, and Δti = the ith age group range.
Statistical analyses
Estimation of LE and LoLE
Survival functions of psoriatic patients were estimated by the Kaplan–Meier method, beginning with the first date of the diagnosis of psoriasis and followed survival until the last date of follow-up, the date of death or December 31, 2017, whichever came first. For subgroup analysis, follow-up began from the diagnosis of MSP or PsA for patients with MSP and PsA, respectively. Estimates of lifetime survival function beyond the follow-up period were conducted using the semi-parametric survival extrapolation method proposed by Hwang and Wang 20 and Hwang et al. 25 and validated mathematically by Fang et al. 26 First, for each patient with psoriasis, we generated an age, gender, and calendar year of diagnosis-matched referents according to the life table of the general population from the Taiwan National Vital Statistics using the Monte Carlo method 20 and estimated their survival functions. Second, the survival ratio between the cohorts of psoriasis and the referents at each time point was calculated. We then logit-transformed the survival ratio. Third, we fitted the logit-transformed relative survival curve into a restricted cubic splines model to extrapolate the survival curve for one extended month. Fourth, by applying a rolling extrapolation algorithm, we extrapolated the survival function of patients with psoriasis and referents to their lifetimes. LE of patients with psoriasis, MSP, and PsA was defined as the summation of the area under the estimated lifetime survival curve after the diagnosis of psoriasis, MSP, and PsA, respectively. LoLE was calculated by subtracting the LE of the psoriasis cases from that of the corresponding age- and sex-matched reference population. The calculations of LE and LoLE were done using the R package, iSQoL2.
Estimation of lifetime HEs
We collected reimbursement data from the NHIRD to calculate the mean monthly cost for each psoriatic patient after diagnosis by dividing the total healthcare expenditures of all survived patients by the total number of survived patients in that month. Next, we summed up the product of average monthly cost and the corresponding monthly survival rate over a lifetime to obtain the lifetime cost. Annual NHIRD expenditures were adjusted with the consumer price index (CPI), and the costs of extrapolated months were adjusted with an annual discount rate of 3%. Monetary value was expressed in 2017 US dollars (1 US = 30.44 TWD).
Results
CIR of psoriasis
A total of 217,924 new psoriasis cases were identified during 2000–2017. The characteristics of study population are shown in Supplementary Table S1. The CIR in the general Taiwanese population ages 18–80 years (CIR18–80) shows a declining trend during 2000–2017 in all psoriasis patients and in the male and female subgroups. The CIR18–80 in all psoriasis patients decreased from 7.93% in 2000 to 3.25% in 2017 (Supplementary Table S2).
Estimation of LE and LoLE
The overall mortality rate was 14.7 per 1,000 person-years in psoriasis patients. The estimated overall mean LE after psoriasis diagnosis was 34.38 years, and the estimated mean LoLE was 1.19 years. The survival function in patients with mild psoriasis did not differ significantly from general population reference (Supplementary Figure S1a). However, the survival rate in patients with MSP was significantly lower than in patients with mild psoriasis (log-rank test p-value = 0.005). Table S3 shows the leading ten causes of death in patients with MSP. Thus, we next focused on the LE and LoLE in MSP patients. Among MSP patients, the mean LE and LoLE were 27.11 and 6.41 years, respectively. Stratified by age and gender, the mean LE and LoLE after diagnosis of MSP were 30.6 and 6.42 years, respectively, for females. Male patients with MSP had LE of 26.83 years and LoLE of 4.97 years (Figure 1 and Table 1). The younger the patients, the greater the LE and LoLE.

Moderate-to-severe psoriasis leads to mean loss of life expectancy.
Life expectancy, loss-of-life expectancy, lifetime healthcare expenditures, and cost per life-year of patients with moderate-to-severe psoriasis, stratified by sex and age at diagnosis.
