Abstract
Background:
Consensus among dermatologists and rheumatologists in the diagnosis and assessment of musculoskeletal diseases in psoriasis (PsO) patients is needed. This study assesses characteristics of musculoskeletal pain in patients with PsO for the presence of psoriatic arthritis (PsA) and evaluation of a novel 16-item visual instrument (PsA-Disk).
Methods:
Data were collected from eight dermatological/rheumatological centres across Italy. Patients with PsO completed PEST (Psoriasis Epidemiology Screening Tool) and PsA-Disk questionnaires during the first visit. A rheumatological visit was performed to confirm the presence of PsA. Both validity and reliability of PsA-Disk were assessed.
Results:
A total of 573 patients with PsO were examined at the first visit, and 120 (21%) were diagnosed with PsA. Patients with PsA compared with patients with PsO (n = 119) presented statistically significant differences for: nail involvement, PEST score ⩾3, higher erythrocyte sedimentation rate (ESR), Nail Psoriasis Severity Index (NAPSI)-feet, NAPSI-(hands + feet) and PsA-Disk scores (73.9 ± 32.1 versus 58.1 ± 39.8, p < 0.001). Patients with PsA with knee arthritis had higher PsA-Disk scores (98.4 ± 26 versus 71.5 ± 31.9, p = 0.006) that were also correlated with number of swollen (r = 0.2, p < 0.05) and tender joints (r = 0.24, p = 0.021), patient (r = 0.4, p < 0.001) and physician-pain-visual analogue scale (VAS; r = 0.33, p < 0.001), patient global assessment (PGA)-VAS (r = 0.23, p = 0.025), physician-health assessment questionnaire (HAQ; r = 0.38, p = 0.011), Disease Activity Score (DAS)-44 (r = 0.25, p = 0.023) and Disease Activity in Psoriatic Arthritis (DAPSA; r = 0.31, p = 0.005). The instrument had excellent reliability in terms of internal consistency (Cronbach’s alpha = 0.90) and stability (intraclass correlation = 0.98). Moderate agreement between PsA-Disk and PEST (Cohen’s kappa = 0.46) was observed, while construct validity appeared appropriate [PsA + patients: PsA-Disk score (interquartile range; IQR) =71 (50–96); PsA-patients: PsA-Disk score (IQR)=50 (20–90); p < 0.001].
Conclusion:
PsA-Disk may be considered a valid novel instrument aiding both dermatologists and rheumatologists in the rapid detection and assessment of musculoskeletal disease characteristics.
Introduction
Psoriatic arthritis (PsA) is a chronic inflammatory disease typically associated with psoriasis (PsO), involving peripheral joints, entheses, tendons and axial skeleton affecting 0.16‒0.25% of the world population.1–3 The prevalence of PsA in PsO patients varies from 6% to 41% based on different clinical settings.4–7 Diagnosis relies mainly on clinical evaluation and classification is based on CASPAR (ClASsification criteria for Psoriatic ARthritis), often used in epidemiological and research studies. 8
PsO and PsA have a strong impact on physical and mental functions, similar to that observed in other severe chronic diseases 9 with high levels of stress, poor self-esteem, increased rates of mood disorders and detrimental effects on social functioning, interpersonal relationships and productivity. 10
Patients with PsA may develop severe musculoskeletal involvement in addition to cutaneous signs, with a further negative impact on function and quality of life (QoL).11,12 A delay in the diagnosis of PsA is associated with adverse long-term outcome, such as progressive erosive and osteoproliferative joint damage and associated disabilities; the early detection of PsA instead can potentially prevent these significant complications. 13
The Group for Outcome Measures in Rheumatology (OMERACT), and for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) refined and assessed the different PsA domains in order to identify accurate, reliable and feasible activity measures for clinical trials and daily practice. 14 The OMERACT group originally proposed a core set of six core domains, represented by peripheral joint and skin activity, pain, patient global assessment (PGA), physical function, and health-related (HR)QoL. 15 This has since been updated to now include musculoskeletal disease activity, skin disease activity, fatigue, pain, PGA, physical function, HRQoL and systemic inflammation. 16
Concurrently, the GRAPPA highlighted that the assessment of PsA should include all major disease domains, including peripheral arthritis, axial disease, enthesitis, dactylitis, PsO, and nail disease. However, the characteristics of musculoskeletal pain and its impact on patients with PsA should also be examined. 17 In addition, disease activity and other potentially related conditions should also be considered, including cardiovascular disease, uveitis and inflammatory bowel disease. 17
A number of screening questionnaires [e.g. Toronto Psoriatic Arthritis Screen, Psoriasis Epidemiology Screening Tool (PEST), psoriatic arthritis screening and evaluation and Early ARthritis for Psoriatic patients (EARP)] used by dermatologists or general practitioners have been developed to identify PsO patients who refer symptoms suggestive of PsA.18–21 Although the PEST questionnaire was shown to have the highest area under the curve, it is limited by its simplicity.22,23
Frequently, these instruments are not systematically performed in daily clinical practice, probably due to the lack of time available in routine clinical practice. Another limitation of these tools is that they do not provide any information on the global impact of the musculoskeletal disease on patients with PsO.24,25 Therefore, a rapid and visually intuitive instrument is needed to screen and assess PsA in patients with PsO for dermatologists for a correct rheumatological referral. The aim of this study was to assess characteristics of musculoskeletal pain in patients with PsO for the presence of PsA and evaluation of a novel 16-item visual instrument (PsA-Disk) for the characterization and description of PsA clinical manifestations.
