Abstract
Objective
Psoriasis is an immune-mediated chronic inflammatory disease with skin and joint manifestations. Smoking is considered an unfavorable lifestyle factor for psoriasis. We aimed to explore the association between smoking, disease risk, and treatment efficacy in relation to psoriasis.
Methods
We searched the Cochrane Library, Embase, and PubMed databases for studies examining the relationship between smoking and psoriasis, up to 12 April 2020. We then conducted a meta-analysis using a fixed-effects model to obtain odds ratios (ORs) with 95% confidence intervals (CIs).
Results
Ever, current, and former smokers all had higher risks of developing psoriasis than non-smokers (pooled ORs (95% CIs): 1.60 (1.51–1.69), 1.63 (1.48–1.80), and 1.36 (1.13–1.64), respectively). Ever smokers were less likely to show disease improvement at 6 months following treatment with biologic agents than non-smokers (pooled OR (95% CI): 0.80 (0.67–0.95)).
Conclusions
Smoking increases psoriasis risk and negatively impacts the benefits of biologic agents. However, the number of reports is limited and more studies are needed to confirm the effects of smoking and smoking cessation on therapeutic response in patients with psoriasis. Education to encourage a healthy lifestyle remains a valuable approach in clinical practice.
Introduction
Psoriasis is an immune-mediated chronic inflammatory skin disease with manifestations including erythema, which can occur on the scalp, elbows, knees, and lower back. Joint damage, referred to as psoriatic arthritis (PsA), can also occur in patients with psoriasis. PsA is a progressive and often destructive joint disease; however most patients only experience joint symptoms several years after the onset of cutaneous psoriasis. 1 Both genetic and environmental factors are involved in the development of psoriasis. Unhealthy lifestyle habits, such as smoking, drinking, and lack of exercise, are suggested risk factors for psoriasis, and have thus attracted attention from researchers. Dietary interventions and exercise have been reported to reduce the severity of psoriasis. 2 Increased risks of cardiovascular disease and metabolic syndrome, such as coronary heart disease, hypertension, hypercholesterolemia, and diabetes, 3 , 4 have also been identified in patients with psoriasis, and smoking is a known risk factor for these cardiovascular and metabolic diseases.
Psoriasis results from uncontrolled keratinocyte proliferation and dysfunctional differentiation. Dendritic cells, macrophages, T cells, and neutrophils also promote inflammatory infiltrates in the skin, and neovascularization has also been shown to occur in psoriasis. 5 Smoking has been reported to worsen psoriasis in several ways, including via interactions with genetic susceptibility loci such as the HLA-Cw6 allele. 6 Smoking initially produces deleterious free radicals, leading to intracellular signaling involving mitogen-activated protein kinase/activator protein 1, nuclear factor kappa B, and Janus kinase signal transducers and activators of transcription. 7 Active immune cells and keratinocytes trigger increased secretion of inflammatory cytokines, such as tumor necrosis factor (TNF)-α and interferon-γ, which in turn exacerbate the psoriasis. 8 Nicotine, as the main component of cigarettes, also induces overexpression of vascular endothelial growth factor, thus worsening pathological angiogenesis in patients with psoriasis.9–11 In addition to phototherapy and conventional drug therapies such as cyclosporin and methotrexate, emerging targeted biological drugs, such as adalimumab 12 and infliximab, 13 have been shown to be effective and safe for the treatment of psoriasis in clinical trials. However, despite overall promising clinical trial results, varied drug survival and therapy discontinuation can occur, and some patients might fail to respond to certain biologic therapies and change to other strategies. We noted a high proportion of smokers among psoriasis patients at our clinic. However, the results of clinical trials and real-world data regarding the impact of smoking on therapeutic responses in patients with psoriasis remain contradictory, and the impact of smoking on psoriasis needs careful examination.
We therefore conducted a literature review and meta-analysis to investigate if smoking (ever, current, or former) might be an independent risk factor for the development of psoriasis, and to explore the impacts of smoking on psoriasis treatment response.
