Abstract
Background:
Individuals diagnosed with relapsed or refractory large B-cell lymphoma (R/R LBCL) typically exhibit a dismal prognosis when treated with conventional therapeutic modalities. CD19—targeted chimeric antigen receptor T (CAR-T) cell therapy has brought about a paradigm shift in the treatment paradigm of this disease. Nevertheless, a comprehensive assessment of the efficacy and safety profiles of diverse CAR-T products (e.g., axicabtagene ciloleucel (axi-cel), tisagenlecleucel (tisa-cel), and lisocabtagene maraleucel (liso-cel)) is imperative.
Objective:
This systematic review aims to systematically summarize and evaluate the efficacy and safety of CAR T-cell therapies for R/R LBCL through systematic review and meta-analytic approaches.
Design:
This is a systematic review and meta-analysis.
Data sources and methods:
Relevant studies were identified by systematically searching PubMed and Web of Science until November 29, 2023. Cohort studies and clinical trials were incorporated, with the inclusion of single-arm studies. CAR T-cell therapies are involved in tisa-cel, axi-cel, and liso-cel. Proportions and their 95% confidence intervals were calculated using standard meta-analytic approaches.
Results:
Thirty-seven studies were included in meta-analyses, liso-cel demonstrated equivalent overall survival rates (70.3%) to axi-cel (65.6%) at 12 months post-treatment, whereas tisa-cel was 48.0%. Objective response rate for liso-cel was comparable to axi-cel (79.0% vs 76.8%,
Conclusion:
Based on the available evidence, liso-cel has shown promising efficacy and a manageable safety profile in patients with R/R LBCL, when compared to axi-cel and tisa-cel. However, real-world data on liso-cel are limited.
Plain language summary
This study provides a comprehensive evaluation of three CAR T-cell therapies for treating aggressive lymphoma that has returned or stopped responding to treatment. The analysis shows that lisocabtagene maraleucel (liso-cel) performed similarly to axicabtagene ciloleucel (axi-cel) in helping patients survive for at least one year, while tisagenlecleucel (tisa-cel) showed lower survival rates. In terms of tumor shrinkage, both liso-cel and axi-cel worked better than tisa-cel. When looking at side effects, liso-cel caused fewer severe immune reactions than the other two treatments, though it had slightly more neurological complications than tisa-cel. These findings suggest liso-cel may offer the best balance between effectiveness and safety for this difficult-to-treat cancer, though more real-world experience is needed to confirm these results.
Keywords
Introduction
Diffuse large B cell lymphoma (DLBCL) is the most common type of non-Hodgkin lymphoma worldwide, accounting for approximately 30%–40% in different geographic regions. 1 Although the majority of DLBCL patients respond to first-line chemotherapy or a comparable regimen, approximately one-third of patients will develop relapsed or refractory large B-cell lymphoma (R/R LBCL), which remains a major cause of morbidity and mortality. 2 Treatment options are limited for patients with R/R LBCL, and only a minority of patients can be cured by the second-line chemotherapy followed by consolidative autologous stem cell transplantation (ASCT). 3 However, the prognosis remains dismal for patients who are ineligible for ASCT or relapse even after ASCT. Since chimeric antigen receptor (CAR) T-cell therapy was introduced, up to 40% of R/R DLBCL patients could achieve a high response rate and long-term remission. 4 To date, only three CAR T-cell therapies are available for third- or later-line treatments of LBCL: tisagenlecleucel (tisa-cel), axicabtagene ciloleucel (axi-cel), and lisocabtagene maraleucel (liso-cel), all of which are CD19-directed CAR T-cell therapies.
Three randomized controlled trials regarding these three CAR T-cell therapies have demonstrated their clinical benefits in R/R LBCL, compared with standard of care (SOC).5–7 As no head-to-head studies comparing various CD19-directed CAR T cell therapies, some indirect treatment comparison studies have been conducted between axi-cel and tisa-cel,8–16 axi-cel and liso-cel,17,18 as well as tisa-cel and liso-cel.19,20 Additionally, moderate numbers of studies based on real-world data and single-arm trials were also published.21–33 A systematic review has been performed to evaluate the benefits and harms of CAR T-cell therapy for people with R/R LBCL, identifying 13 eligible uncontrolled studies evaluating a single or multiple arms of CAR T-cell therapies, but only tisa-cel and axi-cel were included, and no meta-analysis was conducted. 34 Another two previous reviews utilized the meta-analysis to evaluate the efficacy and safety of CAR T-cell therapies, including axi-cel, tisa-cel, and liso-cel,3,35 but only three randomized controlled studies were included,5–7 which found that CAR T-cell therapy has superior outcomes as compared to SOC. Therefore, for better evaluation of the efficacy and safety of CAR T-cell therapies, we aimed to conduct a systematic review and meta-analysis for available clinical evidence of three CAR T-cell therapies in the treatments of R/R LBCL, including tisa-cel, axi-cel, and liso-cel.
Methods
Literature searching strategies
This systematic review and meta-analysis were conducted per the reporting guidelines in Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 36 Our researchers conducted a literature search to identify relevant studies assessing the efficacy and safety of CAR T-cell treatments in R/R LBCL in two electronic databases, including PubMed (National Library of Medicine, National Center for Biotechnology Information, Bethesda, MD, USA) and Web of Science (Clarivate Analytics, Boston, MA, USA) through November 29, 2023. Relevant searches utilized various combinations of relevant terms, such as (“large B-cell lymphoma” OR “large B cell lymphoma” OR “LBCL” OR “DLBCL”) AND (“chimeric antigen receptor T” OR “chimeric antigen receptor therapy” OR “chimeric antigen receptor T-cells” OR “chimeric antigen receptor-modified T” OR “chimeric antigen receptor-engineered T” OR “CAR-T” OR “CAR T-Cell” OR “CAR T Cell” OR “Axicabtagene” OR “tisagenlecleucel” OR “lisocabtagene”). The methodologies employed for the search of each database were delineated in Table S1.
