Abstract
The burden of chronic hepatitis C virus (HCV) infection is significant and growing. HCV is considered one of the leading causes of liver disease worldwide and the leading cause of liver transplantation globally. While those infected is estimated in the hundreds of millions, this is likely an underestimation because of the indolent nature of this disease when first contracted. Approximately 20% of patients with HCV infection will progress to advanced fibrosis and cirrhosis. Those that do are at risk of decompensated liver disease including GI bleeding, encephalopathy, severe lab abnormalities, and hepatocellular carcinoma. Those individuals with advanced fibrosis and cirrhosis have historically been difficult to treat. The backbone of previous HCV regimens was interferon (IFN). The outcomes for IFN based regimens were poor and resulted in increased adverse events among those with advanced fibrosis and cirrhosis. Now, in the era of new direct acting antiviral (DAA’s) medications, there is hope for curing chronic HCV in everyone, including those with advanced fibrosis and cirrhosis. This article provides a review on the most up to date data on the use of DAA’s in patients with advanced fibrosis and cirrhosis. We are at a point where HCV could be truly eradicated, but to do so will require ensuring there are effective and safe treatments for those with advanced fibrosis and cirrhosis.
Introduction
Chronic hepatitis C virus (HCV) infection is one of the most common liver diseases in the world with 130–150 million people infected and is the leading cause of liver transplantation globally [Mohd Hanafiah et al. 2013; Wilder and Patel, 2014; World Health Organization, 2014]. Natural history studies suggest that 20% of patients with chronic HCV will develop cirrhosis [Di Bisceglie, 2000; Wilder and Patel, 2014]. The worldwide burden of cirrhosis is increasing and advanced fibrosis or cirrhosis accounts for the majority of the morbidity and mortality associated with HCV, including hepatocellular carcinoma (HCC) [Lavanchy, 2011; Mohd Hanafiah et al. 2013; Wilder and Patel, 2014]. Once cirrhosis develops, effective treatment reduces the risk of hepatic decompensation, HCC, liver transplantation, and overall mortality [Backus et al. 2011; Van Der Meer et al. 2012; Morgan et al. 2013].
Historically, a pillar of treatment for HCV has been interferon (IFN). Treatment with IFN-based regimens when patients had reached the point of cirrhosis was less effective and carried increased risk of adverse events [McHutchison et al. 2009]. Early experience treating decompensated cirrhosis revealed potential life-threatening complications [Crippin et al. 2002]. The associated morbidity from anemia, bleeding, and poor renal function has historically and currently continues to limit treatment options. Subsequent efforts initiated low doses of IFN and ribavirin with gradual increases, resulting in improved safety profiles but low response rates [Everson et al. 2005]. The morbidity and mortality of HCV-associated cirrhosis in the setting of poor treatment options prioritized identifying IFN-free regimens.
Direct-acting antivirals
Direct-acting antivirals (DAAs) act directly on various structural and nonstructural components of the 9.6 kb HCV genome, and based on their mechanism of action, have been combined to create treatment paradigms which form the basis for treatment regimens today.
The first wave of DAAs included the protease inhibitors boceprevir and telaprevir in 2011 (Table 1). They targeted genotype 1 infection and required combination with peginterferon and ribavirin [Bacon et al. 2011; Jacobson et al. 2011; Poordad et al. 2011; Sherman et al. 2011; Zeuzem et al. 2011]. Outcomes with these regimens were lower in compensated cirrhosis for both treatment-naive and experienced patients. The telaprevir REALIZE trial of treatment-experienced patients highlighted the challenges for patients with cirrhosis. The sustained virologic response (SVR) rate for prior null responders with cirrhosis was 14% (7/50) [Zeuzem et al. 2011]. The phase III clinical trials for boceprevir and telaprevir suggested generally similar safety profiles for treatment-experienced patients with and without cirrhosis, but safety concerns emerged among patients with cirrhosis outside of clinical trials. The CUPIC study group evaluated 674 genotype 1 patients with compensated cirrhosis prescribed 48 weeks of triple therapy [Hezode et al. 2013]. There were a significant amount of serious adverse events (40.0%) and severe complications including death and hepatic decompensation (6.4%). There was also a high incidence of severe anemia requiring erythropoietin (50.7%) or transfusion (12.1%) [Hezode et al. 2013]. Death and severe complications were more likely in subjects with thrombocytopenia (platelets count ⩽ 100,000/mm) and low serum albumin (albumin < 35 g/day) [Hezode et al. 2013]. These findings stressed the need to develop IFN-free regimens.
New approved direct-acting antivirals.

GT, genotype; IFN, interferon; RBV, ribavirin.
The next major protease inhibitor simeprevir was initially approved for combination with peginterferon and ribavirin in 2013 [Janssen Therapeutics, 2013] (Table 1). This regimen offered little beyond boceprevir and telaprevir, and simeprevir’s impact would be as part of an IFN-free regimen, which later received regulatory approval.
Another important breakthrough in the treatment of HCV was sofosbuvir, a pangenotypic nucleotide analog NS5B polymerase inhibitor approved initially in 2013 (Table 1). Sofosbuvir was the first DAA to receive an indication for both HCV and HIV/HCV coinfected patients at the time of initial approval and was also the first approved in an IFN-free regimen. In addition to combination therapy with peginterferon and ribavirin for genotypes 1 and 4, sofosbuvir and ribavirin was approved for genotype 2 and 3, genotype 1 IFN-ineligible patients, and those with HCC awaiting transplant [Gilead Sciences, 2013]. The pangenotypic coverage, high efficacy rates and barrier to resistance, and safety profile made sofosbuvir a leader among the new class of DAAs.
