Abstract
Background:
The urinary sodium potassium (NaK) ratio is associated with dietary sodium and potassium intake and blood pressure, and it also reflects the activity of aldosterone. Herein we evaluated the value of the urinary NaK ratio in predicting the surgical outcomes of patients with unilateral primary aldosteronism (uPA).
Methods:
This non-concurrent prospective cohort study was conducted from 2011 to 2017 and included 241 uPA patients who had undergone adrenalectomy. Predictors of successful clinical outcomes were analyzed using logistic regression.
Results:
Among the 241 uPA patients, 197 (81.7%) achieved clinical complete or partial success. A urinary sodium potassium ratio <3 (odds ratio (OR): 2.5; 95% confidence interval (CI): 1.2–5.4; p = 0.015), body mass index <25 kg/m2 (OR: 2.82; 95% CI: 1.31–6.06; p = 0.008), renin <1 ng/mL/h (OR: 2.51; 95% CI: 1.01–6.21; p = 0.047) and mean preoperative blood pressure >115 mmHg (OR: 5.02; 95% CI: 2.10–11.97; p < 0.001) could predict clinical success after adrenalectomy. Furthermore, higher pre-treatment plasma aldosterone (OR: 1.014; 95% CI 1.005–1.024; p = 0.002) or lower serum potassium (OR: 0.523; 95% CI: 0.328–0.836; p = 0.007) were correlated with lower urinary NaK ratio (<3), and log urinary NaK ratio was positively correlated with serum C-reactive protein (β value 2.326; 95% CI 0.029–4.623; p = 0.047).
Conclusions:
uPA patients with a lower urinary NaK ratio, due to high plasma aldosterone and low serum potassium concentrations, were more likely to have clinical success after adrenalectomy. uPA patients with a higher urinary NaK ratio were associated with more severe inflammatory status, and possibly more resistant hypertension post-operatively.
Keywords
Introduction
Primary aldosteronism (PA), the most common form of secondary hypertension, is caused by an excess and unregulated production of aldosterone. The characteristics of PA include hypokalemia, hypertension and metabolic alkalosis. The early identification of PA is important, because it has been associated with higher rates of cardiovascular and cerebrovascular complications1,2 and a higher risk of developing metabolic syndrome compared to age and sex-matched patients with essential hypertension. 3 In addition, early targeted treatments can improve blood pressure (BP) control and completely or partially reverse these complications.4,5
Ideally, unilateral adrenalectomy could correct the over-production of aldosterone and further resolve the hypertension in patients with unilateral primary aldosteronism (uPA), but BPs of some uPA patients did not improve after adrenalectomy. Few studies have investigated the predictors of successful clinical outcomes after adrenalectomy for uPA.6–8 Moreover, the definition and classification of clinical outcomes adopted by these studies have varied widely.
It is well known that a 24-hour urinary sodium potassium (NaK) ratio can reflect dietary sodium and potassium intake, 9 both of which impact significantly on BP.10,11 Besides, a 24-hour urinary NaK ratio may also be an indicator of aldosterone function because the epithelial sodium channel (ENaC) in the renal collecting duct is responsible for sodium reabsorption and subsequent potassium excretion under the action of aldosterone. Furthermore, the urinary NaK ratio has been reported to be strongly associated with cardiovascular outcomes, renal outcomes and all-cause mortality in the general population.11,12
In this study, we evaluated the role of the urinary NaK ratio in predicting clinical success after adrenalectomy in patients with uPA. We used the consensus on surgery outcomes for uPA as defined by the Primary Aldosteronism Surgery Outcome (PASO) study, and classified clinical outcomes as complete, partial and absent clinical success. 6 As several studies have reported that there are positive correlations between the urinary NaK ratio and inflammation, in this study we also examined if there is any association between the urinary NaK ratio and the severity of inflammation in the uPA population.