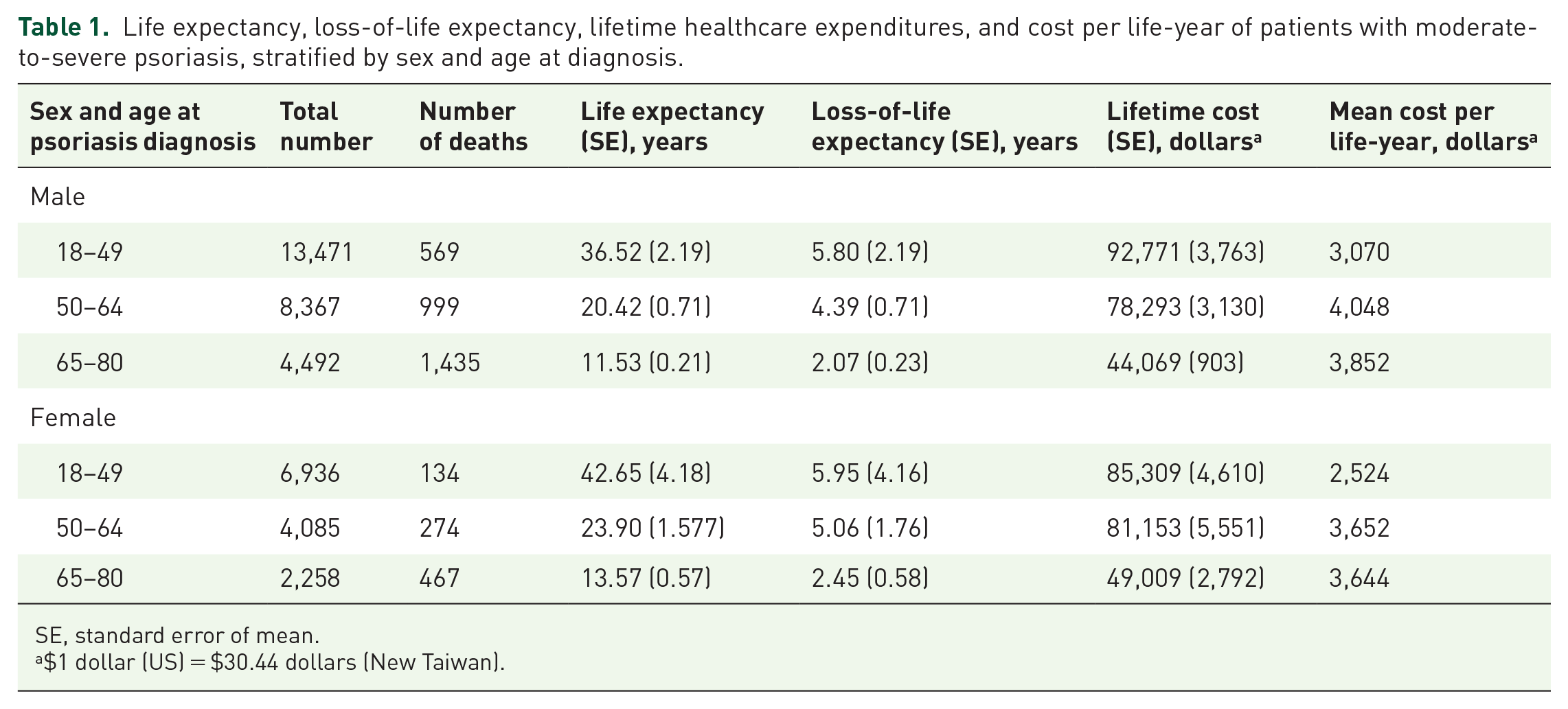
SE, standard error of mean.
$1 dollar (US) = $30.44 dollars (New Taiwan).
We next stratified patients by the presence of PsA. There were no statistical significant differences between the survival functions of patients without PsA and general population (Supplementary Figure S1b). However, patients with PsA had an expected loss of 6.48 life-years compared with the general population. The main causes of mortality in PsA patients are presented in Supplementary Table S3. Figure 2 and Table 2 summarize the LE and LoLE of patients with PsA, stratified by age and sex. Similar to MSP patients, in patients with PsA, the LE and LoLE decreased gradually as age increased, although the trend is less pronounced between male patients aged 18–49 and 50–64 (Figure 2 and Table 2).

Psoriatic arthritis: mean life expectancy and expected loss-of-life expectancy.
Life expectancy, loss-of-life expectancy, lifetime healthcare expenditures, and cost per life-year of patients with psoriatic arthritis, stratified by sex and age at diagnosis.

SE, standard error of mean.
$1 dollar (US) = $30.44 dollars (New Taiwan).
