Abstract
Background:
Chimeric antigen receptor T-cell therapy represents a revolutionary advancement in personalized treatment for diffuse large B-cell lymphoma (DLBCL). However, data regarding the use of tisagenlecleucel (tisa-cel) outside of clinical trials remain scarce, especially in East Asia. Therefore, we conducted this study to provide insights into the logistics, efficacy, and safety profile of tisa-cel treatment in Korea.
Methods:
Clinical data were collected from 46 patients with DLBCL who underwent leukapheresis for commercial tisa-cel manufacturing at Seoul National University Hospital between January 2022 and December 2023. Response evaluations were conducted at 1, 3, 6, and 12 months after tisa-cel infusion.
Results:
Overall, 44 patients received tisa-cel infusion. The median time from tisa-cel order placement to product delivery and subsequent infusion was 28 days (range, 24–84) and 42 days (range, 29–118), respectively. The overall response rate was 70.5%, with a complete response rate of 47.7%. Patients who achieved an overall response at 3 months post-infusion had longer overall survival (median, not reached vs 2.6 months, p < 0.001) and progression-free survival (median, 13.0 months vs 1.4 months, p < 0.001) compared to those who did not. Additionally, an elevated baseline LDH was associated with poor survival. Cytokine release syndrome was observed in 70.5% of the patients (grade ⩾3, 4.5%), while immune effector cell-associated neurotoxicity syndrome occurred in 15.9%.
Conclusion:
To the best of our knowledge, this study presents the first real-world data on tisa-cel outcomes in Korean patients with DLBCL. Commercial tisa-cel is a feasible treatment option for patients with DLBCL in real-world settings.
Introduction
Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of non-Hodgkin lymphoma and is treated with immunochemotherapy as the standard first-line approach, incorporating rituximab-based regimens.1,2 Recently, the incorporation of polatuzumab vedotin, an anti-CD79b drug conjugate, has further extended clinical benefits in previously untreated patients. 3 However, approximately one-third of patients ultimately experience relapse or develop refractory disease despite initial treatment.4,5 In these relapsed or refractory (R/R) patients, outcomes remain poor.6,7 This unmet clinical need has driven the development of novel therapeutic strategies, and chimeric antigen receptor (CAR) T-cell therapy has marked a paradigm shift in immunotherapy for R/R DLBCL.8,9
Tisagenlecleucel (tisa-cel) is a second-generation CD19-directed CAR T-cell therapy approved for the treatment of R/R DLBCL, 10 that has played a pivotal role in expanding the therapeutic landscape for this challenging patient population. The landmark trial for tisa-cel in R/R DLBCL is the single-arm phase II JULIET trial, which reported an overall response rate (ORR) of 53%, a median progression-free survival (PFS) of 2.9 months, and a median overall survival (OS) of 11.1 months. 11 While other CD19 CAR T-cell therapies, such as axicabtagene ciloleucel and lisocabtagene maraleucel, have demonstrated superior outcomes,12–14 tisa-cel remains the only commercially available CAR T-cell therapy in Korea, as in many other countries where access to multiple CAR T-cell therapies is limited. 15 This underscores the need for continuous generation of real-world data to optimize the use of CAR T-cell therapy implementation across diverse healthcare settings.
Despite the increasing global interest in CAR T-cell therapies,16,17 real-world data on tisa-cel use remains relatively scarce, particularly in East Asian populations. Given the challenges in accessibility and the need for evidence-based optimization of CAR T-cell therapy, generating robust real-world data is essential to bridge existing knowledge gaps and refine treatment strategies. To contribute to this growing body of evidence, we conducted a retrospective single-center study evaluating the real-world efficacy and safety of commercial tisa-cel in Korean patients with R/R DLBCL. In addition, we analyzed factors associated with response and survival outcomes to provide insights for optimizing CAR T-cell therapy in routine practice.