Materials and methods
Literature search strategy
This meta-analysis was not registered in PROSPERO but was conducted in strict accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines.
14
Ethical committee review was not applicable for this meta-analysis. The Cochrane Library, Embase, and PubMed databases were searched up to 12 April 2020 for studies examining the relationship between psoriasis and smoking using the MeSH terms “psoriasis” and “smoking”, with no restrictions on language, time, or race. The detailed search strategy is shown in the
Inclusion and exclusion criteria
Studies were included for analysis if they met the following criteria: (i) examined the effect of smoking on psoriasis incidence, disease exacerbation, and treatment efficacy (case-control study or cohort study); (ii) included original case numbers; and (iii) included sufficient data to calculate odds ratios.
Studies were excluded if they met any of the following criteria: (i) no relevant content; (ii) duplicate or overlapping data; or (iii) editorials, reviews, protocols, notes, and letters. Conference abstracts were excluded for examination of smoking and psoriasis risks, but retained for potential use in the analysis of the impact of smoking on treatment efficacy.
Study selection and quality assessment
The identified studies were organized using NoteExpress 3.2 software and Microsoft Office Excel 2016. Identified references from different databases were merged in NoteExpress 3.2, and duplicate studies were removed. The title and abstract of each reference was screened to determine if it met the inclusion and exclusion criteria, and decisions were made based on full-text examination if the information from the title and abstract was insufficient. Potentially eligible studies were classified using Microsoft Office Excel 2016 for further analysis.
Each selected paper was assessed according to the Newcastle–Ottawa Quality Assessment Scale by two independent researchers (H.Z. and R.W.). Advice from a third individual (Y.K.) was solicited if the two researchers could not reach a consensus. Studies with seven stars or more were considered high quality, and studies with five stars or less were considered low quality.
Data extraction
Data including the first author’s last name, publication year, country, participants’ demographic characteristics, research type, and sample size were extracted from each candidate study by two independent researchers (H.Z. and R.W.). Smoking status was classified as ever, current, and former smoking; if no detailed information on smoking status was available, patients were classified as ever smokers. Current smokers were defined as individuals who were smoking at the time of the study or who had stopped smoking less than 1 year ago. Former smokers were defined as those who had not smoked for at least 1 year. Information on smoking quantity and smoking duration, such as smoking years, pack-years, daily cigarettes quantity, and cigarette type was extracted where possible. Any disagreements were solved by a third researcher (Y.K.).
Data analysis
The total effect size of the odds ratio (OR) between patients with psoriasis and controls was computed or raw case numbers were extracted from each study using a fixed-effects model. Forest plots were adopted to estimate ORs and 95% confidence intervals (CIs). Heterogeneity of the included studies was tested by the Q-statistic and quantified using I2 = 100% × [(Q − degrees of freedom)/Q]. I2 ≥ 50% was considered to indicate significant heterogeneity, I2 ≥ 25% but < 50% was considered to show moderate heterogeneity, and I2 ≥ 0 but < 25% was considered as minor heterogeneity. A visual funnel plot was adopted to show publication bias. All statistical analyses were conducted using Review Manager 5 software.
Results
Literature selection
A flowchart of the literature selection process is depicted in Figure 1. A total of 2638 potentially eligible references were identified using the previously described search strategy. Among these, 642 duplicated articles were excluded, and a further 1749 articles were excluded after examining the titles and abstracts. The full texts of the remaining 237 citations were screened for eligibility, and 130 references were excluded because they did not meet the inclusion criteria. Overall, 66 conference abstracts were identified. Thirty-four references were selected to analyze smoking and psoriasis risk, and 16 studies were selected to analyze the impact of smoking on psoriasis treatment efficacy in this meta-analysis.

Flowchart of literature selection. The Cochrane Library, Embase, and PubMed databases were searched for potentially eligible studies. Duplicate or unrelated studies were excluded. Full-text examination was conducted to distinguish different study types. Thirty-four references were selected to examine smoking and psoriasis risk, and 16 studies were assessed for smoking and psoriasis treatment efficacy.