Materials and methods
Patients and study design
Data were collected prospectively from eight dermatological/rheumatological centres in Italy.
Consecutive adult patients with PsO at their first dermatological visit from September 2017 to March 2018 presenting with musculoskeletal pain were invited to complete both PEST 19 and PsA-Disk questionnaires.
Inclusion criteria were: male and female patients aged ⩾18 years, with a diagnosis of PsO and presence of musculoskeletal pain but without a previous diagnosis of PsA. Exclusion criteria were: patients diagnosed with PsA, patients with PsO treated with systemic steroids, nonsteroidal anti-inflammatory drugs (NSAIDs), conventional systemic disease-modifying antirheumatic drugs (cDMARDs) or biological agents and patients affected by other autoimmune or chronic inflammatory diseases. Patients with psychiatric comorbidities and not capable of cooperating in the study were also excluded. A visit with a rheumatologist confirmed the presence or absence of PsA. Diagnosis of PsA was made according to CASPAR criteria. 8 A range of clinimetric and disease activity variables were recorded. The Ethical Committee of each site evaluated and approved the study (RS 228/16). An informed consent form was presented to all eligible and willing patients, who signed their agreement to participate.
Clinical measures
PsO severity was measured using the Psoriasis Area and Severity Index (PASI), 26 the Physician Global Assessment 25 and Body Surface Area (BSA). 27 Nail disease activity was measured using the Nail Psoriasis Severity Index (NAPSI). 28 Joint disease activity was assessed using the number of tender and swollen joint count (68 joints were assessed for tenderness and 66 joints were assessed for swelling), the 44-joint-count Disease Activity Score (DAS-44), Disease Activity in Psoriatic Arthritis (DAPSA), composite psoriatic arthritis disease activity index (CPDAI), the visual analogue scale (pain-VAS) and Bath Ankylosing Spondylitis Disease Activity Index (BASDAI).29–31 The presence or absence of axial involvement and number of fingers/toes with dactylitis were also recorded. Axial involvement was defined in patients who had a physician-reported presence of spinal involvement at enrolment, based on clinical judgment of clinical features thought to be representative of active inflammatory spondylitis, or radiographs or magnetic resonance imaging showing sacroiliitis. 32 Enthesitis was assessed by physical examination as painful (positive confirmation) or without pain (negative confirmation) according to the expanded Leeds index. The following entheses were bilaterally evaluated: lateral epicondyle, medial femoral condyle, Achilles tendon insertion, insertion of quadriceps tendon on the superior pole of the patella, proximal insertion of the patellar tendon on the inferior pole of the patella and the insertion of the plantar fascia on the calcaneus, as previously described. 33 Laboratory tests included erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP). ESR was measured by the Westergren method and CRP was determined using standard commercial kits. Data on demographic and anamnestic characteristics and concomitant diseases were also recorded.