Based upon impressive outcomes using sofosbuvir and simeprevir in the relatively small COSMOS study, there was a paradigm shift towards IFN-free regimens [Lawitz and Gane, 2013]. In 2014, two additional IFN-free regimens arrived for genotype 1. They are the fixed dose combination of sofosbuvir and ledipasvir and the combination of fixed dose ombitasvir, paritaprevir and ritonavir plus dasabuvir. In 2015, data now have emerged with two new DAA regimens, including daclatasvir (pangenotypic NS5A inhibitor) in combination with the fixed dose tablet of asunaprevir (NS3 protease inhibitor), and beclabuvir (nonnucleoside NS5B thumb-1 polymerase inhibitor) and the fixed-dose combination of grazoprevir (NS3/4A protease inhibitor) and elbasvir (NS5A inhibitor) [Lawitz et al. 2015a; Muir et al. 2015; Zeuzem et al. 2015] (Table 1). These regimens show promise for treating patients with compensated cirrhosis and these data will be summarized.
Compensated cirrhosis
Genotype 1
Sofosbuvir plus ledipasvir
The ION clinical trials (Table 2) evaluated the efficacy and safety of the all oral fixed dose combinations of sofosbuvir (nucleotide polymerase inhibitor) and ledipasvir (NS5A inhibitor) for the treatment of HCV. ION-1 evaluated treatment-naive genotype 1 patients with or without ribavirin for 12 or 24 weeks. There were 865 patients randomized with 67% genotype 1a and 16% (138/865) compensated cirrhosis [Afdhal et al. 2014b]. The SVR rates among the different cohorts ranged from 94% to 100% among patients with cirrhosis [Afdhal et al. 2014b]. The most common adverse events were fatigue, headache, insomnia, and nausea, and these were most commonly associated with ribavirin use. Among the treatment-naïve patients with cirrhosis, sofosbuvir–ledipasvir had excellent SVR rates and was very well tolerated. The ION-2 study evaluated this combination among treatment-experienced patients with or without ribavirin for 12 or 24 weeks [Afdhal et al. 2014a]. There were 440 patients, of which 20% (88/440) had cirrhosis and 79% had genotype 1a.The SVR rates with and without cirrhosis were 86% (19/22) and 95% (83/87) respectively in the 12-week cohort, and 100% (22/22) and 99% (86/87) for patients with and without cirrhosis respectively in the 24-week cohort. The regimen was well tolerated with no discontinuations for adverse events. The most common adverse events were fatigue, headache, and nausea. The ION-4 study evaluated sofosbuvir–ledipasvir in patients with HIV coinfection and included 67 (20%) patients with compensated cirrhosis. The overall SVR rate for patients with cirrhosis was 94% (63/67) and there were no significant differences in safety [Naggie et al. 2015].
DAAs and compensated cirrhosis.

DAA, direct acting antiviral; RBV, ribavirin; SVR, sustained virologic response.
Following the ION trials, questions concerning the ideal length of the regimen, the need for the addition of ribavirin, and overall safety in patients with cirrhosis remained. The SIRIUS phase II study (Table 2) evaluated patients with genotype 1 and compensated cirrhosis who had failed to respond to treatment with IFN and a protease inhibitor [Bourliere et al. 2015]. Patients were treated for 12 or 24 weeks with sofosbuvir–ledipasvir with or without ribavirin. The SVR rates after 12 and 24 weeks were similar with an SVR12 of 96% (74/77) for patients in the ledipasvir–sofosbuvir plus ribavirin group and 97% (75/77) in the ledipasvir–sofosbuvir group without ribavirin [Bourliere et al. 2015]. Here, high SVR rates were achieved using this 12-week IFN-free regimen without ribavirin in treatment-experienced patients with compensated cirrhosis. This study design compared ledipasvir–sofosbuvir versus placebo in the first 12 weeks and demonstrated that the regimen was well tolerated with no increased risk of decompensation or serious adverse events.
An integrated safety and efficacy study was performed on this regimen to further define the ideal duration, need for ribavirin, and safety. This study used a post hoc analysis of seven clinical trials on sofosbuvir–ledipasvir [Reddy et al. 2015]. The SVR rates were similar among subjects receiving 12 versus 24 weeks of treatment [95% (305/322) versus 98% (188/191 respectively] and those who did and did not receive ribavirin [97% (254/262) versus 95% (239/251) respectively]. Treatment-experienced patients without ribavirin did have a relatively lower but still impressive SVR of 90% (64/71). The safety analysis raised no major concerns and the most common adverse events were headache, fatigue, and asthenia. Therefore, sofosbuvir–ledipasvir appears effective and safe in treatment-naïve patients with compensated cirrhosis. Taking all these data into account, the AASLD-IDSA HCV Guidance Panel as well as European Association for the Study of the Liver (EASL) recommend that treatment-experienced patients with genotype 1 cirrhosis could be treated with sofosbuvir–ledipasvir without ribavirin for 24 weeks or consider the addition of ribavirin in a 12-week course American Association for the Study of Liver Diseases/ Infectious Diseases Society of America/ International Antiviral Society- USA [AASLD/IDSA/IAS-USA, 2015]. Candidacy for ribavirin should consider baseline hemoglobin, adequate renal function, and cardiopulmonary comorbidities that might not tolerate anemia.
