Abstract
Background:
Elevated arterial stiffness in patients with primary aldosteronism (PA) can be reversed after adrenalectomy; however, the effect of medical treatment with mineralocorticoid receptor antagonist (MRAs) is unknown.
Objectives:
The aim of this study was to evaluate the effect of MRAs and compare both treatment strategies on arterial stiffness in PA patients.
Design:
Prospective cohort study.
Methods:
We prospectively enrolled PA patients from 2006 to 2019 who received either adrenalectomy or MRA treatment (spironolactone). We compared their baseline and 1-year post-treatment biochemistry characteristics and arterial pulse wave velocity (PWV) to verify the effects of treatment and related determinant factors.
Results:
A total 459 PA patients were enrolled. After 1:1 propensity score matching for age, sex and blood pressure (BP), each group had 176 patients. The major determinant factors of baseline PWV were age and baseline BP. The adrenalectomy group had greater improvements in BP, serum potassium level, plasma aldosterone concentration, and aldosterone-to-renin ratio. The MRA group had a significant improvement in PWV after 1 year of treatment (1706.2 ± 340.05 to 1613.6 ± 349.51 cm/s, p < 0.001). There were no significant differences in post-treatment PWV (p = 0.173) and improvement in PWV (p = 0.579) between the adrenalectomy and MRA groups. The determinant factors for an improvement in PWV after treatment were hypertension duration, baseline PWV, and the decrease in BP.
Conclusion:
The PA patients who received medical treatment with MRAs had a significant improvement in arterial stiffness. There was no significant difference in the improvement in arterial stiffness between the two treatment strategies.
Central illustration

Introduction
Primary aldosteronism (PA) is a curable hypertensive disease characterized by the excessive production of aldosterone, accounting for about 10% of all patients with hypertension. 1 Long-term exposure to high levels of aldosterone leads to increased arterial wall stiffness and atherosclerosis through endothelial dysfunction and collagen synthesis in the vascular wall.2–5 This can be assessed by pulse wave velocity (PWV), which is calculated as the distance between two points in the body divided by the pulse wave transit time. A higher PWV indicates a stiffer arterial wall. PWV is widely used as a non-invasive method to evaluate arterial wall stiffness, 6 as arterial sampling for direct measurements of wall stiffness in human subjects is not practical in clinical settings.
In addition to vascular damage, excess aldosterone can also cause cardiac and renal dysfunction and structural damage beyond that caused by high blood pressure (BP).7–13 Compared with essential hypertensive patients, PA patients have more cardiovascular events including stroke, coronary artery disease, and even atrial fibrillation. 14 However, the adverse consequences resulting from long-term exposure to high aldosterone levels in PA patients, including myocardial fibrosis, atrial fibrillation, and arterial wall stiffness, are reversible after treatment.15–17
The standard treatment for PA is either surgery (adrenalectomy) or medical therapy (mineralocorticoid receptor antagonists, MRAs), depending on the clinical diagnosis (laterization) and patients’ preference. 18 Arterial stiffness measured by PWV in patients with aldosterone-producing adenoma (APA; a major subtype of PA) has been demonstrated to significantly improve after adrenalectomy. 19 MRA was also proposed as an effective agent in reversal of arterial stiffness represented by PWV. 20 However, few studies have investigated and compared treatment outcomes as assessed by PWV after adrenalectomy and MRA therapy in primary aldosteronism. Therefore, we conducted this study to (1) understand the effect of MRA treatment on improving arterial stiffness in primary aldosteronism, and (2) compare the improvements in arterial stiffness between the two treatment strategies.
Methods
Patients
We prospectively enrolled PA patients from 2006 to 2019 at National Taiwan University Hospital. All of the patients were registered in the Taiwan Primary Aldosteronism Investigation (TAIPAI) registry. The inclusion criteria were patients: (1) with a confirmed diagnosis of PA and (2) who underwent baseline brachial-ankle PWV measurements at enrollment. The exclusion criteria were (1) patients age < 18-year-old, (2) patients refused to participate in the study, (3) patients lost to follow-up 1-year after adrenalectomy or on MRA treatment, and (4) patients with other secondary hypertension, including but not limited to renovascular hypertension, Cushing’s syndrome, hyperthyroidism, and pheochromocytoma.