Duplicate publications were excluded each title and/or abstract was checked for relevance. The full text of an article was reviewed only if the abstract indicated that the article was potentially relevant. Only original studies conducted among human participants were considered for this review. Cross-referencing was employed to complement the study identification process of the systematic review. Two researchers (Q.L. and S.X.) conducted an independent assessment of the eligibility of all retrieved literature using EndNote 20 software (Clarivate). Their findings underwent comparison at each selection stage, and any disagreements were resolved through discussions. In instances where additional inconsistencies emerged, the senior researchers (H.H. or Y.H.) were consulted to address them.
Inclusive and exclusive criteria
Studies were included in this systematic review if they met the following criteria: (1) subjects must be 18 years or older; (2) clinical trials or observational cohort studies, whatever single arm and double arms; (3) the sample sizes for any individual CAR T-cell treatment group ought to be 10 or greater; (4) axi-cel, tisa-cel, or liso-cel were used as an independent treatment group, whatever treating dose levels; (5) all subjects were diagnosed as positron emission tomography-positive relapsed or refractory diffuse large B-cell lymphoma (de novo or transformed from any indolent lymphoma), high-grade B-cell lymphoma with rearrangements in MYC a proto-oncogene encoding a transcription factor regulating cell growth and proliferation and either BCL2, BCL6, or both (double-hit or triple-hit lymphoma), primary mediastinal B-cell lymphoma, or follicular lymphoma grade 3B 37 ; (6) at least one efficacy or safety outcome was reported, including overall survival (OS), progression-free survival (PFS), objective response rate (ORR), complete response rate (CRR), partial response rate (PRR), cytokine release syndrome (CRS), and neurologic events (NE).
Conversely, studies were excluded if (1) studies include patients included juveniles (age <18 years); (2) article types are case reports, animal experiments, editorials, conference abstracts without full-text publications; (3) studies with any two or three CAR T-cell therapies mixed in one group were also excluded; (4) studies combining CAR T-cell therapy with other investigation treatments were excluded to ensure treatment effect specificity; (5) only CAR T-cell therapy was mentioned, but axi-cel, tisa-cel, liso-cel were not be referred; (6) reviews or meta-analytic reviews were excluded, but corresponding individual studies were judged for eligibility per the above-mentioned inclusive criteria; (7) the duration of response (DOR), event-free survival (EFS), and treatment-emergent adverse events (TEAE) were not assessed due to the shortage of reported studies, which are insufficient to compare the differences among these CAR T-cell therapies; (8) repeated reports were removed, and a singular, representative paper was chosen for each study that met the criteria for inclusion. Furthermore, the paper with the longest follow-up period was preferentially selected, as a general rule.
Outcome measures
Overall survival was defined as the time from the first infusion of any one of CAR T-cell therapies to death from any cause. PFS was defined as the time from first infusion of any one of CAR T-cell therapies to disease progression or death from any cause at or after assessment by independent review committee (IRC) or investigators. ORR is calculated as the proportion of patients who achieved a best overall response of complete response or partial response, based on assessment by the IRC per Lugano criteria. 38 CRR was computed as the proportion of patients achieving a complete response. PRR was computed as the proportion of patients achieving a partial response.
Data extraction
At least two authors independently extracted the following data in a standardized manner for each eligible study: study abbreviation name, study period and countries, clinical trial phase, registered number in ClinicalTrialsgov, indications of R/R LBCL, age and sex proportions of patients, drug dosages, sample size, duration of follow-ups, as well as primary and secondary endpoints, including OS, PFS, ORR, CRR, PRR, CRS, and NE. Any disagreement was resolved in a study team after additional review of the articles or further discussion with a senior researcher, if necessary.
Quality assessments
The two researchers independently performed the quality assessment of the included studies using the Newcastle–Ottawa scale (NOS) 39 for cohort studies, phase I trials, and phase II trials, and Jadad score scale 40 for three randomized controlled trial (RCT). Accounting for eight items across three domains (selection, comparability, and outcome), a study was awarded one point for each item, with the exception of the item related to comparability, which allowed the assignment of two points. Hence, NOS ranges from 0 to 9, and a score of 7 or greater was considered good quality. Jadad score includes randomization (2 points), double-blinding (2 points), and withdrawals and dropouts (1 point). If the study did not employ double-blinding, then the use of blinded or independent central review would also be rewarded with a score of 2 points. The sum of the above-mentioned items was computed for each study and ranged from 0 to 5 points. Low quality was defined as 2 points or less, and high quality as ⩾3 points.
Statistical analyses
Only outcomes were extracted from axi-cel, tisa-cel, and liso-cel treatment groups to assess the efficacy and safety in treating R/R LBCL, and the outcomes from other treatment groups were ignored. Finally, proportions (or rate) with their 95% confidence intervals (CI) calculated as effect sizes (ES) of meta-analysis. Cochran’s
Results
Study screening and selection
A flow diagram of the study search and selection process is presented in Figure 1. We found 1738 publications via a systematic literature search, of which 258 records were deleted as duplicates. The remaining 1480 titles/abstracts were screened for eligibility, and 1266 publications were excluded, including 505 non-original articles, 323 conference abstracts, 305 irrelevant, 125 case reports or series with sample size <10, and 8 animal experiments. Two hundred and fourteen studies appeared to be potentially relevant for inclusion, but 177 were excluded during the full-text evaluation due to the following reasons: 61 literatures reported the outcomes of multiple CAR T-cell therapies; 49 did not defined which CAR T cell therapies were used; 20 literatures are repeated papers,5,6,44–61 which was the same studies with included study62–68; 15 studies being meta-analytic reviews or indirect comparison3,16–20,34,35,69–75; 15 targeted outcomes not reported76–90; 12 studies reporting combined results of CAR T-cell therapies and other treatments91–102; 5 for other CAR T-cell therapies.103–107 The literature listing of studies excluded from the analysis is presented in Table S2. Ultimately, 37 studies were included, involving in 20 for axi-cel,24–33,64,65,68,108–114 8 for direct comparisons between axi-cel and tisa-cel,8–15 5 for tisa-cel,7,21–23,67 and 4 for liso-cel.62,63,66,115

Flowchart of literature selection.