PsA-Disk
The PsA-Disk is a visual intuitive instrument, aimed to describe the musculoskeletal symptoms of patients with PsA. It consists of 16 questions developed using (dermatology and rheumatology) focus groups and a Delphi procedure 34 exploring the presence and characteristics of pain in different body sites and quality of sleep [Figure 1(a)]. All 16 items included in the questionnaire were agreed by the group of eight ‘derma-rheuma’ centres, based on their expertise in the clinical presentation of PsA.

The PsA-Disk questionnaire with an example of a polygon derived from answers to the 16 items. The 16 questions (a) correspond to the 16 items visualised in the disk (b). Most items relating to inflammatory joint symptoms are positioned in the upper half of the disk (items 1–4 and items 13–16; light pink through to dark blue) and most items relating to myalgia and fatigue are positioned in the lower half of the disk (items 5–10; light green through to dark green). Answers to questions vary from 0 to 10, 0 being ‘absolutely no’ and 10 ‘definitely yes’. The exception to this scale is item #1, ‘Morning joint stiffness’, that is measured in minutes on a scale of 0–100.
Answers to questions are marked on an 11-point VAS, each varying from 0 to 10, 0 being ‘absolutely not’ and 10 ‘definitely yes’. The exception to this scale was item #1, ‘Morning joint stiffness’, that is measured in minutes on a scale of 0–100 [Figure 1(b)]. Points are represented as rays of a circle, such that by connecting them, a polygon is obtained whose area may be intuitively understood as the size of the symptoms [Figure 1(b)]. The majority of items presented in the upper half of the disk include items involving inflammatory joint symptoms (apart from knee and shoulder pain), whereas the lower half of the disk includes items associated with myalgia and fatigue. Colours used in the disk have no clinical/scientific purpose.
Validation and cut-off scores of PsA-Disk
The overall and item-specific reliability of the PsA-Disk questionnaire was evaluated for internal consistency and stability (test-retest of 45 patients within a time-span of 3 days) using the Cronbach’s alpha and the intraclass correlation (ICC), respectively. Internal consistency ranges between 0–1. A commonly accepted rule of thumb is that an α of 0.6–0.7 indicates acceptable reliability, and 0.8 or higher indicates good reliability. We used the receiving operating characteristics curve with Youden’s J statistic to assess the cut-off value of the total PsA-Disk score that better predicts clinical diagnosis of PsA (Supplemental Material Figure 1). Based on this cut-off value (PsA-Disk score = 44: sensitivity, 87.2%; specificity, 46.4%; J index = 0.34; Supplemental Material Table 1), we assessed the criterion validity of the PsA-Disk evaluating its agreement with the PEST screening tool through the Cohen’s kappa coefficient (concurrent validity). The construct validity of the PsA-Disk was evaluated by comparing the overall score between patients with and without clinical diagnosis of PsA using the Wilcoxon rank-sum test (known-groups validity).
Statistical analysis
Data are presented as mean ± standard deviation for continuous variables, and number and percentage for categorical variables. The association between PsA diagnosis with demographic, anamnestic and clinical characteristics was evaluated using the Chi-square test for categorical variables and the Wilcoxon rank-sum test for continuous variables. After verification of the assumptions of normality and homogeneity of variance through the Shapiro–Wilk test and Levene’s test, respectively, the overall PsA-Disk score was compared between groups of patients with confirmed PsA using the Student’s t test or the one-way analysis of variance. The Spearman correlation coefficient was used to describe the association between the overall PsA-Disk score and continuous clinical variables. A p value <0.05 was considered statistically significant. All analysis was performed using Stata/MP version 13 (Stata Corp. LP, TX, USA).
Results
Baseline characteristics of patients with PsO and PsA
From a total of 573 patients with PsO, 279/573 (48.7%) patients were referred for joint symptoms of pain and 242/279 (86.7%) accepted to respond to the questionnaire. 239/242 (98.8%) patients with PsO met the inclusion criteria and participated to the study. Demographic and clinical characteristics for patients with both PsA and PsO are summarized in Table 1. The majority of patients were female (n = 130; 54.4%), the mean age was 51.2 ± 13.4 years and body mass index (BMI) was 27.4 ± 5.3 kg/m2. Following a rheumatological visit, we observed an estimated prevalence of PsA of 21% (120/573) for the Lazio region among the patients included and 120/239 (50.2%) were diagnosed positive for the presence of PsA.
Demographic and clinical characteristics of patients with PsO and PsA.

