Abstract
Multiple myeloma (MM) is a B-cell malignancy involving monoclonal plasma cells that produce nonfunctional immunoglobulins. It is still incurable due to de novo or acquired resistance of malignant cells to standard chemotherapies. Thus, understanding the mechanism of therapeutic resistance is essential to develop better interventions and identifying new therapeutic targets. MM is a heterogeneous disease characterized by complex genetic alterations, which are critical for prognostic stratification. Due to the heterogeneity and clinical challenges associated with MM, there remains a critical need for novel biomarkers to improve both early diagnosis and prognostic accuracy. Antioxidants participate in protecting cancer cells from the damaging effects of oxidative stress by neutralizing reactive oxygen species. These molecules may have their own role in myeloma progression and drug resistance. However, studies reported contradictory ideas regarding the levels and roles of antioxidants in MM. The main aim of this review is to conceptualize the diagnostic, prognostic, and therapeutic significance of common antioxidants for MM. The most common antioxidant systems discussed in this study were DJ-1, glutathione, thioredoxin, and superoxide dismutase. The review revealed that antioxidants are overexpressed in myeloma cells compared to normal plasma cells. This excess expression of antioxidants increases tumor cells’ survival and their chemoresistance leads to poor prognosis and short survival of myeloma patients. Thus, antioxidants and their synthesis pathways may have a promising prognostic, diagnostic, and therapeutic role in MM. Vitamin C (ascorbic acid), on the other hand, has a two-faced role in cancer, including MM. It can function as an antioxidant (increase myeloma cell resistance to chemotherapy) or pro-oxidant (enhance the therapeutic effect of myeloma drugs), depending on its dose and route of administration. Pharmacological (ultra-high) doses of vitamin C administered intravenously may act as a pro-oxidant that can selectively kill myeloma cells by elevating the cellular labile iron pool and generating extracellular H2O2.
Introduction
Multiple myeloma (MM), a malignancy of plasma cells, is characterized by the uncontrolled proliferation of monoclonal plasma cells in the bone marrow (BM), resulting in the production of nonfunctional intact immunoglobulins. 1 It is the second most common hematological malignancy next to non-Hodgkin lymphoma, accounting for 1% and 10% of all and blood cancers, respectively. 2 The incidence of MM has increased by as much as 120% since 1990, and now around 200,000 cases are reported annually worldwide. 3 The disease is mostly preceded by two asymptomatic stages, benign monoclonal gammopathy of undetermined significance (MGUS) and smoldering multiple myeloma. 4 The hallmark end-organ damage of MM, called “CRAB” symptoms, standing for hypercalcemia, renal involvement, anemia, and bone lesions are not the feature of these two asymptomatic stages. 5
MM is characterized by poor prognosis, remission, and relapses, and only about 47% of patients with MM have a 5-year survival rate. 6 Clinical data showed that roughly half of initially bortezomib (BTZ; one of the well-known proteasome inhibitors, PI) sensitive MM patients do not respond to BTZ once the disease has relapsed. 7 Thus, it is essential to understand the mechanism underlying the resistance to chemotherapy and to identify those patients with poor prognosis. 8 Moreover, a deeper understanding of unique biochemical targets and molecular mechanisms involved in drug-resistant MM disease provides a clue to discover a novel target and design innovative drug combinations to improve survival and remission rates of MM patients. 9
It is well known that myeloma cells produce large quantities of immunoglobulins, and this results in the production of many misfolded and/or unfolded proteins.
An efficient protein degradation system is necessary to maintain myeloma cell homeostasis, which may be disrupted due to these proteins. 10 Inhibition of the protein degradation system called ubiquitin/proteasome system by PIs results in the accumulation of unfolded/misfolded proteins in the endoplasmic reticulum (ER). 11 This leads to the occurrence of events such as loss of the transmembrane potential, cytochrome c release, caspase 9 activation, and generation of reactive oxygen species (ROS), which may induce myeloma cell death. 12
ROS are essential in multiple signaling pathways and involved in a variety of processes, including inflammation and pathogen elimination, cell growth, and differentiation. 13 The role of ROS in cancer pathogenesis and development depend on their amount and exposure time. 14 At low level, ROS play a crucial role in tumorigenesis and progression by inducing DNA mutations and genomic instability. They promote cell proliferation and differentiation by activating transcription factors, such as nuclear factor-κβ (NF-κβ) and activator protein-1. 15 On the other hand, high levels of ROS are toxic to cancer cells and can potentially induce cell death. 10 ROS induces cell death by caspase activation, activation of Bcl-2 family proteins, and modulation of protein kinases.10,15 Tumor cells are generally characterized by having higher metabolic demands due to their highly proliferative nature and increased production of ROS. 16 To balance the state of oxidative stress, cancer cells need their own escape mechanisms. One of these escaping mechanisms to prevent further cellular damage is increasing the production of ROS-scavenging molecules called antioxidants and stress-response proteins. 16 Cancer cells take advantage of upregulated antioxidant production for the alleviation of ROS-induced cell damage.15,17 Studies on large randomized samples reported that antioxidants are upregulated in many human cancer types, correlating with tumor proliferation, survival, and drug resistance.7,15,18–20
Although there is an advanced understanding of the pathogenesis and improved landscape of MM treatment, the incidence of MM is increasing, which may be due to the development of measurement techniques or the aging of the population. Resistance to standard myeloma therapies, including PI, immunomodulatory drugs, and monoclonal antibodies, and subsequent relapse of the disease are the main reasons for mortality in MM patients. Overexpression of antioxidants in MM may be one of the reasons for the relapse of the disease, chemoresistance of myeloma cells, and poor prognosis of MM patients.7,17,21,22 Antioxidants may participate in protecting myeloma cells from the damaging effects of oxidative stress by neutralizing ROS. 17 Thus, focusing on the antioxidant systems may be one of the potential molecular targets to develop new single or combinatory anti-myeloma therapy to reverse myeloma chemoresistance and improve myeloma patients’ survival. However, the role of antioxidants in MM pathogenesis and drug resistance is not well elucidated, even though MM is the second most common hematological malignancy.