Methods
Patients and study design
Patients with histologically confirmed DLBCL who had a history of at least two prior lines of systemic chemotherapy were deemed eligible for tisa-cell therapy. Initially, 46 patients with R/R DLBCL who underwent leukapheresis for commercial tisa-cel manufacturing at Seoul National University Hospital between January 2022 and December 2023 were identified. Per the JULIET trial, 3 leukapheresis was recommended for patients with an absolute lymphocyte count (ALC) of ⩾300/µL. Lymphodepleting chemotherapy was initiated upon the arrival of tisa-cel at the treatment site. In cases where the tisa-cel product did not meet commercial release criteria (out-of-specification, OOS) but was deemed acceptable by the physician, patients received infusion, and their outcomes were included in the analysis. Tisa-cel infusion was administered during hospitalization to enable close monitoring for adverse events (AEs). Finally, 44 patients received tisa-cel infusion (Figure 1), and efficacy and safety analyses were conducted among these 44 patients. Response evaluations using positron emission tomography/computed tomography were recommended at 1, 3, 6, and 12 months following tisa-cel infusion. 18 Follow-up continued until December 2024. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 19

Cohort consort diagram.
All patients received standardized prophylactic regimens. Trimethoprim/sulfamethoxazole 400/80 mg and acyclovir 400 mg were administered orally once daily from day 5 until 6 months post-infusion to prevent Pneumocystis jirovecii pneumonia and viral infections, respectively. Fungal prophylaxis consisted of fluconazole 200 mg once daily, initiated on day 5 and continued until the absolute neutrophil count exceeded 1000/μL for three consecutive days. Patients who were hepatitis B core antibody positive received entecavir 0.5 mg once daily, administered from day 5 until 12 months post-infusion. 20 The serum cytomegalovirus (CMV) polymerase chain reaction was performed at least once weekly from conditioning until discharge and at outpatient visits when clinically indicated. Preemptive therapy was considered when the viral load reached ⩾500 copies/mL, based on clinical judgment.
Definition
Primary refractory DLBCL was defined as the failure to achieve a partial response (PR) or complete response (CR) either during or at the end of first-line immunochemotherapy. 21 The cell of origin was classified as GCB or non-GCB according to the Hans algorithm. 22 Bridging therapy was defined as any treatment targeting lymphoma that was given between leukapheresis and lymphodepleting chemotherapy.
Grading and management of cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS) were done according to the American Society for Transplantation and Cellular Therapy (ASTCT) criteria. 23 AEs were assessed according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0.
Response evaluation was performed according to the Lugano criteria. 24 ORR was defined as the percentage of patients who achieved PR or CR after tisa-cel infusion. PFS was defined as the time from tisa-cel infusion until relapse, progression, or death from any cause. OS was defined as the time from tisa-cel infusion to death from any cause. The duration of response (DoR) was defined as the time from PR or CR to relapse, progression, or death from any cause.
Statistical analysis
Descriptive statistics are presented as median values with ranges or numbers with percentiles. Differences between groups were assessed using Fisher’s exact test or Pearson’s chi-square test for categorical variables and Student’s t-test or Wilcoxon rank-sum test for continuous variables. The Clopper-Pearson method was used for calculating the 95% confidence interval (CI) for the response rate. Survival outcomes were estimated using the Kaplan-Meier method. Univariate and multivariate Cox regression analyses were conducted for survival outcomes. Variables included in the Cox analyses were age, sex, performance status, disease extent and stage, prior treatment, lactate dehydrogenase (LDH), and disease status. Multivariate analyses were performed using a Cox proportional hazards model with stepwise variable selection, in which variables with p values of <0.1 in univariate analyses were initially included. Hazard ratios (HRs) with 95% CIs were calculated. The statistical software “R” version 4.4.1 (www.r-project.org) was used for all statistical analyses. p Values of <0.05 were considered statistically significant.
Results
Patients and product characteristics
A total of 46 patients underwent leukapheresis (Figure 1). Thirty-nine completed the process with a single successful collection. Three experienced manufacturing failures; one discontinued, and two underwent a second leukapheresis, both yielding successful products. Of the remaining four patients, one had a repeat leukapheresis due to a delivery issue (second product classified as OOS), and three had OOS classification on the initial product. Among the four OOS products, one was not used, while three were deemed acceptable and infused. In total, 49 leukapheresis procedures were performed, and 44 patients (95.7%) ultimately received tisa-cel infusion.