Data are presented as mean ± SD or n (%), as indicated.
BMI, body mass index; BSA, body surface area; CRP, C-reactive protein; DLQI, Dermatology Quality of Life Index; ESR, erythrocyte sedimentation rate; NAPSI, Nail Psoriasis Severity Index; PASI, Psoriasis Area and Severity Index; PEST, Psoriasis Epidemiology Screening Tool; PsA, psoriatic arthritis; PsO, psoriasis; SD, standard deviation.
A higher proportion of patients with PsA had a PEST score ⩾3 compared with patients with PsO (77.5 versus 37.8%, p < 0.001). Furthermore, patients with PsA presented with significantly higher levels of ESR (21.9 ± 17.1 versus 15.9 ± 10.2 mm/h, p = 0.041), PsA-Disk scores (73.9 ± 32.1 versus 58.1 ± 39.8, p < 0.001), nail PsO (54.2 versus 42, p = 0.05), NAPSI (feet; 10.2 ± 9.7 versus 3.9 ± 7.8, p = 0.002), NAPSI (hands + feet; 9.3 ± 6.8 versus 4.6 ± 7.9, p = 0.007), compared with patients with PsO (Table 1).
While other clinical characteristics remained similar between the two groups, a slightly higher proportion of patients with PsA had cardiovascular disease (30.8 versus 22.7%, p = 0.16) and PsO localized at the limbs or trunk or face (85 versus 78.2%, p = 0.12; Table 1).
Association between PsA-Disk scores and rheumatological variables
The impact of a range of rheumatological disease activity measures (presence or absence of a specific feature) on PsA-Disk scores was next evaluated in patients with PsA (Table 2). This analysis revealed that patients presenting with knee joint involvement had significantly higher PsA-Disk scores (98.4 ± 26 versus 71.5 ± 31.9, p = 0.006) compared with patients without knee joint involvement (Table 2). Furthermore, patients presenting with oligoarthritis (⩾5 painful joints) had significantly higher PsA-Disk scores compared with those without (81.4 ± 32.8 versus 66.4 ± 30, p = 0.023). Stratifying DAPSA scores into low, moderate and high disease activity was also found to be associated with increasing PsA-Disk scores (Table 2). Patients with axial involvement also had a higher PsA-Disk score, although this difference did not attain statistical significance (Table 2). We observed several positive correlations between disease activity and PsA-Disk score in patients with PsA including number of swollen and tender joints, patient and physician pain-VAS, PGA-VAS, physician-HAQ, DAS-44 and DAPSA (Table 3). However, CRP and ESR were not correlated with PsA-Disk score (Table 3).
