Abstract
Background:
Hemophilia A (HA) management remains challenging in northeastern India due to limited access to specialized care. Emicizumab, a bispecific monoclonal antibody, offers prophylactic benefits for patients with or without factor VIII (FVIII) inhibitors.
Objectives:
The study aimed to assess the long-term effectiveness, safety, and patient-reported outcomes of emicizumab prophylaxis in people with HA with or without inhibitors.
Design:
This 12-month follow-up from a noninterventional, real-world observational study builds on previously published 6-month data and was conducted at the Department of General Medicine, Assam Medical College and Hospital, India.
Methods:
Forty pediatric patients with moderate-to-severe HA, including those with inhibitors, receiving emicizumab prophylaxis were followed at a treatment center. The outcomes assessed at baseline, 6 months, and 12 months included annualized bleeding rate (ABR), Hemophilia Joint Health Score (HJHS), Functional Independence Score in Hemophilia (FISH), European QoL-5 dimensional-5 level (EQ-5D-5L) score and visual analog scale (VAS). Subgroup analyses explored age- and inhibitor-status based differences. Data were analyzed using appropriate statistical tests to compare outcomes across time points and subgroups.
Results:
The mean ABR reduced significantly from 11.48 at baseline to 0.05 at 12 months (
Conclusion:
This 12-month follow-up study from northeastern India confirms the sustained efficacy and safety of emicizumab in reducing bleeds and improving outcomes in children with HA. The findings underscore its value in underserved settings, supporting broader implementation in similar healthcare environments.
Plain language summary
Hemophilia A (HA) is a genetic condition where the blood cannot clot properly due to very low or missing levels of clotting factor VIII (FVIII). This causes frequent bleeding, especially into joints, leading to pain and long-term joint damage. Treatment usually involves replacing FVIII. However, about 30% of people with severe HA develop antibodies called inhibitors, which block the treatment and make it harder to stop bleeding. Emicizumab is a laboratory-made monoclonal antibody that helps blood to clot by mimicking the action of FVIII. It is given as a regular injection under the skin and is effective even in people who have developed inhibitors. Emicizumab has been shown to reduce bleeding episodes in clinical trials, but there is limited information on how well it works in real-life healthcare settings, particularly in areas with fewer resources. In this study, 40 children with moderate to severe HA were treated with emicizumab at a hemophilia treatment center in northeastern India. All participants were followed for 12 months to assess the treatment’s effectiveness and safety. The number of bleeding episodes dropped significantly, from an average of approximately 11 bleeds per year at the beginning of the study to nearly none after 12 months. Joint bleeds were completely eliminated. Children experienced better joint health, increased independence in daily activities, and an improved quality of life. These benefits were seen in all children, regardless of age or inhibitor status. The treatment was well tolerated, with no serious side effects reported. This 12-month follow-up study builds on previously published results from the same group of children observed over 6 months. The findings confirm that emicizumab continues to be a safe and effective treatment for preventing bleeds in children with HA. The results support broader use of emicizumab, especially in areas with limited access to specialized hemophilia care.
Introduction
Hemophilia is an X-linked genetic bleeding disorder caused by defects in the F8 or F9 gene that lead to a deficiency of factor VIII (FVIII) or IX. 1 Most hemophilia bleeds occur in joints and muscles, leading to musculoskeletal changes caused due to hemophilic arthropathy. 2 Hemophilia A (HA), the most common X-linked bleeding disorder, affects 1 in 5000 men and accounts for 80% of all hemophilia cases. Globally, it affects over 400,000 men, with many undiagnosed in developing countries. 3 India is estimated to have 80,000–100,000 individuals with severe HA, yet only about 19,000 are registered with the Hemophilia Federation of India. 4
The primary goal of hemophilia treatment is to prevent or manage bleeding, traditionally through on-demand or prophylactic FVIII replacement. Enhancing health-related quality of life (HRQoL) is also a critical objective. 5 However, challenges with FVIII concentrates include diminishing drug efficacy, difficulties with administration and adherence, breakthrough bleeding, venous access issues, and the development of inhibitors. 4
Alloantibodies that inhibit FVIII develop in approximately 30% of individuals with severe HA, usually within the first 20 exposures to FVIII in early life. These inhibitors reduce the effectiveness of FVIII replacement, increase bleeding risk, and require alternative hemostatic strategies. 6 The development of inhibitors significantly increases treatment costs and reduces QoL, particularly in low- and middle-income countries (LMICs), where access to standard treatments such as immune tolerance induction, bypassing agents, or nonfactor replacement therapies is limited. 7