Materials and methods
Patients
This non-concurrent prospective cohort study was based on data from the Taiwan Primary Aldosteronism Investigation (TAIPAI) study group.13–21 The patients in this study group were recruited from two medical centers, three affiliated hospitals and two regional hospitals in different cities in Taiwan. Patients who were suspected of having PA were referred to the TAIPAI study group for aldosterone-to-renin ratio (ARR) screening and confirmation tests. We excluded patients with other causes of secondary hypertension including renovascular hypertension, Cushing’s syndrome, hyperthyroidism, and pheochromocytoma. From 2011 to 2017, we enrolled 241 uPA patients who further underwent a unilateral adrenalectomy and have been followed for at least 1 year postoperatively.
Screening, diagnosis and subtype classification
Diuretics and mineralocorticoid receptor antagonists were withheld for more than 6 weeks and all the other antihypertensive medications were withheld for at least 21 days before tests. Diltiazem and/or doxazosin were administered to control hypertension during those periods when required. Other medications which may have interfered with the renin–angiotensin–aldosterone system, including steroids, oral contraceptives, and non-steroidal anti-inflammatory drugs were also withheld. 19 Patients were instructed to maintain their usual salt intake during the study.
The diagnosis of PA was confirmed if all the three of the following criteria were fulfilled: (a) autonomous excess aldosterone production evidenced with a 24-h urinary aldosterone level (Uald-24 h) > 20.3 µg; 22 (b) a TAIPAI score > 60%; (c) seated post-saline infusion test (SIT) aldosterone level > 16 ng/dL, or ARR > 35 (ng/dL)/(ng/mL/h) obtained in a post-captopril test. 23
The TAIPAI score is a score calculated by a logistic regression model, which can predict the probability of having PA.
where β = (PAC [ng/dl] × [0.063] + (PRA [ng/ml/h] × [−0.205]) + ([ARR × 0.001] + BMI [kg/m2] × [0.067]) + (male × [−0.738] + SK [mmol/l] × [−1.512]) + (eGFR [ml/min/1.73 m2] × [0.017]) + ([propensity score] × [−0.539] + [1.851])
Where PAC = plasma aldosterone concentration, PRA = plasma renin activity, BMI = body mass index, SK = serum potassium level.
Unilateral PA (uPA) was identified in the PA patients who had the following: (a) lateralization of aldosterone secretion at adrenal vein sampling or during dexamethasone suppressing NP-59 SPECT/CT; or (b) pathologically proved adenoma after an adrenalectomy and positive staining for CYP11B2.19,24,25
Laboratory studies
We obtained complete demographic and laboratory data from the study group. Demographic data included age, sex, body mass index (BMI), BP, antihypertension medications and hypertension duration. Laboratory data included levels of potassium, serum creatinine, and 24-h urine albumin, estimated glomerular filtration rate (eGFR; calculated using the Chronic Kidney Disease Epidemiology Collaboration formula), 26 renin and aldosterone levels, ARR, and 24-h urinary NaK ratio. All the laboratory tests of a certain patient were collected and measured at the confirmation stage.
During the confirmation stage, we specifically instructed them to have liberal/usual salt intake as the guideline for the diagnosis of primary aldosteronism recommends.
The aldosterone concentration was measured by radioimmunoassay using a commercial kit (Aldosterone Maia Kit; Adaltis Italia SPA, Bologna, Italy). The lowest detectable concentration of aldosterone was 1.0 ng/dL. The normal range of plasma aldosterone concentration is 7–35 ng/dL with the patient in an upright position. Plasma renin activity (PRA) was measured as the generation of angiotensin I in vitro using a commercially available radioimmunoassay kit (DiaSorin, Stillwater, MN, USA). The normal range of PRA is 2.63 ± 1.32 ng/mL/h with the patient in an upright position. The mean [standard deviation (SD)] intra and interassay coefficients of variation for the PRA assay were 1.9 (5.0%) and 4.5 (5.2%), respectively. The C-reactive protein (CRP) in plasma was measured by a latex-enhanced immunoturbidimetric test/immunoturbidimetry (Beckman Coulter, Brea, CA, USA). The normal range of CRP is <10 mg/L. The correlation coefficient was 0.999.