Paritaprevir boosted with ritonavir, ombitasvir, dasabuvir, and ribavirin
The IFN-free regimen of paritaprevir (NS3/4A protease inhibitor) boosted with ritonavir, ombitasvir (NS5A inhibitor), dasabuvir (nonnucleoside polymerase inhibitor), and ribavirin was evaluated in a large development program, and the TURQUOISE-II study specifically evaluated this regimen for treatment-naïve and treatment-experienced patients with compensated cirrhosis and genotype 1 infection (Table 2). In TURQUOISE-II, 380 patients with Child Pugh A cirrhosis were given this regimen for 12 or 24 weeks [Poordad et al. 2014]. All patients, including genotype 1b patients, received ribavirin. Among these patients with compensated cirrhosis the SVR rate after 12 weeks of treatment was 92% (191/208), and the SVR after 24 weeks of treatment was 96% (165/172). Subgroup analyses suggested lower SVR rates for genotype 1a patients treated with 12 weeks (88.6%, 124/140) compared with 24 weeks (94.2%, 114/121). Genotype 1b patients did very well with both 12 weeks (99%, 67/68) and 24 weeks (100%, 51/51) [Poordad et al. 2014]. Differences in SVR were also noted for the treatment-experienced patients with SVR rates of 90.2% (110/122) at 12 weeks and 96.6% (95/98) at 24 weeks. This difference was driven by the prior null responders with 86.7% (65/75) and 95.2% (59/62) in the 12- and 24-week regimens respectively. Upon further examination, the genotype 1a prior null responders did not do as well with SVR rates of 80% (40/50) for 12 weeks and 92.9% (39/42) for 24 weeks [Poordad et al. 2014]. These findings in the genotype 1a patients with cirrhosis have led the AASLD-IDSA and EASL to recommend 24 weeks of treatment with ribavirin for this regimen with genotype 1a with cirrhosis. Genotype 1b patients are recommended to receive 12 weeks with ribavirin, and studies are ongoing to see if ribavirin is really necessary for this group. The regimen was well tolerated in these patients with cirrhosis and the most common adverse events were fatigue, headache, and nausea. Ribavirin was associated with a hemoglobin level less than 10 g per deciliter in 7.2% (12 week) and 11.0% (24 week) of patients [Poordad et al. 2014]. This large study with its focus on patients with compensated cirrhosis provided important safety and efficacy data.
Simeprevir and sofosbuvir
The regimen of simeprevir plus sofosbuvir has been evaluated in treatment-experienced and treatment-naïve patients with compensated cirrhosis. Cohort-2 within the COSMOS study included 87 subjects with cirrhosis randomized to simeprevir–sofosbuvir for 12 or 24 weeks with or without ribavirin. The SVR rates for genotype 1a (94%) and 1b (95%) among patients with cirrhosis were similar [Lawitz et al. 2014]. The SVR rate was similar at 12 weeks whether ribavirin was (93%) or was not included (93%). There was a slight improvement in SVR at 24 weeks without ribavirin (100%) over with ribavirin (93%). The regimen was safe and well tolerated with few serious adverse events [Lawitz et al. 2014].
OPTIMIST-2 was a larger study that evaluated the safety and efficacy of 12 weeks of simeprevir–sofosbuvir in patients with genotype 1 compensated cirrhosis, and this study design did not include ribavirin [Lawitz B et al. 2015]. There were 103 patients with 70% genotype 1a and 51% treatment experienced. The overall SVR rate was lower than previous studies at 83% (86/103). The SVR rate appeared to be impacted by the presence of the Q80K mutation, a resistance-associated amino acid substitution conferring simeprevir resistance in genotype 1a. This variant was initially reported in the simeprevir studies in combination with peginterferon–ribavirin but was not clearly identified as a concern in the COSMOS studies [Lawitz et al. 2014; Zeuzem A et al. 2014]. In OPTIMIST-2, genotype 1a SVR rates without the Q80K were 92% (35/38), while this dropped to 74% (25/34) in the presence of the Q80K mutation. The overall SVR rate for genotype 1b was 84% (26/31), a finding lower than typical of genotype 1b in other analyses. Treatment-naïve patients (SVR rate 88%, 44/50) did better than treatment-experienced patients (SVR rate 79%, 44/53) [Lawitz B et al. 2015]. These findings suggest that Q80K testing should be considered prior to treatment of genotype 1a patients with this regimen and raises the question of the need for ribavirin with this regimen.