The baseline characteristics of infused patients at the time of leukapheresis are summarized in Table 1. As there is no age limitation for CAR T-cell therapy, the patient’s age ranged widely from 39 to 86 years old (median, 66.5). Overall, 30 patients (68.2%) had a performance status ⩾2, 33 (75.0%) were categorized as stage III/IV, and 9 (20.5%) were classified as germinal B-cell type according to the Hans algorithm. More than half of the patients were classified as International Prognostic Index (IPI) high-risk. The patients had received a median of three prior lines of chemotherapy (range, 2–6), and all had been exposed to rituximab. Before undergoing leukapheresis, six patients had prior exposure to polatuzumab, and three to acalabrutinib. There were nine (20.5%) patients who underwent hematopoietic stem cell transplantation (eight autologous; one allogeneic). All but three patients had an ALC of ⩾300/µL at the time of leukapheresis. The ALC values for these three patients were 257, 226, and 86/µL, respectively, with the latter requiring three leukapheresis procedures to achieve adequate cell collection. None of these three patients with an ALC <300/µL received OOS.
Baseline characteristics.

ECOG, Eastern Cooperative Oncology Group; GCB, germinal center B-cell; IPI, International Prognostic Index; LDH, lactate dehydrogenase; TNC, total nucleated cell; ULN, upper limit of normal.
The median time from tisa-cel order placement to product delivery to the site was 28 days (range, 24–84). On the other hand, due to logistic issues and varying patients’ conditions, the median time from tisa-cel order placement to infusion was 42 days (range, 29–118). There were two patients who did not receive bridging therapy prior to tisa-cel infusion. For those who underwent bridging therapy, nine received cisplatin-based chemotherapy (either DHAP or GDP), while eight patients each were treated with CHOP, pola-BR, ICE, or immunomodulatory drug-based regimens. A single patient received selinexor (Table S1). All patients were admitted at the start of lymphodepletion. All but one patient received FC (fludarabine 25 mg/m2/day plus cyclophosphamide 250 mg/m2/day for three consecutive days). The remaining patient underwent bendamustine lymphodepletion (90 mg/m2/day for two consecutive days) due to cyclophosphamide intolerance.
Efficacy and survival outcome
Response evaluation was available for 42 out of 44 patients 1-month post-infusion, while 2 patients died within the first month due to disease progression. At 1 month after infusion, 18 patients achieved CR, and 13 achieved PR (Figure 2). At 3 months after infusion, 21 out of 31 patients (67.7%) remained responsive. Among the 13 patients who achieved PR at 1 month, 3 (23.1%) converted to CR at 3 (1 patient) and 6 (2 patients) months, respectively; 7 patients experienced disease progression 3 months post-infusion. By 12-month post-infusion, nine patients were confirmed to remain responsive, of whom eight were in CR. In summary, among the 44 infused patients, 21 patients (47.7%) achieved a best response of CR, and 10 (22.7%) achieved PR, resulting in an ORR of 70.5% (95% CI: 54.8–83.2; Table 2). The outcomes of the three patients who received OOS are summarized in Table S2.

Response evaluation results by time point.
Clinical efficacy of tisa-cel.

These two patients were clinically assessed as having PD.
CI, confidence interval; CR, complete response; NA, not assessed; PD, progressive disease; PR, partial response; SD, stable disease.
With a median follow-up duration of 19.0 months (95% CI: 12.8–32.3), 32 patients experienced disease progression, and 22 patients died; of the 22 deaths, 17 were attributed to disease progression (Figure S1). Among the 31 patients who achieved a response, the median time to response was 1.2 months (range, 0.2–2.9), and the median DoR was 10.0 months (95% CI: 4.4 months–not assessed; Figure 3(a)). At the time of data cutoff, 12 of 31 patients maintained their response. The estimated 6- and 12-month DoR rates were 62.1% (95% CI: 46.7–82.7) and 47.2% (95% CI: 31.9–70.0), respectively. Patients who achieved a CR at 1-month post-infusion had a longer DoR compared to those with a PR at the same time point, although the difference was not statistically significant (median, 19.4 months vs 3.3 months, p = 0.2; Figure S2).

Kaplan-Meier curve of clinical outcome: (a) duration of response, (b) progression-free survival, and (c) overall survival.