We recorded the patients’ detailed medical information including medical history, biochemistry, BP, and PWV at the first evaluation (defined as the baseline), and at a scheduled follow-up evaluation 12-months after PA-specific treatment (adrenalectomy or MRA therapy). All patients received spironolactone as the MRA treatment. The study was approved by the Institutional Review Board of National Taiwan University Hospital, and informed consent in a written format was obtained from all patients.
Diagnosis of primary aldosteronism
According to previously published protocols and algorithms,21,22 the patients who met the following criteria were diagnosed with PA: (1) autonomous excess aldosterone production with an aldosterone-to-renin ratio (ARR) > 35, (2) a TAIPAI score > 60%, and (3) post-saline loading plasma aldosterone concentration (PAC) > 10 ng/dL, or ARR > 35 (ng/dL)/(ng/mL-h) in a post-captopril test, or PAC > 6 ng/dL in a fludrocortisone suppression test.
The PAC was measured using a commercial radio-immune assay kit (Aldosterone Maia Kit; Adaltis Italia, Bologna, Italy). Plasma renin activity (PRA) was measured as the generation of angiotensin-I in vitro using a commercial radio-immune assay kit (Cisbio, Bedford, MA).
Diagnosis of APA/ idiopathic adrenal hyperplasia
A diagnosis of APA in the PA patients was based on the following criteria: (1) adenoma on a pre-operative computed tomography (CT) scan, (2) lateralization of aldosterone secretion confirmed by adrenal vein sampling or dexamethasone suppression NP-59 single photon emission computed tomography (SPECT)/CT, or (3) pathologically proven adenoma after adrenalectomy/cure of hypertension or improvement in hypertension, biological parameters after adrenalectomy. 22
A diagnosis of idiopathic adrenal hyperplasia (IHA) PA patients was based on the following criteria: (1) bilateral diffuse enlargement of adrenal glands on a CT scan, (2) non-lateralization of aldosterone secretion in adrenal vein sampling or during a dexamethasone suppression NP-59 SPECT/CT, or (3) pathologically reported diffuse cell hyperplasia for those who underwent adrenalectomy. 22
PWV measurements
We used an automatic waveform analyzer (Colin VP-2000, Omeron Inc., Japan) to measure the patients’ PWV. Each measurement was done after a 15-minute resting period in the supine position. The analyzer simultaneously recorded pulse waves of bilateral brachial arteries, phonocardiogram and electrocardiogram. Occlusive cuffs were wrapped around the upper arms and ankles and connected to oscillatory and plethysmography sensors to measure and analyze BP and pulse waveforms. Wave front theory was applied to estimate the conduction time differences. The brachial-ankle PWV at each side was obtained by dividing the brachial-ankle distance by the differences in conduction time. The average PWV from both sides was used in the analysis.
Statistical analysis
For sample size calculation, based on the results from a previous study, 23 a minimum 74 patients in adrenalectomy and 74 patients in MRA treatment group is required to achieve a type I error of 0.05 and a power of 99%.
Continuous variables were expressed as mean ± SD. The pre- and post-treatment results were compared using the paired t-test. To balance confounders between the two treatment strategy groups, we performed 1-to-1 propensity score matching for age, sex, pre-treatment systolic BP (SBP) and diastolic BP (DBP) in order to eliminate the influences of these factors on PWV.
We analyzed the relationship between treatment method and outcomes with univariable analysis. We used Pearson’s correlation analysis and univariable analysis to determine the potential candidates associated with pre- and post-treatment PWV and ∆PWV. Factors with a significant correlation (p < 0.05) were further tested in multivariable linear regression analysis with stepwise selection to identify the independent predicting factors. We used 2-way repeated measures ANOVA to evaluate the effect of treatment type on outcome. PAC, PRA and ARR were log-transformed because of their non-normality, as tested by the Kolmogorov-Smirnov test. p values less than 0.05 were considered statistically significant. All statistical analyses were conducted on IBM® SPSS® Statistic 25th ed.
Results
Clinical and biochemical characters before and after propensity score matching
Before propensity score matching, compared with the MRA group, the patients in the adrenalectomy group were younger, and used more kinds of anti-hypertensive medications. For the biochemical findings, the adrenalectomy group had a lower serum potassium level, lower log-transformed PRA, higher log-transformed ARR, and lower PWV (Table 1).
