Abstract
Background:
Development of inhibitors to transfused factor VIII in patients with hemophilia A continues to be a challenge for professionals involved in hemophilia care. The majority of patients in India receive ‘on-demand’ rather than prophylactic therapy. The present study was done to assess the prevalence of factor VIII inhibitors in patients with hemophilia A (PWHA) receiving ‘on-demand’ therapy in a North Indian population and to study the clinicopathological parameters influencing the development of inhibitors.
Methods:
The study group comprised of 300 PWHA. Detailed clinical parameters, treatment history, bleeding profile including family history were recorded. Diagnosis of hemophilia A was confirmed by relevant coagulation tests. Inhibitors were screened using mixing based studies followed by quantification by Bethesda assay and Nijmegen modified Bethesda assay. Samples were collected from five cities in North India where a free supply of factor VIII was available and effectively used in three of these cities.
Results:
In the 300 PWHA, disease phenotype was severe in 219 (73%), moderate in 62 (20.67%) and mild in 19 (6.34%), based on the factor VIII bioassay. Inhibitor prevalence was 9.6% (
Conclusions:
Factor VIII inhibitor prevalence in PWHA receiving ‘on-demand’ therapy was 9.6%. Clinicopathological correlates of inhibitor development in such PWHA have been analyzed in this novel study.
Introduction
Hemophilia A is a recessively inherited X-linked bleeding disorder which results from a deficiency of blood coagulation factor VIII. It affects one in 5000 male births worldwide.
1
The mainstay of treatment of hemophilia A is usually protein replacement using plasma-derived or recombinant factor VIII concentrates to achieve a prophylactic status and to prevent bleeding. A recent United Kingdom guideline recommended prophylactic administration of factor VIII or factor IX concentrate 2–3 times a week as the standard of care for persons with severe hemophilia A or B to prevent hemarthrosis.
2
However, prophylactic replacement therapy is expensive and not affordable in developing countries where an on-demand factor replacement is provided to achieve hemostasis during bleeding episodes or prior to surgical procedures. The exogenous factor VIII may cause an alloantibody or inhibitor formation.
3
These factor VIII inhibitors develop in approximately 25–30% of patients with hemophilia A (PWHA) with a severe phenotype. Presence of inhibitors renders replacement therapies ineffective, limits patient access to a safe and effective standard of care and predisposes them to an increased risk of morbidity and mortality.
4
Factors that may increase the risk of inhibitor development comprise genetic factors including the type of mutation in the
Materials and methods
The dose of factor VIII was calculated as: Factor VIII (IU) required = body weight (in kg) × desired factor VIII increase (% normal) × 0.5. The therapy protocol is in Table 1. 15
On-demand therapy protocol for PWHA.

PWHA, patients with hemophilia A.
Pooled normal plasma (PNP) was prepared by an identical protocol following collection of blood from 20 normal (10 male/10 female) volunteers. Each individual platelet-poor plasma sample was screened [activated partial thromboplastin time (APTT)/prothrombin time (PTs)] prior to pooling to provide a given lot of PNP.
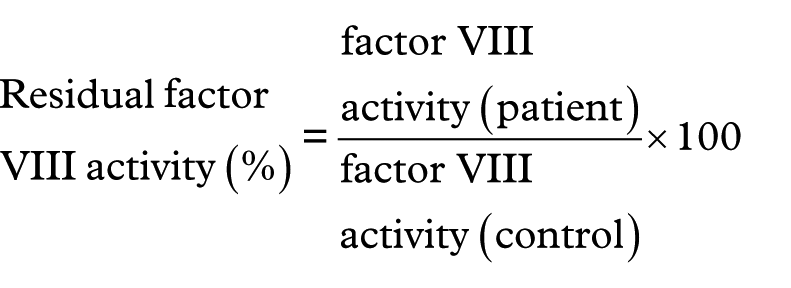
Factor VIII activity (control)
Results
Clinicopathological characteristics of study population.

PWHA, patients with hemophilia A.

Comparison of inhibitor-positive versus inhibitor-negative patients with reference to: (a) phenotype of patients with hemophilia A (b) blood products and factor administered, and (c) city-level distribution in relation to the annual factor intake.