Change in PsA-Disk score across different rheumatological variables in patients with PsA.
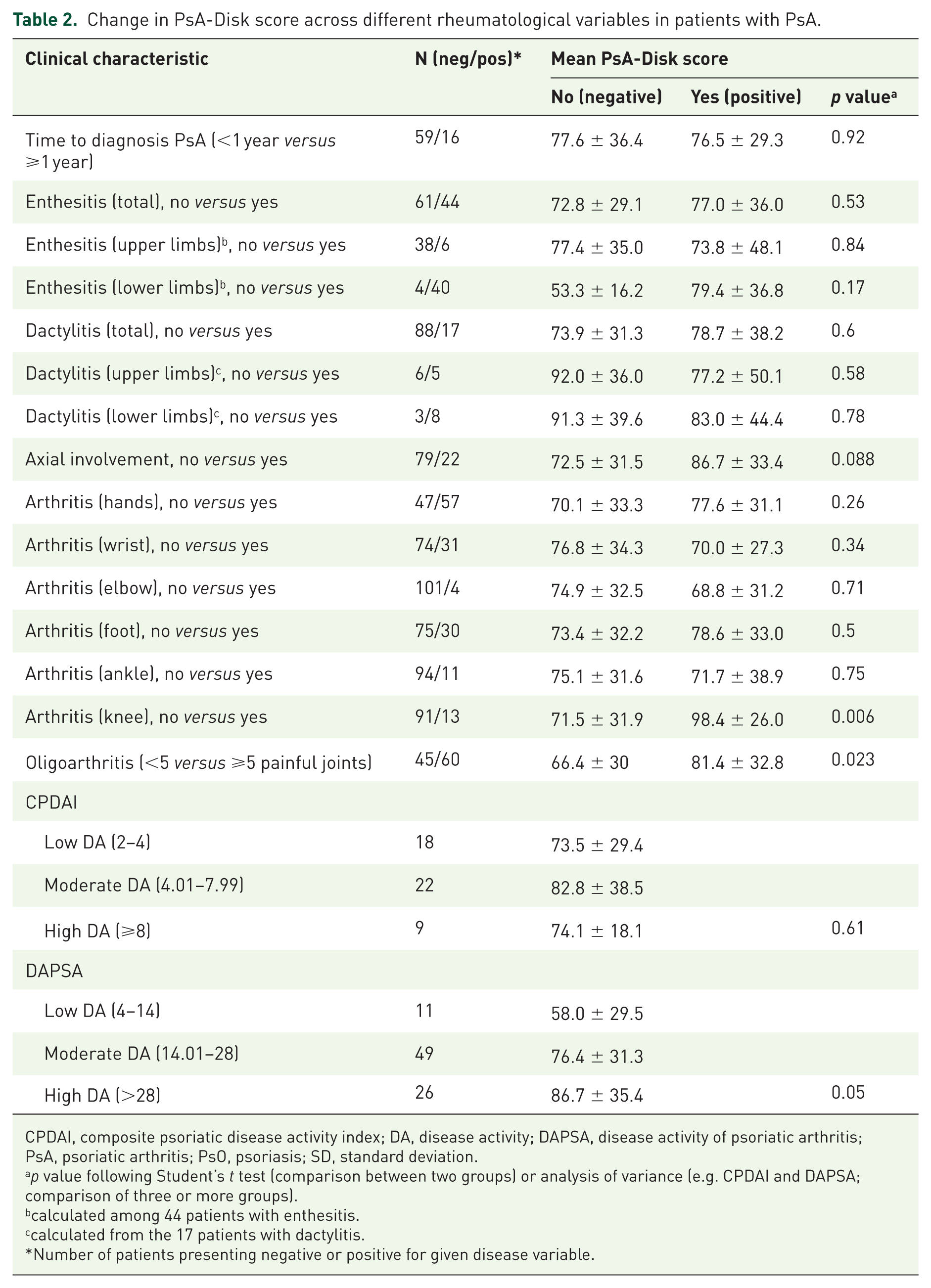
CPDAI, composite psoriatic disease activity index; DA, disease activity; DAPSA, disease activity of psoriatic arthritis; PsA, psoriatic arthritis; PsO, psoriasis; SD, standard deviation.
p value following Student’s t test (comparison between two groups) or analysis of variance (e.g. CPDAI and DAPSA; comparison of three or more groups).
calculated among 44 patients with enthesitis.
calculated from the 17 patients with dactylitis.
Number of patients presenting negative or positive for given disease variable.
Correlation between clinical variables and PsA-Disk score in patients with PsA.

BASDAI, bath ankylosing spondylitis disease activity index; BSA, body surface area; CPDAI, composite psoriatic disease activity index; CRP, C-reactive protein; DAPSA, disease activity index for psoriatic arthritis; DAS-44; Disease Activity Score of 44 joints; ESR, erythrocyte sedimentation rate; HAQ, health assessment questionnaire; PASI, psoriasis area severity index; PGA, physician global assessment; PsA, psoriatic arthritis; SD, standard deviation; VAS, visual analogue score.
Spearman rank correlation coefficient (r). N refers to number of patients diagnosed with PsA.
Impact of clinical characteristics on PsA-Disk 16-item visual output
Examples of the powerful visual use of this instrument can be seen in Figure 2. Differences in clinical characteristics across the 16 items can be visualised quickly. Following subgroup analysis, clinical features such as age, sex and presence of PsA resulted in a clear difference in the size of the two or more polygons [Figure 2(a, b and d)]. The polygon derived from the PsA group of patients overlaps with that of patients with PsO in the lower and upper halves of the disk suggesting increased fibromyalgia and articular pain, respectively, consistent with a positive PsA diagnosis [Figure 2(d)]. Other clinical features such as BMI did not impact upon any of the 16 items, apart from a significant difference in severity of neck pain [Figure 2(c)].