Outcomes
We defined the clinical outcomes according to the PASO consensus: clinical success (complete and partial clinical success), indicating normal BP without antihypertension medication (hypertension remission; complete clinical success), the same BP with less antihypertension medication, or a reduction in BP with either the same amount or less antihypertension medication (partial clinical success) at 1 year after surgery. 27 Resistant hypertension was defined as unchanged or increased BP levels with either the same amount or an increase in antihypertension medication at 1 year after surgery. 6
Statistical analysis
For the baseline characteristics, continuous variables with normal distribution were expressed as mean ± SD; non-normally distributed variables were reported as median (interquartile range), and categorical variables were expressed as frequency and percentage. The Mann–Whitney U test or Student’s t-test was used to compare differences among the means of continuous variables, and the chi-squared or Fisher’s exact test was used for categorical variables. Multiple logistic regression analysis was used to identify the independent predictors of successful clinical outcomes. Variables which have been reported to be known predictors, including age and sex and those with a p value < 0.1 (including BMI, mean BP, renin, eGFR, urinary NaK ratio) in univariate logistic regression analysis were included into multivariate logistic regression analysis. Pearson’s correlation test was used to evaluate correlations between continuous variables, and multivariate linear regression analysis was used to evaluate the predictors for continuous variables.
Computationally, we used the vgam() function with the default values of the smoothing parameters [e.g. s(age, df = 4, spar = 0) for the cubic smoothing splines] in the vector generalized additive model (GAM) package to fit the GAMs for continuous, binary, and count responses respectively, and then used the plotvgam() function of the same package to draw the GAM plots for visualizing the linear or non-linear effects of continuous covariates in R.14,28 The GAM plots were adjusted with age, sex, BMI, mean blood pressure, eGFR and renin.
We used R software version 3.4.4 (Free Software Foundation, Inc., Boston, MA, USA) 29 and IBM SPSS statistics version 24 (IBM Corp., Armonk, NY, USA) software. A two-sided p value < 0.05 was considered to be statistically significant.
Ethics statement