Baseline characters.

ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; ARR, aldosterone to renin ration; BMI, body-mass index; BSA, body surface area; CCB, calcium channel blocker; Cre, serum creatinine level; DBP, diastolic blood pressure; HTN, hypertension; K, serum potassium level; MAP, mean arterial blood pressure; MRA, mineralocorticoid antagonist; log_, log-transformed; PAC, plasma aldosterone concentration; PRA, plasma renin activity; PWV, pulse wave velocity; SBP, systolic blood pressure.
p < 0.05.
p < 0.001.
After 1:1 propensity score matching for age, sex, pre-treatment systolic and diastolic BP, there were 176 patients in each group. Compared with the MRA group, the adrenalectomy group used more kinds of anti-hypertensive medications, had a lower average dose of spironolactone, lower serum potassium level, lower log-transformed PRA, and higher log-transformed ARR. However, the pre-treatment PWV became similar after propensity-score matching (Table 1) [Figure 1(a)].
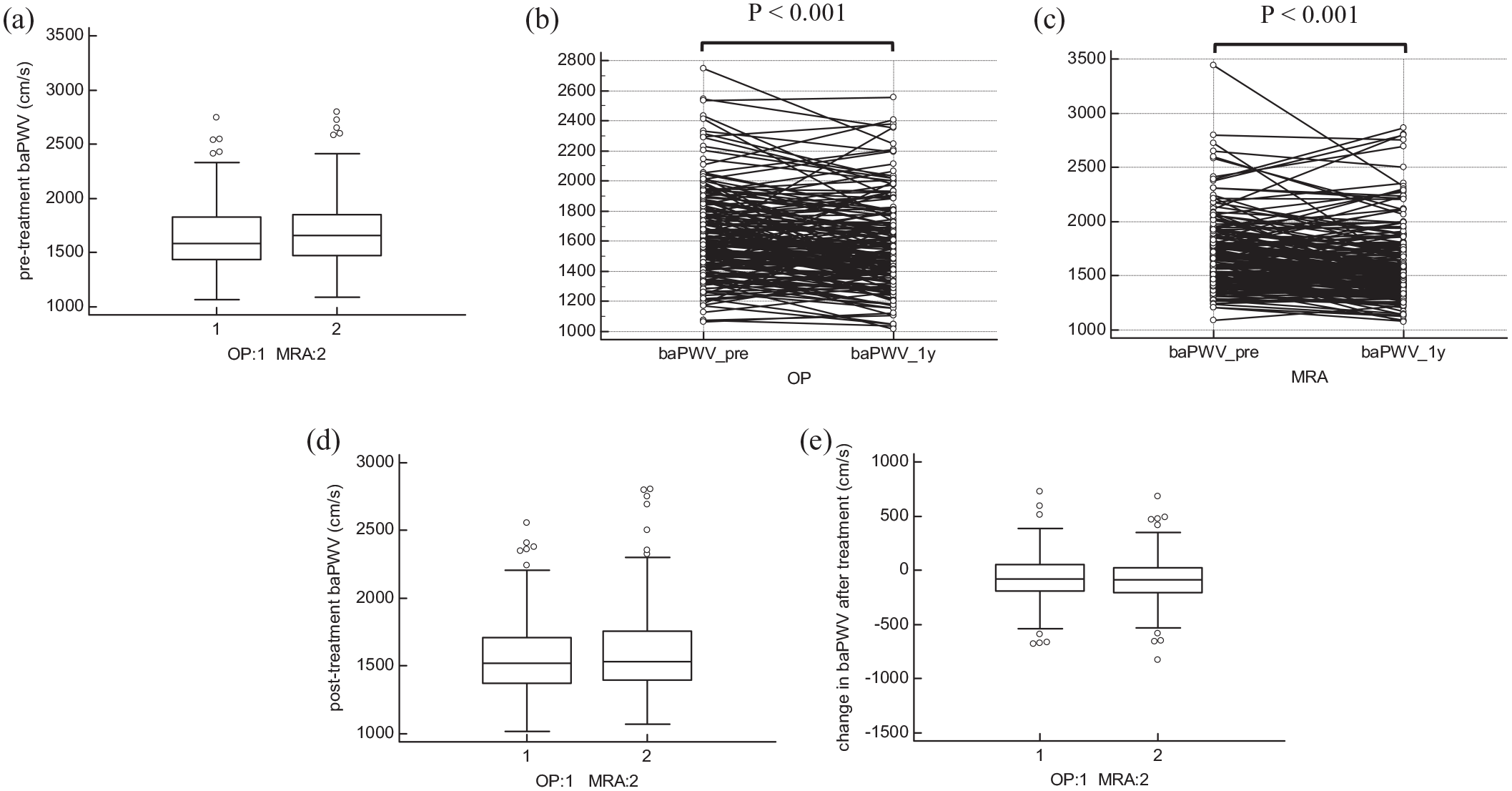
(a) Comparison of pre-treatment PWV, (b) the change in PWV in adrenalectomy group after treatment, (c) the change in PWV in MRA group after treatment, (d) comparison of post-treatment PWV, and (e) comparison of the change in PWV after treatment.