Recently, nonreplacement therapies have emerged as promising alternatives. Emicizumab is a novel nonfactor therapy and the first in its class approved for prophylaxis in patients with hemophilia A (PwHA), with or without inhibitors. 8 As a FVIII mimetic, it improves hemostasis by partially correcting the coagulation defect, by replicating the cofactor function of activated FVIII. 9 Its subcutaneous administration has improved adherence, with safety and efficacy profiles comparable to those of recombinant and plasma-derived FVIII concentrates in HA. 10 Since April 2019, it has been available in India for managing HA, with or without FVIII inhibitors, and is suitable for patients of all ages.4,11
Access to specialized hemophilia care remains limited in northeastern India, where many patients live in remote areas, and the burden of HA is often underrecognized. A recent study from northeastern India assessing hemophilia prevalence and clinical features, confirmed HA as the most common inherited bleeding disorder, with severe cases predominating. Limited diagnostic resources and lifelong care remain major challenges in this region’s rural areas. 12
Given the limited data on long-term real-world outcomes of emicizumab in Indian patients, especially in resource-constrained regions like northeastern India, this 12-month follow-up study builds upon previously published 6-month data. 13 This study aimed to evaluate the sustained effectiveness, safety, and patient-reported outcomes of emicizumab prophylaxis in pediatric PwHA, with or without inhibitors, in a real-world setting. By capturing the clinical, functional, and QoL parameters over time, this study sought to generate context-specific insights to support ongoing use and optimization of emicizumab in resource-limited regions.
Methods
Study design and setting
This hospital-based, observational study, designed as a noninterventional investigation, represents the second follow-up phase carried out at the Department of General Medicine, Assam Medical College and Hospital in northeastern India. 13 The collection of study-specific data was prospective during the follow-up phase, while baseline demographic and clinical details were obtained retrospectively from existing patient records. The current phase extended from June 20, 2024 to December 20, 2024. As patient records and treatment documentation were maintained continuously from the initial enrollment period (October 19, 2023 to November 19, 2023), all 40 participants previously recruited were eligible for inclusion in this extended follow-up.
Study population
All participants previously enrolled in the initial 6-month observational study were included in the extended follow-up. Due to the rare nature of the condition, sampling remained clinically driven, with a total of 40 PwHA continuing in the follow-up phase. Written informed consent was obtained from the parents or guardians in the local language and assent was secured from adolescents aged 7 years and above, as done previously. 13
Data collection
Demographic data, baseline clinical history, and hemophilia-related characteristics were obtained during the original study and updated periodically throughout the follow-up. Ongoing assessments included physical examinations, treatment adherence reviews, and documentation of any new or continuing clinical findings. Procedures for clinical evaluation mirrored those used in the initial phase to ensure comparability.
Treatment protocol
The dosing regimen for emicizumab remained unchanged: patients received 3 mg/kg weekly for the first 4 weeks, followed by 6 mg/kg once every 4 weeks. The initial induction doses had been administered under medical supervision, during which parents and caregivers were trained in proper self-administration techniques. Home-based administration continued during the follow-up period for those deemed competent, with monthly in-clinic reviews to confirm adherence and reassess joint and musculoskeletal status.
Dose titrations
Dosing modifications were undertaken only if clinically necessary and in consultation with the medical monitor. The use of anticoagulants or antiplatelets was discouraged unless clinically justified, in which case individualized decisions were made under close supervision.
Follow-up monitoring
Parents or caregivers were contacted via registered phone numbers in the event of missed doses. Each patient maintained a dosing log, which was reviewed during monthly clinic visits. The hemophilia care team remained available throughout the study to respond to patient queries related to treatment, dosage, or side effects.
Evaluation parameters
The following outcomes were assessed at baseline, 6 months, and 12 months:
All assessment tools used were the same as those applied in the initial study and followed standardized administration protocols to preserve consistency in measurement.
Safety surveillance
Patients were continuously monitored for adverse events, including systemic hypersensitivity, injection site reactions, thromboembolic complications, and other emicizumab-related events. All adverse events were recorded, graded, and analyzed based on their frequency, severity, and outcome.