Lifetime healthcare expenditures and cost per life-year
The estimated total lifetime cost and cost per life-year was $79,205 and $3,327 for patients with MSP, and $54,716 and $2,314 for patients with PsA. Males with MSP had higher lifetime cost ($79,621 versus $71,908, p < 0.01) and cost per life-year than did females ($3,408 versus $2,740, p < 0.01). MSP patients aged 18–49 had the highest lifetime expenditures (Table 1). Among patients with MSP and PsA, lifetime expenditures decreased with advanced age, but healthcare cost per life-year showed an increasing trend along with increasing age for both male and female patients (Tables 1 and 2).
Data validation
To validate our extrapolated data, we used the first 9 years of data to extrapolate another 9 years and then compared these data with the actual Kaplan–Meier estimate using all 18 years of data. Relative bias was calculated by taking the difference between the extrapolated estimate and the actual Kaplan–Meier estimate, then dividing by actual Kaplan–Meier estimate. All relative biases were < 5%, indicating that our estimates are relatively accurate (Supplementary Table S4.
Discussion
Previous epidemiologic studies have provided the prevalence, IR, or annual IR of psoriasis, which only estimates psoriasis risk at a particular time point or over a short period. Information on risk of psoriasis occurrence across the lifespan has been lacking. Here, we calculated the CIR of psoriasis in the population aged 18–80 years as an estimate of lifetime risk, based on the mean LE of Taiwanese in 2019 of 80 years. Thus, CIR18–80 can be interpreted as the lifetime probability of developing psoriasis at some point during life span for Taiwanese, taking into account the competing risk of mortality. We estimate LoLE using a general population, which was matched to the psoriasis cohort with regard to age, sex, and calendar year as a comparison group. Thus, estimates are not affected by the confounding effects of age, sex or a possible time trend (such as trends in medical care advancement or medical costs over time). Finally, by including incidence cases of psoriasis only, the actual survival and health expenditure costs are not confounded by duration of illness and different disease stages. Moreover, the estimate can be more accurately representative of the LE after diagnosis of psoriasis and the LoLE conferred by psoriasis
Although the exact reasons underlying the declining lifetime risk of psoriasis in this study are unclear, several behavioral and environmental factors, such as smoking and alcohol intake, have been implicated in the initiation of psoriasis and may be possible causes.27,28 In support of this, a previous population study noted that cigarette smoking and exposure to secondhand smoke declined from 2001 to 2013 in Taiwan, because of the legislation of Tobacco Hazards Prevention Act in 1997. 29 A national survey in Taiwan also found a significant decrease in the population’s prevalence of alcohol use and binge drinking in recent years. 30 Decreased smoking habits and alcohol intake may partially account for the declining trend in lifetime CIR of psoriasis in Taiwan. Consistent with our results, one recent study revealed a significant decreasing trend in the incidence of psoriasis in Taiwan from 2002 to 2016. 31 Another UK population-based cohort study also showed psoriasis incidence declined from 2008 to 2013. 32 Previous studies reported that the incidence of psoriasis varied from 23 to 65 per 100,000 person years in Taiwan.31,33,34 The difference in the IR of psoriasis between our study and other studies in Taiwan31,33,34 is because our study presented lifetime risk (CIR), but other studies showed annual IR of psoriasis.31,33,34
Accumulating evidence suggests that the prevalence of cardiovascular events is a leading cause of the higher mortality rates among patients with psoriasis.2,4,35 Similarly, our results also showed that cardiovascular diseases are a leading cause of death for psoriasis patients. Moreover, our study showed that LoLE was greater in younger patients with MSP. This finding agrees with previous studies that found that the relative risk of cardiovascular events, cardiovascular death and all-cause mortality was highest in younger patients with MSP.35,36 The higher LoLE in younger patients with MSP may reflect, in part, that an age-dependent increase in overall cardiovascular risk may attenuate the psoriasis-related impact on LoLE in the elderly. Studies examining mortality in patients with PsA have yielded inconsistent results.36–43 Some studies have reported that an increased overall mortality in PsA patients compared with the general population.36–39 In contrast, the University of Toronto cohort, 40 the Denmark nationwide cohort, 41 the Olmsted County cohort 42 and the UK THIN database 43 did not show elevated mortality in PsA patients. Prior studies estimating the number of life-years lost in PsA patients are limited. Ali et al. 44 investigated a PsA cohort from the University of Toronto’s PsA clinic and reported an LoLE of 2.99 years, calculated based on the unadjusted standardized mortality ratios (SMRs). 44 Using standard single-decrement life-table analysis, Mok et al. 45 showed that the LoLE was 6.5 years for female patients with PsA. Similar to the study by Mok et al., 45 our results suggest that patients with PsA have a reduced LE of 6.48 years compared with the general population. Discrepancies in LoLE for those with PsA among studies may stem from differences in study design, study populations and control populations. In contrast to the large-scale, nationwide population of our study, most of studies to date have been confounded by small sample sizes with few events, the potential for selection bias in clinic-/hospital-based studies and the use of SMR to compare with census statistics rather than directly comparing the PsA with internal controls.36–39