Determinant factors for baseline PWV
We used Pearson correlation analysis to identify the potential clinical determinant factors for baseline PWV and tested the significance of the candidates with univariable linear regression analysis. The candidates with significant correlations (p < 0.05) were further entered into a multivariable linear regression model. The results showed that baseline PWV was determined by the patients’ age, and baseline mean BP (Table 2).
Determinant factors for baseline PWV.
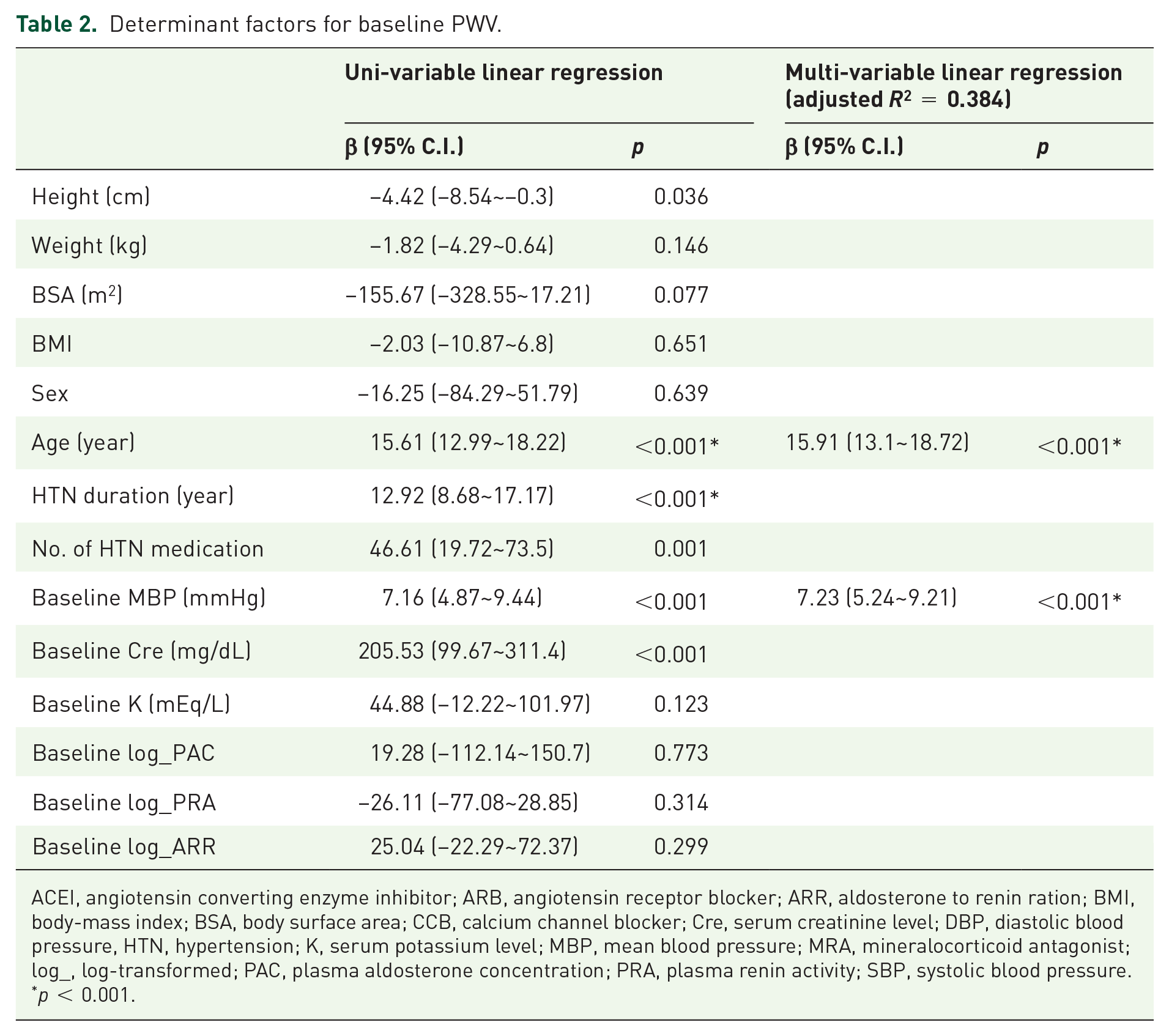
ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; ARR, aldosterone to renin ration; BMI, body-mass index; BSA, body surface area; CCB, calcium channel blocker; Cre, serum creatinine level; DBP, diastolic blood pressure, HTN, hypertension; K, serum potassium level; MBP, mean blood pressure; MRA, mineralocorticoid antagonist; log_, log-transformed; PAC, plasma aldosterone concentration; PRA, plasma renin activity; SBP, systolic blood pressure.
p < 0.001.
Clinical and biochemistry parameters improved after both treatments
BP, serum potassium level, characteristic biomarkers in PA patients (PAC, PRA, ARR) and PWV all significantly improved after treatment in both groups. Serum creatinine level increased significantly 1 year after treatment in both groups; however, the average serum creatinine level was still within normal range (Table 3) [Figure 1(b) and (c)].