Antioxidants found in human cells can be enzymatic (e.g., catalase (CAT), superoxide dismutase (SOD), glutaredoxin (Grx), glutathione peroxidases (GPx), glutathione S-transferases (GST), thioredoxin reductase (TrxR) or non-enzymatic (e.g., glutathione (GSH), DJ-1, and thioredoxin (Trx) systems.9,15. DJ-1 protein is a small (20 kDa) protein comprising 189 amino acid protein dimer located in the nucleus and cytoplasm.23,24 It is encoded by the Parkinson’s disease-associated protein seven (PARK7) gene. 25 The antioxidant role of DJ-1 is mainly due to its ability to upregulate the expression of genes encoding transcription factors and/or proteins with an antioxidant function which is associated with cancer cell survival and poor prognosis.15,26–28 The GSH system is the most abundant intracellular antioxidant system. 14 The system consists of GSH, GPx, Grx, and GST.29,30
GSH is a major antioxidant defense molecule existing in all cellular compartments, which is composed of amino acids cysteine, glutamine, and glycine. GPx and GST contribute to ROS detoxification by reducing hydrogen peroxide or hydroperoxide using GSH as a substrate. 30 The Trx system consists of Trx, TrxR, and reduced nicotinamide adenine dinucleotide phosphate (NADPH).16,30,31 Trx 1 and TrxR 1 are located mainly in the cytoplasm, whereas Trx 2 and TrxR2 are found in the mitochondria. 28 The Trx system maintains cellular redox homeostasis by either directly scavenging ROS or by regulating several redox enzymes. 31 The Trx1 and Trx2 possess two cysteine active sites to serve as hydrogen donors to the Trx-dependent peroxide reductases toward reducing oxidized proteins and protecting cells. SODs are an important free-radical scavenger system functioning as the first line of defense against oxidative damage by catalyzing the dismutation of superoxide anions (O2−), yielding hydrogen peroxide (H2O2) and O2. 32 H2O2 is subsequently transformed into O2 and H2O in a process catalyzed by GPx or CAT. Three groups of SOD are found in mammals: cytoplasmic SOD (Cu/Zn-SOD, SOD1), mitochondrial SOD (Mn-SOD, SOD2), and extracellular SOD (EC-SOD, SOD3). 33
Studies in vitro and vivo data suggested that there are certain antioxidants that could selectively inhibit the growth of tumor cells and thereby enhance the effects of cytotoxic therapy. 34 One of these antioxidants that can act as “pro-oxidants” at elevated levels is vitamin C, ascorbic acid (AA). 35 The role of AA in cancer, including MM, has been debated for decades. Through the reduction of intracellular ROS, AA could shield MM cell lines from the toxicity of arsenic trioxide (As2O3). 36
On the other hand, vitamin C administered intravenously at a pharmacological concentration has potent anticancer activity. 37 Especially in the presence of catalytic metal ions such as iron, pharmacologically dosed vitamin C exerts pro-oxidant effects leading to the formation of highly ROS, resulting in cancer cell death.38,39 Vitamin C readily undergoes pH-dependent autoxidation, producing H2O2.38,40–42 Under high concentration conditions, vitamin C catalyzes the reduction of free transition metal ions, including iron, resulting the formation of oxygen radicals. Reduced iron ions react with H2O2 to form reactive hydroxyl radicals or peroxide ions. 43 High-dose vitamin C may elevate cellular labile iron pool (LIP) concentrations, causing cell death through ferroptosis (a form of iron-dependent type of controlled cell death).37,39,44 Vitamin C induced ferroptosis by depleting GSH and increasing levels of ferrous ions (Fe2+) and ROS.39,45
Moreover, vitamin C produces H2O2 in the extracellular space, consequently inhibiting tumor cells.40,41 Extracellular ascorbate in pharmacologic concentrations mediates death of human Burkitt’s lymphoma cells, but not normal cells, by apoptosis and pyknosis necrosis, independently of intracellular ascorbate. Ascorbate-mediated cell death was due to protein-dependent extracellular H2O2 generation, via ascorbate radical formation from ascorbate as the electron donor. 41 In breast cancer, dose-dependent vitamin C is involved in the suppression of the NF-κB signaling pathway and reduces tumor cell proliferation and survival. 46 Addition of a pharmacological concentration of ascorbate to p53+/+ HTC116 cells (colon cancer) caused an extension of the p53 half-life by induction of MDM2 degradation. The authors suggested that the tumor response to ascorbate treatment may depend on p53 expression, and the efficacy may be enhanced by combining vitamin C therapy with another cancer drug. 47 Furthermore, vitamin C stimulates the production and activation of immune system cells, such as NK cells and CD8+ T cells, which are essential in the defense against tumor cells. 48
Apoptosis-inducing factor 1 (AIF1) plays a critical role in pharmacological AA-induced myeloma cell death. AIF1 induces cell death in caspase-dependent and caspase-independent manners. Pharmacological AA by reacting with LIP generates ROS, inducing mitochondrial AIF1 cleavage. Cleaved AIF1 is released from the mitochondria to the cytoplasm and then translocates to the nucleus to induce chromatolysis, leading to caspase-dependent and caspase-independent cell death. 37
In this review, we will see the role of the most common antioxidant systems (DJ-1, GSH, Trx, SOD, and vitamin C) in MM. The review aimed to understand the diagnostic, prognostic, and therapeutic implications of these antioxidants in MM (Table 1).
Major cellular antioxidant systems relevant to MM.

AA, ascorbic acid; BTZ, bortezomib; GPx, glutathione peroxidase; Grx, glutareductase; GSH, glutathione; GST, glutathione S-transferase; H2O2, hydrogen peroxide; MM, multiple myeloma; ROS, reactive oxygen species; SOD, superoxide dismutase; Trx, thioredoxin; TrxR, thioredoxin reductase.
Common antioxidants and their roles in MM
Diagnostic role
The diagnosis of MM requires ⩾10% clonal BM plasma cells or a biopsy-proven plasmacytoma and evidence of one or more MM defining events (CRAB criteria), BM clonal plasmacytosis ⩾60%, serum involved/uninvolved free light chain ratio ⩾100, urinary monoclonal protein excretion is ⩾200 mg per 24 h and more than one focal lesion on magnetic resonance imaging. 2
However, a large proportion of MM patients experience a prolonged asymptomatic period, and a small proportion are asymptomatic at diagnosis. 49 Identification of novel and specific biomarkers for early diagnosis is urgently needed. Tumor cells have a significant increase in their metabolic demands due to their high proliferative nature, and ROS production is increased. To prevent further cellular damage by ROS, cancer cells overexpress a network of intracellular redox buffering molecules, antioxidants. 50 Studies on primary and cultured myeloma cells indicated that serum levels of antioxidants are higher in patients with MM compared to healthy individuals.