Ethical framework
The study was conducted in accordance with ethical standards and the Declaration of Helsinki. Ethical clearance for the extended follow-up was obtained from the Institutional Ethics Committee of Assam Medical College and Hospital (Approval No. 2023/AMC/EC/10795). All personal health information was anonymized and stored securely, accessible only to authorized research staff. This study is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (Supplemental Material 1).
Data analyses procedures
Analyses were conducted using R version 4.3.2 (R Foundation for Statistical Computing, Vienna, Not applicable, Austria) (General Public License project). Descriptive statistics were calculated to describe participant demographics and clinical profiles, with continuous variables presented as mean ± standard deviation (SD) or median with interquartile range (
Results
Study population demographics
A total of 40 male patients with a median age of 4.5 years (2.35–8.55–15.9) were enrolled, of which 72.5% had severe hemophilia and 27.5% had moderate disease. Inhibitors were present in 30.0% of patients. The majority of the patients (95%) were on low- to moderate-intensity prophylaxis regimens: 10 patients received 15 IU/kg twice weekly, 28 patients received 20 IU/kg thrice weekly, and 1 patient was treated on-demand; the remaining 1 patient had an undetermined treatment status. The mean baseline ABR was 11.50 (SD: 2.67), and the mean number of target joints was 0.83 (SD: 0.55). Although 39 out of 40 patients (97.5%) were recorded as being on FVIII prophylaxis prior to starting emicizumab, The detailed baseline demographic characteristics of the study population are shown in Table 1.
Baseline demographic and clinical characteristics of the study population.

IQR, interquartile range; SD, standard deviation.
Analysis of bleed and joint bleed rates at 6 months and 1 year