Smoking and psoriasis risk
Thirty-four full-text studies consisting of 2315–37 case-control and 1138–48 cohort studies were identified and used to analyze the impact of smoking on psoriasis risk; i.e. to determine if the incidence of psoriasis was increased among smokers. These 34 full-text studies were conducted in Europe (20 studies), Asia (9 studies), North America (4 studies), and South America (1 study). Three 15 , 31 , 36 case-control studies and one 41 cohort study examined smoking and the risk of palmoplantar pustulosis (PPP), a specific type of psoriasis. One 28 case-control study and six38,42–44,48 cohort studies focused on PsA. Notably, smokers accounted for higher percentages of patients with both PPP and PsA. However, adding PPP and PsA data would introduce bias, and these were considered to be more suitable for separate discussion and were not included in the current meta-analysis. We also explored the impact of smoking quantity and smoking duration on psoriasis risk in and a total of five15,17,19,21,37 case-control and three39,40,47 cohort full-text studies. Because measures such as smoking years, pack-years, or daily cigarettes varied, and cigarette type and components differed between areas, a meta-analysis of this topic was not possible. However, there was a trend towards a relationship between heavier smoking and psoriasis.
Except for one 46 study focusing on infants, 2015–26,29–35,37 case-control studies and four39,40,45,47 cohort studies that examined the association between smoking status and psoriasis risk were identified. The characteristics of these studies are listed in Table 1. The four39,40,45,47 cohort studies were excluded because of variations in the psoriasis populations, outcomes, and effect sizes. We further extracted information from the remaining 20 case-control studies and assessed their qualities, as shown in Table 2. Sixteen studies were ranked as good quality (7–9 stars) and four studies were ranked as acceptable quality (6 stars).
Characteristics of literature examining smoking and psoriasis risk.
