Abstract
Objectives
To investigate the clinical and serological features of autoimmunity with chronic hepatitis C virus infection before and after direct-acting antiviral therapy and assess their relation to treatment response.
Methods
A prospective cohort study was performed in adult patients aged ≥18 years who had chronic hepatitis C virus infection, as confirmed by polymerase chain reaction, and were eligible for direct-acting antiviral therapy. Patients with rheumatological disease prior to the onset of hepatitis C virus infection, decompensated cirrhosis, or hepatocellular carcinoma were excluded. All patients were treated with sofosbuvir (400 mg once daily) plus daclatasvir (60 mg once daily) for 3 months. Patients were assessed before and 12 weeks after treatment.
Results
Ninety patients completed the follow-up (66.7% females, 33.3% males, mean age: 49.2 ± 12.3 years); 85.55% of them had immune-mediated manifestations prior to direct-acting antiviral therapy. In patients with sustained virologic response, autoimmune manifestations persisted in 66 of the 71 (92.9%) patients with an observable rise in posttreatment erythrocyte sedimentation rate and C-reactive protein level (p > 0.01). The predictors of persistence of autoimmune manifestation were age ≥49 years (p = 0.009), female sex (p = 0.026), and tobacco use (p = 0.043).
Conclusion
Direct-acting antiviral drugs were not associated with a significant short-term change in the prevalence of autoimmune manifestations in patients who had hepatitis C virus infection with sustained virologic response.
Keywords
Introduction
Chronic hepatitis C virus (HCV) infection is a multifaceted disease that has been associated with a wide spectrum of systemic autoimmune manifestations. Based on the World Health Organization data in 2022, approximately 58 million patients were affected by chronic HCV infection worldwide, with approximately 0.3 million deaths attributable to HCV-related complications, as reported in 2019.1,2 In Egypt, before the launch of the presidential HCV eradication campaign, HCV infection has been one of the most endemic viral infections, with approximately 6.3% of individuals aged 1–59 years displaying HCV seropositivity and 4.4% having active infection, which is among the highest prevalence rates worldwide. The diagnosis of HCV infection has been linked to a myriad of extrahepatic immune-mediated manifestations simulating those observed in patients with primary autoimmune disorders, 3 with evidence reporting a prevalence rate of up to 74%. Among these extrahepatic immune-mediated associations, nondeforming arthralgia and myalgia as well as Sicca syndrome are considered the most common, with reported incidences of 40%–80% and 20%–30% in patients with chronic HCV infection, respectively. 4 In Egypt, the prevalence of HCV-related extrahepatic manifestations (EHMs) reportedly reached 16.39%, with chronic fatigue syndrome in 9.5%, Sicca symptoms in 8.8%, arthralgia in 6.5%, fibromyalgia in 1.9%, myalgia in 1.3%, arthritis in 0.7%, cryoglobulinemic vasculitis in 0.7%, autoimmune hemolytic anemia in 0.7%, and thrombocytopenia in 0.7% of all patients with HCV. 5 The presence of chronic HCV viremia has been associated with seropositivity to several autoantibodies, including cryoglobulins, rheumatoid factor (RF), anti-nuclear antibody (ANA), anti-cyclic citrullinated peptide antibody (anti-CCP), anti-cardiolipin antibodies (ACL), anti-neutrophil cytoplasmic antibody (ANCA), anti-double stranded deoxyribonucleic acid antibodies (anti-dsDNA), anti-thyroid and anti-liver autoantibodies.5,6 Direct-acting antivirals (DAAs) are currently being considered a cornerstone in the HCV treatment armamentarium toward global eradication of HCV by 2030.7–10 The present study aimed to investigate the spectrum of clinical and serological features of autoimmunity in a cohort of patients with HCV before and after DAA therapy and assess their relation to the achievement of sustained virologic response (SVR) to treatment.