At the 6-month and 1-year follow-up, all 40 patients (100.0%) demonstrated improvement in the ABR, annual joint bleed rate, and annual target joint bleed rate. No patients showed no improvement or deterioration in these scores at either time point.
At baseline, the mean ABR was 11.48 (SD: 2.76), with a median of 11.0 (range: 10.0–14.0), which significantly decreased after 1 year of emicizumab treatment to a mean of 0.05 (SD: 0.32) and a median of 0.0 (IQR: 0.00–0.00;
Comparison of ABR, AJBR, and number of target joints at baseline and after 1 year of emicizumab treatment.

ABR, annualized bleeding rate;
At baseline, the median ABR was 11.48 (10.0–14.0). After 1 year, this was reduced to nearly 0 (0.0–0.0), representing a 99.6% reduction in ABR. Similarly, the mean number of target joints decreased from a baseline value (approximately 1) to 0 after 1 year, reflecting a 100.0% reduction (Figure 1).
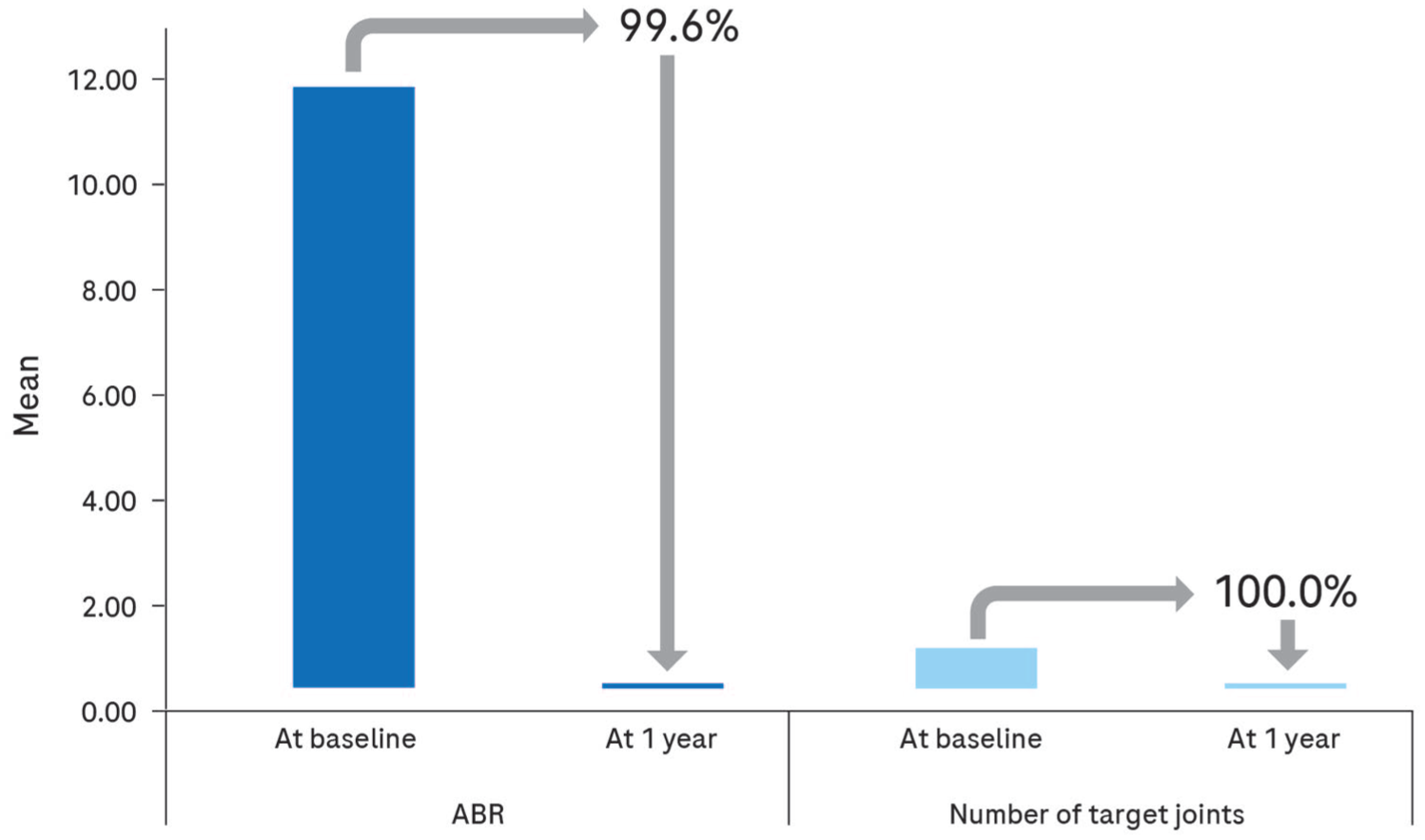
Mean change in ABR and number of target joints from baseline to 1 year of emicizumab prophylaxis.
Bleeding profile/phenotype
At baseline, all patients experienced recurrent bleeding events, with a mean ABR of 11.48. Following emicizumab initiation, 92.5% (
ABR comparison in noninhibitor versus inhibitor groups at baseline and 1 year
At baseline, the mean ABR was 11.32 (SD: 2.88) in the noninhibitor group and 11.83 (SD: 2.55) in the inhibitor group. After 1 year of emicizumab treatment, the noninhibitor group had a mean ABR of 0.00 (SD: 0.00), while the inhibitor group showed a mean ABR of 0.17 (SD: 0.58). This difference was statistically significant (
Comparison of bleeding episodes before and after emicizumab in inhibitor and noninhibitor patients.
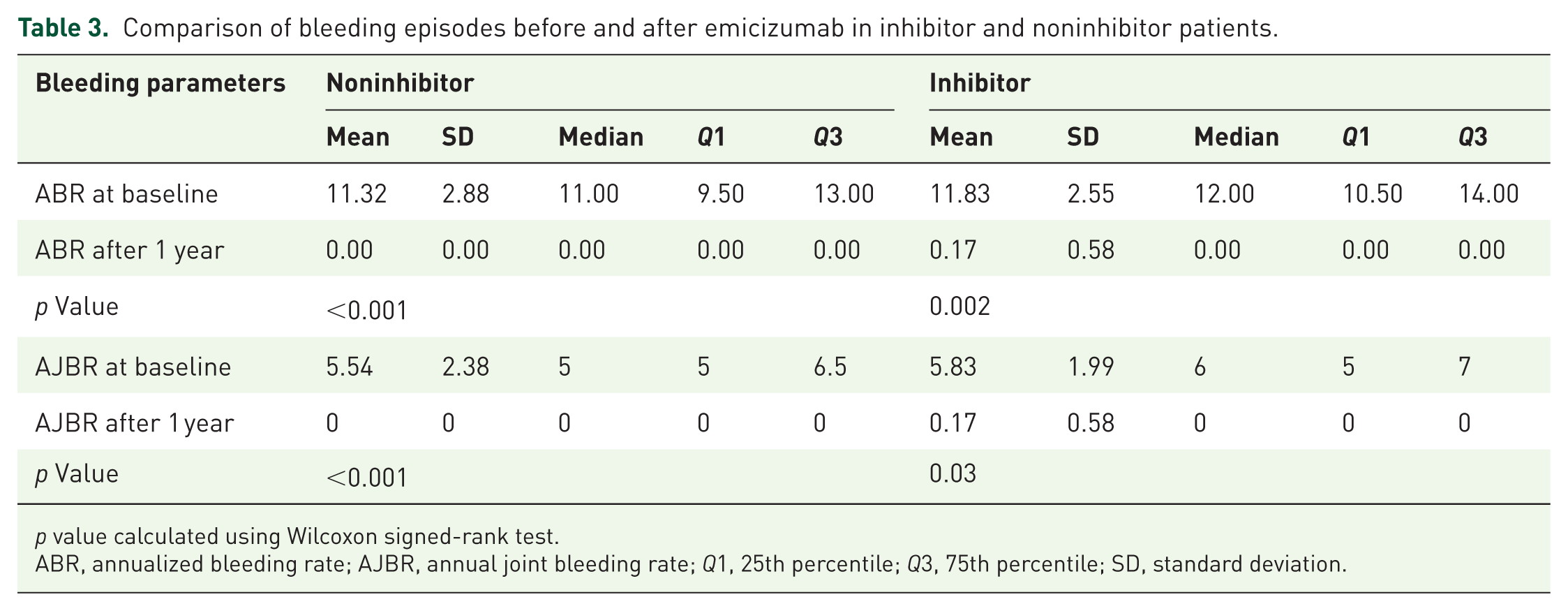
ABR, annualized bleeding rate; AJBR, annual joint bleeding rate;
Reduction in bleeding episodes over 1 year of emicizumab therapy
Following 6 months of emicizumab therapy, 92.5% (

