Abstract
Keywords
Chronic hepatitis C (CHC) is the major cause of liver cirrhosis; traditionally the standard of care was pegylated interferon alpha (PEG-IFNα) and ribavirin. Due to this cytokine’s immunological properties, accurate immunological screening was mandatory before starting therapy and the treatment was contraindicated in patients with concomitant autoimmune diseases. Several reports of the onset of autoimmune hepatic disease after CHC treatment with IFN have been made. 1 Novel IFN-free regimens have dramatically reduced the risk of this complication, and recent guidelines 2 no longer list autoimmune disorders as an absolute contraindication. Nevertheless, we report the first case of a patient in whom hepatitis C virus (HCV) eradication by an IFN-free regimen unmasked primary biliary cholangitis (PBC).
A 54-year-old female patient with CHC was referred to our outpatient unit in July 2015. She had been diagnosed with HCV infection, genotype 1b, in 1997 and failed to respond to two PEG IFNα-based courses. She had been suffering from rheumatoid arthritis since 2008. In 2009, her immunoglobulin (Ig)M and IgA levels were normal, with low positivity for anti-nucleus antibodies (ANAs) 1:80; other autoantibodies tested negative.
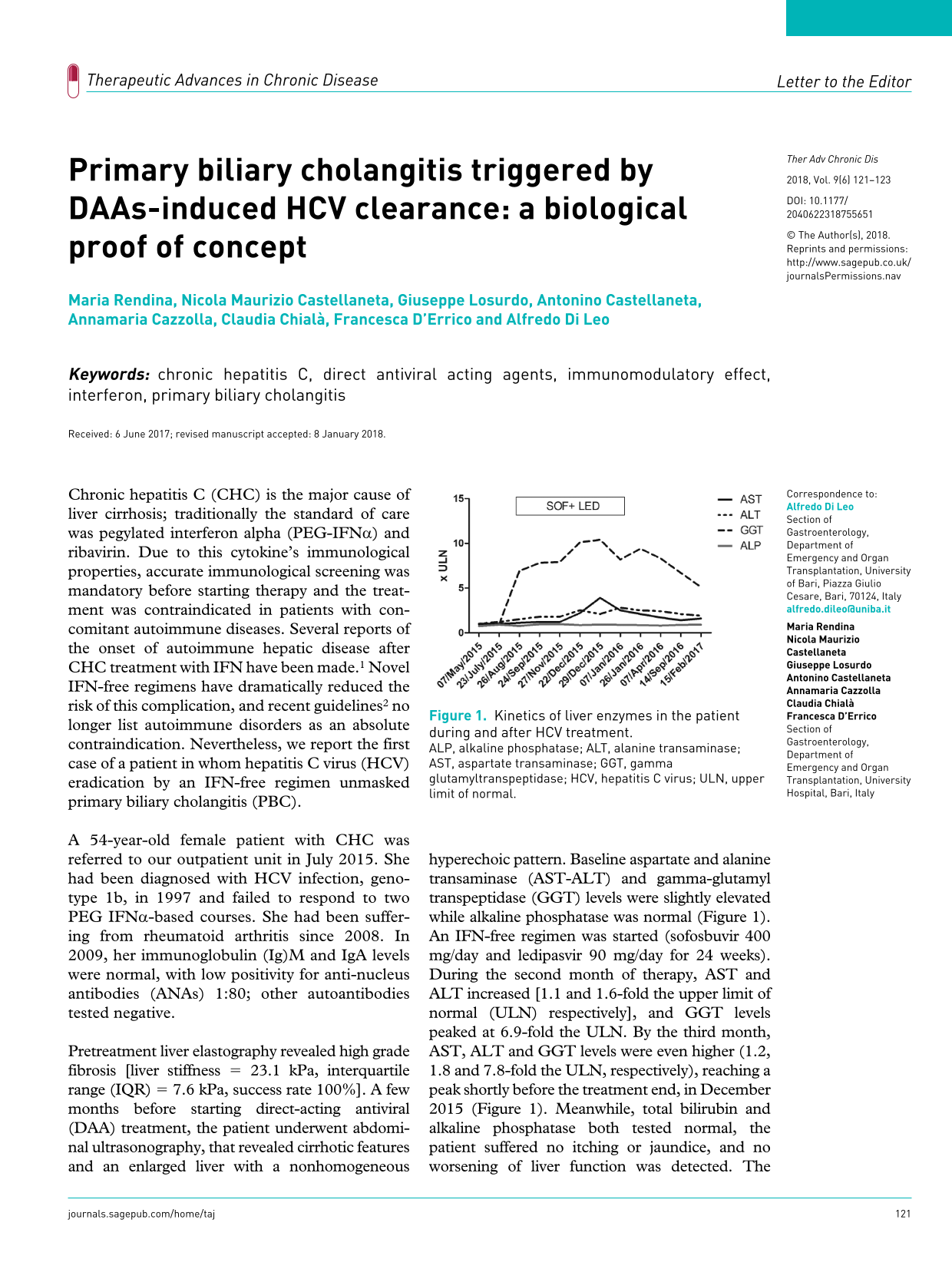
Pretreatment liver elastography revealed high grade fibrosis [liver stiffness = 23.1 kPa, interquartile range (IQR) = 7.6 kPa, success rate 100%]. A few months before starting direct-acting antiviral (DAA) treatment, the patient underwent abdominal ultrasonography, that revealed cirrhotic features and an enlarged liver with a nonhomogeneous hyperechoic pattern. Baseline aspartate and alanine transaminase (AST-ALT) and gamma-glutamyl transpeptidase (GGT) levels were slightly elevated while alkaline phosphatase was normal (Figure 1). An IFN-free regimen was started (sofosbuvir 400 mg/day and ledipasvir 90 mg/day for 24 weeks). During the second month of therapy, AST and ALT increased [1.1 and 1.6-fold the upper limit of normal (ULN) respectively], and GGT levels peaked at 6.9-fold the ULN. By the third month, AST, ALT and GGT levels were even higher (1.2, 1.8 and 7.8-fold the ULN, respectively), reaching a peak shortly before the treatment end, in December 2015 (Figure 1). Meanwhile, total bilirubin and alkaline phosphatase both tested normal, the patient suffered no itching or jaundice, and no worsening of liver function was detected. The patient took no drugs other than sofosbuvir and ledipasvir before and during the treatment. Accordingly, autoantibodies were reassessed, showing positive ANAs (1:320) and anti-mitochondria (1:640), M2 subtype. Liver biopsy showed a cirrhotic appearance with a cholestatic pattern, sprouting bile ducts and destruction of the native biliary tree, a picture suggestive of PBC. A second ultrasound scan during the biopsy procedure showed a similar picture. No dilation of bile ducts was observed. Sustained viral response was achieved by the 12th week, and a course of ursodeoxycholic acid was started, resulting in a partial decrease of liver enzymes. In particular, according to the Paris criteria, 3 our patient was fully responsive to ursodeoxycholic acid.

Kinetics of liver enzymes in the patient during and after HCV treatment.