DJ-1 protein
DJ-1, also called PARK7, Parkinson’s protein 7, is a multifunctional protein of 20 kDa whose gene is located in the distal part of human chromosome 1. 51 It was first associated with a familial form of Parkinson’s disease due to loss of DJ-1 function. Apart from its role in neurodegenerative disorders, DJ-1 acts as a mitogen-dependent oncogene that plays a crucial role in the progression of various types of cancer and is identified as a novel oncoprotein. 52 DJ-1 was initially identified as a putative oncogene that, together with H-RAS, can transform mouse NIM3T3 cells. 23 It is a multifunctional protein protecting cells from oxidative-mediated death through several molecular processes. 15 Reverse transcriptase-quantitative PCR (RT-qPCR) and western blotting analysis, respectively, showed that DJ-1 mRNA and DJ-1 protein expressions are significantly higher in newly diagnosed and relapsed myeloma patients compared to healthy individuals. 22 DJ-1 is also expressed at high levels in primary lung tumors, prostate cancer, and acute leukemia patient samples.53–55 It is also upregulated in 81% of primary ovarian tumors and 80% of solid metastases. 52 DJ-1 functions as an antioxidant when cells experience oxidative stress, and it induces the expression of several antioxidant enzymes. 28 The protein has three redox-sensitive cysteine residues, namely Cys-46, Cys-53, and Cys-106. 56
DJ-1 performs its cyto-protection role mainly due to oxidation of its Cys-106 residue to cysteine sulfinic acid (Cys106-SO2−) to scavenge ROS.56–58 The well-known role of DJ-1 as an antioxidant is through regulating other transcription factors and antioxidants, including GSH, Trx, and SOD.
The GSH system
GSH is an important buffering agent that maintains redox homeostasis in eukaryotic cells, and increased GSH levels are associated with cancer development. 59 The level of GSH is found to be significantly higher in myeloma cells compared to controls. 60 Enzyme-linked immunosorbent assay (ELISA) determination of glutathione S-transferase P1 (GSTP1) from myeloma patients and healthy controls found the mean concentrations of GSTP1 to be 29.64 ± 9.13 µg/l and 16.15 ± 5.64 µg/l, respectively. 61 The study showed that GSTP1 has an excellent diagnostic power for MM (area under the curve, AUC = 0.9) with a cut-off value of 19.45 µg/l. 61 Moreover, increased expression of mRNA of GPx-1 and GST enzymes in MM cell lines is found relative to normal plasma cells. 62 Excess GSH in MM is positively correlated with the elevation of serum glycine. The elevation of glycine in MM patients is caused by the degradation of bone collagen mediated by MM cell-secreted matrix metallopeptidase 13 (MMP13). Myeloma cells use the channel protein solute carrier family 6 member 9 (SLC6A9) to absorb this extrinsic glycine for the synthesis of GSH for their survival and escape from chemotherapy. 63 Furthermore, it may be correlated to the translocation of nuclear factor erythroid 2-related factor 2 (Nrf2) for activation of stress-protective genes, including cystine-glutamate antiporter Xc− for GSH synthesis. 64 DJ-1 protein also increases both the expression and activity of a rate-limiting enzyme called glutamate cysteine ligase (GCL) in the biosynthesis of GSH.15,28,65 Myeloma cells produce high levels of immunoglobulins.
They might rely on increased GSH to reduce protein misfolding due to ROS and to handle toxic protein derivatives. 59 These findings revealed that GSH and its family proteins may serve as diagnostic biomarkers for MM.
The Trx system
Upregulated levels of Trx and TrxR are observed in MM patients’ serum. Both Trx1 and TrxR1 proteins are expressed at significantly higher levels in new and relapsed myeloma patient cells compared to normal plasma cells. 31 Moreover, TrxR1 gene expression analysis data showed that high-risk patients with MM exhibited increased TrxR1 expression relative to low-risk patients. 7 MM cells also overexpress TrxR2, but have low canonical markers of ER stress (ATF4, XBP1s, and CHOP) compared to control cells. 66 Overexpression of Trx and TrxR may be due to activation of their genes in the nucleus at the promoter region after Nrf2 is translocated to the nucleus due to increased DJ-1 in MM (Figure 2(b)).15,26,28 As a result, Trx and TrxR may have a promising diagnostic role for MM.
Superoxide dismutase
SOD1 plays a multifaceted role in signaling pathways, proliferation, and survival of cancer cells. Elevated levels of SOD1 have been observed in various cancers, including leukemia and breast cancer.67,68 A study on human MM cell lines using qPCR analysis found that SOD mRNA was overexpressed in MM cells compared to control cell lines. Cell lines from MGUS and MM, respectively, showed 1.6- and 2.4-fold increased expression of SOD1 compared to normal plasma cells. 9 Pro-proliferative cytokines, including TNF-α, IL-1β, and IL-6 induces upregulation of SOD in myeloma cells via NF-κB-mediated transcription.69,70 However, SOD is found to be significantly lowered in patients with MM as compared to controls in another study.
The reason for the variation may be due to the use of different lab methods; the latter used a commercially available kit to determine whole blood SOD. 71 These findings lead to the possibility of using SOD as a tumor biomarker for the diagnosis of MM.
Although DJ-1 and other antioxidants are elevated in MM cells compared to healthy controls, their expression in multiple cancer types limits their specificity as a diagnostic marker for MM. Consequently, while these antioxidants may help distinguish MM patients from healthy individuals, their diagnostic utility remains limited and requires further investigation alongside established MM biomarkers.