Percentage of patients with different bleeding episodes after 6 months and 1 year of emicizumab therapy.
Reduction in ABR following emicizumab therapy across different age groups
At baseline, the mean ABR was 10.14 in children under 3 years of age and 12.19 in those aged 3 years and above. After 1 year of emicizumab therapy, ABR dropped to 0.14 and 0.00, respectively, with the median ABR at 0.00 in both groups. Reductions were statistically significant within each group (
ABR at baseline and after 1 year of emicizumab treatment, stratified by age group.

ABR, annualized bleeding rate;
Reduction in joint bleed over 1 year of emicizumab therapy
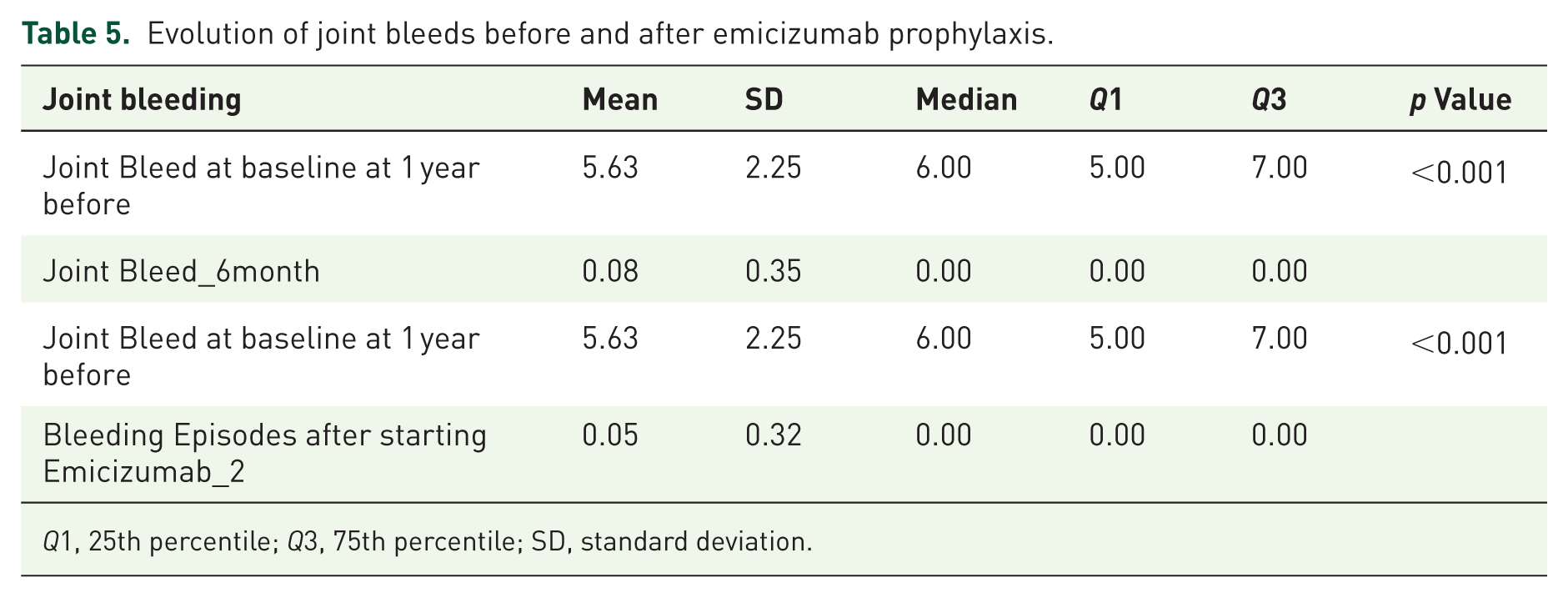
The mean number of joint bleeds significantly decreased from 5.63 ± 2.25 at baseline to 0.08 ± 0.35 at 6 months and 0.05 ± 0.32 at 12 months (
Evolution of joint bleeds before and after emicizumab prophylaxis.
Improvement in HJHS over 1 year of emicizumab therapy
A significant improvement in joint health was observed over time, as measured using the HJHS. At baseline, 50% of patients had HJHS between 11 and 20, with a mean score of 12.0 ± 5.23. By 6 months, the proportion of patients in the 1–10 score range increased markedly from 45.0% to 92.5%, and only one patient (2.5%) remained in the 11–20 range. This improvement was sustained at 1 year, with 90.0% of patients scoring between 1 and 10 (mean score: 4.6 ± 3.13). The change in HJHS from baseline to both 6 months and 1 year was statistically significant (
Improvement in HJHS over 1 year of emicizumab therapy.

HJHS, Hemophilia Joint Health Score; SD, standard deviation.
All patients (100%) showed improvement in HJHS at 6 months and 1 year, with no cases of deterioration or lack of improvement.
Improvement in FISH over 1 year of emicizumab therapy