Treatment results (propensity-score-matched).

ARR, aldosterone to renin ration; BMI, body-mass index; BSA, body surface area; Cre, serum creatinine level; DBP, diastolic blood pressure; K, serum potassium level; log_, log-transformed; MBP, mean blood pressure; PAC, plasma aldosterone concentration; PRA, plasma renin activity; PWV, pulse-wave velocity; SBP, systolic blood pressure.
Comparison of the treatment results between adrenalectomy and MRA treatment
Post-treatment SBP, DBP, log-transformed PAC, and log-transformed ARR were significantly lower in the adrenalectomy group than in the MRA group. The adrenalectomy group also had greater improvements in SBP, DBP, serum potassium level, log-transformed PAC, and log-transformed ARR. The serum creatinine level in the adrenalectomy group increased slightly more than in the MRA group after treatment. As for PWV, there were no significant differences in post-treatment PWV and improvement in PWV between the two treatment types (Table 4) [Figure 1(d) and (e)]. By 2-way repeated measures ANOVA, the type of treatment did not have main effect on PWV (F score 0.379, p value 0.539, partial eta squared 0.002).
Comparison of post-treatment Results, propensity-score-matched.
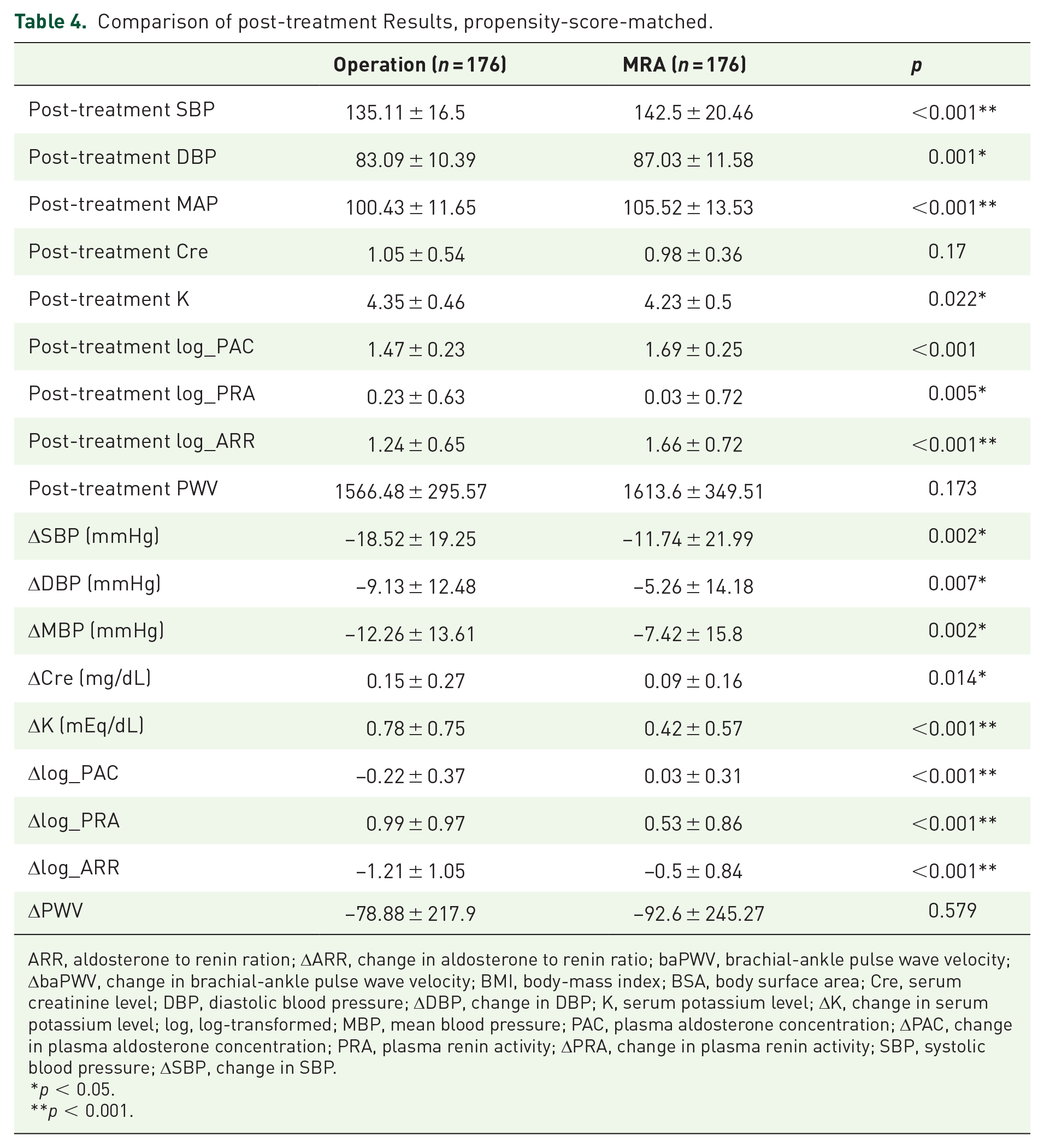