Prognostic role
Although the outcomes for MM patients showed improvement with advanced drugs and therapeutic approaches, most patients still showed poor prognosis, and the disease remains incurable. 49 Thus, it is essential to identify biomarkers that enables to evaluate the prognosis and monitor treatment outcomes of MM patients. Cancer cells exploit antioxidant molecules to maintain intracellular redox homeostasis to prevent further cell damage from the anticancer drugs inducing ROS production. 50 Overexpression of these antioxidants in MM patients may be associated with increased cell proliferation, evasion of apoptosis, chemoresistance, and therefore poor prognosis of the patients.31,50
DJ-1 protein
Overexpression of DJ-1 in tumor cells is positively correlated with tumor metastasis and negatively correlated with patient survival. 27 A study on human MM cell lines shows that elevated DJ-1 levels correlate with tumor metastasis, chemoresistance, and poor prognosis of MM patients. MM patients having higher DJ-1 have a high risk of death compared to those with lower DJ-1 levels. 22 The elevated expression of DJ-1 is also significantly associated with the stage, grade, and poor progression-free survival of ovarian tumor patients. 28 DJ-1 functions as a cytoprotective molecule through direct scavenging of ROS through various mechanisms. 26 It eliminated hydrogen peroxide (H2O2) by oxidizing itself to protect cells against H2O2-induced cell death. 15
Consistent findings of DJ-1 as an antioxidant are mainly through regulating transcription factors and other antioxidants. One of the cyto-protective mechanisms of DJ-1 is through inhibiting the p53-Bax-caspase pathway. 52 DJ-1 binds with the C-terminus of p53, and therefore p53 cannot activate the transcription of the pro-apoptotic factor, Bcl-2-associated X (Bax). This inhibits the downstream caspase activation leading to myeloma cell cycle progression and long survival. 56
Additionally, DJ-1 reduces the expression of Krüppel-like family (KLF) in myeloma cells. 22 KLF induces myeloma cell death by activating the death-associated protein kinase 2 (DAPK2) 72 as well as by increasing cellular ROS via repression of the antioxidant gene TrxR2, resulting in myeloma cell death. 73 This results in myeloma cell survival and proliferation, ultimately leading to poor treatment outcomes of myeloma patients. 22
Regulating the expression of hypoxia inducible factor-1 alpha (HIF-1α) is another important role of the DJ-1 protein in MM. 22 The BM microenvironment in myeloma patients is hypoxic, activating HIF-1α expression. 74 DJ-1 negatively regulates von Hippel-Lindau (VHL) ubiquitination activity by inhibiting HIF-VHL interaction. 28 Then, HIF-1α translocates to the nucleus, dimerizes with HIF-1β, and activates the expression of vascular endothelial growth factor (VEGF), glucose transporter 1 (GLUT1), and antioxidants. This results in proliferation, growth, survival, and chemoresistance of myeloma cells.7,75,76 Hypoxia is also able to activate the NF-κβ signaling pathway in myeloma cells. 7 NF-κβ and its signaling pathways are constitutively activated in MM cells, leading to their proliferation, drug resistance, and survival. 31
Another mechanism of the DJ-1 protein in myeloma cell-protection is through binding with and inhibiting apoptosis signal-regulating kinase 1 (ASK1). 27 ASK1 is a mitogen-activated protein 3 kinase (MAP3K) family member activated by oxidative stress stimuli and stimulating the activation of downstream SAPK/JNK and p38 signaling pathways. 77 Under normal conditions, ASK1 is bound by the reduced form of Trx1, keeping ASK1 in an inactive state. 27 However, under oxidative stress conditions, including cancer, Trx1 is oxidized and releases ASK1, allowing ASK1 activation by auto-phosphorylation. 27 Ask1 activation induces a cascade of the MAPK pathway with subsequent activation of JNK and p38, leading to stress-induced myeloma cell apoptosis (Figure 1(a)).15,78,79 However, in MM, DJ-1 inhibits ASK1 activation by preventing the dissociation of Trx1 from ASK1 and by increasing the transcription of Trx1, thus protecting myeloma cells from apoptosis (Figure 1(b)). 27 DJ-1 performs this inhibitor activity through self-oxidation to scavenge ROS, thus minimizing the dissociation of the Trx1/ASK1 complex. 58 DJ-1 also inhibits ASK1 activation in the cytoplasm through nuclear sequestration of ASK1 activator protein, death domain-associated protein (Daxx).58,80 Studies reported that a lack of ASK1 increased long-lived plasma cell numbers in MM patients compared to controls, and this is related to the poor prognosis of myeloma patients.81,82 Thus, binding of DJ-1 with ASK1 inhibits ASK1-induced apoptosis under oxidative stress conditions and results in cancer cell survival.15,26

Regulation of ASK1/Trx1 by DJ-1 under oxidative stress. (a) ASK1 is dissociated from Trx1 under oxidative stress conditions in the absence of DJ-1 and gets activated. Activated ASK1 again activates JNK/P38 pathways, which result in myeloma cell apoptosis. (b) However, overexpression of DJ-1 under oxidative stress inhibits dissociation of ASK1 from Trx. This inhibits ASK1 activation, and rescues myeloma cells from death.
More importantly, the DJ-1 protein is involved in upregulating the expression of the transcription factor, Nrf2, in cells. 83 Nrf2 is a master regulator of the cellular response to oxidative stress through direct regulation of over 200 genes.14,84 Regulation of these genes gives a response to oxidative stress by binding to an antioxidant-responsive element (ARE) of genes encoding antioxidants. Normally, Nrf2 is found in the cytoplasm, binding with its inhibitor called kelch-like ECH-associated protein 1 (Keap1). This results in degradation of Nrf2 by the ubiquitin-proteasome pathway. 83 However, overexpression of DJ-1in MM prevents the association of Nrf2 protein with Keap1 and eliminate the presence of Nrf2/Keap1 complexes in the cytoplasm. 15 This leads to the translocation of Nrf2 to the nucleus to activate its target genes through binding to ARE.26,83 After Nrf2 protein is translocated to the nucleus, it forms heterodimers with other transcription regulators, such as small musculoaponeurotic fibrosarcoma (Maf) proteins. This induces the expression of different antioxidant genes, including GSH and the Trx system (Figure 2(b)).7,15,16,22,26 Thus, DJ-1 is crucial for Nrf2 stabilization by affecting Nrf2 association with Keap1, leading to the production of several antioxidants, which may have their own role in the development of myeloma. 84

Induction of antioxidant expression by DJ-1 via the Nrf2 pathway. (a) Under normal conditions, Nrf2 binds to Keap1 and is degraded through ubiquitination by proteasomes. (b) However, during oxidative stress, DJ-1 inhibits the interaction of Nrf2 with Keap1 result in activation of Nrf2. Then, activated Nrf2 translocates to the nucleus and binds with the ARE element in the antioxidant gene promoter region to induce its expression.
Overall, the DJ-1 protein participates in the regulation of transcription factors, playing an important role in cell signaling pathways and production of antioxidants, participating in the pathogenesis and development of MM.14,26 Overexpression of these antioxidant molecules in MM may offer an advantage to myeloma cells by eliminating the detrimental effects of ROS. 15 Thus, DJ-1 protein may serve as a potential prognostic biomarker in MM. 26
The GSH system
Although GSH is crucial for the removal and detoxification of carcinogens in healthy cells, its elevated level in MM is associated with myeloma progression, chemoresistance, and poor prognosis of myeloma patients. 59 A study on human MM cell lines demonstrated that the addition of GSH prevents cell cycle arrest after treatment with melphalan and increases resistance to high doses of melphalan. 17 The molecule efficiently blocks melphalan-induced myeloma cell death in patients. Addition of GSH melphalan increases myeloma cell lines’ survival compared to control. 60 GSH protects CD138+ cells from melphalan-induced cytotoxicity by scavenging ROS molecules as well as by inhibiting caspase 3/7 induced cell death. 17 GSH contains a very reactive thiol group (-SH) acting as a ROS scavenger.9,17,22 It also completely abrogated lipid peroxidation induced by melphalan and reduced the level of ROS. 17
The GPx, Grx, and GST contribute to ROS detoxification by reducing hydrogen peroxide or hydroperoxide using GSH as a substrate. 30 GST is also involved in increasing myeloma cell proliferation through the induction of angiogenesis via the regulation of HIF-1α.74,85,86 GSTP1 is an important enzyme having a great function in myeloma cell tumorigenesis and ROS detoxification. 62 Higher levels of GSTP1 are associated with MM clinical stage and shorter overall survival time of patients with MM. 61 Furthermore, upregulation of H2O2-detoxifying enzyme, GPx-1, in MM patients is linked to anti-myeloma drug resistance. Myeloma cells that are resistant to BTZ showed increased GPx-1 activity compared to their drug naïve counterparts. 9 Overall, these findings demonstrated that GSH, GST, and GPx may function as novel biomarkers in predicting MM prognosis and its drug response.