FISH improved significantly following emicizumab treatment. At baseline, 92.5% of patients had scores between 24 and 32, with a mean score of 27.5 ± 2.11. By 6 months and sustained at 1 year, all patients (100%) achieved scores in the 24–32 range, with the mean increasing to 30.6 ± 1.32 and 30.6 ± 1.39, respectively. This improvement was statistically significant (
Improvement in FISH over 1 year of emicizumab therapy.

FISH, Functional Independence Score in Hemophilia; SD, standard deviation.
An improvement in FISH was observed in all patients (100%) at both 6 months and 1 year, with no instances of score deterioration or lack of improvement.
Improvement in HRQoL score over 1 year of emicizumab therapy
A significant improvement of HRQoL was observed at 6 months, as measured using the EQ-5D-5L index and VAS scores. The mean EQ-5D-5L index significantly increased from 0.794 ± 0.108 at baseline to 0.961 ± 0.044 at 6 months (
Evaluation of EQ-5D-5L and EQ-VAS score changes over 6 months and 1 year of emicizumab therapy.

EQ-5D-5L, European QoL 5-dimensional 5-level; SD, standard deviation; VAS, visual analog score.
Improvement in patient-reported outcomes over time
Patient-reported outcomes improved over 1 year (Figure 3(a)–(d)). Functional status, measured by FISH, increased from median value of 27 at baseline to 31 at 6 months and remained stable. Joint health, assessed by HJHS, improved significantly, with scores decreasing from median value of 12 to 4 at 1 year. QoL also improved, as reflected by the median EQ-5D-5L score rising from 0.80 to 0.95 in 6 months and remaining steady for 1 year. Patient-reported self-health, measured by the VAS score, increased from 70.5 to 94.5 at 6 months and further to 95 at 1 year.

Changes in patient-reported outcome measures from baseline to 6 months and 1 year. (a) Median HJHS indicating a reduction in joint-related complications; (b) median FISH showing increased functional ability; (c) median EQ-5D-5L score showing improvement in health-related quality of life; (d) median VAS score reflecting improved overall health.