ARR, aldosterone to renin ration; ΔARR, change in aldosterone to renin ratio; baPWV, brachial-ankle pulse wave velocity; ΔbaPWV, change in brachial-ankle pulse wave velocity; BMI, body-mass index; BSA, body surface area; Cre, serum creatinine level; DBP, diastolic blood pressure; ΔDBP, change in DBP; K, serum potassium level; ΔK, change in serum potassium level; log, log-transformed; MBP, mean blood pressure; PAC, plasma aldosterone concentration; ΔPAC, change in plasma aldosterone concentration; PRA, plasma renin activity; ΔPRA, change in plasma renin activity; SBP, systolic blood pressure; ΔSBP, change in SBP.
p < 0.05.
p < 0.001.
Determinant factors for the improvement in PWV
We used the same method as in the previous section to identify the determinant factors for the improvement in PWV after treatment. The results showed that hypertension duration, baseline PWV, and improvement in mean arterial BP were strongly associated with the improvement in PWV (Table 5).
Determinant factor to ΔPWV.

ARR, aldosterone to renin ration; ΔARR, change in aldosterone to renin ratio; BMI, body-mass index; BSA, body surface area; Cre, serum creatinine level; ΔCre, change in serum creatinine level; DBP, diastolic blood pressure; ΔDBP, change in DBP; K, serum potassium level; ΔK, change in serum potassium level; log, log-transformed; MBP, mean blood pressure; PAC, plasma aldosterone concentration; ΔPAC, change in plasma aldosterone concentration; PRA, plasma renin activity; ΔPRA, change in plasma renin activity; PWV, pulse wave velocity; SBP, systolic blood pressure; ΔSBP, change in SBP.
p < 0.05.
p < 0.001.