Figure showing distribution of levels of inhibitors in PWHA; six patients show black bars, where the inhibitor was detected by Nijmigen modified bethesda asssay; others were detected by classical Bethesda assay.
Comparison of clinicopathological parameters in inhibitor-positive

Applied unpaired Student’s
PWHA, patients with hemophilia A.
Mean factor VIII level (%)
The mean factor VIII level (%) of the full study group was 3.99 ± 2.59 while in the severe subgroup it was 0.64 ± 0.19. Inhibitor-positive PWHA had a mean factor VIII level of 0.64 ± 0.19 which when compared with the full study group was significantly lower (
Mean age of onset of bleeding
Mean age of onset of bleeding for the full study group was 5.69 ± 3.73 years while in the severe subgroup it was 1.98 ± 1.5 years. Inhibitor-positive PWHA had a mean age of onset of bleeding of 1.00 ± 0.82 years which when compared with the full study group as well as the severe subgroup did not show any significant difference.
Family history and pedigree analysis
Investigation of the family history and pedigree analysis in the full study group revealed a positive family history in 195/300 patients (65%) while in the severe subgroup it was 141/219 (64.3%). Inhibitor-positive PWHA had a family history in 17/29 (58.6%) patients which when compared with the full study group as well as the severe subgroup did not show any significant difference.
Type of treatment
The patients were treated with plasma-derived factor VIII when available and also with blood products including cryoprecipitate or fresh frozen plasma. A total of 121 patients were treated with factor VIII only, 152 with both blood products and factor VIII, and 27 received blood products only. Table 3 compares the type of treatment given to the full study group and to the severe PWHA in inhibitor-positive
Average number of treatments administered
PWHA were grouped on the basis of mean number of treatment courses given annually and were categorized into PWHA receiving 1–5, 6–10 and >10 courses per year. The average number of treatment courses, in other words the frequency of treatment course given to the PWHA significantly correlated with the development of inhibitors both in the full study (
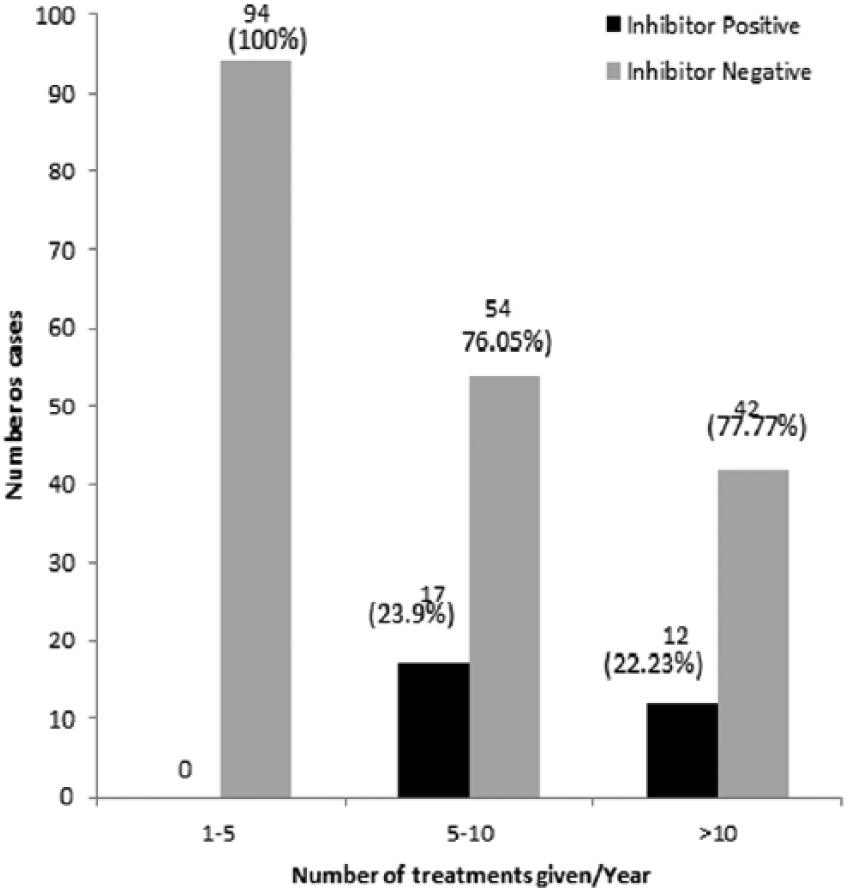
Comparison of frequency of average number of bleeds per year in inhibitor-positive and inhibitor-negative severe patients.
Mean age at first exposure to factor VIII
The mean age at exposure to factor VIII for the full study group was 6.17 ± 3.45 years while in the severe subgroup it was 4.24 ± 5.27 years. Inhibitor-positive PWHA had a mean age of 2.08 ± 0.83 years at the time of exposure to factor VIII which when compared with the full study group, as well as the severe subgroup, was significantly lower (
Age at first factor VIII replacement therapy
PWHA with and without inhibitors were further categorized into age groups above and below 5 years. Development of inhibitors correlated significantly with exposure at <5 years of age in both the full group (
City-level distribution of factor VIII intake and development of inhibitors
Samples were collected from PWHA from five cities in which three cities (A, B, D) had a well-used free government supply of factor VIII for on-demand therapy to resident PWHA, while city C and E had poor availability of factor VIII. It was interesting to note on analysis that the prevalence of inhibitors was higher in city A, B and D at 10.2%, 12% and 11.55% respectively and lower at 4.87% and 5.88% in city C and E [Figure 2(c)]. The mean factor consumption in city A, B, C, D and E was 2818.27, 2925, 1129.2, 1921 and1518 (IU/kg/year) respectively. The annual mean factor intake in inhibitor-positive PWHA was found to be significantly higher at 4353.45 ± 3713.82 IU/kg/year, as compared with a lower annual factor intake in the full study group at 1636.35 ± 1352.29 IU/kg/year (
Bleeding manifestations
The frequency of bleeding manifestations is shown in Table 4. Bleeding in joints and ecchymoses was the most common manifestation found in the study population whereas when inhibitor-positive and negative PWHA were compared, central nervous system (CNS) bleeds and hematuria were found to be significantly higher in inhibitor-positive PWHA (
Bleeding manifestations in inhibitor-positive and negative patients.