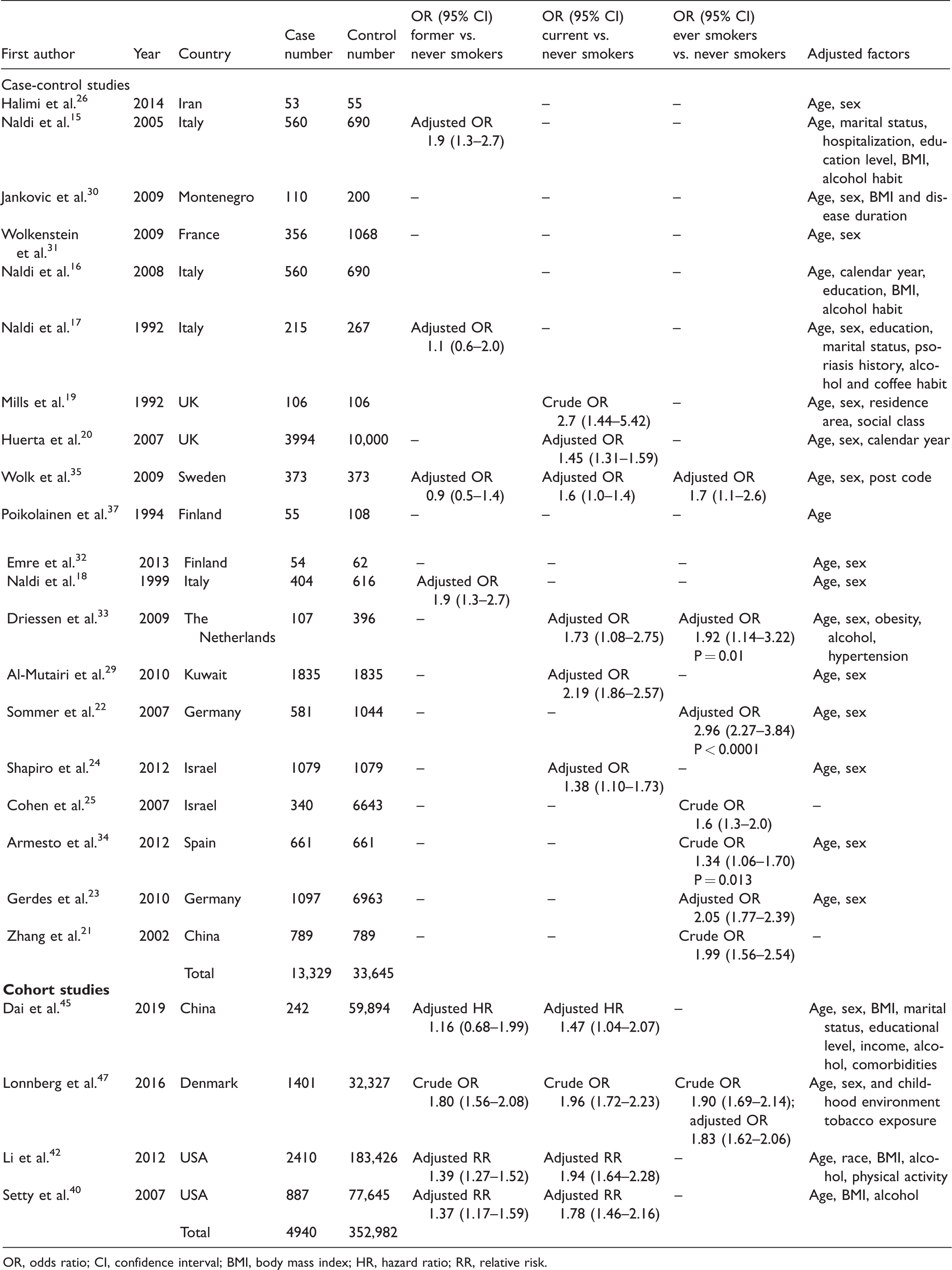
OR, odds ratio; CI, confidence interval; BMI, body mass index; HR, hazard ratio; RR, relative risk.
Further information extracted from 20 case-control studies and quality assessment.

Among the 20 case–control studies, 1617–23,25,26,29–31,34,35,37 examined the association in ever smokers vs. non-smokers, nine 15 , 17 , 18 , 24 ,29–32 examined the association in current smokers vs. non-smokers, five 15 , 17 , 18 , 30 , 31 examined the association in former smokers vs. non-smokers, and two 15 , 30 case–control studies and three 40 , 46 , 47 cohort studies examined the effect of passive smoking on psoriasis.
We first analyzed the risks of psoriasis in ever smokers vs. non-smokers. Three16–18 out of 16 studies were excluded from this analysis because of overlapping data, in which case we retained the most recent and complete of the overlapping studies. 15 We then conducted a meta-analysis of the remaining 13 studies to examine the association between ever smoking and psoriasis risk, as shown in Figure 2. The pooled OR for the risk of psoriasis in ever smokers vs. never smokers was 1.60 (95% CI: 1.51–1.69) with significant heterogeneity (I2 = 82%, P < 0.00001). We identified five studies that introduced heterogeneity because of their small sample size, 19 , 26 sex imbalance, 21 or sample specialty.22,23 The pooled OR was 1.41 (95% CI: 1.32–1.51) with insignificant heterogeneity (I2 = 15%) (Figure 3a). No significant visual asymmetry was identified in the funnel plot, as demonstrated in Figure 3b.

Forest plots of smoking status in relation to psoriasis risk. (a) Ever smokers vs. non-smokers; (b) current smokers vs. non-smokers; and (c) current smokers vs. non-smokers.

Forest and funnel plots for ever or current smokers vs. non-smokers, with excluded studies. (a) Forest plot and (b) funnel plot for ever smokers vs. non-smokers with five studies excluded. (c) Forest plot and (d) funnel plot for current-smokers vs. non-smokers with one study excluded.