Grazoprevir and elbasvir
The once-daily regimen of grazoprevir (NS3/4A protease inhibitor) and elbasvir (NS5A inhibitor) has completed phase III clinical trials and is expected to undergo regulatory review in the United States and Europe in 2015. The C-WORTHY trial evaluated this regimen with or without ribavirin for 12 or 18 weeks in treatment-naïve patients with cirrhosis and previous null responders with or without cirrhosis [Lawitz et al. 2015]. Of the prior null responders, 67 (52%) were F3/F4. The presence of ribavirin and prolonged duration of treatment did not clearly impact the SVR for treatment-naive patients with cirrhosis. The SVR at 12 weeks with and without ribavirin was 90% (28/31) and 97% (28/29) respectively. Similarly, the SVR rates at 18 weeks with and without ribavirin were 97% (31/32) and 94% (29/31) respectively. Among those who were null responders and had cirrhosis, the overall SVR for 12 and 18 weeks was 92% (23/25) and 100% (23/23) respectively. SVR rates were not impacted by the presence of ribavirin [Lawitz et al. 2015]. The overall SVR rate for genotype 1a was 94% (31/33) versus 100% (15/15) for genotype 1b [Lawitz et al. 2015]. The regimen of grazoprevir–elbasvir was also evaluated in a randomized placebo controlled trial with 421 treatment-naïve patients, 92 (22%) of whom had cirrhosis [Zeuzem et al. 2015]. Among those with genotype 1a, the SVR rate was 92% (144/157) after 12 weeks of this regimen. Those with cirrhosis achieved an SVR of 97% (68/70). The most common adverse events were headache, fatigue, and nausea [Zeuzem et al. 2015]. This regimen has also shown promise for treating patients with chronic kidney disease. The C-Surfer study evaluated this regimen for 12 weeks in treatment-naïve and treatment-experienced subjects with genotype 1 infection. The small population included 6% (7) of subjects with compensated cirrhosis and 75% (92) were on hemodialysis [Roth et al. 2015]. Among those with cirrhosis and chronic kidney disease, 100% achieved SVR. The SVR rates for those with chronic kidney disease stage 4 and 5 were 100% (22/22) and 99% (93/94) respectively [Roth et al. 2015]. This regimen awaits regulatory review but appears to be another effective option for patients with compensated cirrhosis, even those with chronic kidney disease.
Daclatasvir, asunaprevir, and beclabuvir
The all oral IFN-free regimen of daclatasvir (NS5A inhibitor), asunaprevir (NS3 protease inhibitor), and beclabuvir (nonnucleoside NS5B inhibitor) was evaluated in the ribavirin-free UNITY-1 study in patients with genotype 1 HCV without cirrhosis. The UNITY-2 study evaluated this regimen in treatment-naïve and treatment-experienced patients with compensated cirrhosis and also evaluated the role of ribavirin in a randomized design [Muir et al. 2015] (Table 2). Approximately 74% of patients in UNITY-2 had genotype 1a. The SVR after 12 weeks among the 112 treatment-naïve patients with or without ribavirin was 98% (54/55) and 93% (53/57) respectively. The 90 patients in the treatment-experienced group achieved SVR rates at 12 weeks with or without ribavirin of 93% (42/45) and 87% (39/45) respectively. Among those with genotype 1a, the SVR was 95% (70/74) and 88% (66/75) respectively, with and without ribavirin. These findings suggests a role for ribavirin in this regimen, but the study size was too small for definitive conclusions. There were three serious adverse events considered treatment related and four adverse event related discontinuations. Four patients exhibited treatment-emergent grade 3 or 4 alanine aminotransferase elevations and one had concomitant bilirubin elevation [Muir et al. 2015]. The liver enzyme elevations are under investigation and highlight the risks and challenges of developing efficacious and safe regimens for patients with cirrhosis.
Sofosbuvir and GS-5816
Currently, phase III studies are evaluating GS-5816, a new NS5A inhibitor as a fixed dose combination with sofosbuvir. This regimen has been studied as a 12-week course with or without ribavirin in genotype 1 patients with cirrhosis. Among the treatment-experienced patients, 41% had cirrhosis, and the overall SVR for the fixed-dose combination of sofosbuvir and GS-5816 was 100% (27/27) without the addition of ribavirin. In combination with ribavirin, the SVR for this regimen was 96% (27/28) [Pianko et al. 2014]. The regimen was well tolerated with increased lab abnormalities and adverse events associated with the inclusion of ribavirin in the regimen [Pianko et al. 2014].
Sofosbuvir and GS-5816 and GS-9857
The fixed dose combination of sofosbuvir plus GS-5816 has been combined with a new NS3/4A protease inhibitor (GS-9857) to evaluate this regimens efficacy as a shorter duration regimen (6 weeks) as well as an option for prior DAA failures. Among treatment-naïve genotype 1 patients with cirrhosis, this regimen achieved an SVR of 87% (13/15) after only 6 weeks of treatment [Gane et al. 2015]. Among those who were treatment experienced (including DAA failures), the SVR rates were lower with an SVR of 68% (17/25) among treatment-experienced patients without cirrhosis and 60% (3/5) for treatment-experienced patients with cirrhosis [Gane et al. 2015].
Collectively, these studies have demonstrated that patients with genotype 1 and compensated cirrhosis have multiple effective options (Figure 1). Regimens as short as 12 weeks among treatment-naïve patients with cirrhosis have excellent SVR rates. These regimens have been proven to be successful in the sickest subjects, including those with renal impairment. There are data to support the use of sofosbuvir plus simeprevir, sofosbuvir plus ledipasvir, grazopevir plus elbasvir, and the fixed dose combination of paritaprevir/ritonavir/ombitasvir plus dasabuvir for individuals with mild to moderate renal impairment [AASLD/IDSA/IAS-USA, 2015; Roth et al. 2015]. There are limited data on the use of paritaprevir/ritonavir/ombitasvir plus dasabuvir and grazoprevir plus elbasvir for individuals with severe renal dysfunction (creatinine clearance less than 30ml/min) [Wilder and Patel, 2014; AASLD/IDSA/IAS-USA, 2015; Pockros et al. 2015; Roth et al. 2015]. In the setting of treatment-experienced patients with cirrhosis, the SVR may benefit from the addition of ribavirin or a prolonged duration of treatment.