This study complied with the Declaration of Helsinki and was approved by the National Taiwan University Hospital Research Ethics Committee (no. 200611031R). All participants were given comprehensive written information and signed a consent form before inclusion into the study.
Results
During the study period, 241 uPA patients who underwent a unilateral adrenalectomy were included. The mean age of the patients was 50.6 ± 11.2 years, and 41.1% were men (Table 1). The types, numbers and percentages of various antihypertensive medications these patients were prescribed before the study enrollment were: angiotensin-converting enzyme inhibitors in 11 (4.6%), angiotensin receptor blockers in 92 (38.2%), alpha blockers in 57 (23.7%), beta-blockers in 102 (42.3%), calcium antagonists in 177 (73.4%), vasodilators in 15 (6.2%), and diuretics in 27 (11.2%).
Basic characteristic of the PA patients with outcomes of clinical success and absent clinical success.
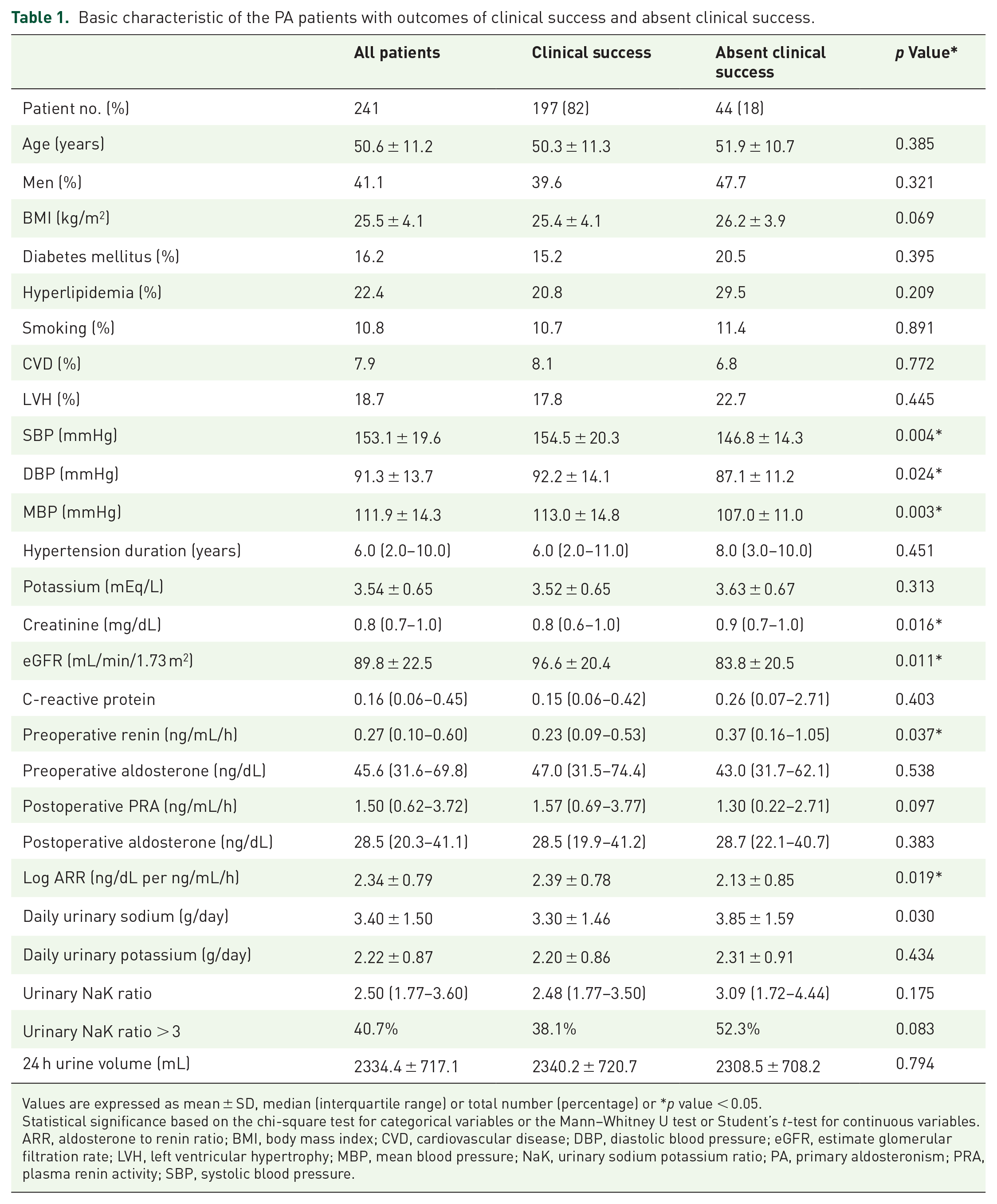
Values are expressed as mean ± SD, median (interquartile range) or total number (percentage) or *p value < 0.05.
Statistical significance based on the chi-square test for categorical variables or the Mann–Whitney U test or Student’s t-test for continuous variables.
ARR, aldosterone to renin ratio; BMI, body mass index; CVD, cardiovascular disease; DBP, diastolic blood pressure; eGFR, estimate glomerular filtration rate; LVH, left ventricular hypertrophy; MBP, mean blood pressure; NaK, urinary sodium potassium ratio; PA, primary aldosteronism; PRA, plasma renin activity; SBP, systolic blood pressure.
Among the 241 patients, 197 (81.7%) achieved clinical success (including complete and partial success) and 44 (18.3%) failed to achieve clinical success (absent clinical success group) after adrenalectomy. During the confirmatory period, the clinical success group had a higher preoperative systolic BP (154.49 ± 20.30 versus 146.84 ± 14.34 mmHg; p = 0.004) and diastolic BP (92.22 ± 14.10 versus 87.07 ± 11.20 mmHg; p = 0.024) than the absent clinical success group. The clinical success group also had better renal function than the absent clinical success group (eGFR: 96.60 ± 20.44 versus 83.84 ± 20.47 mL/min/1.73 m2; p = 0.001). Moreover, renin [0.23 (0.09–0.53) versus 0.37 (0.16–1.05) ng/mL/h; p = 0.037] was lower and log ARR (2.39 ± 0.78 versus 2.13 ± 0.85; p = 0.019) was higher in the clinical success group (Table 1).
There were no statistically significant differences in age, sex, history of diabetes mellitus, hyperlipidemia, left ventricular hypertrophy, duration of hypertension and potassium level between the two groups (Table 1).