Patients and methods
Methods
This prospective cohort study was conducted among adult patients with chronic HCV infection who fulfilled the following eligibility criteria: (a) adult patients (>18 years old) of both sexes with detectable HCV RNA via polymerase chain reaction (PCR) and (b) who consented for participation and were eligible for antiviral treatment. The exclusion criteria were as follows: (a) patients with decompensated cirrhosis (Child-Pugh score B and C); (b) those with a platelet count <50,000/mL; (c) those with hepatocellular carcinoma or extrahepatic malignancy; and (d) those with other contraindications related to study drug administration, such as pregnancy, lactation, and inability to use contraception. All patients were recruited from a single specialized viral hepatitis center affiliated to the National Committee for the Control of Viral Hepatitis (NCCVH)/Ministry of Health and Population (MOHP) from October 2019 to June 2020. A total of 130 patients were recruited, and 90 patients completed the study. All patients were treated with sofosbuvir (400 mg once daily) plus daclatasvir (60 mg once daily) for 3 months.
For all patients, complete clinical history and thorough clinical examination were performed. Baseline laboratory evaluation was performed, including liver profile serum alanine transaminase (ALT), aspartate transaminase (AST), serum alpha fetoprotein, serum bilirubin, serum albumin, prothrombin time and international normalized ratio, complete blood count, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), random blood sugar, glycosylated hemoglobin level for patients with diabetes mellitus (DM), serum creatinine, hepatitis B surface antigen and quantitative HCV RNA PCR, ANA (titer of ≥1/80) via indirect immunofluorescence (IIF), RF via latex agglutination, ACL IgG via enzyme-linked immunosorbent assay, ANCA via IIF and anti-ds DNA via IIF in patients with positive ANA. Patients were evaluated at baseline before starting treatment and reevaluated for target laboratory and serological variables 12 weeks after the end of treatment.
Ethical statement
All procedures involving human participants were conducted according to the ethical standards of the institutional and/or national research committee as well as the Helsinki declaration and its later amendments. The study was approved by the Research Ethics Committee for human subject research at the Faculty of Medicine, Cairo University (number MD-162-2019) on 28 September 2019. All patients consented to the inclusion of patient data in this study and were ensured regarding anonymization of patient data. We confirm that we have fully anonymized the data. The reporting of this study conforms to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines. 8
Statistical methods
Sample size was calculated statistically by comparing two proportions of paired samples using the McNemar test, α-error of 0.05, and a power of 80%. Sample size calculation was performed using PS Power and Sample Size Calculations software (version 3.0.11) for MS Windows (William D Dupont and Walton D, Vanderbilt University, Nashville, TN, USA). 11 The collected data were coded, tabulated, and statistically analyzed using IBM SPSS statistics (Statistical Package for Social Sciences) software version 22.0, IBM Corp., Chicago, USA, 2013. Qualitative data were presented as numbers and percentages. Quantitative data were described as means, standard deviation (mean SD), and ranges after testing for normality using the Shapiro–Wilk test. The comparison between two independent groups was performed using the independent samples t-test, whereas paired comparisons were performed using the paired samples t-test. Qualitative data were compared using the Fisher’s Exact test for independent variables with small, expected numbers, while McNemar test was used for comparing paired data. The confidence interval was set to 95% and the acceptable margin of error was set to 5%. A p-value of <0.05 was considered to indicate statistical significance.
Results
Of the 130 recruited patients, 90 completed the study follow-up and were included in the analysis; 66.7% of them were females. The mean patient age was 49.2 ± 12.3 years, while the mean baseline HCV viral load was 1398.8 ± 3870.1 × 103/mL. Moreover, SVR was achieved in 81/90 patients in the study cohort. The detailed baseline characteristics of all patients are shown in Table 1.
Baseline characteristics of the study population.

BMI: body mass index.
Factors such as age >49 years, female sex, presence of DM, direct bilirubin level of >0.12 mg/dL, hemoglobin A1c level (HbA1C%) of >5.5%, and hemoglobin level ≤14.1 g/dL increased the probability of the existence of pre-treatment autoimmune manifestations, whereas tobacco consumption and drug abuse decreased this probability (Table 2).
Predictors of autoimmunity before direct-acting antiviral therapy.