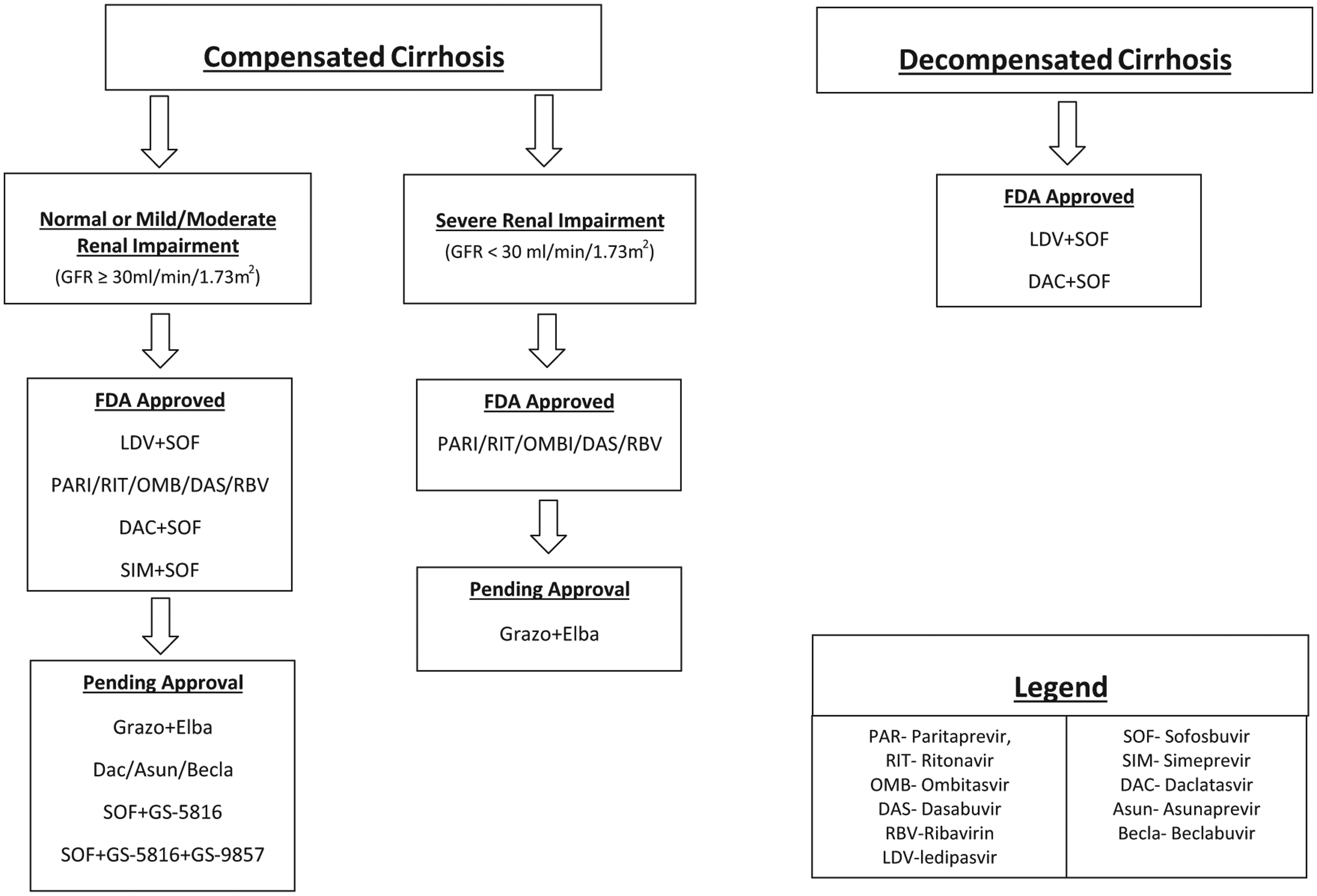
Treating genotype 1 hepatitis C virus infection with cirrhosis. FDA, US Food and Drug Administration; GFR, glomerular filtration rate.
Genotype 2
The introduction of sofosbuvir and ribavirin brought impressive outcomes for genotype 2 patients with an IFN-free regimen. As the experience with the regimen has matured, questions have emerged in genotype 2 patients with cirrhosis concerning the duration of treatment. Efforts have also focused on development of ribavirin-free regimens.