Output from visual PsA-Disk questionnaire depicting variation in polygon shapes among a range of clinical variables. Subgroups of patients according to different clinical features (e.g. male and female sex) exhibit different PsA-Disk scores, resulting in unique polygon profiles. Statistically significant differences between variables (e.g. b, male versus female) for specific items (questions) are represented by an asterisk positioned outside the disk, where p < 0.05. PsA-Disk scores are based on 219 patients.
Validation and cut-off scores of PsA-Disk
Overall, the reliability of the PsA-Disk questionnaire was excellent for both internal consistency (Cronbach’s alpha = 0.90) and stability [ICC = 0.98, 95% confidence interval (CI): 0.97–0.99; Table 4]. In particular, according to test–retest, we observed a good stability (ICC > 0.8) for all items, except shoulder pain (ICC = 0.64, 95% CI: 0.39–0.80; Table 4). The analysis of criterion validity showed a moderate level of agreement between PsA-Disk and PEST (Cohen’s kappa = 0.46), while the construct validity appeared appropriate [PsA + patients: median PsA-Disk score (IQR) = 71 (50–96); PsA-patients: median PsA-Disk score (IQR) = 50 (20–90); p < 0.001].
Reliability of PsA-Disk for 16 items (internal consistency and stability).

The item-specific Cronbach’s alpha was calculated removing the item from the calculation of the overall coefficient.
ICC (absolute agreement, two-way mixed effects model).
CI, confidence interval; ICC, intraclass correlation; PsA, psoriatic arthritis.
Discussion
In this multicentre study, we validated the PsA-Disk in a large group of patients with PsO from eight dermatological/rheumatological centres across Italy. We demonstrated that the PsA-Disk can be used rapidly and easily to evaluate the characteristics of musculoskeletal symptoms in patients with PsA.
The present study also evaluated the prevalence of PsA in patients with PsO. Indeed, we demonstrated that 48.7% (279/573) of patients with PsO with joint pain were diagnosed with PsA (prevalence of 21% based on all 573 patients who presented at their first visit). This prevalence is higher than would be expected, even in a secondary care setting 7 and is likely due to the fact that the eight centres that participated in this study already routinely undertook shared dermatology-rheumatology clinics, therefore raising their level of expertise and facilitating diagnosis.
The phenotypic characteristics of patients with PsO and PsA were also different: a greater proportion of patients with PsA had localized PsO at the limbs or trunk or face and more nail PsO. Moreover, a significantly higher proportion of patients who were PsA-positive had elevated PEST scores, ESR, nail PsO, NAPSI and PsA-Disk scores compared with patients who were PsA-negative. PsA-Disk scores were also positively correlated with tender and swollen joints, physician-HAQ, patient- and physician-VAS (pain), PGA-VAS, DAS-44 and DAPSA. Results of this first phase developmental study are particularly promising in that a range of classical disease activity measures were significantly correlated with PsA-Disk scores.
While this questionnaire cannot be used independently to diagnose PsA, we have found that using a PsA-Disk cut-off value of ⩾44 could be used to identify and monitor patients with PsA. The internal consistency was also very satisfactory, with Cronbach’s alpha value of 0.90 and internal stability of 0.98.
The PsA-Disk offers potential advantages over already existing instruments and tools used to evaluate PsA. The relative simplicity of the PEST questionnaire is regarded as a weakness, since this tool cannot detect pure axial forms of the disease. 23 The PsA-Disk questionnaire was specifically designed to address this limitation, by including specific items for neck and back pain. In addition, many other instruments are completed by the physician, whereas the PsA-Disk was designed to be completed by the patient, promoting discussion between patient and specialist, often neglected in a medical consultation. Improved communication between patient and physician may in turn lead to improved adherence to treatment. Furthermore, implementation of the PsA-Disk will also promote discussion between dermatologist and rheumatologist, particularly in clinics where a dermatology-rheumatology multidisciplinary approach is not already established, in turn facilitating early referral and early diagnosis.
Another potential benefit afforded from the PsA-Disk is that it is a visual instrument. Answers to the 16 questions yield scores are graphically displayed as a polygon implying that both patient and physician can literally observe changes over the course of the disease. The PsA-Disk is also a simple and intuitive instrument allowing the patient to complete it rapidly.