Characteristics of included studies
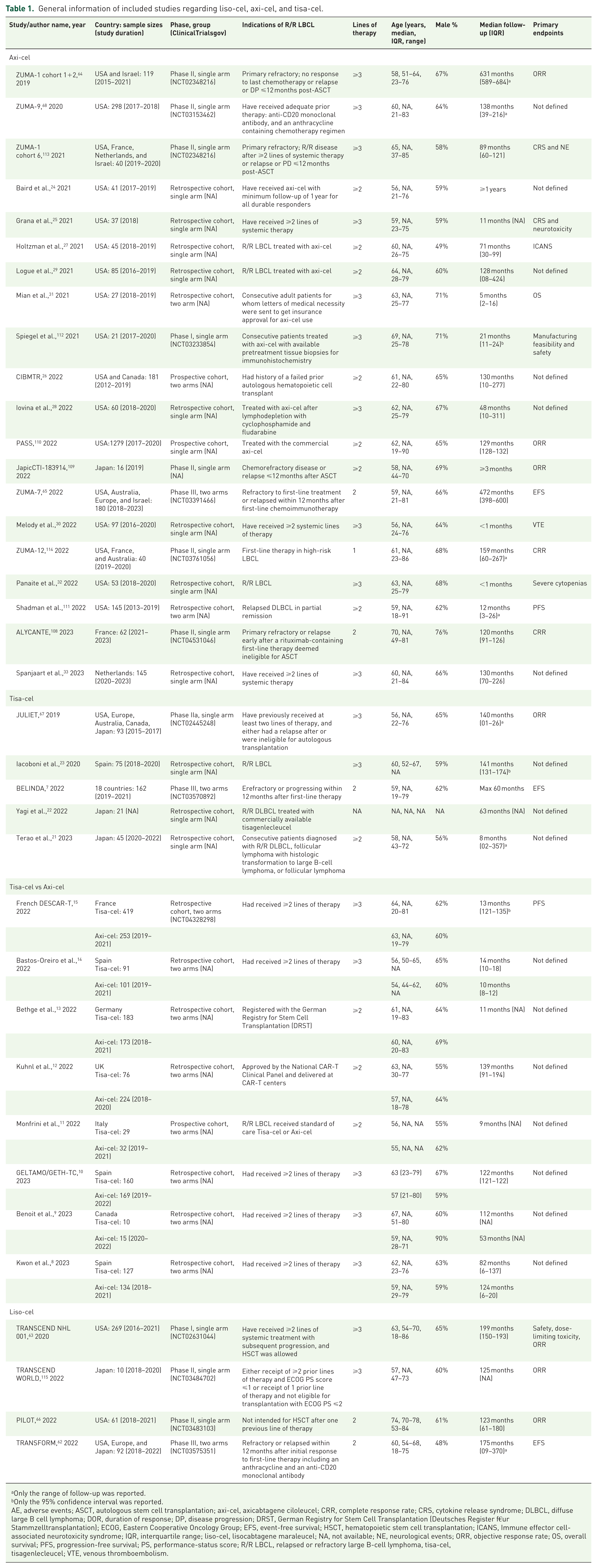
Details on the general characteristics of included studies are shown in Table 1. These studies were conducted in the USA, Canada, Europe, Australia, Japan, and Israel. Among 37 eligible studies, 28 individual studies assess the efficacy and safety of the axi-cel in R/R LBCL, involving 4072 patients.5,24–33,55,57,64,65,68,108–114 Of 28 axi-cel studies, 17 were retrospective studies,8–10,12–15,24,25,27–32,111 6 phase II trials,64,68,108,109,113,114 3 prospective cohort studies,11,26,110 1 phase I trial, 112 and 1 phase III trial. 65 The longest follow-ups were 5 years, 64 and the shortest were <1 month. 30 A total of 13 studies among 1491 patients reported the outcomes of tisa-cel, 4 of which were single-arm studies,21–23,67 and another 9 were two-arm studies.7–15 Only one was randomized controlled trial (phase III trial), 7 others were observational studies comparing tisa-cel with axi-cel.8–15 Since liso-cel was approved for third-line R/R LBCL in 2021, after axi-cel in 2017 and tisa-cel in 2018, only four studies of 432 R/R LBCL patients reported the efficacy and safety of liso-cel so far, including one phase I trial, 63 two phase II trials,66,115 and one phase III trial. 62
General information of included studies regarding liso-cel, axi-cel, and tisa-cel.
Only the range of follow-up was reported.
Only the 95% confidence interval was reported.
AE, adverse events; ASCT, autologous stem cell transplantation; axi-cel, axicabtagene ciloleucel; CRR, complete response rate; CRS, cytokine release syndrome; DLBCL, diffuse large B cell lymphoma; DOR, duration of response; DP, disease progression; DRST, German Registry for Stem Cell Transplantation (Deutsches Register f€ur Stammzelltransplantation); ECOG, Eastern Cooperative Oncology Group; EFS, event-free survival; HSCT, hematopoietic stem cell transplantation; ICANS, Immune effector cell-associated neurotoxicity syndrome; IQR, interquartile range; liso-cel, lisocabtagene maraleucel; NA, not available; NE, neurological events; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; PS, performance-status score; R/R LBCL, relapsed or refractory large B-cell lymphoma, tisa-cel, tisagenlecleucel; VTE, venous thromboembolism.