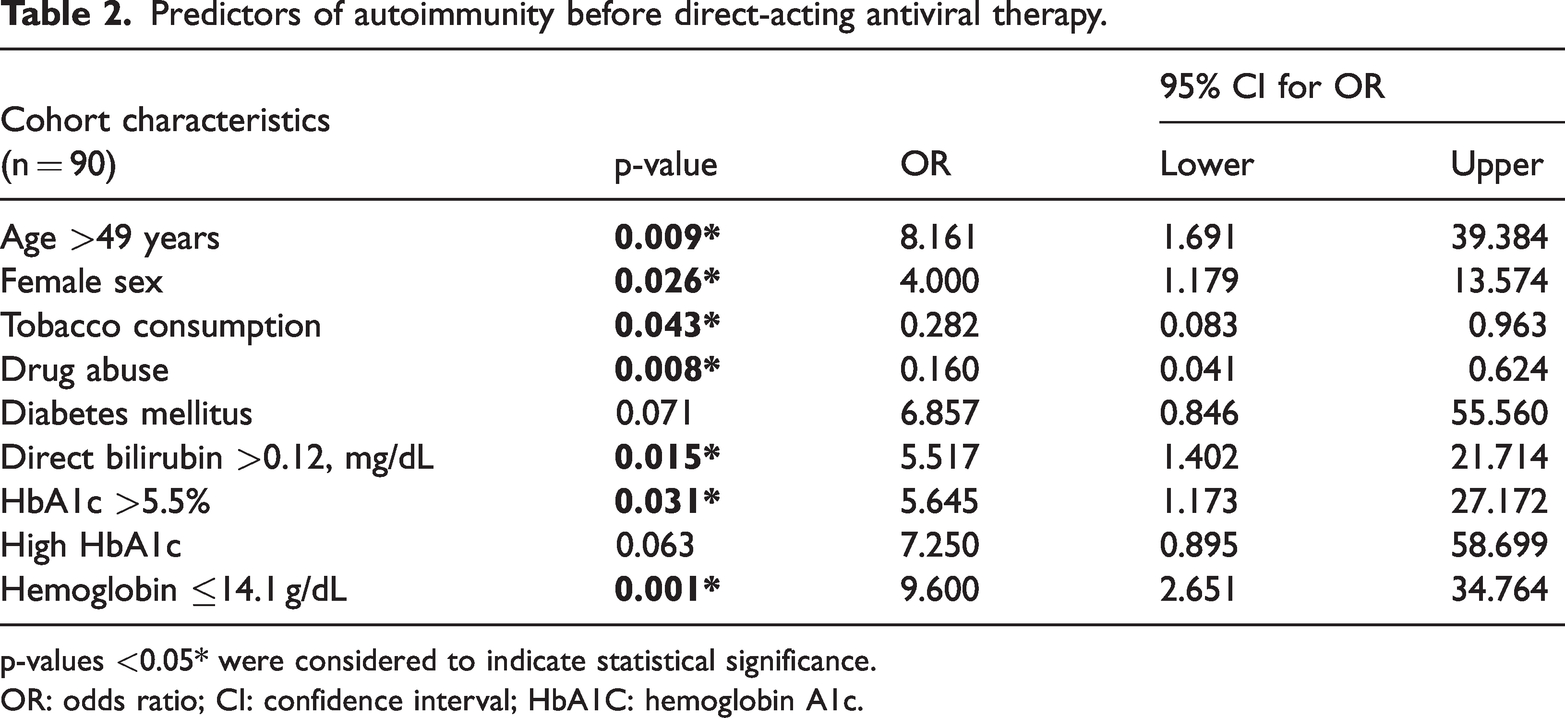
p-values <0.05* were considered to indicate statistical significance.
OR: odds ratio; CI: confidence interval; HbA1C: hemoglobin A1c.
No significant difference was observed in the clinical manifestations suggestive of autoimmunity among patients with SVR at baseline versus 12 weeks after the end of treatment. The most commonly observed manifestations at baseline versus 12 weeks after the end of treatment were musculoskeletal manifestations in the form of arthralgia (58% vs. 48.1%), fatigue (50.6% vs. 46.9%), myalgia (43.2% vs. 40.7%), and arthritis (8.6% vs. 2.5%), followed by ocular (23.5% vs. 18.5%) and neurological (23.5% vs. 29.6%) manifestations (Figures 1 to 3; Table 3).