We then examined psoriasis risk in current smokers vs. non-smokers based on nine15,17,18,24,29–32 cohort studies. Two 17 , 18 overlapping studies were removed. The pooled OR for the risk of psoriasis in current smokers vs. never smokers was 1.63 (95% CI: 1.48–1.80) with significant heterogeneity (I2 = 78%, P = 0.0001) (Figure 2b). We identified one 29 study that introduced heterogeneity, and omission of this study resulted in a pooled OR of 1.35 (95% CI: 1.19–1.53) with non-significant heterogeneity (I2 = 21%) (Figure 3c). The addition of more related studies in the future might reduce the potential study bias, as indicated by the funnel plot (Figure 3d).
Regarding the risk of psoriasis in former smokers vs. never smokers, out of the five related case-control studies, three15,17,18 overlapping studies were identified and the most recent study 15 was retained. The pooled OR from these three15,30,31 studies was 1.36 (95% CI: 1.13–1.64) without heterogeneity (I2 = 0%) (Figure 2c).
Two15,30 case–control and three40,46,47 cohort full-text studies examined the relationship between psoriasis risk and passive smoking. Unfortunately however, these five studies included diverse ages, populations (e.g., twins), and data types and were therefore not suitable for the meta-analysis. Nevertheless, these studies indicated that passive smoking in both early life and adulthood may be a risk factor for psoriasis.
Smoking and treatment efficacy
We also identified 1649–64 studies for examination of the relationship between smoking and treatment efficacy in patients with psoriasis. We further examined these 16 studies and identified five49–53 studies closely associated with this topic and with data that qualified for the meta-analysis. Original case numbers or calculated ORs and 95% CIs were available. The characteristics and qualities of these five papers are listed in Table 3. Two full-text full-cohort studies and three conference abstracts with enough original data and descriptions were included, all published between 2016 and 2019. All five studies examined the efficacy of biologic agents in patients with psoriasis, and one 49 also included non-biologics. Four of the five studies examined ever smokers and non-smokers, but one 50 study that calculated the ORs and 95% CIs of both previous and current smokers compared with non-smokers, without combining current and previous smokers as ever smokers, was excluded. Another study was excluded due to its small sample size. 51 Three studies were therefore finally included in the meta-analysis.
Characteristics and quality assessment of five included studies for assessment of impact of smoking on treatment response for psoriasis.
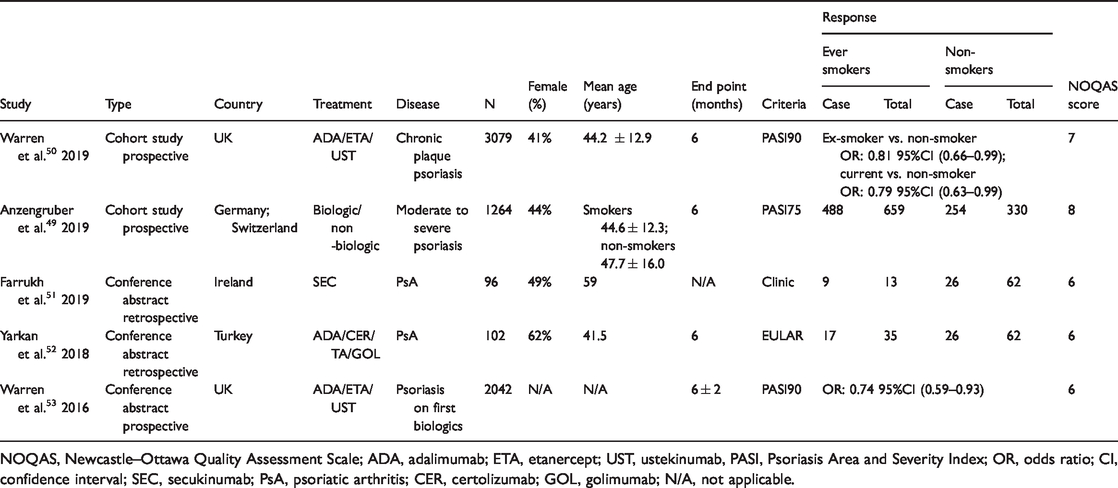
NOQAS, Newcastle–Ottawa Quality Assessment Scale; ADA, adalimumab; ETA, etanercept; UST, ustekinumab, PASI, Psoriasis Area and Severity Index; OR, odds ratio; CI, confidence interval; SEC, secukinumab; PsA, psoriatic arthritis; CER, certolizumab; GOL, golimumab; N/A, not applicable.