Discussion
This 12-month follow-up study reaffirms the safety and effectiveness of emicizumab in PwHA, with or without FVIII inhibitors, within a real-world clinical setting. Building on findings from the previously published 24-week analysis,
13
the current results demonstrate sustained bleed control, reduced need for factor replacement therapy, and a favorable safety profile over a longer treatment duration. The ABR decreased from a mean of 11.48 at baseline to 0.05 after 1 year (
The findings corroborate international research, emphasizing the role of emicizumab in HA. Pivotal trials have demonstrated significant reductions in ABRs, with HAVEN 1 showing an 87% reduction, 17 HAVEN 2 a 99% reduction, 18 and HAVEN 3 a 96%–97% reduction. 19 Further studies confirmed its efficacy in severe HA, with HAVEN 4 reporting a model-based ABR of 2.4 20 and HAVEN 5 showing a 96% reduction in ABRs with no thrombotic events. 21 A retrospective study in pediatric patients showed a decrease in ABR from 4 to 0.5, 22 while HAVEN 6 reported improvements in ABRs and target joint issues in mild/moderate HA patients. 23 In infants with severe congenital HA, HAVEN 7 reported an ABR of 0.4 and a favorable safety profile. 24
It should be noted that in this study, the disproportionately high bleed rates were largely driven by a small subset of heavy bleeders (clinically nonresponders) who experienced recurrent bleeding in multiple joints. This clustering effect inflated the overall ABR, while the actual number of target joints remained relatively low. Moreover, these patients transitioned from low-dose prophylaxis to full-dose emicizumab, which likely contributed to the marked improvement and favorable statistical outcomes observed.
In this study, 72.5% of participants had severe HA, reflecting the proportion of severe cases treated with emicizumab, consistent with other Indian studies reporting a high prevalence of severe HA, such as a recent prospective analysis which found 68% of HA cases to be severe. 12 Most patients (95%) were on prior FVIII prophylaxis, with 10 patients on 15 IU/kg twice weekly and 28 patients on 20 IU/kg thrice weekly; 1 patient was on on-demand therapy. These low- to moderate-intensity regimens, often influenced by cost or adherence challenges, likely contributed to the high baseline ABR, and the improvements observed with emicizumab should be interpreted in this context. Additionally, a recent cross-sectional study showed that inhibitor prevalence was higher in patients with severe HA (52.95%). Complications are more common in those with inhibitors. 25 In patients with severe HA, late diagnosis, lack of prophylaxis, and frequent bleeding episodes significantly reduce work productivity, particularly in LMICs. 26 Long-term treatment effectiveness in PwHA requires assessing both short-term outcomes and sustained benefits of emicizumab. While early improvements in clinical and functional measures may plateau, the persistence of these improvements is key to confirming a treatment’s long-term impact. In this study, the improvements observed in 6 months were sustained through 12 months of follow-up. Although most clinical and functional gains, such as reductions in ABR, target joints, and improvements in FISH, were evident by 6 months, these effects were maintained through 12 months, indicating a sustained benefit over time. The consistency between the 6- and 12-month outcomes highlights the durability of emicizumab’s real-world effectiveness in this population. Interestingly, the proportion of patients with zero bleeds increased from 92.5% at 6 months to 97.5% at 12 months, emphasizing continued clinical stabilization over time.
HRQoL improvements in PwHA are rarely documented, but pivotal studies such as HAVEN 2 and HAVEN 3 demonstrated significant gains in physical health 18 and a 16-point increase in the Hemophilia A QoL (Hem-A-QoL) score. 19 Clinically important respondents’ thresholds across HAVEN 1, 3, and 4 indicated significant improvements in QoL by 24 weeks. 27 Additionally, an Indian study reported that emicizumab reduced bleeding episodes and improved daily functioning across all Pediatric Hemophilia Activities List domains, 28 while a resource-limited country study showed improvements in joint health and functional independence. 29 Other studies have similarly highlighted emicizumab’s positive effects on HRQoL, with improvements in both physical health and work productivity.30 –32 The findings of this study were in line with the above-discussed studies, showing marked improvement at 6 months, sustained at 12 months.