Monoarthritis of the second proximal interphalangeal joint of the left index in a patient with chronic hepatitis C virus infection developing 1 month after receiving direct-acting antiviral therapy.

Erythema nodosum in a patient with chronic hepatitis C virus infection before (a) and (b) after direct-acting antiviral therapy.

A patient with diabetes mellitus and chronic hepatitis C infection and palpable purpura before direct-acting antiviral therapy (a) and (b) improvement of the lesions following treatment.
Comparison of the suggestive clinical manifestations of autoimmunity before and after direct-acting antiviral therapy among SVR patient subgroup.
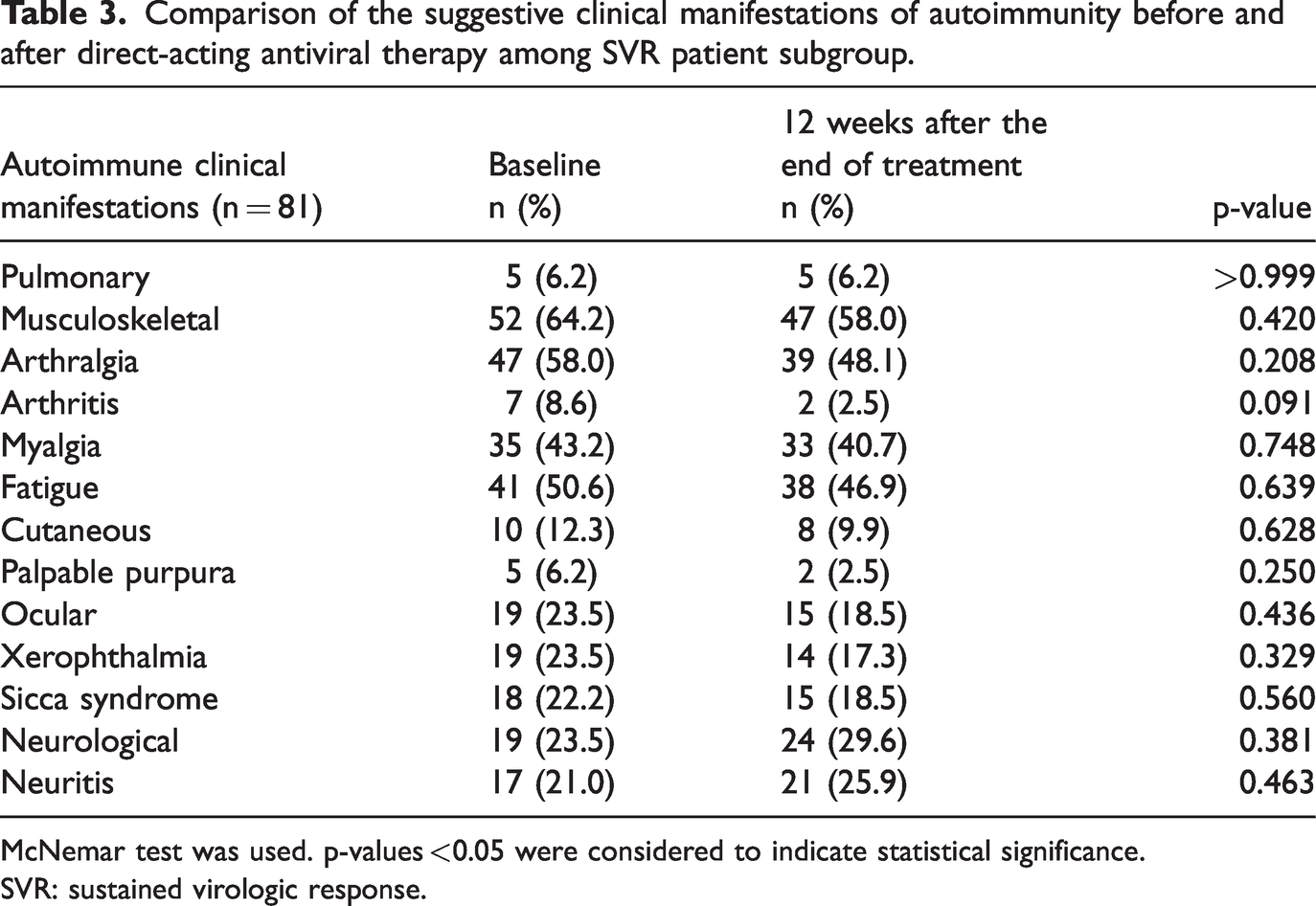
McNemar test was used. p-values <0.05 were considered to indicate statistical significance.