The Trx system
The Trx is involved in multiple cellular pathways of myeloma cell survival and proliferation.16,30,31 The Trx system maintains intracellular redox homeostasis by directly scavenging intracellular ROS or by regulating various redox enzymes. 28 The transcription factor ASK1 continuously forms an inactive complex with reduced Trx1 in resting cells. 78 The reduced form of Trx1 inhibits ASK1 by binding to its N-terminal region. Thus, high Trx1 levels lead to inhibition of cancer cell apoptosis by inhibiting ASK-1 (Figure 1). 87 Studies showed that malignancies, including MM, have high Trx and TrxR expressions that correlate with increased cell proliferation, growth, and chemoresistance.7,16,20,31,50,88
Furthermore, higher TrxR1 gene expression is associated with high-risk disease, adverse overall survival, and decreased responsiveness to the treatment.7,28 Patients having higher TrxR1 expression are at a higher death risk compared to patients with low TrxR1 expression. 7 A MM xenograft mouse model study indicated that overexpression of TrxR2 decreased oxidative stress production by PIs and reduced MM cell death by approximately 50%. 66 The Trx system performs cytoprotection by either directly scavenging ROS or by regulating enzymes and redox-sensitive transcription factors that promote cancer cell proliferation. 16 The Trx system downregulates the integrin very late activation antigen-4 (VLA-4), responsible for cell adhesion-mediated drug resistance in MM by regulating the DNA binding capacity of NF-κB. 31 This enables NF-κB binding to its target genes involved in angiogenesis, cell differentiation, and proliferation, as well as drug resistance. 50 The Trx1 is also able to control HIF-1α activity and induce the expression of hypoxia-responsive genes, including VEGF and nitric oxide synthase 2, for angiogenesis in cancer cells. 89
Superoxide dismutase
SOD is an important part of the free-radical scavenger system that functions as the first line of defense against oxidative damage by catalyzing the dismutation of O2−, producing H2O2 and O2. 15 Increased gene expression of SOD correlated with cancer progression, high-risk disease, and drug resistance. Myeloma patients having increased SOD1 expression have shorter overall and event-free survival compared to patients with lower SOD1 levels. The risk of death for MM patients with high SOD1 levels is higher compared to patients with low levels. The risk of a progression event occurrence for MM patients with high SOD1 levels is greater than for patients with low levels. 9
Vitamin C
The relationship between vitamin C and tumor progression is complex and controversial. Vitamin C can have both “anticancer” and “pro-cancer” effects. 90 A study in vitro using myeloma cell lines found that vitamin C could reverse the cell death caused by BTZ and suppress the drug’s ability to block proteasome function. The study also demonstrated that vitamin C, at orally achievable concentrations using a xenograft mouse model of human MM, significantly reduces the activity of BTZ treatment. 91 This finding suggested that vitamin C may have a negative effect on BTZ-mediated anticancer activity.
In contrast, ascorbate is a cofactor for a group of enzymes called hydroxylases that are involved in regulation of HIF1. 92 The lack of this necessary cofactors (ascorbate) interferes with the activity of hydroxylases, leading to increased stabilization and activation of HIF1. 93 HIF1 regulates the transcription of different genes coding for proteins involved in various aspects of cancer biology, including cell immortality, angiogenesis, or resistance to treatment. 94 This fact indicates that a lack of vitamin C supplement may result in poor prognosis of cancer patients, including those with MM.
In addition, vitamin C (ascorbate) regulates the demethylation of DNA and histones. DNA demethylation favors the expression of tumor suppressor genes and improve chemo-sensitivity. 95 Ten-eleven translocation (TET) dioxygenases initiate DNA demethylation by converting 5-methylcytosine (5mC) into 5-hydroxymethylcytosine (5hmC). Vitamin C is essential for the function of TET by providing Fe (II), a cofactor of TET. Loss of 5hmC is accompanied by malignant cellular transformation. 92 This suggested that vitamin C shortage may worsen TET function and increase cancer cell survival and chemoresistance. 95
Therapeutic role
Despite the developments of different therapeutic strategies for MM, including chemotherapy, radiotherapy, and stem cell transplantation, the disease prognosis is still poor and remains incurable.61,96 This highlights the need for novel therapy approaches to improve the overall survival of MM patients. Today, the discovery of anticancer drugs targeting redox signaling pathways has become a hot research field in cancer. 87 Myeloma cells display high expression of antioxidant molecules that may correlate with drug resistance. 15 Focusing on developing therapies that target the antioxidant systems may be a promising area to reduce the rate of relapsed/refractory MM and to enhance chemo-sensitivity of myeloma cells.
DJ-1 protein
DJ-1 plays a significant role in chemoresistance by eliminating the intracellular ROS produced in response to drug treatment and therefore provides cytoprotection. 26 Targeted knockdown of DJ-1 induced myeloma cell necroptosis and resensitized chemo-resistant cells to anti-myeloma drugs.24,28,83 Inhibition of DJ-1 using small interfering RNA (siRNA) induced necroptosis in myeloma cells. 22 Inhibition of DJ-1 also overcomes acquired BTZ resistance via upregulation of KLF6. DJ-1 reduction by siRNA significantly increased KLF6 levels in MM cells and induced their apoptosis. 28 Furthermore, inhibition of DJ-1 reduces the growth of myeloma cells residing under a hypoxic microenvironment and overcomes hypoxia-induced BTZ resistance. 22 A study on human cell lines reported that inhibition of constitutively expressed Nrf2 reduced MM cell viability. 97 Thus, DJ-1 may be considered as a possible therapeutic target in newly diagnosed as well as relapsed/drug-resistant MM patients. Targeted inhibition of DJ-1 in combination with chemotherapeutic agents also enhances cell apoptosis in acute leukemia. 55
The GSH system
In cancer cells, including hematological malignancies, increased GSH levels have been correlated with chemo- and radio-resistance. Enhancing the production of ROS within mitochondria with simultaneous inhibition of GSH production leads to oxidative stress and the induction of apoptosis of myeloma cells. 98 Depletion of GSH can also reverse these resistances and improve the outcome of cancer patients. 70 Addition of GSH to melphalan in cells from myeloma patients increased the number of CD138+ cells compared to GSH-untreated myeloma cells. This showed that GSH treatment protected myeloma cells from melphalan-induced toxicity resulting in drug resistance. 17 Reducing the overexpression of intracellular GSH in myeloma cells by preventing GSH synthesis inhibited myeloma cell proliferation and enhanced their response to chemotherapies.99,100
Manipulation of GSH metabolism with buthionine sulfoximine (inhibitor of GCL) sensitizes MM cell lines to BTZ. Combined treatment of myeloma cells with BTZ and buthionine sulfoximine reduced myeloma cell lines viability. 12
Betaine-based deprivation of glycine, one of the amino acids for GSH synthesis, inhibited the proliferation of myeloma cells and enhanced the anticancer effects of BTZ both in vivo and in vitro. 63 This indicates that targeting GSH and its synthesis pathways reduces the production of GSH level which exposes tumor cells to ROS-induced death.