The median PFS was 3.3 months (95% CI: 2.5–8.4), with 6- and 12-month PFS rates of 35.4% (95% CI: 23.6–53.1) and 25.3% (95% CI: 15.0–42.7), respectively (Figure 3(b)). The median OS was 9.7 months (95% CI: 4.5 months–not assessed), with 6- and 12-month OS rates of 60.1% (95% CI: 46.9–76.9) and 45.9% (95% CI: 32.6–64.7), respectively (Figure 3(c)).
We compared the survival outcomes of patients based on disease status and response at 3 months post-infusion (Figure 4; Table S3). Patients with controlled disease (CR or PR) prior to infusion exhibited longer PFS (median, 8.4 months vs 2.5 months, p = 0.240) and OS (median, not reached vs 6.7 months, p = 0.079) compared to those with uncontrolled disease, although neither difference was statistically significant. Patients who achieved an overall response at 3 months post-infusion had significantly longer PFS than nonresponders (median, 13.0 months vs 1.3 months, p < 0.001), and a similar benefit was observed in those achieving CR (median, 13.0 months vs 1.5 months, p < 0.001). A significant difference was also observed for OS. Patients with an overall response at 3 months demonstrated longer OS compared with nonresponders (median, not reached vs.2.6 months, p < 0.001), and those achieving CR had longer OS than those without CR (median, not reached vs 3.0 months, p = 0.005). Consistent results for PFS and OS were observed when patients were stratified by overall response or CR status at 1-month post-infusion (Figure S3; Table S3).

Kaplan-Meier curve of PFS and OS. (a) PFS by disease status prior to infusion; (b) PFS by overall response at 3 months post-infusion; (c) PFS by complete response at 3 months post-infusion; (d) OS by disease status prior to infusion; (e) OS by overall response at 3 months post-infusion; and (f) OS by complete response at 3 months post-infusion.
Predictors of survival outcomes
Table 3 presents the outcomes of the Cox analysis for PFS. In the univariate analysis, extranodal involvement at >1 site, bone marrow involvement, number of prior lines of therapy, baseline LDH, and overall response at 3 months post-infusion were identified as potential factors associated with PFS. In the multivariate Cox model, ⩾4 prior lines of therapy (HR 2.79; 95% CI: 1.06–7.31; p = 0.037), elevated LDH (HR 2.48; 95% CI: 1.13–5.45; p = 0.023), and overall response at 3 months post-infusion (HR 0.02; 95% CI: 0.00–0.10; p < 0.001) emerged as independent predictors of PFS.
Cox analysis for progression-free survival.

BM, bone marrow; CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; LDH, lactate dehydrogenase; ULN, upper limit of normal.
For OS, extranodal involvement at >1 site, bone marrow involvement, number of prior lines of therapy, baseline LDH, disease status before tisa-cel infusion, and overall response at 3 months post-infusion were identified as potential factors (Table 4). In the multivariate analysis, ⩾4 prior lines of therapy (HR 3.05; 95% CI: 1.05–8.88; p = 0.041), elevated LDH (HR 2.90; 95% CI: 1.17–7.18; p = 0.021), and overall response at 3 months post-infusion (HR 0.10; 95% CI: 0.03–0.30; p < 0.001) were identified as independent predictors of OS.
Cox analysis for overall survival.