SVR: sustained virologic response.
The ESR and CRP values were significantly higher at the end of treatment than at baseline (p < 0.001). Similarly, the posttreatment RF and ACL IgG levels were significantly higher than those at baseline (p = 0.008 and 0.002, respectively). Conversely, ANA positivity and titer as well as anti-ds DNA and ANCA positivity demonstrated no significant differences at baseline versus follow–up (Table 4).
Comparison of the suggestive inflammatory and immunological markers of autoimmunity before and after direct-acting antiviral therapy among SVR patient subgroup.
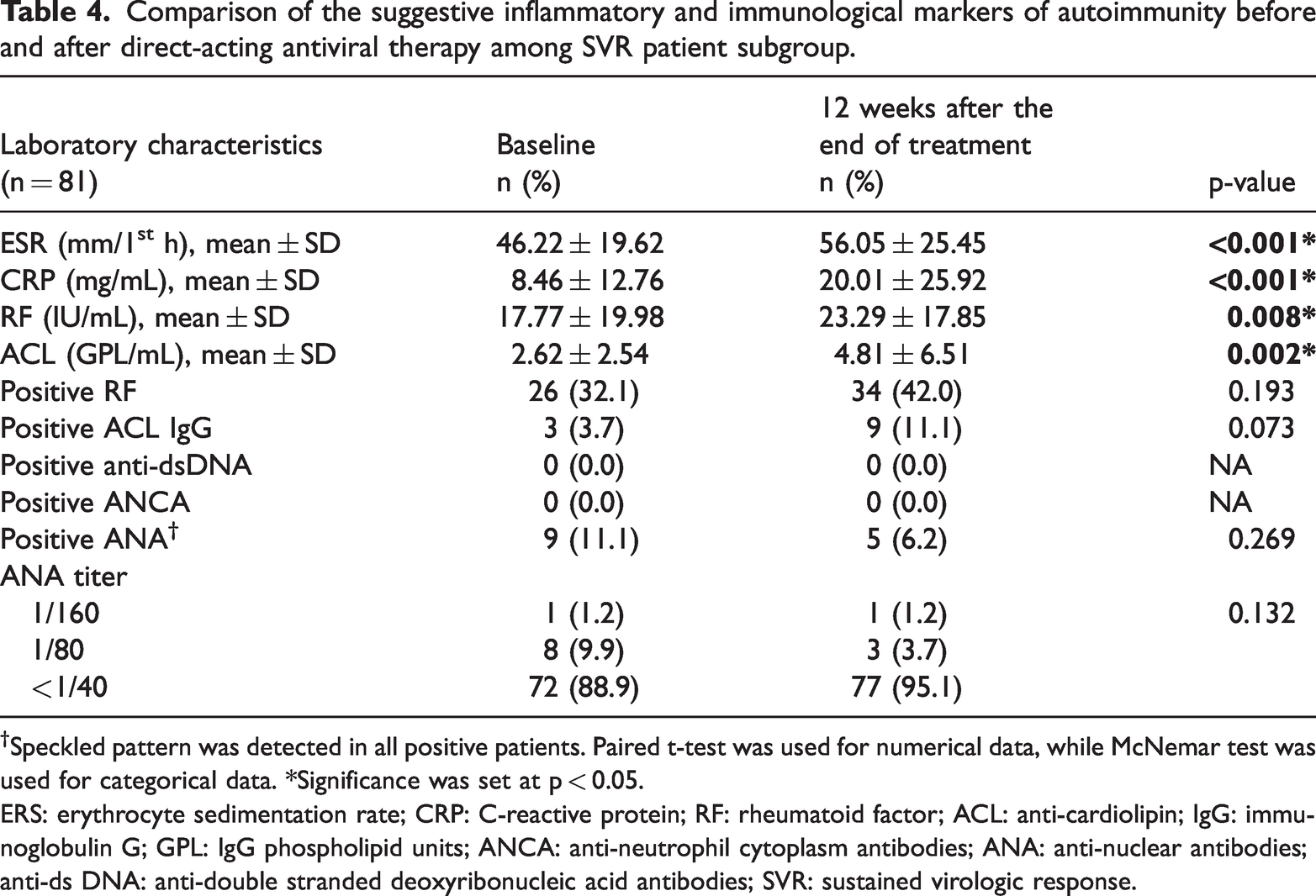
Speckled pattern was detected in all positive patients. Paired t-test was used for numerical data, while McNemar test was used for categorical data. *Significance was set at p < 0.05.