The prevalence of PBC in CHC patients is very low, around 1.5%; typically the diagnosis of CHC precedes PBC, 4 suggesting a role for HCV in the PBC pathogenesis. Several case reports have described the onset of PBC after IFN-based therapies to eradicate HCV. 5 The immunostimulating properties of IFN have been considered the major triggering factor, since this cytokine may induce an imbalance in the immune system, driving the activation of T helper 1 lymphocytes (Th1) over T helper 2 (Th2). In this regard, enhanced activation of Th1 may be a possible pathogenic mechanism causing biliary damage in PBC. 6
In the present case, HCV was eradicated with an IFN-free regimen. To the best of our knowledge, this is the first case of PBC induced by HCV clearance. This event is partially explained according to the ‘hygiene hypothesis’, which postulates that autoimmune responses, when simultaneously elicited by two distinct antigens, tend to be mutually inhibitory 7 and when one disappears (HCV in our case), the immune system may mount a response against self-antigens, causing autoimmune disorders (notably PBC). Moreover, HCV treatment has been demonstrated to lead to lymphocyte polarization towards Th1 over Th2, a mechanism implicated in the pathogenesis of PBC. 8 Some obscure points need to be considered. For example, normal alkaline phosphatase levels are unusual in PBC, but some reports in literature have highlighted this eventuality.9,10 The increased ANA titers could raise the suspicion of an overlap syndrome. However, histological analysis did not reveal any sign of autoimmune hepatitis. Moreover, ANA positivity is a frequent finding in PBC 11 not associated with autoimmune hepatitis, as described in our patient. On the other hand, ANAs are frequently positive when PBC is associated with other autoimmune disorders, 11 and our patient was affected by rheumatoid arthritis. Another limitation of the present manuscript is that it is only a single report of an event, therefore cohort studies investigating the possibility of autoimmunity onset after viral eradication by DAAs are necessary.
Our report could represent a biological proof of concept of HCV eradication as a potential triggering factor for autoimmune activation. In conclusion, we believe that patients with autoimmune factors (female sex, young age, concomitant autoimmune disorders) should continue to be screened carefully for autoimmune liver diseases before and during HCV treatment even with IFN-free regimens. Furthermore, several warnings of the risk of hepatitis B reactivation 12 when using novel DAAs have been issued by the United States Food and Drug Administration, supporting our observation illustrating the potential immunomodulatory effects of such drugs.
Footnotes
Acknowledgements
The authors thank Mary Pragnell for linguistic revision.
M. Rendina, N.M. Castellaneta and A. Castellaneta followed the patient. F. D’Errico, A. Cazzolla and C. Chiala’ collected the data. M. Rendina and G. Losurdo wrote the manuscript. A. Di Leo critically revised the manuscript. All authors read and approved the final version.
Written informed consent was obtained from the patient for publication of this case report and any accompanying images
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