The younger the age of psoriasis diagnosis, the higher the LE and the lifetime healthcare expenditure predicted from our data. The greater healthcare expenditure is presumably because early onset cases with longer survival after diagnosis of psoriasis, lead to a higher accumulated lifetime healthcare cost. Although prior studies evaluated the healthcare expenditure in patients with psoriasis, direct comparison of our results with data from previous studies may not be appropriate because many of these studies only estimated the cost over a short period instead of over the entire lifespan.16–19 Moreover, the present study and prior studies included patients of different ages and sex, and examined different calendar periods.16–19 Finally, previous studies did not investigate incident psoriasis and estimate the cost after the development of psoriasis exclusively.16–19 Our study also found that the healthcare costs per life-year were higher among males than females with MSP, which might be explained partly by a higher risk of developing a severe course of psoriasis in men than in women.28,46,47
Some limitations of our study must be acknowledged. First, this study included only direct medical costs and did not include out-of-pocket money or lost productivity costs due to psoriasis or premature death. This has likely led to an underestimate of the total cost of psoriasis to society as a whole. Second, we used treatment with systemic therapies as a surrogate for identification of patients with MSP; however, prior research has demonstrated the reliability and validity of using this method for grouping MSP.22–24,48,49 Third, the lifetime extrapolation was based on currently available data from 2000 to 2017. The active development and adoption of newer technologies in recent years for psoriasis management were not taken into consideration, which probably led us to underestimate LE and overestimate the LoLE. Fourth, our results on HE may not be generalized to other health systems. Because of the government-run single-payer National Health Insurance that is effective for containing costs in Taiwan, 50 Taiwan’s total HE as a percentage of gross domestic product and per capita HE are significantly lower compared with those in the United States, Japan, Canada, the United Kingdom, Germany, and France.50,51 Fifth, NHIRD did not contain information on the types of psoriasis that identified by ICD-9-CM code (696.0, 696.1, 696.8) before 2016. Compared with psoriasis vulgaris that constitutes the main population in this study, pustular psoriasis may contribute with a different weight to the various aspects that have been studied. 52
Conclusion
In conclusion, the lifelong health impacts (LE after diagnosis, LoLE) and financial burdens on patients with MSP and PsA remain high. Patients with mild psoriasis and patients without PsA did not differ significantly in survival from the general population. The mean LE, LoLE, and lifetime cost were higher for patients at younger age. Our study provides a reliable and easily understandable estimation of the lifetime burden of psoriasis on the survival and health costs at both individual and society levels. These findings can be integrated with quality of life data to generate disability-adjusted life-years in the future. In addition to quantify the lifetime burden between diseases, countries, regions, and population groups, these measures can also evaluate the impact of specific public health interventions to guide health authorities and policy makers for efficient allocation and planning of services resources, financial preparation, and research support to minimize LoLE and HE in patients with psoriasis.
Supplemental Material
sj-docx-1-taj-10.1177_20406223231168488 – Supplemental material for Lifetime risk, life expectancy, loss-of-life expectancy, and lifetime healthcare expenditures for psoriasis in Taiwan: a nationwide cohort followed from 2000 to 2017
Supplemental material, sj-docx-1-taj-10.1177_20406223231168488 for Lifetime risk, life expectancy, loss-of-life expectancy, and lifetime healthcare expenditures for psoriasis in Taiwan: a nationwide cohort followed from 2000 to 2017 by Hsien-Yi Chiu, Joung-Liang Lan and Ying-Ming Chiu in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
The authors are grateful to Health Data Science Center, China Medical University Hospital for providing administrative, technical, and funding support, and Ya-Chu Yang for data analysis. They thank the staff of Department of Medical Research, National Taiwan University Hospital Hsin-Chu Branch for their assistance in study design, statistical analysis, and providing careful review, and insightful comments regarding the articles.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