Study quality assessments
Study quality scores based on NOS among 34 non-randomized studies ranged from 1 to 8 (mean 51 ± 18), but no study had a full score of 9. Seven studies were awarded the status of good quality,8,10,13,15,63,66,111 whereas six studies were rated as bad quality.21,22,25,30,32,109 The remaining 21 studies were rated as fair quality9,11,12,14,23,24,26–29,31,33,64,67,68,108,110,112–115 (Table 2). All three phase III randomized controlled trials were rewarded as a full score of 5 due to complete randomization strategies, blinded or independent central review, as well as objective records for an account of all patients.7,62,65
Assessment of Newcastle–Ottawa scale for 34 non-randomized studies. a
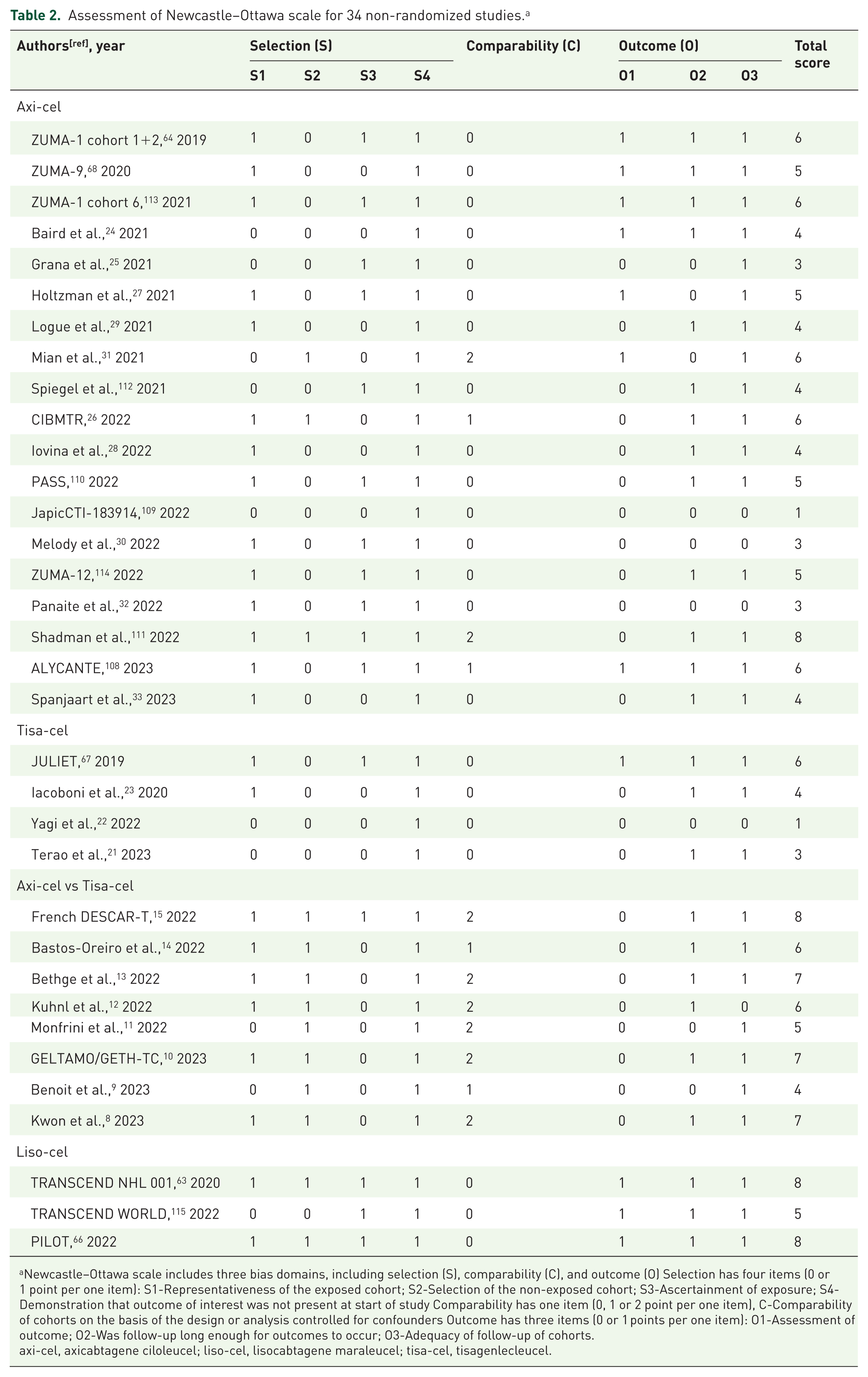
Newcastle–Ottawa scale includes three bias domains, including selection (S), comparability (C), and outcome (O) Selection has four items (0 or 1 point per one item): S1-Representativeness of the exposed cohort; S2-Selection of the non-exposed cohort; S3-Ascertainment of exposure; S4-Demonstration that outcome of interest was not present at start of study Comparability has one item (0, 1 or 2 point per one item), C-Comparability of cohorts on the basis of the design or analysis controlled for confounders Outcome has three items (0 or 1 points per one item): O1-Assessment of outcome; O2-Was follow-up long enough for outcomes to occur; O3-Adequacy of follow-up of cohorts.