Discussion
There are four major findings in this study. First, the PA patients who received medical treatment with a MRA had significant improvements in arterial wall stiffness. Second, there was no significant difference between the two treatment strategies. Third, baseline PWV was determined by age and baseline BP. Fourth, the improvement in PWV after treatment was determined by hypertension duration, pre-treatment PWV, and improvement in BP.
PWV is widely used as a non-invasive method to measure arterial stiffness. 6 High BP is well known to have a major impact on vascular physiology, causing vascular remodeling, endothelial dysfunction, and arterial wall stiffness through complex molecular and cellular mechanisms. 24 This is consistent with our results, in that we found that baseline BP was a strong determining factor for both baseline PWV and post-treatment improvement in PWV, similar with a previous study. 25
In addition to the effects of high BP, aldosterone can influence vascular smooth muscle and endothelium via genomic and non-genomic mechanisms,26,27 impair vascular tone, cause systemic inflammation, and lead to early atherosclerosis. 28 Small artery remodeling (increased collagen, fibronectin and intercellular adhesion molecule-1 [ICAM-1]) has been reported in aldosterone-treated rats, 29 and increased fibronectin, elastin, and collagen have been reported in the large arteries of hyperaldosteronism rats. 30 Various mediators have been shown to participate in mineralocorticoid receptor (MR)-related vascular remodeling, including platelet-derived growth factor (PDGF), angiotensin II, and endothelin-1, mostly by activating the receptors located in the walls of vascular smooth muscle cells. 31 In animal studies, excessive aldosterone has been demonstrated to induce perivascular macrophage infiltration, 32 vascular remodeling with direct effects on smooth muscle cells, 33 and periaortic collagen deposition. 34
Endothelial cell MRs (EC-MRs) have been correlated with vascular stiffness and dysfunction,35,36 and to contribute to arterial wall stiffness by promoting the expressions of sodium ion channels. This may cause EC stiffness and affect adjacent smooth muscle cells.37–39 MRAs may reduce arterial stiffness by blocking EC-MRs. 40 In addition, MRAs may limit extracellular matrix turnover, and hence reduce tissue fibrosis. 41
In our present and previous studies, we found that PAC, PRA and ARR were not determining factors for baseline or post-treatment improvement in PWV. This indicates that these characteristic biomarkers are not appropriate parameters to evaluate the exact physiological impacts of chronic exposure to elevated aldosterone on the cardiovascular system of PA patients. Twenty-four-hour urine aldosterone has been proposed to be a better predictor of cardiovascular damage in PA patients than traditional biochemical markers. 42
In the present study, both surgery and medical treatment resulted in significant improvements in BP and arterial wall stiffness, as well as in other PA characteristic biochemical parameters, including PRA, ARR and serum potassium level. Both groups had significant elevations in post-treatment serum creatine level (although still within normal limits), and the increase in the adrenalectomy group was slightly higher than that in the MRA group. However, a previous study reported a better-long term outcome with adrenalectomy than MRA treatment. 43 Therefore, our data may represent only a short-term effect rather than long-term outcomes.
We found greater improvements in BP, serum potassium, PAC and ARR in the adrenalectomy group, but similar improvements in arterial stiffness between the adrenalectomy and MRA groups. This is consistent with a previous study which reported that spironolactone treatment could also reduce arterial stiffness independently of controlling BP. 44
To focus on the influence of treatment method and eliminate other confounding factors, we used 1:1 propensity score matching for age, sex and BP. Before propensity score matching, the MRA group had higher brachial-ankle PWV than the adrenalectomy group. After matching, there was no significant difference in PWV between the two groups, showing that PWV is highly correlated with those factors. To the best of our knowledge, this is by far the largest prospective cohort study directly comparing the outcome of arterial stiffness after surgical and medical treatment in PA patients.