Diallyl disulfide (DADS), a major organosulfur compound of garlic, disrupts intracellular redox homeostasis by decreasing GSH levels and increasing ROS production. DADS reduces intracellular GSH levels by targeting GST, complexes II–IV in the electron transport chain, and the mitochondrial ATP-synthase, leading to an increase in ROS levels. The GSH level is significantly reduced by the addition of DADS in myeloma cells, and induces their apoptosis. 101 A combination of DADS and melphalan showed a more synergistic anti-MM effect compared to a single drug dose. Apoptosis-related proteins analysis using western blotting revealed that DADS enhances the cleavage of caspase 9 and caspase 3 while reducing the expression of the anti-apoptotic protein Bcl-2. 101 Previous studies also reported that DADS has an antitumor effect in solid cancers, including breast cancer and colon cancer.102,103
Similarly, diallyl trisulfide (DATS), another natural organosulfur compound found in garlic, widely used in China to stimulate the immune system, showed a comparable effect on MM cell lines with BTZ. DATS reduced myeloma cell survival rate in a dose- and time-dependent manner. It increased the percentage of cells in the G1-phase significantly, preventing their transition from the G1- to the S-phase. 104 Note that the combination of chemical agents obtained from garlic with chemotherapies reduce the dose of anti-myeloma drugs needed. This is highly beneficial since high doses of anti-myeloma drugs are very toxic not only to tumor cells but also to normal cells, such as hematopoietic stem cells. 101 Therefore, the use of these natural products in combination with anti-myeloma chemotherapies may be a novel, promising therapeutic regimens for MM.
DATS also showed a promising pro-apoptotic effect on human lung cancer cells by inducing the expression of Bax and Bak proteins and inhibiting the anti-apoptotic proteins Bcl-2 and Bcl-xL protein expression. 105
Pretreatment of MM cell lines with medical food enriched with fish oil (eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA)), called Omega-3 polyunsaturated fatty acids, before BTZ activates GSH degradation and induces apoptosis of BTZ-resistant MM cells by ROS accumulation within cells. GSH degradation by DHA/EPA may be triggered by the expression of cation transport regulator-like protein 1 (CHAC1). CHAC1 is a family of enzymes responsible for the degradation of intracellular GSH in response to different types of stress. DHA/EPA selectively killed patient-derived primary MM cells but showed almost no effect on normal plasma cells. 106
EPA and DHA are also involved in the generation of lipid peroxidation products such as malondialdehyde, resulting in oxidative stress within the cell. Cell death is also mediated by the activation of caspase-3 and by altering the gene expression of NF–κB. 107 However, simultaneous treatment of MM cells with DHA/EPA and BTZ had no anti-myeloma effect. These fatty acids may impede drug influx in MM cells when they are used concomitantly with chemotherapeutic drugs. Additionally, since DHA/EPA can affect cell membranes, these fatty acids may interfere with the uptake of BTZ in MM cells. Therefore, the anticancer activity of DHA/EPA in MM cells depends on the timing of the treatment.106,107 These findings highlight the potential use of DHA/EPA as adjuvants for combating drug resistance in MM.
The Trx system
As discussed earlier, increased expression of antioxidants in the Trx system is related to decreased responsiveness to myeloma treatment. A study on human myeloma cell lines revealed that BTZ-resistant cells contained higher Trx1 protein levels compared to BTZ-sensitive myeloma cells. 31 Trx plays a critical role in BTZ resistance through increased mTOR and ERK1/2 phosphorylation. 108 These findings allow investigators to focus on developing therapies that inhibit Trx. Inhibition of Trx using PX-12 sensitizes MM cells to undergo cell death in response to NF-κβ inhibition and induces apoptosis of BTZ-resistant MM cells. 31 PX-12 treatment decreased the expression of the NF-κβ subunit p65 in MM cells. Addition of PX-12 to myeloma cells significantly increased caspase-3 activity in myeloma cells compared to nontreated MM cells. 31 PX-12 is a commercially available Trx-specific inhibitor that binds irreversibly to the Cys73 residue of Trx, causing it to become biologically inactive. 108 In addition, genetic inhibition of Trx using shRNA-specific gene knockdown techniques sensitizes BTZ-resistant myeloma cells to BTZ. 108 Inhibition of Trx also induces mitophagy in BTZ-resistant myeloma cells. Mitophagy is a mitochondrial autophagy that can act as a tumor-suppressive mechanism through initiation of cell death or senescence. 108
Moreover, overexpression of TrxR in MM cells suppresses BTZ-induced cell death efficiently. 66 Thus, developing new anti-myeloma therapies that can efficiently eradicate TrxR-based drug-resistant myeloma cells is needed. A study on the MM xenograft mouse model found that treatment with only BTZ decreased tumor volume by around 43% compared to their controls. However, combined treatment of MM-vector xenografts with TrxR2 and BTZ decreased the average volume of MM cells only by approximately 16%. 66 Gold compounds have a high affinity for thiol groups, which makes the Trx system vulnerable to these compounds. 50
A study on human MM cell model indicated that TrxR1 inhibition using auranofin (an FDA-approved gold-based drug for arthritis) overcomes hypoxia-induced BTZ resistance by inhibiting hypoxia-induced nuclear accumulation of NF-κβ p65. 7 Another study found that treatment of MM cells with 2 μM auranofin significantly increased caspase-3 activity by 2.3- to 2.5-fold in myeloma cells compared to parental cells. 31 The use of 4 μM auranofin significantly reduced the proliferation of hypoxic-myeloma cells by approximately 85% after 24 h treatment. Furthermore, higher caspase-3 and 9 activities are observed when cells are treated with auranofin and BTZ together compared to the treatment with auranofin or BTZ alone. Fortunately, cotreatment with auranofin and BTZ significantly reduced myeloma cell proliferation without a significant cytotoxic effect on normal plasma cells. 7 This may be related to the fact that the susceptibility of ROS damage-related cell death becomes more apparent in cancer cells since these cells have a narrower margin in reaching the maximum cytotoxicity threshold as compared to normal cells when treated with an anticancer drug.16,28
Furthermore, the use of another improved gold complex compound, [Au (d2pype)2]Cl inhibits myeloma cell proliferation and induces apoptosis in myeloma cells via TrxR inhibition. [Au (d2pype)2]Cl downregulated the expression of the MYC oncogene, which drives myeloma progression both in vitro and vivo. 50 The compound also significantly increased caspase-3 activity in myeloma cell lines and induced apoptosis in BTZ-resistant tumor cells. 16 Inhibiting either Trx or TrxR reduces clonogenic activity of myeloma cells. A significantly decreased number of colonies was seen in MM cell lines after treating with the above agents. 31 These findings suggest that Trx and TrxR systems may serve as one of the therapeutic targets for MM without causing much collateral damage to surrounding normal cells. 16
Superoxide dismutase
Investigators also proposed that BTZ resistance in MM is associated with adaptation to oxidative stress via upregulation of the SOD antioxidant system. 9 Targeting SOD may induce MM cell killing by increasing their susceptibility to chemotherapies and re-sensitize BTZ-resistant myeloma cells. A study on human MM cell lines reported that a copper chelating agent and inhibitor of SOD1, disulfiram (DSF), showed anti-myeloma effect. The study showed that DSF reversed BTZ resistance and increased BTZ cytotoxicity in MM cells. 9 In addition, SOD2 depletion with 2-methoxyestradiol induced MM cell death. A study on human MM cell models examined the combined effect of 2-methoxyestradiol and BTZ in the killing of MM cells. The study found that 2-methoxyestradiol elicited significant cytotoxicity (15%–25%) as a single agent, but the combination of both agents induced apoptosis more than twofold greater than by each compound alone. 109 Thus, inclusion of DSF and 2-methoxyestradiol with other frontline anti-MM drugs augments their cytotoxic effects by inhibition of SOD and may have promising importance in MM treatment.