axi-cel, axicabtagene ciloleucel; liso-cel, lisocabtagene maraleucel; tisa-cel, tisagenlecleucel.
Meta-analyses for survival
Because of the significant variation in sample size and follow-up duration, we provided survival probabilities specific to each time point. Only a few studies reported survival outcomes at 3, 18, 24, and 60 months. A publication mapping related to patient cohorts at different follow-ups after CAR T-cell therapies was shown in Table S3.
Figure 2 illustrates the forest plots of the pooled OS at 6- and 12-month post-treatments of three CAR T-cell therapies. A total of 11 original studies published their 6-month OS after treatment, involving 6 studies for axi-cel,9,12,25,28,108,113 3 for tisa-cel,9,12,22 and 4 for liso-cel.62,63,66,115 In our meta-analysis, OS in liso-cel (ES, 82.2%, 95% CI, 70.4%–84.1%) was comparable to that in axi-cel (ES, 82.1%; 95% CI, 72.9%–91.2%) and tisa-cel (ES, 67.0%; 95% CI, 58.1%–75.8%), with
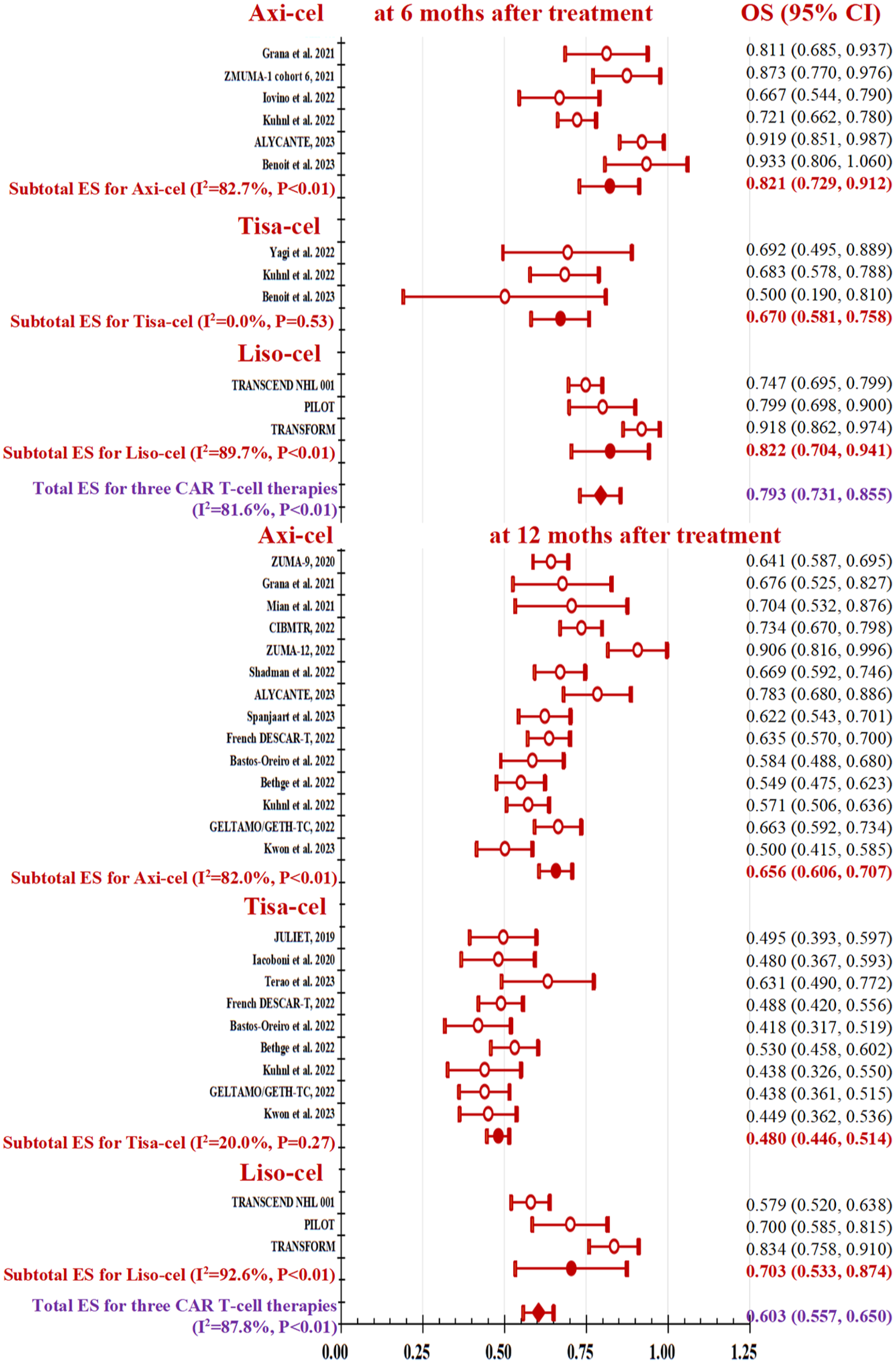
Overall survival by follow-up months after three CAR T-cell therapies.
Subgroup analysis results by patient selection.
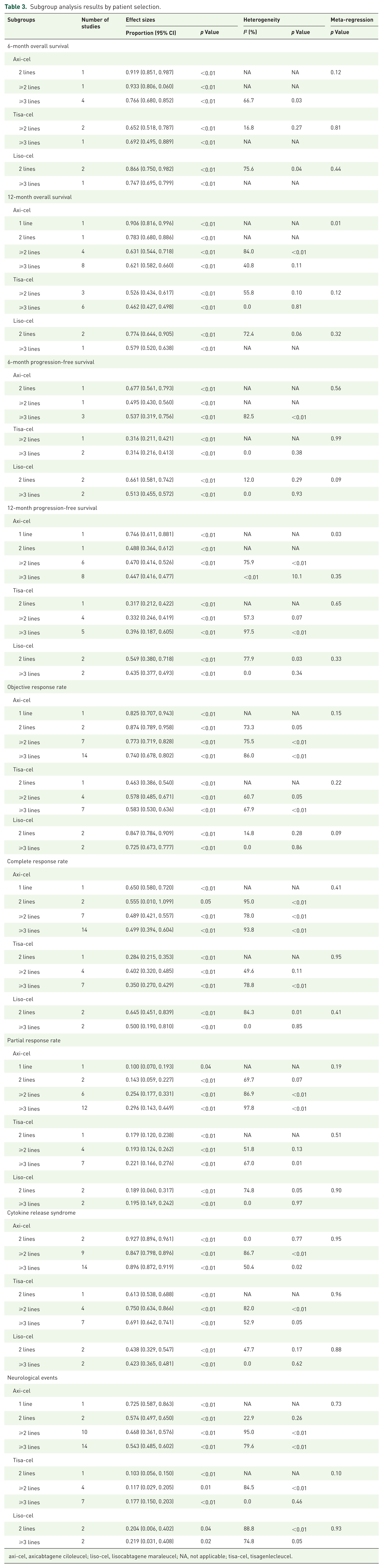
axi-cel, axicabtagene ciloleucel; liso-cel, lisocabtagene maraleucel; NA, not applicable; tisa-cel, tisagenlecleucel.