ERS: erythrocyte sedimentation rate; CRP: C-reactive protein; RF: rheumatoid factor; ACL: anti-cardiolipin; IgG: immunoglobulin G; GPL: IgG phospholipid units; ANCA: anti-neutrophil cytoplasm antibodies; ANA: anti-nuclear antibodies; anti-ds DNA: anti-double stranded deoxyribonucleic acid antibodies; SVR: sustained virologic response.
Discussion
The mega HCV-screening campaign in Egypt reported that the HCV antibody seroprevalence was 4.61% (76.5% of patients had active viremia).12,13 Approximately 16%–31% of patients with HCV experience various EHMs, including metabolic, cardiovascular, renal, lymphoproliferative, and neurologic complications.5,14 The introduction of DAAs dramatically improved HCV infection treatment outcome with higher SVR rates, acceptable tolerability, and shorter durations of therapy. 15 In contrast to the potential immunogenic effects of interferon, DAA-induced SVR was associated with a decrease in various chronic HCV EHMs.16–18 We herein evaluated the impact of DAAs on EHMs and serum biomarkers in patients with chronic HCV.
Our study included 130 patients; only 90 patients completed the study with a female-to-male ratio of 2:1. An SVR 12 weeks after treatment completion (SVR12) was achieved in 81 (90%) patients, which was consistent with the finding of previously published prospective studies in Egyptians who showed up to 100% SVR12.17–23 The most commonly reported autoimmune manifestations prior to therapy included musculoskeletal syndromes (64.2%) in the form of arthralgia (58.0%), fatigue (50.6%), and myalgia (43.2%) as well as neurological manifestations (23.5%) and neuritis (21.0%). Ocular manifestations (conjunctivitis and xeropthalmia) were observed in 23.5% of the population studied. Clinical features of Sjogren–Sicca syndrome (xeropthalmia and xerostomia) were observed in 22.5% of the population studied. Cutaneous manifestations (xerosis, purpura, cutaneous ulcers, and Raynaud’s phenomenon) were observed in 12.3% of the population studied, whereas pulmonary manifestations (cough and dyspnea) developed in 6.2% of the patients. The results of the study by Fathi et al. are consistent with our results; they detected fibromyalgia in 74.5% of patients with HCV, arthralgia in 73.5%, peripheral neuritis in 29.4%, chronic fatigue syndrome in 24.5%, arthritis in 16.7%, and Sicca symptoms in 9.8%. 19 However, Mohammed et al. detected EHMs in only 16.3% of their cohort. The most common clinical manifestations were chronic fatigue syndrome (9.5%), Sicca symptoms (8.8%), and arthralgia (6.5%). 5
Although the frequency of different autoimmune manifestations reduced following treatment, our findings were not statistically significant. Said et al. reported that the frequency of posttreatment fatigue showed only some improvement after DAA therapy compared with that before DAA therapy. 15 Other studies reported fatigue as a side effect of treatment, but they did not address the difference before and after treatment. Hence, fatigue could not be attributed to treatment with certainty.18,21 Conversely, Fathi et al. reported significant reduction in the frequency of EHMs following DAA therapy: arthralgia (73.5% vs. 19.6%), arthritis (16.7% vs. 7.8%), fibromyalgia syndrome (74.5% vs. 24.5%), purpura (24.5% vs. 2.0%), and peripheral neuropathy (PN; 29.4% vs. 19.6%). 19 Although the frequencies of pre- and posttreatment arthralgia and arthritis were not significantly different (p > 0.05), Alian et al. reported posttreatment remission of articular symptoms (80% complete remission vs. 5% partial remission). 21 Similarly, Kumthekar et al. demonstrated an overall trend toward improvement, with moderate improvement in subjective pain in 8 of the 10 patients who visited the rheumatology clinic following DAA therapy. This considerable improvement in cohort symptoms compared with those in our study could be attributed to the synergistic use of opioids in addition to DAAs during their study. 