Chi-square//Fisher’s exact test; *significant (
CNS, central nervous system; GI, gastrointestinal; PWHA, patients with hemophilia A.
Analysis of risk factors associated with inhibitor development.

Chi-square/Fisher’s exact test; *significant (
PWHA, patients with hemophilia A.
(a): ABO blood group and inhibitor development. (b) Statistical significance between ABO blood groups.
(a).

(b).

Chi-square test, *Significant.
Discussion
In the current case series of 300 PWHA, we have analyzed clinicopathological parameters associated with inhibitor development. The study is novel in the estimation of inhibitor occurrence in a study population which has received plasma-derived factor VIII on demand. Since the factor VIII supplied was through a government supply system, all PWHA received the same plasma-derived factor VIII and hence were a homogenous group with respect to the type of factor used. Most studies in literature describe the occurrence of inhibitors in PWHA on prophylactic therapy. Our study aimed at estimating the prevalence of inhibitors and evaluating various risk factors associated with the development of inhibitors in PWHA taking on-demand therapy in the North Indian population. It may be interesting to note here that the North Indian population is genetically closer to Eurasians and Central Asians than populations of other geographical regions of India.19,20 In terms of the prevalence of hemophilia A, no significant difference exists between the various states in India. 21 In contrast with other studies on inhibitor development which have been performed in developed countries where the recombinant factor VIII was also available to the patients, our patients received only plasma-derived factor VIII. This is an important difference as Peyvandi and Mannucci and colleagues in the SIPPET (survey of inhibitors in plasma-product exposed toddlers) study22,23 concluded that patients treated with plasma-derived factor VIII which contains vWF had a lower incidence of inhibitors than those treated with recombinant factor VIII. Peyvandi and colleagues 22 and Mannucci and colleagues 23 speculated that epitope masking by vWF causes the protection of the factor VIII molecule from endocytosis by antigen-presenting cells, thereby preventing immunologic response against it in a fraction of patients. The plasma-derived factor used in our study was not enriched with von Willebrand factor.
Etiology of development of inhibitors is multifactorial and includes environmental and genetic factors. In the present research, we have studied in addition to conventional clinicopathological parameters, the effect of age at start of therapy, type of replacement therapy and the annual factor consumption. A total of 300 PWHA from the North Indian population were included in the study and an inhibitor prevalence of 9.67% was observed. A detailed analysis of correlates of inhibitor development in PWHA receiving on-demand therapy schedules may contribute to generating novel information. We have adhered to the guidelines for reporting inhibitors in manuscripts recommended by Iorio and colleagues.
24
All PWHA in our study who developed inhibitors had a severe deficiency of factor VIII. In a systematic review Wight and colleagues have reported an overall inhibitor prevalence of 5–7% which when limited to patients with severe disease was much higher at 12–13%.
10
We compared the age at first exposure to factor concentrates and observed that inhibitor development occurred in PWHA where the age at first exposure was <5 years (
The intensity and duration of factor VIII treatment covers a wide spectrum of clinical practices, ranging from single prophylactic infusions of factor VIII concentrate 2–3 times a week to frequent administration of high doses of factor VIII for several consecutive days in the case of major bleeding or surgical procedures. A number of studies have investigated the possible link between intensity (high dosage or prolonged) of treatment and increased inhibitor risk. 25 It has been established in some studies that factor intake and intensity of treatment is an important risk factor for inhibitor development. We have also observed a significant increase in inhibitors in PWHA who received intense therapy. Some studies have demonstrated an increased rate of inhibitor formation after intensive treatment (i.e. surgical procedure or high-frequency treatment);20,28–30 however, a case-control study by Santagostino and colleagues did not confirm these findings. 9
The mean annual factor intake in inhibitor-positive PWHA in our study was higher at 4353.45 ± 3713.82 IU/kg/year as compared with a lower annual factor intake of 1636.35 ± 1352.29 IU/kg/year in inhibitor-negative PWHA. This difference was statistically significant (