The pooled OR for disease improvement at 6 months in ever smokers vs. non-smokers based on these three studies was 0.80 (95% CI: 0.67–0.95) with no heterogeneity (I2 = 0%, P = 0.01) (Figure 4). This OR was close to that in the excluded study, which demonstrated an OR for former smokers vs. non-smokers of 0.81 (95% CI: 0.66–0.99) and an OR for current vs. non-smokers of 0.79 (95% CI: 0.63–0.99). However, there was a potential risk of bias because of the small number of studies, and this could be reduced by adding more related studies in the future.

Forest plot of psoriasis-treatment efficacy in ever smokers vs. never smokers at 6 months.
No comparable data could be extracted from 11 other related papers. One cohort study reported no measurable effect of smoking on the short-term response of severe psoriasis to conventional therapies. 54 A randomized controlled study indicated that smoking did not influence the effect of adalimumab treatment on chronic plaque psoriasis. 55 However, two 56 , 57 studies involving drug survival concluded that current smoking negatively impacted the duration and response of TNF-α inhibitor treatment. Ávila-Ribeiro et al. 58 found that active smoking was related to a loss of remission in patients treated with biologics. Honda et al. 59 stated that smokers with psoriasis changed biologics because of poor efficacy more often than non-smokers, and Walsh et al. 60 reported that a higher percentage of smokers than non-smokers was exposed to at least three immune regulators. Faraawi et al. 61 and Miller et al. 62 focused on patients with PsA and found that a lower percentage of smokers achieved biologic remission at 6 months compared with non-smokers. Gupta et al. 63 found a lower percentage of smokers met the Psoriasis Area and Severity Index 75 criteria at 3 months compared with abstainers regarding conventional drug therapy for plaque psoriasis, while Umezawa et al. 64 reported that smoking 20 cigarettes a day was related to a worse response to ustekinumab.
Discussion
Sixteen case–control studies were identified and included in the current meta-analysis, which demonstrated an increased risk of psoriasis among ever smokers compared with never smokers. Similar results were found for current smokers vs. never smokers, and an increased psoriasis risk was also identified in former smokers compared with never smokers, despite a limited number of studies. Overall, smoking was shown to increase the risk of psoriasis.
Although a previous meta-analysis 65 also examined psoriasis risk among smokers, we conducted a more stringent analysis including only one case-control study. Despite the different criteria, we achieved similar results indicating that smoking was negatively associated with the development of psoriasis.
We noted a trend towards a relationship between heavier smoking and psoriasis. Zhang et al. 21 reported that smoking quantity was related to the severity of psoriasis in a Chinese population. We suggest that more well-designed studies conducted within the same countries are needed to minimize geographical differences. Further exploration of the relationship between decreasing smoking quantity and psoriasis severity might be of clinical value; however, no studies investigating this relationship were found.
Similarly, few studies have assessed the impact of quitting smoking on psoriasis. Based on the pooled OR of psoriasis risk (current smoking vs. never smoking and former smoking vs. never smoking), we hypothesized that quitting smoking might benefit psoriasis patients and/or decrease the incidence of psoriasis. Two39,40 studies reported that quitting smoking decreased psoriasis risk, and Setty et al. 40 found that the risk of psoriasis among former smokers who had quit smoking for 20 years was decreased, similar to that of never smokers. However, both these studies were limited to women, and the benefit of smoking cessation in relation to psoriasis thus remains unclear.