24 Interestingly, another Egyptian study on rheumatoid arthritis (RA) with HCV reported significant improvement of arthritis, arthralgia, morning stiffness, and myalgias following HCV elimination, with the post SVR12 DAS28 score significantly lower than the baseline score (p < 0.001). They also detected a significant decline in the mean levels of anti-CCP after achieving SVR12 (p < 0.05), which might be explained by the fact that the included patients with RA were concomitantly treated with disease-modifying antirheumatic drugs (DMARDs) during DAA therapy. 25 El Kassas et al. found significant improvement of extrahepatic cutaneous manifestations in Egyptian patients with HCV. 26 Notably, Tawfik et al. reported the same results with pruritis showing the most significant clinical improvement in their study. 27 The frequencies of neurological manifestations before and after treatment were 23.5% and 29.6%, respectively, in the current study. El Steiha Bahaa et al. documented DAA-induced PN in their cohort. They detected an increase in the frequency of patients with clinically evident PN from 10% to 30% (p < 0.001), while electrophysiological evidence of PN was detected in 15% of the patients prior to treatment (p = 0.062). 28
The laboratory and immunological markers prior to DAA therapy in the present study revealed that the ESR and CRP values were significantly higher following the end of treatment than at baseline (p < 0.001). Similarly, the posttreatment RF and ACL IgG values were significantly higher than those at baseline (p = 0.008 and 0.002, respectively). Conversely, ANA positivity and titer as well as anti-ds DNA and ANCA positivity demonstrated no significant differences at baseline versus follow-up. The increase in the mean ESR values following treatment could be explained by the skewness of the data; only seven patients had strikingly elevated ESR ranging from 80 to 100 mm/1st h. However, Lashen et al. reported significant improvement of disease activity (DAS28) following treatment in patients with RA. There was a significant decline in the mean serum anti-CCP antibody levels, RF titer, ESR, and CRP values in patients who achieved SVR12.19,26 Similar results were documented by Alian et al. in patients with RA with concomitant chronic HCV infection. Notably, these patients continued DMARDs and nonsteroidal anti-inflammatory drugs during DAA therapy. 21 Moreover, Lauletta et al. reported a significant decline in the RF titers following 12 weeks of DAA therapy. Furthermore, the RF titer increased again at SVR12; however, the increment was insignificant. 29 Approximately 3.7% of our patients had positive ACL, a frequency that is almost similar to that reported by Ordi-Ros et al. (3.3% of Spanish patients). However, the difference in ACL positivity between patients (3.3%) and healthy controls (0%) was not statistically significant in their study (p = 0.07). 30 A significant increase in the ACL titer was observed following DAA therapy (2.62 ± 2.54 vs. 4.81 ± 6.51 GPL/mL; p = 0.002). These findings are supported by another hypothesis reported by Romano et al., who stated that chronic immune stimulation caused by HCV may result in dysregulated immune responses that could cause autoimmune phenomena. 31 Consistently, the cessation of chronic immunological activation caused by HCV may reduce the risk of developing subclinical or overt autoimmunity. Careful long-term follow-up may be necessary to determine the significance of persistently positive serum autoantibodies in the small percentage of patients whose autoreactive immunoglobulins did not disappear after DAA treatment; in these patients, autoreactivity may not be linked to HCV infection or it may have disengaged from the triggering viral infection. This description explains immune response in general, with no clear specific explanation for ACL. ANA was detected in 11.1% of the patients at baseline. However, Gilman et al. reported positive ANA in a higher percentage (21.8%) compared with ours. Of interest, they stated that there is no evidence that patients with autoantibodies are more likely to develop EHMs compared to those without. 32 No significant difference regarding ANA positivity before and after treatment (11.1% vs. 6.2%; p = 0.269). This result is in agreement with that of Simoes et al. who reported improvement of the serologic and histopathological features of autoimmune hepatitis in patients with chronic HCV following DAA treatment. They found positive ANA in 28.6% patients before treatment versus 19.05% patients after treatment without a significant difference between the pre- and posttreatment results. 