Moreover, the findings of this study align with the World Federation of Hemophilia Guidelines (third edition), which recommend initiating long-term prophylaxis with FVIII or other hemostatic agents before the age of 3 years to prevent joint damage.33,34 In the current study, in the preemicizumab era, FVIII prophylaxis showed an age-related difference in bleeding rates, supporting the rationale for early intervention. However, with emicizumab, this age-related disparity was eliminated, suggesting that emicizumab may offer even more consistent protection than FVIII from infancy onward. This supports its role as a highly effective early prophylactic option, regardless of age at initiation.
Access to emicizumab remains highly inequitable and is closely linked to national income (42.1% in high-income countries vs 7.5% in low-income countries). The World Federation of Hemophilia humanitarian program has partially addressed this gap, distributing over 2.2 million mg of emicizumab to 34 countries and enabling prophylaxis in more than 1100 patients. 35 Building on this, studies from resource-limited settings have explored low-dose regimens to reduce costs while maintaining efficacy. In Malaysia, maintenance dosing was reduced to 1.7–1.9 mg/kg every 2–4 weeks after standard loading doses, and the patients largely remained bleed-free except for a single hematuria episode. 36 Similarly, in Thailand, low-dose emicizumab (2.0–2.5 mg/kg every 4 weeks after two fortnightly loading doses) decreased the median ABR by 2 bleeds/year, improved joint health and quality of life, and achieved zero bleeds in 46.7% of patients. 37 Supporting this approach, an Indian study of low-dose emicizumab (2.6–3.6 mg/kg), emicizumab (2.6–3.6 mg/kg) in eight patients with severe HA reported marked reductions in bleeding (median ABR 1.95, AJBR 1.8) over 12 months, with no serious adverse events, providing proof of concept and real-world evidence for cost-effective low-dose therapy. 38
This study provides a meaningful long-term real-world evaluation of emicizumab in PwHA from northeastern India, highlighting emicizumab’s clinical effectiveness in an underrepresented, underserved population with limited access to specialized hemophilia care. Against this backdrop, the findings of this study not only reaffirm the clinical efficacy of emicizumab in achieving near total bleed suppression and improving joint health, functional independence, and QoL but also underscore its early and uniform effectiveness across age groups. The elimination of baseline age-related disparities in ABR, seen with prior FVIII therapy, further reinforces the potential of emicizumab as a transformative prophylactic agent. These results emphasize the critical value of integrating such therapies into routine care, especially in regions with constrained access to hemophilia treatment infrastructure.
This study encountered several constraints. The small sample size and single-center design may limit the broader applicability of the results. Additionally, the observational nature of the study, lacking a control group, means the observed improvements could be primarily attributed to emicizumab therapy. To validate these findings, larger, multicenter randomized controlled trials are recommended.
Conclusion
This study underscores the long-term effectiveness of emicizumab, demonstrating sustained clinical benefits beyond the initial 6-month period. Over the study period, emicizumab showed an exemplary safety and tolerability profile, providing compelling real-world evidence for its use in routine clinical practice. By incorporating patient-reported outcomes, the study provides a comprehensive understanding of emicizumab’s impact on QoL and caregiver burden. Moreover, the uniform effectiveness observed across age groups supports the early initiation of emicizumab as a means to close persistent treatment gaps associated with conventional FVIII therapy. These improvements should be interpreted in the context of a small subset of heavy bleeders and prior low- to moderate-intensity FVIII prophylaxis, which contributed to both the high baseline ABR and the observed favorable outcomes following full-dose emicizumab. As one of the few long-term studies from northeastern India, this research is particularly valuable in underserved and resource-limited regions where specialized hemophilia care is often unavailable. It fills a significant gap in hemophilia research and highlights the urgent need for inclusive healthcare strategies. These findings may help inform policy decisions and advocate for broader access to emicizumab in underserved regions.
Supplemental Material
sj-docx-1-tah-10.1177_20406207261419325 – Supplemental material for Twelve-month follow-up of a real-world, noninterventional study evaluating the impact of emicizumab on bleeding episodes, joint health, and quality of life in people with hemophilia A
Supplemental material, sj-docx-1-tah-10.1177_20406207261419325 for Twelve-month follow-up of a real-world, noninterventional study evaluating the impact of emicizumab on bleeding episodes, joint health, and quality of life in people with hemophilia A by Anupam Dutta, Taniya Sarkar Dutta, Luish Borboruah, Yash Duseja, Juhi Borah and Papori Gogoi in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