Sofosbuvir combined with ribavirin to treat genotype 2 was evaluated in three clinical trials (FISSION, POSITRON, and VALENCE) and across all three trials 201/214 (94%) patients achieved SVR with sofosbuvir–ribavirin. Patients received daily peginterferon and ribavirin for 24 weeks or sofosbuvir–ribavirin for 12 weeks in FISSION [Jacobson et al. 2013; Lawitz and Gane, 2013; Zeuzem B et al. 2014]. Approximately 20% of patients had cirrhosis, and 83% (10/12) achieved SVR with sofosbuvir–ribavirin, much better than the 62% (8/13) for the IFN–ribavirin group [Gilead Sciences, 2013; Lawitz and Gane, 2013]. POSITRON randomized genotype 2 patients to receive sofosbuvir–ribavirin versus placebo for 12 weeks. The overall SVR was 94% (16/17) among those who had cirrhosis [Gilead Sciences, 2013; Jacobson et al. 2013]. The VALENCE (419 patients, 21% with cirrhosis) evaluated the combination of sofosbuvir–ribavirin in patients with genotype 2 and 3 in treatment-naive and treatment-experienced patients [Gilead Sciences, 2013; Zeuzem B et al. 2014]. The study included only 11 genotype 2 patients with cirrhosis. Both treatment-naïve and seven of nine (78%) of the treatment-experienced patients with cirrhosis achieved SVR. The FUSION study evaluated the impact of a longer treatment duration on SVR in treatment-experienced genotype 2 patients with cirrhosis (34%). Patients received 12 versus 16 weeks of sofosbuvir–ribavirin [Gilead Sciences, 2013; Jacobson et al. 2013]. The SVR improved with longer durations of therapy, with a SVR rate of 82% (32/39) for 12 weeks and 89% (31/35) for 16 weeks. Among patients with cirrhosis, the SVR improved from 60% (6/10) at 12 weeks to 78% (7/9) after 16 weeks [Gilead Sciences, 2013; Jacobson et al. 2013].
Although the emphasis has been on IFN-free regimens, the role of peginterferon was explored in treatment-experienced patients in the LONESTAR-2 and BOSON studies [Foster et al. 2015; Lawitz C et al. 2015]. In LONESTAR-2, patients received sofosbuvir, peginterferon, and ribavirin for 12 weeks. This small study included 23 subjects, 61% of whom had compensated cirrhosis. The overall SVR was 96% (22/23), while the SVR for subjects with cirrhosis was 93% (13/14) [Lawitz C et al. 2015]. While promising, the overall number of subjects in this study was small. The BOSON study further evaluated peginterferon in combination with sofosbuvir and ribavirin for 12 weeks compared with a 16- or 24-week sofosbuvir–ribavirin regimen. The study included genotype 2 and 3 patients, and all the genotype 2 patients were treatment experienced and had cirrhosis. All genotype 2 patients did well in all cohorts. The SVR rate following 12 weeks of triple therapy was 94% (15/16). The SVR rates following 16 and 24 weeks of sofosbuvir–ribavirin were 87% (13/15) and 100% (17/17) respectively [Foster et al. 2015].
The optimal duration and approach remains uncertain for treatment of genotype 2 patients with cirrhosis. Studies to date have included small numbers of patients with cirrhosis. The FUSION study provides the support for the current guideline from the AASLD-IDSA HCV Guidance Panel to recommend 16 weeks of treatment with sofosbuvir and ribavirin in patients with cirrhosis. The addition of peginterferon is reasonable to consider for treatment-experienced patients with cirrhosis. EASL guidelines support this regimen for 16–20 weeks in the setting of compensated cirrhosis [EASL, 2015]. Ongoing studies are evaluating these questions and should offer more guidance in the future.
Genotype 3
In the IFN-free era, genotype 3 has emerged as one of the most difficult populations to treat. Multiple studies with a variety of regimens have identified lower SVR rates with cirrhosis and particularly treatment-experienced patients.
The initial studies of sofosbuvir and ribavirin reported low SVR rates with 12 and 16 weeks of treatment, particularly among patients with cirrhosis [Jacobson et al. 2013; Lawitz and Gane, 2013]. The FUSION study (Table 3) evaluated 12 versus 16 weeks of sofosbuvir–ribavirin in treatment-experienced patients and supported the role for longer treatment. The overall genotype 3 SVR rates were 30% and 62% for 12 and 16 weeks of treatment, respectively. Among those patients with cirrhosis with genotype 3, the SVR rate was 19% (5/26) at 12 weeks and 61% (14/23) at 16 weeks [Jacobson et al. 2013]. As a result of this finding, the VALENCE study (Table 3) was amended to extend treatment with sofosbuvir and ribavirin for genotype 3 patients to 24 weeks [Zeuzem B et al. 2014]. Genotype 3 treatment-naïve patients with compensated cirrhosis did well with SVR rates of 92% (12/13) [Gilead Sciences, 2013; Zeuzem B et al. 2014]. Among treatment-experienced patients, the SVR rate dropped to 60% (27/45) for those with compensated cirrhosis [Zeuzem B et al. 2014].
Direct-acting antivirals, genotype 3 infection, and cirrhosis.

DAA, direct-acting antiviral; RBV, ribavirin; SVR, sustained virologic response.