Factors predicting clinical outcomes
A GAM was used to identify the best cut-off point of the urinary NaK ratio to predict the clinical outcomes. The results showed that a urinary NaK ratio of <3 was the best cut-off point to predict clinical success with a high positive predictive value (0.85) (Figures 1 and 2). We used 25 kg/m2 as the cut-off point for BMI and 1 ng/mL/h as the cut-off point for plasma renin activity according to the general definitions of obesity and suppressed renin activity, respectively. 30 We used 115 mmHg as the cut-off point for mean BP according to our previous study. 27

Generalized additive model (GAM) plot for the association between urinary NaK ratio and absent clinical success.

Association between potassium and urinary NaK ratio. r2 = 0.044, p = 0.015. NaK, urinary sodium potassium ratio
We further used logistic regression to identify the factors associated with clinical success. In multivariate logistic regression, in addition to low BMI (<25 kg/m2) (odds ratio (OR): 2.82; 95% confidence interval (CI): 1.31–6.06; p = 0.008), high mean BP (>115 mmHg) (OR: 5.02; 95% CI: 2.10–11.97; p < 0.001), low renin (<1 ng/mL/h) (OR: 2.51; 95% CI: 1.01–6.21; p = 0.047), and low urinary NaK ratio (<3) (OR: 2.53; 95% CI:1.19–5.36; p = 0.015) were also significantly independently associated with clinical success (Table 2).
Factors associated with clinical success after adrenalectomy for lateralized primary aldosteronism.

p Value < 0.05.
Statistical significance based on logistic regression.
BP, blood pressure; CI, confidence interval; eGFR, estimate glomerular filtration rate; NaK, urinary sodium potassium ratio.
The accuracy, area under the curve (AUC), sensitivity and specificity of the model including urinary NaK ratio only were 81.7%, 0.571, 100% and 0%, respectively. The accuracy, AUC, sensitivity and specificity of a model that included urinary NaK ratio, age, sex, eGFR, BMI, renin, mean blood pressure and renin were 83%, 0.747, 98% and 15%, respectively.
Factors associated with urinary NaK ratio
Patients with a low urinary NaK ratio (<3) had a higher aldosterone level (62.02 ± 39.00 versus 48.08 ± 29.10 ng/dL; p = 0.002), higher log ARR (2.45 ± 0.80 versus 2.18 ± 0.74 ng/dL per ng/mL/h; p = 0.008), lower renin (0.82 ± 2.64 versus 1.34 ± 4.87 ng/mL/h; p = 0.049) and lower serum potassium level (3.45 ± 0.65 versus 3.68 ± 0.62 mEq/L; p = 0.035) than the patients with a high urinary NaK ratio (Supplemental Table 1). In multivariate logistic regression, higher aldosterone (OR: 1.013; 95% CI 1.004–1.022; p = 0.002) and lower potassium (OR: 0.538; 95% CI: 0. 0.342–0.845; p = 0.007) levels were associated with a low urinary NaK ratio (Supplemental Table 2). Pearson’s correlation test also showed a positive correlation between potassium (Figure 3) (r = 0.044, p = 0.015) and the urinary NaK ratio, as well as a negative correlation between aldosterone and the urinary NaK ratio (r = 0.021, p = 0.011) (Figure 4).

Association between aldosterone and urinary NaK ratio. r2 = 0.021, p = 0.011. NaK, urinary sodium potassium ratio.

Generalized additive model (GAM) plot for the association between C-reactive protein and log urinary NaK ratio.
Association between the urinary NaK ratio and CRP
Multiple linear regression analysis revealed that log urinary NaK ratio could predict CRP with significant statistical significance (β value = 2.449; 95% CI: 0.018–4.880; p = 0.048) (Supplemental Table 3). A non-linear model showed that the urinary NaK ratio was positively associated with the serum CRP level, especially when the urinary NaK ratio was >0.49 (p = 0.025) (Figure 4).