Note that targeting DJ-1 and other antioxidants may offer therapeutic benefits in MM by overcoming drug resistance and reducing tumor cell survival. However, these molecules also play essential roles in maintaining redox homeostasis in normal cells. Systemic inhibition could therefore disrupt physiological antioxidant defenses, potentially leading to severe adverse effects (oxidative stress). Consequently, therapeutic strategies aimed at these targets require careful design to maximize anti-myeloma efficacy while minimizing toxicity, such as selective targeting of tumor cells or transient modulation of antioxidant activity.
Vitamin C
Vitamin C treatment of cancer patients initially conducted by Pauling and Cameron 110 showed conflicting results with regard to efficacy. There is a number of evidence suggesting vitamin C-rich foods play a protective role against the development of cancer due to its antioxidant properties, as it protects DNA from free radicals. 111 Reduced vitamin C levels within cells may promote the transformation of healthy cells into leukemia cells. 112 A study in vitro, determining the effect of vitamin C on the growth of MM cell lines, concluded that vitamin C at physiological concentrations blocks BTZ anti-myeloma effect. The study also evaluated the inhibitory effect of vitamin C on BTZ in vivo of xenograft mouse model of human MM. The study revealed that oral administration of vitamin C antagonized anti-MM activity of BTZ and suggested that patients receiving treatment with BTZ should avoid taking vitamin C dietary supplements. Interestingly, the inhibitory effect of vitamin C was restricted to PIs classes containing a peptide boronic acid, including BTZ and MG262. 91 Vitamin C abrogates the anticancer activity of BTZ through a direct binding between the hydroxyl group of vitamin C and the boronic acid of the BTZ. 113 Thus, it is necessary to consider the negative effect of vitamin C on the anticancer activity of BTZ when BTZ is used in the clinic for myeloma treatment. 113
However, different studies demonstrated that high-dose intravenous administration of vitamin C may improve leukemia patients’ health-related quality of life. 114 Vitamin C inhibits cellular signaling and enhances autophagy, which might decrease leukemic cell survival and increase sensitivity to conventional chemotherapy. 115 Ascorbate causes H2O2 dependent cytotoxicity in lymphoma cells without deleteriously affecting healthy cells. 95 The Linus Pauling Institute’s 9th International Conference on Diet and Optimum Health took place in September 2017 in Corvallis, OR, USA reported that pharmacologic intravenously administered vitamin C, but not oral vitamin C, had the potential to decrease cancer growth in humans. The report suggested that pharmacologic ascorbate is cytotoxic to cancer, but not to normal cells in vitro, both in animals and humans studies. 92 Ascorbate can selectively kill cancer cells by acting as a pro-oxidant by generating high fluxes of H2O2 that can react with intracellular labile iron via Fenton chemistry, while it is believed to simultaneously protect normal tissue by acting as a donor antioxidant. 116 The cell death depends only on extracellular but not intracellular vitamin C, through the formation of extracellular H2O2 with ascorbate radical as an intermediate.41,92 Similarly, pharmacologic ascorbate has an additive or synergistic promise with other therapies in myeloma. 45 When vitamin C is pharmacologically dosed in the presence of iron, it produces a high ROS concentration that ultimately causes myeloma cell death. A multicenter, open-label, phase I/II trial study indicated that combined use of As2O3/BTZ/vitamin C (ABC) in patients with relapsed/refractory MM showed preliminary evidence of efficacy with an objective response rate of 27%, and was well-tolerated by most patients. 117 Similarly, a multicenter phase II study to evaluate the safety and efficacy of steroid-free combination treatment with melphalan, As2O3, and vitamin C (MAC) for patients with relapsed or refractory MM showed that the MAC regimen is active and well-tolerated even in a group of patients who had either relapsed or refractory MM to standard and/or investigational MM treatments. 118
Another study using the MM xenograft mouse model demonstrated that intravenous administration of vitamin C increased myeloma cells’ sensitivity to melphalan, suggesting a synergistic activity. The study revealed that the combined use of vitamin C and melphalan may significantly reduce the dose of melphalan needed. This is beneficial since high doses of melphalan are very toxic not only to tumor cells but also to normal tissues, such as hematopoietic stem cells. However, the therapeutic effect of pharmacological AA depends on cellular iron and ROS. 37 High cytosolic iron would catalyze pharmacological AA autooxidation, leading to cell death. The AA anti-myeloma effect is dependent on LIP because MM tumor cells have a higher LIP than non-tumor cells. 37 Pharmacological dosed vitamin C induced early necrosis and then late apoptosis of myeloma cells. It induced mitochondria-mediated apoptosis with a marked increase in caspases 3, 8, and 9 activity. 113 Using vitamin C may also help to manage the negative effects of BTZ-induced peripheral neuropathy since vitamin C almost entirely rescues the Schwann cells. 119 These and related findings indicated that vitamin C at pharmacologic concentrations administered intravenously may be a pro-oxidative drug mainly depending on LIP for the delivery of H2O2, revealing its therapeutic implication in MM (Table 2).
Summary on the role of common antioxidants in MM.