Given low SVR rates observed with sofosbuvir–ribavirin, the efficacy of sofosbuvir–ribavirin combined with peginterferon for 12 weeks was evaluated in the LONESTAR-2 study [Lawitz et al. 2015]. The total population for this study was relatively small, but the SVR rate (83%) was the same among those with (12 subjects) and without (12 subjects) cirrhosis [Lawitz et al. 2015]. There were no significant safety concerns identified in this study. The BOSON study built upon this literature to study IFN-containing and IFN-free regimens for genotype 3 head to head. Patients were randomized to sofosbuvir–ribavirin for 16 or 24 weeks versus sofosbuvir–ribavirin plus peginterferon for 12 weeks [Foster et al. 2015]. There were 219 (37%) patients with cirrhosis in this analysis. For genotype 3, the overall SVR for sofosbuvir–ribavirin for 16 weeks was 72% (128/181), sofosbuvir–ribavirin for 24 weeks was 85% (153/182), and sofosbuvir–ribavirin plus peginterferon for 12 weeks was 93% (168/181). Among genotype 3 patients with cirrhosis, the SVR for sofosbuvir–ribavirin for 12 weeks was 51% (29/57), sofosbuvir–ribavirin for 24 weeks was 79% (44/56), and the combination of sofosbuvir–ribavirin plus peginterferon for 12 weeks achieved an SVR of 88% (51/58) [Foster et al. 2015]. Among treatment-experienced genotype 3 subjects with cirrhosis, the combination of sofosbuvir–ribavirin and peginterferon for 12 weeks achieved an SVR of 86% (30/35). This study supports either the 24-week regimen of sofosbuvir–ribavirin or 12-week triple therapy with sofosbuvir–ribavirin and peginterferon for genotype 3 patients with cirrhosis. While the best outcomes may come with the addition of peginterferon, the risks and impact on quality of life must be considered.
The regimen of sofosbuvir–ledipasvir is currently available and recommended for genotype 1 and 4 patients, and this regimen has also been studied in genotype 3. The early work suggested that ledipasvir was not pangenotypic and particularly less effective for genotype 3 [Gilead Sciences, 2015]. The ELECTRON-2 study (Table 3) evaluated treatment-experienced genotype 3 patients using the fixed dose regimen of sofosbuvir–ledipasvir plus ribavirin. The cohort of 50 patients included 40% with cirrhosis, and they achieved SVR in 89% (25/28) and 73% (16/22) of patients with and without cirrhosis, respectively [Gane et al. 2014]. These outcomes are similar to those observed with other genotype 3 regimens, but the small sample size warrants caution in drawing conclusions. With large studies lacking, there will not be a regulatory indication granted or endorsement in society guidelines for this regimen. As a result, access to this regimen would be limited for genotype 3 patients, and there are other reasonable options to consider.
The ALLY-3 clinical trial (Table 3) evaluated sofosbuvir and the NS5A inhibitor daclatasvir for 12 weeks in genotype 3 patients who were treatment naïve and treatment experienced [Nelson et al. 2014]. There were 152 subjects, and 21% had cirrhosis [Nelson et al. 2014]. Overall 90% (91/101) and 86% (44/51) of treatment-naive and treatment-experienced patients respectively achieved SVR12 without IFN or ribavirin [Nelson et al. 2014]. Among those with cirrhosis, the overall SVR rates decreased to 58% (11/19) for treatment-naïve and 69% (9/13) for treatment-experienced patients. Although small subsets, these findings raise concern yet again for a genotype 3 regimen with cirrhosis. Current guidelines from EASL recommend treating genotype 3 patients with cirrhosis with sofosbuvir–daclatasvir plus weight-based ribavirin for 24 weeks [EASL, 2015]. Ongoing studies should provide more data to understand the best approach with this regimen.
A new treatment option under study for treatment of genotype 3 is the fixed dose combination of sofosbuvir and GS-5816 (a new NS5A inhibitor). This regimen has been studied as a 12-week regimen in genotype 3 patients with or without ribavirin. In the analyses treatment-experienced patients with cirrhosis were included. Among treatment-experienced genotype 3 patients without cirrhosis, this regimen achieved an SVR of 100% (27/27) without ribavirin [Pianko et al. 2014]. Among those who were treatment experienced with cirrhosis, this regimen achieved a SVR of 88% (23/26) without ribavirin, and 96% (25/26) with ribavirin [Pianko et al. 2014]. The regimen was well tolerated with increased adverse events and lab abnormalities associated with use of ribavirin.
Decompensated cirrhosis
The development programs of new regimens in HCV begin with patients without cirrhosis and then demonstrate safety in patients with compensated cirrhosis before moving to decompensated cirrhosis. Few regimens have made it to this point. The hope and main question for treatment of decompensated cirrhosis is if the course of liver disease can be impacted by eradication of HCV. Treatment of decompensated cirrhosis from hepatitis B infection does improve clinical outcomes, but it is unclear if HCV treatment will have the same effect [Liaw et al. 2011]. When treating patients with decompensated cirrhosis, a major consideration is the exposure and safety of the medication in Child Pugh B and C. Preclinical studies have identified exposure and safety concerns with hepatic impairment with some agents. Simeprevir is metabolized by the liver and the mean steady state area under the curve (AUC) of simeprevir is 2.4-fold higher with moderate hepatic impairment (Child Pugh Class B) and 5.2-fold higher with severe hepatic impairment (Child Pugh Class C) [Janssen Therapeutics, 2013]. The AUC values of ombitasvir, ritonavir, and dasabuvir decrease by 30%, 30%, and 16% respectively and paritaprevir AUC values increase by 62% in subjects with moderate hepatic impairment [AbbVie, 2014]. Paritaprevir, ritonavir, and dasabuvir AUC values increase by 945%, 13%, and 325% respectively and ombitasvir AUC values decrease by 54% in subjects with severe hepatic impairment [AbbVie, 2014]. Simeprevir and the regimen of ombitasvir, paritaprevir/ritonavir and dasabuvir are therefore not recommended in patients with decompensated cirrhosis.