Discussion
To the best of our knowledge, this is the first study to examine the role of the urinary NaK ratio in predicting clinical outcomes after adrenalectomy in patients with uPA. We found that a lower urinary NaK ratio, largely due to high aldosterone concentration and low serum potassium level, with an optimal cut-off point of 3, could predict the clinical success of hypertension after adrenalectomy. Besides, we demonstrated that mean BP > 115 mmHg, BMI < 25 kg/m2, and renin < 1 ng/mL/h could predict clinical success in uPA patients receiving adrenalectomy. We also found a positive correlation between the urinary NaK ratio and the serum CRP level, an indicator of inflammation.
The role of the urinary NaK ratio in predicting clinical success after adrenalectomy
Previous studies have shown that the 24-hour urinary NaK ratio can reflect dietary sodium and potassium intake, 9 and thus that it is associated with BP, 31 stroke32,33 and the progression of chronic kidney disease.9,11 The primary function of aldosterone is to act on epithelial sodium channels on renal distal tubules and collecting ducts to stimulate sodium absorption and potassium excretion. An increased plasma aldosterone concentration will decrease the urinary NaK ratio, and therefore the urinary NaK ratio can also reflect the activity of aldosterone. 34 Taken together, the urinary NaK ratio can be regarded as reflecting the combined effect of dietary salt intake and aldosterone activity.
The mean urinary NaK ratio in our study was 2.86 ± 1.44. This value is lower than that in a large prospective study conducted in China, which represented the NaK ratio with a general Chinese diet. 35 The result is compatible with our expectation that due to the impact of a higher plasma aldosterone concentration, the urinary NaK ratio in uPA patient is lower than that in the general population. In our study, we also demonstrated that a high plasma aldosterone concentration and low serum potassium were associated with a low urinary NaK ratio, and that a low urinary NaK ratio (<3) could forecast clinical success after adrenalectomy. That is, among uPA patients, those with low urinary NaK ratio (<3), largely due to high aldosterone and low serum potassium, are more likely to benefit from surgery.
In addition to uPA patients, several observational studies have also shown that patients with many other diseases have better outcomes if their urinary NaK ratio is low. Averrill et al. found that a NaK ratio of ⩽1 may be related to a clinically relevant reduction in the risk of stroke. 32 In addition, Koo et al. found that the higher the urinary NaK ratio, the greater the risk of chronic kidney disease progression. 9
As the 24-h urinary NaK ratio is related to dietary sodium and potassium and patients with a low urinary NaK ratio (<3) had better surgical outcomes in this study, it is possible that those who had low salt and high potassium intake were also more likely to have successful outcomes after surgery. This is the first study to show that the clinical outcomes of uPA after adrenalectomy could also be affected by dietary salt intake.
Through complex and multifaceted interactions between white blood cells and vessel walls, systemic inflammation can accelerate atherosclerosis, impair endothelial function, cause arterial stiffness and finally increase the risk of cardiovascular disease.36,37 We found that a high log urinary NaK ratio was associated with a high CRP level, and these uPA patients with a higher urinary NaK ratio were also associated with a higher failure rate to have resistant hypertension after adrenalectomy. This could suggest that the mechanism underlying absent clinical success in uPA patients with a high urinary NaK ratio may at least partially be due to a more severe inflammatory status in these patients, thereby increasing the likelihood of developing atherosclerosis, vascular calcification, arterial stiffness, and to have residual hypertension after surgery. In line with our findings, a previous study also demonstrated that a high salt diet and therefore high urinary sodium and urinary NaK ratio was accompanied by the excessive production of inflammatory markers, and that this may have resulted in hypertension and poor cardiovascular outcomes. 38
Preoperatively high blood pressure and low renin could also predict clinical success after adrenalectomy
In this study, we demonstrated that high BP and suppression of renin were independently associated with clinical success after adrenalectomy. Both high BP and suppression of renin are indicators of mineralocorticoid receptor over-activation and/or fluid overload.39,40 Our results suggest that patients with higher mineralocorticoid receptor activation may have a better chance of achieving BP control after surgery. The mechanisms underlying this phenomenon are not entirely clear. However, it is possible that in patients with uPA, hyperaldosterone is the predominant cause of elevated BP, and that the removal of the adrenal gland effectively and completely cures the problem. Our findings are similar to those of the study by Weigel et al., 41 which also reported better outcomes in patients with more severe uPA with post-SIT aldosterone levels of 100 ng/l and higher systolic BP.