Few studies have focused on the effects of passive smoking. However, despite different assessment methods, the limited studies in the current analysis indicated that passive smoking might increase the risk of psoriasis. A large cohort study by Groot et al. 46 published in 2020 reported ORs of psoriasis risk of 1.55 (95% CI: 1.19–2.01) for prenatal tobacco exposure compared with non-exposure, 1.58 (95% CI: 1.16–2.15) for infants, and 1.46 (95% CI: 1.07–1.98) for children. A cohort study by Lonnberg et al. 47 in 2016 focused on twins, and found an OR for childhood tobacco exposure of 1.28 (95% CI: 1.1–1.49). Setty et al. 40 calculated the relative risk of passive smoking in adults as 1.10 (95% CI: 0.95–1.28). Evidence from large prospective cohort studies has thus demonstrated a close association between passive smoking and psoriasis.
Regarding the effect of smoking on psoriasis treatment, a recent meta-analysis by Mourad et al. 66 in 2019 aimed to determine the impact of smoking on biologic drugs in patients with psoriasis, but failed because of limited reports. To resolve this issue, we assessed both full-text studies and data-extractable conference abstracts, which enabled us to collect more data in the current meta-analysis. We concluded that smoking negatively affected the efficacy of psoriasis treatment at 6 months, mainly involving biologic agents. However, the value of smoking cessation remains to be explored.
Future studies of the effects of smoking on psoriasis-therapy response should take more factors into consideration. Most psoriasis patients have a mild disease course that can be controlled by conventional drugs or external applications, without the need for biologic therapies. However, only one study 49 examined the effect of smoking on the efficacy of traditional anti-psoriasis treatments, such as acitretin, cyclosporin, and methotrexate, and showed that smoking had no effect on the efficacies of these agents. However, the numbers of patients taking acitretin, cyclosporin, and methotrexate were small (n < 100 in most cases), and the non-smokers were significantly older than the active smokers (44.6 vs 47.7 years, P ≤ 0.001), both of which might have influenced the treatment outcomes in this study. No other studies to date have reported on the impacts of smoking on systemic psoriasis treatments such as acitretin, cyclosporin, and methotrexate. Further investigations are therefore needed to answer this question. More attention should be paid to mild psoriasis and traditional therapies, while evidence from more homogeneous studies is also needed to clarify the impact of smoking on the efficacy of biologic agents.
This meta-analysis of the effects of smoking on treatment efficacy was mainly limited by the nature of the published literature. Most available studies of smoking and psoriasis treatment efficacy involved the use of biologics, particularly TNF-α inhibitors. In addition, this meta-analysis was not registered in PROSPERO, and we will ensure that future studies are registered accordingly. Thus although the results revealed a negative impact of smoking on psoriasis treatment response, more real-world evidence is needed to confirm the findings.
The strength of this meta-analysis was the updating of previous studies of a higher risk of psoriasis among smokers, using more stringent criteria. We also found that smoking might have a negative impact on the efficacy of psoriasis treatment, mainly biologic agents. Smoking is associated with multiple factors, including age, sex, educational background, economic status, and lifestyle. We recommend that more thorough studies should be conducted with larger sample sizes to allow the examination of smoking as an independent risk factor for psoriasis. The value of smoking cessation in relation to psoriasis also needs to be further explored. Given that smokers comprise a large percentage of psoriasis patients, we recommend that clinicians educate patients about healthy lifestyles.
Conclusion
Smoking increases the risk of psoriasis and has a negative impact of the efficacy of biologic agents. More well-designed studies are necessary to assess the value of smoking cessation on psoriasis and the effect of smoking on therapeutic responses. Healthy lifestyle education remains a valuable approach in clinical practice.
Footnotes
Data accessibility
All of the data in this meta-analysis are published data and are available in the article.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China [grant number 81872534].
Supplemental Material
Supplemental material for this article is available online.