Although we have limited data on treatment for decompensated HCV cirrhosis, recent studies have reported promising regimens for treating HCV in patients with decompensated cirrhosis. A multicenter study by Charlton and colleagues (Table 4) evaluated ledipasvir–sofosbuvir plus ribavirin for 12 and 24 weeks to treat genotype 1 and 4 treatment-naïve and treatment-experienced patients with Child Pugh B or Child Pugh C decompensated cirrhosis. For Child Pugh B, SVR rates for 12 and 24 weeks were 87% (26/30) and 89% (24/27) respectively. The SVR for Child Pugh C was 86% (19/22) for 12 weeks and 90% (18/20) for 24 weeks of treatment [Charlton et al. 2015]. Changes in bilirubin, albumin, and Child Pugh score from baseline were evaluated. There were statistically significant drops in bilirubin and rises in albumin for both Child Pugh B and C patients. A majority of patients had a drop in their Child Pugh and Model for End-Stage Liver Disease (MELD) scores [Charlton et al. 2015]. There was one discontinuation in a Child Pugh B patient treated for 24 weeks and two discontinuations among Child Pugh C patients treated for 24 weeks. Early discontinuations occurred due to septic shock, hepatic encephalopathy, and peritoneal hemorrhage. There were six deaths in total: septic shock (two), septic shock with multiorgan failure (two), oliguric renal failure, and cardiac arrest [Charlton et al. 2015].
Direct-acting antivirals and decompensated cirrhosis.

DAA, direct acting antiviral; RBV, ribavirin; SVR, sustained virologic response; CTP,Child-Turcotte-Pugh Class.
The ALLY-1 phase III clinical trial (Table 4) evaluated 12 weeks of sofosbuvir–daclatasvir plus ribavirin in treatment-naïve and treatment-experienced patients with advanced cirrhosis and DAA failures were allowed with the exception of NS5A [Poordad et al. 2015]. The majority of patients were genotype 1 and 80% were considered Child Pugh B or C. The overall SVR rate for genotype 1 was 82% (37/45). The Child Pugh class did impact SVR rates, with Child Pugh A, B, and C having overall SVR rates of 92% (11/12), 94% (30/32), and 56% (9/16) respectively. Overall the regimen was safe and well tolerated [Poordad et al. 2015].
The use of sofosbuvir in combination with both ledipasvir and daclatasvir for the treatment of genotype 1 and 3 in patients with decompensated cirrhosis was evaluated by the HCV Research UK-EAP Group (Table 4). Medications were provided for free and clinicians were free to select a regimen of their choice in this analysis. The inclusion criteria were quite broad, including treatment-experienced patients (47.1%), patients with HIV coinfection (5.7%), and 66% Child Pugh B [Foster et al. 2015]. Among those with genotype 1 and decompensated cirrhosis, the SVR rate was 86% (141/164) for sofosbuvir–ledipasvir plus ribavirin, 81% (17/21) for sofosbuvir–ledipasvir, 82% (37/45) for sofosbuvir–daclatasvir plus ribavirin, and 60% (3/5) for sofosbuvir–daclatasvir. The SVR for genotype 3 was significantly lower with a SVR rate for sofosbuvir–ledipasvir plus ribavirin of 59% (36/61), sofosbuvir–ledipasvir 43% (3/7), sofosbuvir–daclatasvir plus ribavirin 70% (80/114), and sofosbuvir–daclatasvir 71% (5/7). Overall 40% of patients showed improvement in liver function as measured by MELD score [Foster et al. 2015].
These studies demonstrated that virologic cure of HCV can be achieved even with decompensated cirrhosis. Although they have demonstrated that some markers of liver function do improve, such as bilirubin and albumin, the degree of recovery with decompensated cirrhosis remains unclear. There are likely some patients with cirrhosis who can improve their liver function and acquire a reasonable quality of life. Other patients may be better served by proceeding to liver transplantation and then receiving antiviral treatment after transplant. Long-term studies are underway to address these questions. In the meantime, clinicians and patients will need to consider the role of treatment of patients with decompensated cirrhosis using their best judgment, and the opportunity for transplantation will likely be a major consideration.
Conclusion
The new regimens for HCV demonstrate impressive responses in patients with compensated and decompensated cirrhosis, but gaps in knowledge persist. A wealth of data exists for genotype 1 infection, but robust studies with genotype diversity are needed. The duration of treatment remains a question for genotype 2 patients with cirrhosis, and genotype 3 patients with cirrhosis stand out with their lower response rates. The approach to treatment of decompensated cirrhosis remains an area of great uncertainty. When patients are not transplant candidates, eradication of their HCV infection is the best available option. The transplant-eligible patient will need to consider the role of treatment before or after transplantation. Identifying regimens for decompensated cirrhosis which are IFN free and preferably ribavirin free is a priority for future regimens in HCV.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Andrew Muir has received research grants or is on advisory boards for AbbVie, BMS, Gilead, and Merck. Julius Wilder has no conflicts of interest to report.