The forest plots of PFS at 6- and 12-month after CAR T-cell therapies are presented in Figure 3. Eleven individual studies reported PFS at 6 months post-treatment, among which five reported for axi-cel,9,12,64,108,113 four for tisa-cel,9,12,22,23 and four for liso-cel.62,63,66,115 In meta-analysis using random effects model, the liso-cel (ES, 59.2%, 95% CI, 48.5%–69.8%) and axi-cel (ES, 55.9%, 95% CI, 44.5%–67.2%) exhibited better PFS than tisa-cel (ES, 34.1%, 95% CI, 25.3%–42.9%). Large heterogeneity was observed across included studies (
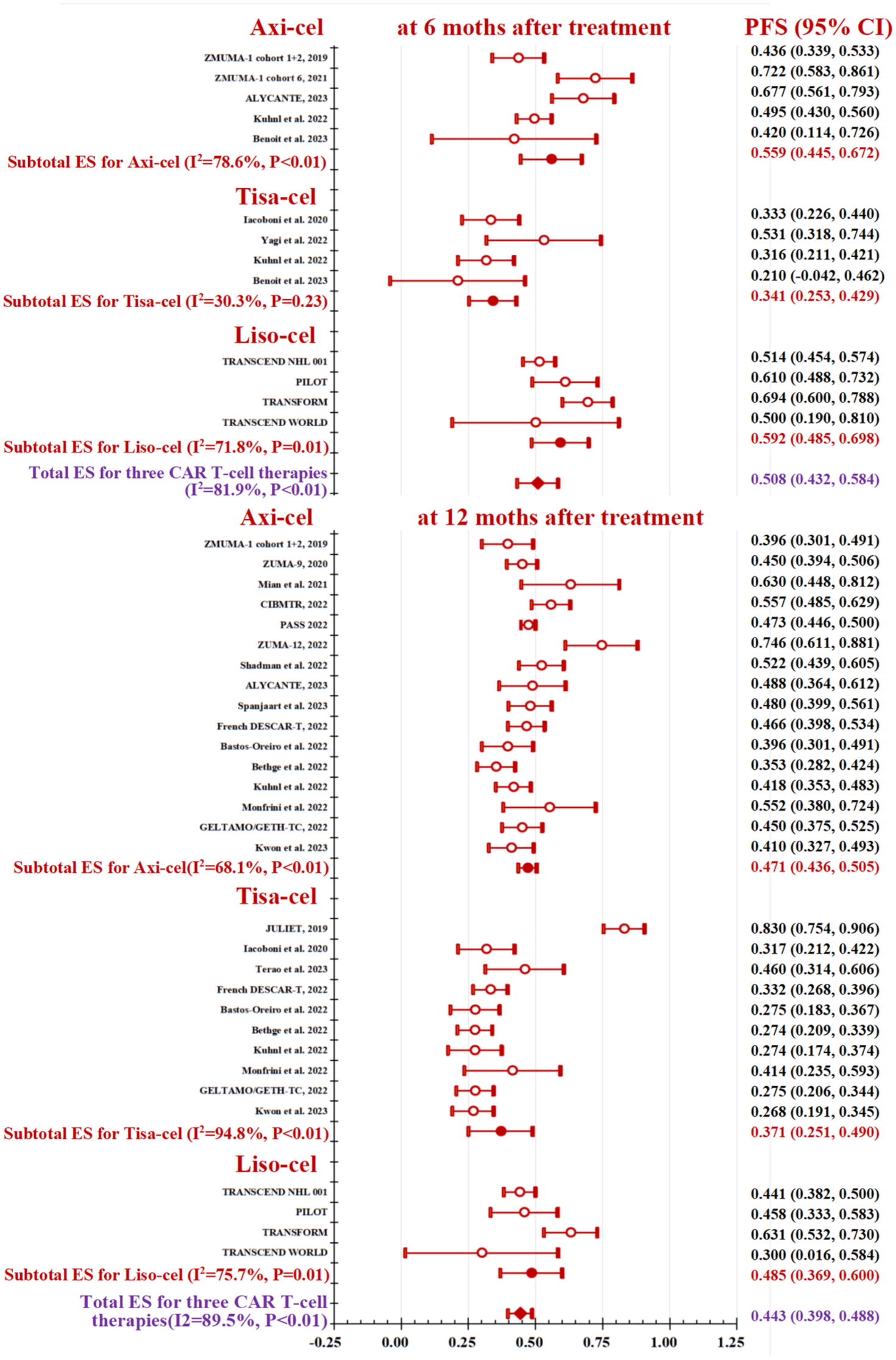
Progression-free survival by follow-up months after three CAR T-cell therapies.
Meta-analysis for responses
A total of 32 individual studies were extracted the ORR of CAR T-cell therapies, comprising 24 for axi-cel, 14 for tisa-cel, and 4 for liso-cel. The combined ORR was estimated as 71.4% (95% CI, 67.6%–75.3%;

Objective response rate of three CAR T-cell therapies.
Analogous findings from meta-analyses were shown for CRR in Figure 5. Pooled CRR was calculated as 59.2% in liso-cel and 51.0% in axi-cel, both of which were equivalent in the meta-regression (
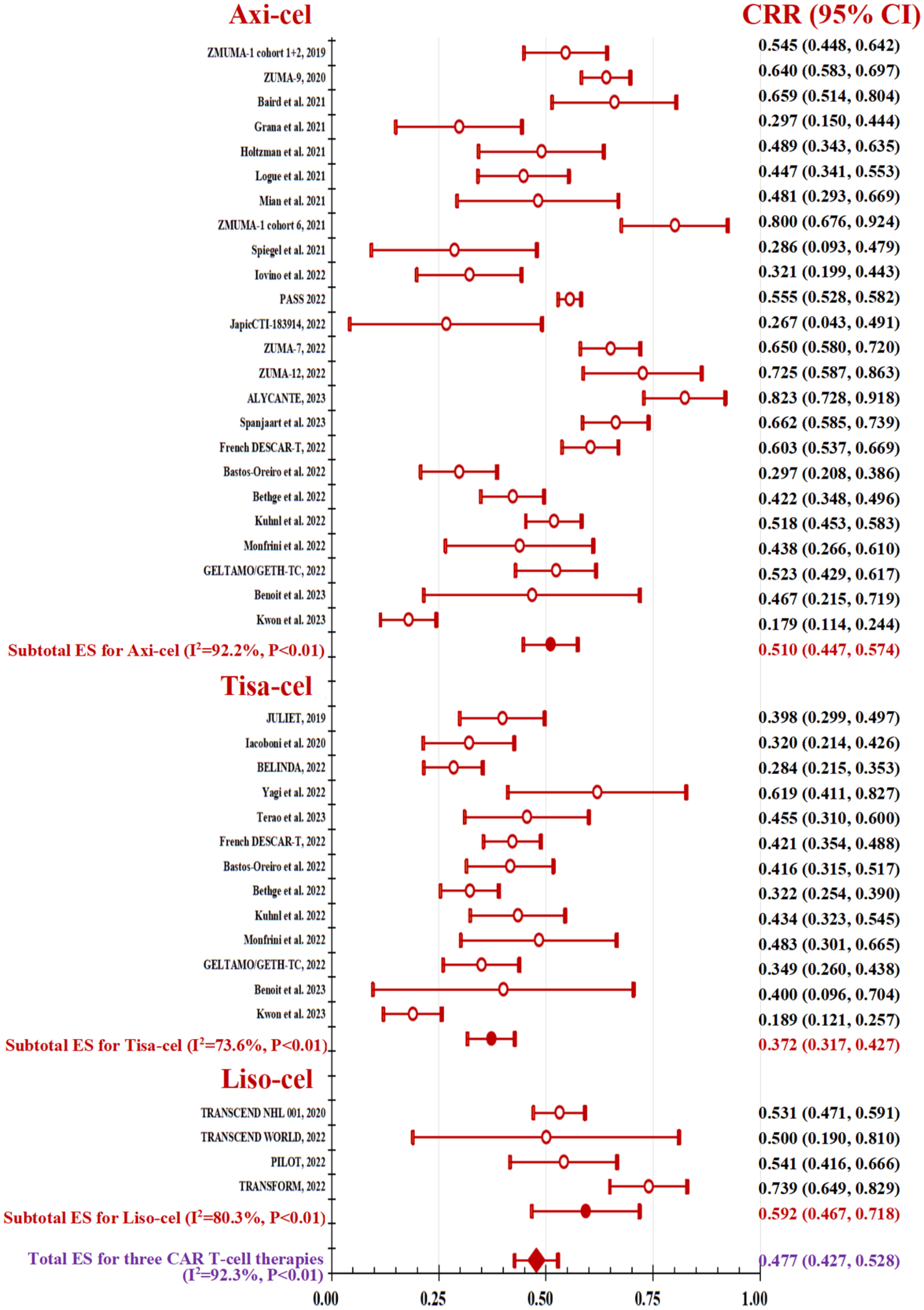
Complete response rate of three CAR T-cell therapies.