Obesity and clinical outcomes
Another finding of our study was a higher success rate after surgery in the patients with a BMI < 25 kg/m2. In the aldosteronoma resolution score established by Zarnegar et al., a BMI ⩽ 25 kg/m2 was one of the four factors that could predict the resolution of hypertension after surgery. 42 Excess fat tissue is associated with changes in several endocrine, inflammatory and endothelial substances, including adiponectin, leptin, and angiotensin II.43–45 These substances may cause renal sodium reabsorption, activation of the renin–angiotensin–aldosterone system and sympathetic systems, and finally induce arterial hypertension. In addition, adipose tissue hormones, known as adipokines, may directly affect the adrenal gland and increase aldosterone and mineralocorticoid receptor activity. Through these mechanisms, obese patients are associated with dysregulated homeostasis that may influence BP and aldosterone, leading to the persistence of hypertension despite adrenalectomy. 45
eGFR and clinical outcomes
Although the eGFR was significantly different between the clinical success and absent clinical success groups, we found the eGFR was not correlated with clinical outcome by way of multivariate logistic regression. We thought that the higher eGFR in the clinical success group may be confounded by other risk factors.
Strengths and limitations
This study has some important strengths. First, it was prospectively designed. Second, the screening and diagnosis of PA were standardized using the protocol of the Taiwan Society of Aldosteronism. Third, clinical outcome classification was based on the PASO criteria. 19 Fourth, it is the first study to evaluate the role of the urinary NaK ratio in predicting outcomes after adrenalectomy. Finally, and most importantly, we collected 24-h urine samples to calculate urinary sodium and potassium levels. As the urinary NaK ratio fluctuates with a diurnal pattern and is higher in the morning and evening and lower in the daytime, 46 our results are more reliable than results using spot urine samples. However, there are also some limitations to this study. First, our sample size was relatively small and the follow-up duration was relatively short, even though we had at least a 1-year postoperative period according to the PASO criteria. Second, we did not have information about some of the predictors mentioned in other studies, such as gene mutations, or control of patients’ sodium or potassium intake before the 24-h urine collection.
Conclusion
We found that the urinary NaK ratio could independently predict clinical outcomes after surgery in uPA patients. The patients with a low urinary NaK ratio, which was largely due to high aldosterone and low serum potassium, were more likely to have successful outcomes after surgery. The uPA patients with a high urinary NaK ratio were associated with severe inflammatory status. Preoperative BP, renin and BMI were also important predictors of clinical outcomes. Our findings may provide useful information to both clinicians and patients while evaluating the risks and benefits of adrenalectomy.
Supplemental Material
sj-pdf-1-taj-10.1177_2040622321990274 – Supplemental material for Urinary sodium potassium ratio is associated with clinical success after adrenalectomy in patients with unilateral primary aldosteronism
Supplemental material, sj-pdf-1-taj-10.1177_2040622321990274 for Urinary sodium potassium ratio is associated with clinical success after adrenalectomy in patients with unilateral primary aldosteronism by Ming-Jse Lee, Chiao-Yin Sun, Ching-Chu Lu, Yuan-Shian Chang, Heng-Chih Pan, Yen-Hung Lin, Vin-Cent Wu and Jeff S. Chueh in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
The authors greatly appreciate the technical support from the Second Core Lab in the Department of Medical Research in National Taiwan University Hospital for technical assistance. They also greatly appreciate English editing from Eric B. Chueh of Case Western Reserve University.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Taiwan National Science Council (MOST 106-2314-B-002-166-MY3, 106-2321-B-182-002, 107-2314-B-002-026-MY3), National Taiwan University Hospital–Taipei Veterans General Hospital Joint Research Program (#NTUH-TVGH VN103-01, #NTUH-TVGH VN104-07, #NTUH-TVGH VN105-04) and the National Taiwan University Hospital (105-S3061, 107-S3809, UN103-082, UN106-014, 105-P05, 106-P02, 107-T02).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