In our study, inhibitors to factor VIII developed in 29 PWHA of which all patients were exposed to factor VIII concentrates and 16 had also taken blood products. A small group of PWHA who had taken only blood products did not develop inhibitors. Further we also observed that inhibitor formation is associated with high intensity exposure to factor VIII protein. The CANAL cohort study addresses treatment-related risk factors of inhibitor development in previously untreated patients with hemophilia A. The authors conclude that treatment increases the risk of inhibitor formation and this may be explained by the intensity of treatment. Peak treatment moments may trigger inhibitor formation and have been considered the most significant determinant of inhibitor development. 29 In a study, Kurnik and colleagues observed that standard prophylaxis started at or after the first joint or other severe bleed, led to the formation of inhibitors in 47% of patients, compared with only 3.8% in patients given a low-dose prophylactic regimen started at manifested bleeding tendency, with no long or intensive treatment. 31 The presence of severe bleeds, trauma, surgery are associated with the use of high-dose factor VIII or prolonged treatment which leads to up-regulation of the cellular T and B-cell lymphocyte response and an increased risk of inhibitor development. In contrast, a lower dose of factor VIII protein with regular prophylaxis carries a decreased risk of inhibitor development. 29
During data analysis we observed that blood group A was most common in inhibitor-positive PWHA whereas in the study group as a whole the ABO subtype B was most frequent. This association of blood group A with inhibitor development needs validation in larger studies from different geographic regions. In a recent multivariate logistic regression analysis by Franchini and colleagues, blood group O was shown to have a lower (approximately twofold) inhibitor risk, but the sample size was too small for conclusive results and further the patients were confined to the Italian population.
32
In our study, the proportion of inhibitor-positive PWHA was higher in blood group A (
Evaluation of test methods for inhibitor development is difficult, because there is no gold standard against which to compare them. In practice, both laboratory and clinical evidence is used to determine whether a patient has an inhibitor. The inhibitor-negative population is defined largely by the screening test, although non-neutralizing antibodies that are not detected in clot-based assays may be suspected in some patients. 34 Quantification of inhibitor titers is important for deciding the therapy for PWHA. The Bethesda assay, which is the most frequently used method, lacks sensitivity especially in the lower titer range resulting in unreliable data. In our study those samples which were found to be inhibitor-positive after the screening assay were confirmed for presence of inhibitors and their titers quantified by the Bethesda assay. In cases with suspected low titers the Nijmegen modification was used. This modification involves replacement of the imidazole buffer in the control mixture by immunodepleted factor VIII-deficient plasma and allows better discrimination between positive and negative samples with improvement in reliability. The quantification of inhibitor titers by CBA and NBA went through rigorous standardization processes and results were validated by external quality assurance. Association with factor VIII replacement therapy near the time of vaccination, presence of comorbid conditions like immune disorders and malignancy was attempted. However, the number of PWHA with these associations was low or nil and conclusions could not be derived.
The current study has comprehensively evaluated clinicopathological parameters and risk factors involved in the development of inhibitors in PWHA on-demand therapy in a large cohort with interesting results. It is paradoxical that while the availability of factor VIII has helped PWHA overcome bleeding complications, it has introduced this new complication in therapy with development of inhibitors in one tenth of patients, a number which is fast growing.
Supplemental Material
Supplementary_Material – Supplemental material for Clinicopathological parameters influencing inhibitor development in patients with hemophilia A receiving on-demand therapy
Supplemental material, Supplementary_Material for Clinicopathological parameters influencing inhibitor development in patients with hemophilia A receiving on-demand therapy by Sanya Arshad, Anshima Singh, Namrata Punit Awasthi, Swati Kumari and Nuzhat Husain in Therapeutic Advances in Hematology
Footnotes
Funding
Authors wish to acknowledge Department of Science and Technology (DST), New Delhi, India for providing SERB grant support (Ref. no SB/SO/HS/025/2014).
Conflict of interest statement
The authors declare that there is no conflict of interest.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