33 Romano et al. reported the prevalence and outcome of serum autoantibodies (ANA, anti-smooth muscle antibody, and antimitochondrial antibodies) in 77 patients with HCV undergoing DAAs treatment tested for autoantibodies. They found positive autoantibodies at baseline in 31 patients; and only 20/31 patients completed their treatment. ANA was positive in 13/20 patients. After treatment, nine patients turned negative; and three patients showed reduction to 1:80. Seven patients did not show any change. The impact of DAA on the improvement of the EHMs was also highlighted by many studies including34–38 where they showed how HCV infection affects a variety of organs, therefore inducing EHMs and they concluded that introduction of interferon-alpha–free antiviral regimens are safe, effective, had a good prognosis for patients with HCV infection, and demonstrated reduction in both the incidence and severity of EHMs of HCV infection.
Age >49 years, female sex, DM, direct bilirubin level > 0.12, mg/dL, high HbA1C exceeding 5.5%, and hemoglobin ≤14.1 g/dL were associated with increased probability of existence of pre-treatment autoimmunity features while tobacco consumption and drug abuse were linked with decreased probability. Furthermore, female sex increased the probability of persistence of autoimmunity features following the administration of DAA therapy.
Limitations
The small sample size as well as the single center approach of the current study are limiting factors; thus, large-scale studies are warranted to explore the effect of DAAs on the different EHMS using a larger sample size, with a multi-center approach to include different patient cohorts. The small number of non-responders in the patient group did not support appropriate analysis or comparison with the responders group, a future larger sample size could overcome this limitation. Additionally, lack of assessment of other immunological markers such as complement components and cryoglobulins is another limitation in the current study that was attributed to technical difficulties and feasibility; nevertheless, this could offer a great added value in the exploration of DAAs effects on the autoimmune phenomena related to HCV infection.
Recommendations
The question of research provides further input about the possible immunostimulatory effect of DAAS in patients with HCV and emphasizes the need to follow up these patients after the introduced therapy for progress or development of de novo autoimmune manifestations. Complete rheumatological and immunological assessment and follow-up should be offered to all patients with chronic HCV before, during, and after DAA therapy for early diagnosis and management of EHMS. Studying the effect of DMARDs on chronic HCV infection-related autoimmunity in cured patients is recommended.
Conclusions
In the present study, DAA therapy was not associated with significant improvement in the autoimmune phenomena associated with chronic HCV infection at early follow-up 12 weeks after the end of treatment. However, the most prevalent autoimmune manifestations before therapy were musculoskeletal manifestations regardless of SVR and arthralgia.
Footnotes
Acknowledgments
None.
Author contributions
All authors have contributed equally to the manuscript. Reem Hamdy A Mohammed, Hesham I Elmakhzangy, Mai A Rabie, Ahmed ET Elsayed, Doaa HS Attia, and Dina MR Bahgat: conceptualization, planning, and protocol writing. Mai A Rabie, Doaa HS Attia, Reem Hamdy A Mohammed, and Hesham I Elmakhzangy: clinical part of the study. Hesham I Elmakhzangy, Dina MR Bahgat, and Mai A Rabie: laboratory testing. Reem Hamdy A Mohammed and Mai A Rabie: manuscript writing and peer review.
Data availability statement
The data presented are available and can be provided upon reasonable request.
Declaration of conflicting interests
The authors declare no conflict of interest associated with the preparation of this article.
Disclosures
Authors have no disclosures.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