No significant differences were observed in PRR among patients treated with axi-cel, tisa-cel, and liso-cel, with respective rates of 26.5%, 20.6%, and 18.6% (refer to Figure 6). Sensitivity analyses did not find exceptional study in the three above-mentioned response rates. Upon conducting a sensitivity analysis, it was determined that no anomalies were present (Figure S7). Upon the execution of meta-regression analyses, no corroborative evidence was identified to support the alteration of patient selection criteria in PRR for axi-cel (

Partial response rate of three CAR T-cell therapies.
Meta-analysis for safety
Among the various TEAEs, CRS and neurological events emerged as the two most concerning adverse effects, whose forest plots are illustrated in Figures 7 and 8. In these three CAR T-cell therapies, liso-cel exhibited the lowest CRS rate at 43.0% (95% CI, 38.3%–47.6%), followed by tisa-cel at 70.9% (95% CI, 66.1%–75.6%), and axi-cel at 87.9% (95% CI, 85.4%–90.3%), with all

Cytokine release syndrome of three CAR T-cell therapies.

Neurologic events of three CAR T-cell therapies.
Discussions
Although CAR T-cell therapy has been regarded as a promising therapeutic strategy, only three randomized controlled trials reported that CAR T-cell therapies were superior to SOC in R/R DLBCL, and no study demonstrated which CAR T-cell therapy was the most recommended regimen so far. To our knowledge, this meta-analytic review represents the first study aimed at consolidating diverse clinical evidence regarding the efficacy and safety outcomes of three CAR T-cell products in patients with R/R DLBCL, which also compares the differential benefits and risks among axicabtagene ciloleucel, tisagenlecleucel, and lisocabtagene maraleucel employing standard meta-regression and subgroup analytic methodologies. In this study, 37 eligible cohort studies and clinical trials were included, comprising of 4072 patients in 28 studies of axi-cel, 1491 patients in 13 studies of tisa-cel, and 432 patients in 4 studies of liso-cel. Based on these available data, liso-cel has demonstrated promising efficacy and a manageable safety profile in patients with R/R LBCL, when compared to axi-cel and tisa-cel. Axi-cel exhibits comparable efficacy to liso-cel, yet it is associated with more severe CRS and NE. Tisa-cel shows a relatively conservative therapeutic effect, but it holds significant advantages in NE. Even though our meta-analysis incorporated some observational studies with poor quality, conducting a sensitivity analysis for each outcome did not reveal any studies as outliers, and no evidence of publication bias was detected.
Due to the large variability in sample size and the duration of follow-up, we presented time point-specific survival probabilities. The highest number of studies reported a 12-month survival rate, followed by a 6-month survival rate. A small number of studies reported survival outcomes at 3 months,57,109 18 months,62,66 and 24 months.7,55,63,110,111 To date, only ZUMA-1 reported their 5-year follow-up results, demonstrating axi-cel has sustained OS at 42.6% and PFS at 36.6% without new safety signals in patients with refractory LBCL. 64 In our meta-analysis, we calculated OS and PFS at both the 6- and 12-month post-treatment, and observed that approximately 60% of patients treated with axi-cel and liso-cel maintained PFS at 6 months post-treatment, which decreased to <50% by the 12-month point. In contrast, the PFS for patients treated with tisa-cel was less encouraging, with only around 40% maintaining PFS at 6 months post-treatment; however, their PFS appeared to stabilize by the 12-month follow-up. Three CAR T-cell products exhibited around 80% and 60% of overall survival at 6- and 12-month post-treatment, respectively. Even though the overall survival of the three CAR T-cell therapy products did not exhibit any statistically significant differences after a 6-month treatment period, the therapeutic benefits of liso-cel and axi-cel were markedly superior to those of tisa-cel when evaluated at 12 months. Several previous studies utilized a matching-adjusted indirect comparison, suggesting axi-cel had superior survival outcomes to tisa-cel, 16 and liso-cel, 17 only based on three phase I and II trial data from ZUMA-1, JULET, and TRANSCEND NHL 001. However, this indirect comparison inherently possesses certain unavoidable limitations, primarily due to the presence of unknown amounts of residual bias. When analyzing single-arm or non-comparative studies, there will always be a degree of uncertainty surrounding the influence of unknown or unmeasured prognostic factors and effect modifiers. Our study offers additional evidence by synthesizing various study design types and employing standard meta-analytic methods to compare three CAR T-cell products in the treatment of R/R DLBCL. In order to enhance comprehension of the variations in patient selection across these three therapies, additional subgroup analyses and meta-regression analyses were conducted for all outcomes under evaluation. Nonetheless, it was observed that only the overall survival and PFS rates at 12 months post-treatment for axi-cel exhibited a significant decline among patients who had undergone two or more lines of systemic therapy, as compared to those who had received earlier lines. However, the available evidence remains insufficient to establish a definitive conclusion.