Numerous studies regarding the effects of anti-hypertensive medication on arterial wall stiffness have been proposed. Among them the medication targeting on renin-angiotensin (RAS) system are generally viewed superior in reducing arterial stiffness than other classes.45,46 There are no significant differences in prescribed with angiotensin-converting enzyme inhibitor (ACEI) or angiotensin-receptor blocker (ARB) between our two treatment groups after propensity-score matching. Beyond the BP lowering effects, ACEI might also reduce inflammation, oxidative stress, cause smooth muscle relaxation, vascular wall recomposition and increase arterial compliance. 46 ARB could decrease central BP and arterial stiffness as well.47,48 Calcium-channel blocker (CCB) such as amlodipine is effective in improving arterial stiffness. 49 This might result from reduction in terminal peripheral resistance and stiffness of smaller muscular arteries. However, CCB present weaker effects in decreasing arterial stiffness than ARB. 50 Diuretics other than MRA (mainly thiazide) are one of the major first-line anti-hypertensive medication classes. 51 Despite their significance in decreasing BP, they exerted less effects on arterial stiffness than other classes. The depletion in sodium and water while treated with hydrochlorothiazide may trigger the RAAS system which offset the BP-lowering benefits to arterial stiffness. 52 ACEI/ARB, CCB or diuretics had been observed with more favorable effects on arterial stiffness than conventional beta-blockers. However, newer beta-blocker such as nebivolol which exerts protective effects on oxidative stress, increases NO and improved small artery distensibility index and endothelial function, presents more prominent benefits on arterial stiffness than the older atenolol. 53
In PA patients, aldosterone has been shown to induce vascular wall inflammation, 54 vascular calcification 55 and perivascular fibrosis. 56 Aldosterone also increases early atherosclerosis and promotes plaque formation by activating the MR on smooth muscle cells. 57 This activation could also enhance active stiffness by direct increase in vascular myogenic tone and agonist-dependent contraction. 58 Aldosterone may also increase arterial stiffness through interfering serum potassium level while hypokalemia in PA patients is correlated with their arterial stiffness 59 as well as cardiac structural change and dysfunction. 60 The reversal of arterial stiffness by MRAs potentially results from blocking MR activation and regulating serum potassium level. 61 Our present and previous studies confirmed that the arterial stiffness caused by excess aldosterone in PA patients is effectively reversed after treatment with MRAs.
There are several limitations to this study. First, the choice of treatment was influenced by the result of lateralization. When diagnosed with APA, many patients are treated by adrenalectomy, while those diagnosed with idiopathic hyperaldosteronism are always treated with MRAs. This may have interfered with our results, and further studies are needed to clarify this issue. Second, we only provided the results after 1 year of follow-up. A longer follow-up period is needed to clarify the long-term effects of each treatment in PA patients. Third, based on our prospective-enrollment-retrospective-analysis design the vast variety of medication combination in the study population could not adequately avoid the confounding effect of each drug.
Conclusion
The PA patients who received medical treatment with MRAs had a significant improvement in arterial stiffness. There was no significant difference in the improvement in arterial stiffness between the two treatment strategies.
Footnotes
Appendix 1
Membership of the Taiwan Primary Aldosteronism Investigation (TAIPAI) Study Group: Tai-Shuan Lai; Vin-Cent Wu; Shao-Yu Yang; Kao-Lang Liu; Chin-Chen Chang; Bo-Chiag Lee; Shuo-MengWang; Kuo-How Huang; Po-Chih Lin; Yen-Hung Lin; Lian-Yu Lin; Shih-Cheng Liao; Ruoh-Fang Yen; Ching-Chu Lu; Shih-Chieh Jeff Chueh (National Taiwan University Hospital, Taipei, Taiwan); Chieh-Kai Chan (NTUH Hsin-Chu branch); Leay-Kiaw Er; Ya-Hui Hu; Chia-Hui Chang; Che-HsiungWu; Yao-Chou Tsai (Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, Taipei, Taiwan); Chen-Hsun Ho (Taipei Medical University-Shuang Ho Hospital, Ministry of Health and Welfare); Wei-Chieh Huang (Taipei Veterans General Hospital); Ying-Ying Chen (MacKay Memorial Hospital); Kwan-Dun Wu (National Taiwan University Hospital, Taipei, Taiwan NTUH, Director of Coordinating Center).
Acknowledgements
We are greatly thankful to Hsing-Fen Lin for his advises to the statistical analyses while completing the manuscript and revision. We also appreciate the assistance from the staff of the Second Core Lab in the Department of Medical Research in National Taiwan University Hospital and all the members in the TAIPAI study group (![]() ).
).