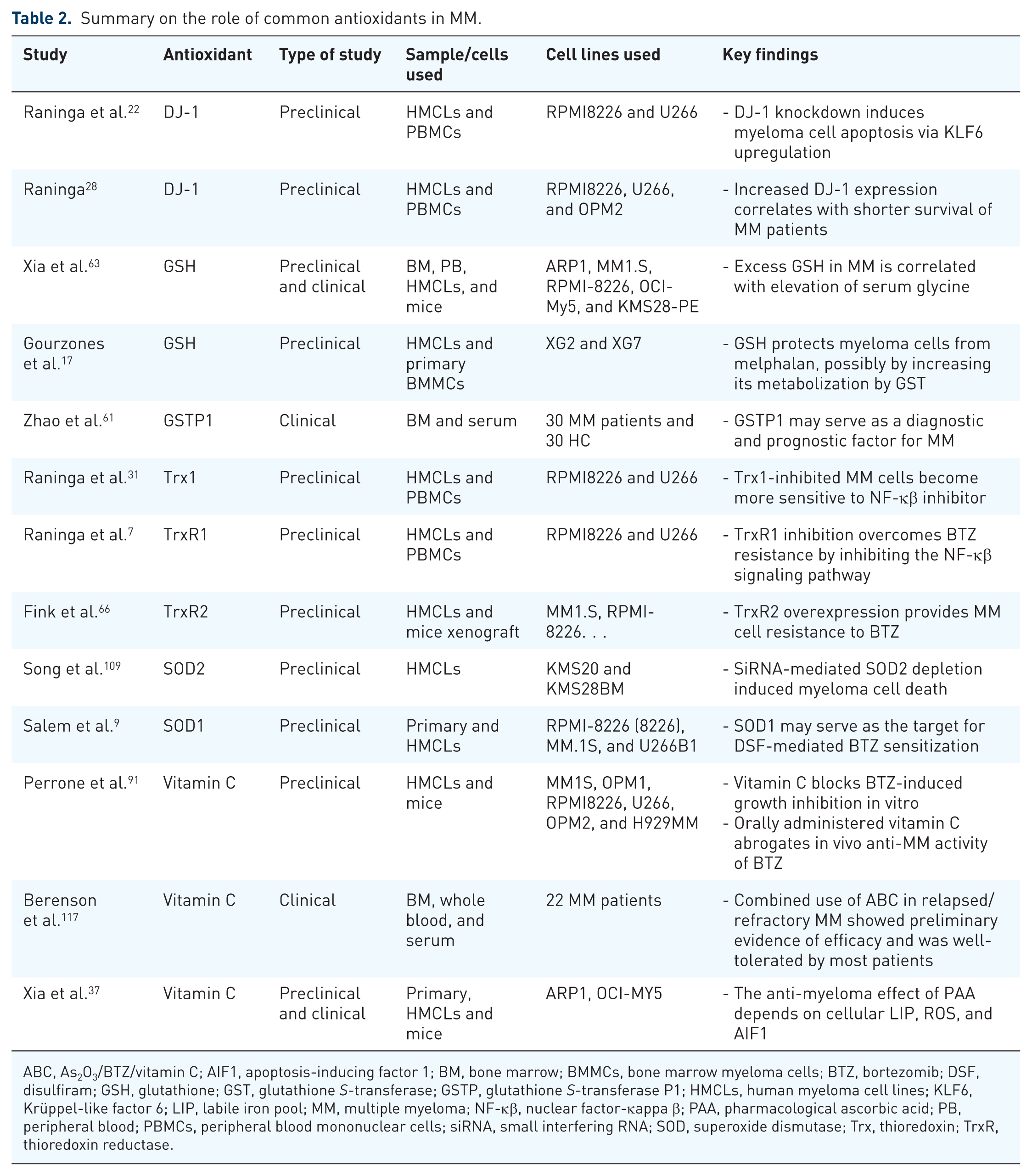
ABC, As2O3/BTZ/vitamin C; AIF1, apoptosis-inducing factor 1; BM, bone marrow; BMMCs, bone marrow myeloma cells; BTZ, bortezomib; DSF, disulfiram; GSH, glutathione; GST, glutathione S-transferase; GSTP, glutathione S-transferase P1; HMCLs, human myeloma cell lines; KLF6, Krüppel-like factor 6; LIP, labile iron pool; MM, multiple myeloma; NF-κβ, nuclear factor-κappa β; PAA, pharmacological ascorbic acid; PB, peripheral blood; PBMCs, peripheral blood mononuclear cells; siRNA, small interfering RNA; SOD, superoxide dismutase; Trx, thioredoxin; TrxR, thioredoxin reductase.
Limitation of the study
The main limitation of this review is that most available evidence on antioxidant systems in MM originates from in vitro cell line experiments, preclinical models, or small comparative studies between MM patients and healthy individuals. Robust clinical trials evaluating the diagnostic, prognostic, or therapeutic value of antioxidants in MM remain lacking. This gap underscores the need for future large-scale, well-designed clinical investigations to validate the clinical relevance of antioxidant systems in MM management. Another limitation of this review is the lack of studies assessing the diagnostic accuracy of individual antioxidants in MM, which limits the ability to draw definitive conclusions about their diagnostic performance.
Conclusion and future perspectives
MM remains incurable and leads to death mainly due to the chemoresistance of myeloma cells. The current review provides an important insight into the cytoprotective role of antioxidants in MM. Antioxidants such as GSH, Trx, TrxR, and SOD participate in myeloma cell survival, growth, and chemoresistance. An increased level of antioxidants is found in MM patients compared to healthy controls. This overexpression of antioxidants in MM patients is associated with poor prognosis, cancer cell survival, proliferation, and resistance to their treatment. These findings provide justification for the development of new diagnostic, prognostic, and therapeutic approaches. Using antioxidant-inhibiting drugs either alone or in combination may reverse chemoresistance myeloma cells to improve the survival of MM patients. In addition, natural antioxidant-modulating compounds derived from sources such as garlic and fish oil may enhance the efficacy of conventional anti-myeloma therapies. However, therapeutic strategies aimed at targeting antioxidant systems require careful design to maximize anti-myeloma efficacy while preserving redox balance in normal cells. While preclinical data suggest potential prognostic and therapeutic roles, the safety and efficacy of targeting these molecules in clinical settings remain to be established. Therefore, further well-designed clinical studies are needed to validate these findings and determine their clinical utility. Furthermore, we highlighted that future research should focus on combining antioxidants with other diagnostic markers to improve the specificity and clinical applicability of these antioxidants for MM.
The role of Vitamin C in MM is a “double-edged sword.” On one hand, pharmacological (ultra-high) doses administered intravenously act as a pro-oxidant that can selectively kill MM cells by exploiting their high iron content and inducing mitochondrial apoptosis. On the other hand, dietary or supplemental vitamin C can chemically neutralize boronate-based PI, such as BTZ, significantly reducing their therapeutic efficacy. This highlights the critical importance of distinguishing between the chemical form, dose, and administration route of vitamin C in the clinical setting.