In addition to ensuring the continuation of life, our meta-analytic review further confirmed that the tumor response rates for axi-cel and liso-cel were found to be quite similar when comparing their effectiveness among who underwent infusion. While tisa-cel exhibits a less favorable response, particularly in terms of ORR (76.8%, 79.0%, vs 58.3%) and CRR (51.0%, 59.2%, vs 37.2%). In earlier research, ORR for CAR-T cell therapies in the treatment of R/R DLBCL has been observed to fall within a broad spectrum, ranging anywhere from 40% up to an impressive 95%.7,11,25,108,113 Although these studies have produced diverse outcomes across various countries and settings, and there has been some apprehension regarding whether patient characteristics influence these results, when compared to historical data from non-randomized control groups, CAR-T cell therapies have demonstrated an impact on the natural progression of this aggressive form of non-Hodgkin lymphoma.16–20,71 Drawing upon the aforementioned evidence, we present an updated systematic review supplemented by a meta-analysis of clinical trials and real-world cohort studies, which aims to compare and evaluate more comprehensively tumor responses among three distinct CAR T-cell products.
Ultimately, we chose CRS and NE as our primary indicators for assessing the safety profiles of the three different products, which are the two most concerned AE closely related to CAR T-cell therapy. Both previous studies and our own research have shown that the use of axi-cel is correlated with a significantly elevated risk of CRS and NE, when compared to the alternative treatments such as liso-cel and tisa-cel.16,17,19,71 Although tisa-cel demonstrates a relatively conservative therapeutic effect, it exhibits significant advantages in terms of its NE in treating non-Hodgkin’s lymphoma. Consequently, tisa-cel is often preferred for older adults because of its lower toxicity, allowing for administration in an outpatient setting. 71 Liso-cel has shown a manageable safety profile in patients with R/R LBCL, despite being approved most recently in 2021. However, the available data on liso-cel were derived solely from four clinical trials, such as TRANSCEND NHL 001,45,63 TRANSFORM,6,62 TRANSORM WORLD, 115 and PILOT. 66 The real-world evidence remains limited in liso-cel, which could aid in translating these clinical trial findings into clinical practice.
One of the significant strengths of our research lies in the utilization of meta-analytic and meta-regression methodologies, which enabled us to compile and synthesize a diverse array of evidence derived from clinical trials as well as real-world data. This comprehensive approach allowed us to conduct a more thorough comparison of the efficacy and safety profiles of three different CAR T-cell products. However, it is crucial to acknowledge certain important limitations when interpreting the outcomes of our study. Firstly, as the original individual data in the eligible articles were inaccessible, relying only on the reported effect sizes might lead to a less precise and potentially inaccurate risk estimation. Secondly, several potentially influential covariates were not taken into account, such as various indications of R/R DLBCL, which could impact the overall effect sizes in the meta-analysis, particularly when dealing with real-world data. Despite the initial classification of patient selection as reported in the articles, we undertook a series of subgroup analyses and a meta-regression to identify potential difference. The heterogeneity in patient selection among the various individual studies remains significant and cannot be overlooked, as it plays a crucial role in the efficacy and safety of various CAR T-cell therapies. Thirdly, in our meta-analysis, some efficacy and safety indicators were omitted, such as the DOR, EFS, and TEAEs. The main reason is the scarcity of documented research and the inconsistency in the selection and reporting of outcomes among these individual studies, which posed challenges for the comparison across CAR T-cell therapies. Notably, this research did not encompass an analysis of survival time because of the varying observational and treatment periods across the studies. Although certain studies present Kaplan–Meier curves, which can reconstruct individual patient data through the Guyot algorithm, the absence of numbers-at-risk/event information and the low-resolution curves in some studies may introduce inconsistencies in the extraction of survival data points, especially for time-to-event outcomes. Fourthly, clinical trials often report diminished toxicity rates and enhanced efficacy durability as a consequence of stringent patient selection criteria, rigorous monitoring, and standardized management protocols. A greater number of real-world studies on axi-cel have been reported to date in comparison with tisa-cel and liso-cel, which could potentially introduce interference in the conclusions drawn. Finally, despite conducting a systematic review that included searches in two databases and implementing thorough cross-referencing to ensure completeness, we cannot rule out the possibility of having overlooked relevant studies.
Conclusion
Despite these limitations, our study furnishes a descriptive summary of the absolute effects observed across studies and is valuable for formulating hypotheses and establishing benchmark rates for future research. CAR-T cell therapy represents a pioneering therapeutic approach for adult patients with R/R DLBCL, who previously had limited treatment options. This meta-analytic review stands as the inaugural study designed to aggregate the varied clinical evidence on the efficacy and safety outcomes of three CAR T-cell products in patients with R/R DLBCL. It also conducts a comparative analysis of the distinct benefits and risks associated with axi-cel, tisa-cel, and liso-cel, utilizing standard meta-regression and subgroup analytic techniques. In conclusion, liso-cel has shown effectiveness and safety in R/R LBCL patients. Axi-cel has similar efficacy but more severe side effects. Tisa-cel has a milder therapeutic effect but advantages in managing AE. Real-world evidence is limited, especially for liso-cel. Ongoing and planned trials may improve CAR-T cell efficacy with combination treatments and could advance CAR-T therapy to earlier lines.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251407511 – Supplemental material for Efficacy and safety of chimeric antigen receptor T-cell therapy in relapsed/refractory large B-cell lymphoma: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tah-10.1177_20406207251407511 for Efficacy and safety of chimeric antigen receptor T-cell therapy in relapsed/refractory large B-cell lymphoma: a systematic review and meta-analysis by Qiang Li, Shenghua Xu, Yun Zhong, Peng Rao, Hui Huang and Yan Huang in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207251407511 – Supplemental material for Efficacy and safety of chimeric antigen receptor T-cell therapy in relapsed/refractory large B-cell lymphoma: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tah-10.1177_20406207251407511 for Efficacy and safety of chimeric antigen receptor T-cell therapy in relapsed/refractory large B-cell lymphoma: a systematic review and meta-analysis by Qiang Li, Shenghua Xu, Yun Zhong, Peng Rao, Hui Huang and Yan Huang in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