This study has some limitations that need to be highlighted. Although we observed some significant associations between disease activity measures and PsA-Disk scores, a higher sample size may have revealed additional significant associations that were not detectable with the current sample size. Our analysis was cross-sectional. It would be interesting to explore the use of this instrument to monitor disease progression or effect of therapeutic intervention on prognosis.
Conclusion
In summary, the PsA-Disk is a novel instrument, which can facilitate in the detection and assessment of disease severity in patients with PsA, particularly in dermatology clinics where joint dermatology-rheumatology clinics are not routinely undertaken. The main advantages of this instrument are the low number of items, the visual nature of the results, and the fact that it is completed together by the patient and dermatologist/rheumatologist. Future studies will be aimed to evaluate the discriminatory power of this instrument in clinical trials and monitoring of patient.
Supplemental Material
Figure_legend_Supplemental_Figure_1 – Supplemental material for PsA-Disk, a novel visual instrument to evaluate psoriatic arthritis in psoriatic patients: an Italian derma-rheuma multicentre study
Supplemental material, Figure_legend_Supplemental_Figure_1 for PsA-Disk, a novel visual instrument to evaluate psoriatic arthritis in psoriatic patients: an Italian derma-rheuma multicentre study by Maria Sole Chimenti, Maria Esposito, Dario Graceffa, Miriam Teoli, Giusy Peluso, Domenico Birra, Gaia Moretta, Alessandra Galossi, Valentina Carboni, Felice Sensi, Annamaria Mazzotta, Rosalba Caccavale, Nicoletta Bernardini, Paola Sessa, Antonio Richetta, Ester Del Duca, Sara Urbani, Severino Persechino, Clara De Simone, Claudio Bonifati, Elisa Gremese, Ketty Peris and Roberto Perricone in Therapeutic Advances in Chronic Disease
Supplemental Material
Supplemental_Material_Figure_1 – Supplemental material for PsA-Disk, a novel visual instrument to evaluate psoriatic arthritis in psoriatic patients: an Italian derma-rheuma multicentre study
Supplemental material, Supplemental_Material_Figure_1 for PsA-Disk, a novel visual instrument to evaluate psoriatic arthritis in psoriatic patients: an Italian derma-rheuma multicentre study by Maria Sole Chimenti, Maria Esposito, Dario Graceffa, Miriam Teoli, Giusy Peluso, Domenico Birra, Gaia Moretta, Alessandra Galossi, Valentina Carboni, Felice Sensi, Annamaria Mazzotta, Rosalba Caccavale, Nicoletta Bernardini, Paola Sessa, Antonio Richetta, Ester Del Duca, Sara Urbani, Severino Persechino, Clara De Simone, Claudio Bonifati, Elisa Gremese, Ketty Peris and Roberto Perricone in Therapeutic Advances in Chronic Disease
Supplemental Material
Supplemental_Material_Table_1_R1_(2) – Supplemental material for PsA-Disk, a novel visual instrument to evaluate psoriatic arthritis in psoriatic patients: an Italian derma-rheuma multicentre study
Supplemental material, Supplemental_Material_Table_1_R1_(2) for PsA-Disk, a novel visual instrument to evaluate psoriatic arthritis in psoriatic patients: an Italian derma-rheuma multicentre study by Maria Sole Chimenti, Maria Esposito, Dario Graceffa, Miriam Teoli, Giusy Peluso, Domenico Birra, Gaia Moretta, Alessandra Galossi, Valentina Carboni, Felice Sensi, Annamaria Mazzotta, Rosalba Caccavale, Nicoletta Bernardini, Paola Sessa, Antonio Richetta, Ester Del Duca, Sara Urbani, Severino Persechino, Clara De Simone, Claudio Bonifati, Elisa Gremese, Ketty Peris and Roberto Perricone in Therapeutic Advances in Chronic Disease
Footnotes
Appendix
Acknowledgements
The authors would like to thank Dr Colin Gerard Egan (CE-Medical Writing, Pisa, Italy) for editorial assistance in the preparation of the manuscript and Dr Massimo Fabiani (Italian National Institute of Health, Rome, Italy), for performing statistical analysis. Maria Sole Chimenti and Maria Esposito contributed equally to this work.
Authors’ note
Maria Esposito is now affliated with Department of Dermatology, Department of Biotechnological and Applied Clinical Sciences, University of L’Aquila, L’Aquila, Italy.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