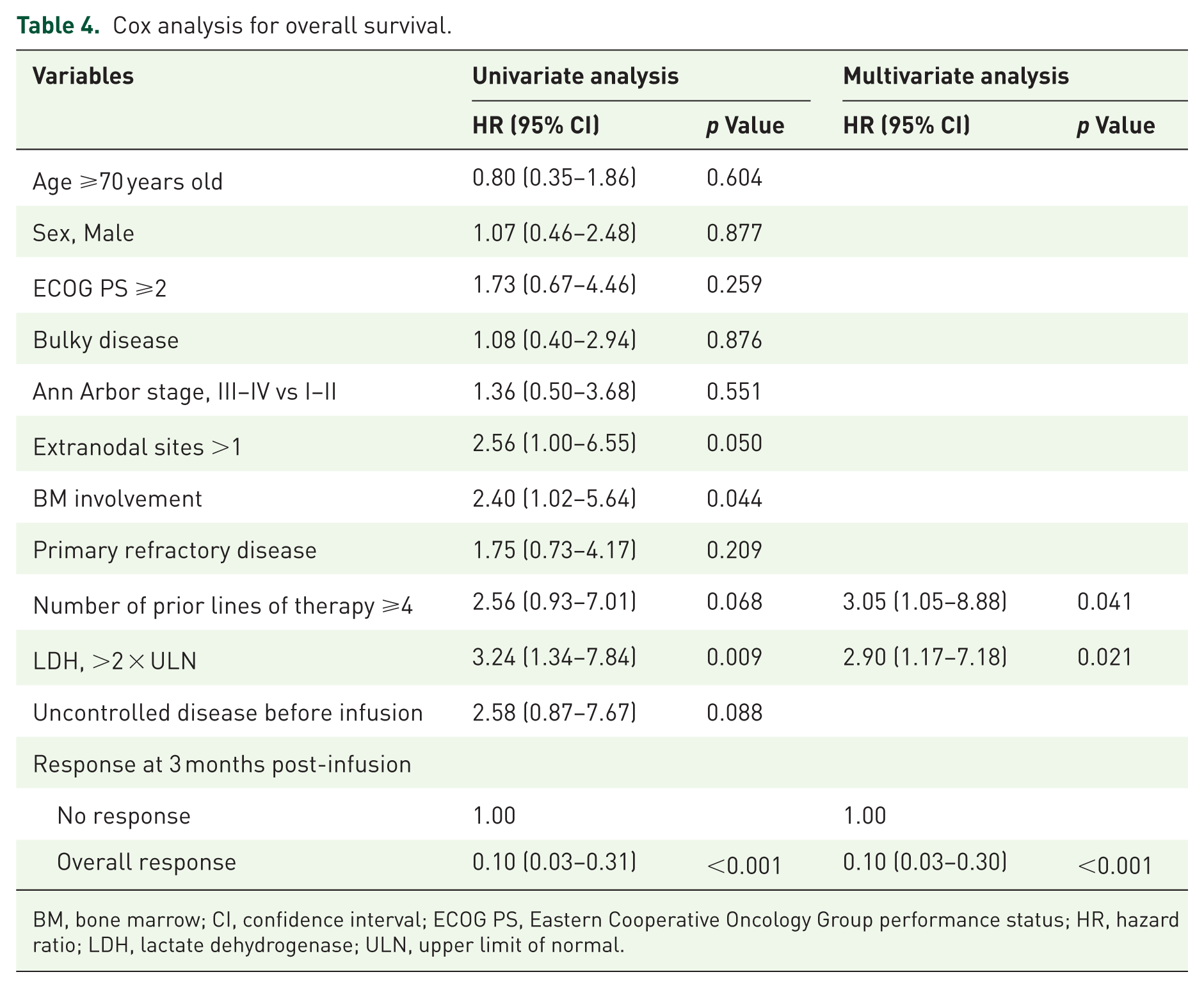
BM, bone marrow; CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; LDH, lactate dehydrogenase; ULN, upper limit of normal.
Safety
Among the infused patients, 31 (70.5%) developed CRS, with a median duration of 3 days (Table 5). Two patients (4.5%) developed grade 3 CRS, and both fully recovered within 10 days with the use of tocilizumab and dexamethasone. Seven patients (15.9%) developed ICANS; four (9.1%) and two (4.5%) developed grade ⩾2 and grade ⩾3 ICANS, respectively. Tocilizumab and corticosteroids were administered to 31 (70.5%) and 19 (43.2%) patients, respectively. There was a single case of CAR T-related hemophagocytic lymphohistiocytosis syndrome (HLH), which occurred in a 63-year-old man 4 days post-infusion. The patient also developed grade 2 CRS and grade 3 ICANS. In addition to the use of tocilizumab and corticosteroids, ruxolitinib 10 mg bid for 5 days was administered (Figure S1). Following ruxolitinib treatment, the patient achieved full resolution.
Safety outcomes.

CAR, chimeric antigen receptor; HLH, hemophagocytic lymphohistiocytosis syndrome.
The infectious events that occurred from the time of infusion to 3 months post-infusion are presented in Table 5. Overall, 18 episodes of infection occurred in 12 of 44 patients (27.3%). 44.4% (8/18) were of bacterial, 44.4% (8/18) were of viral, and 11.1% (2/18) were of fungal origin. Detailed data on infectious events are summarized in Table S4. The most common infectious events were related to CMV, including one case of CMV retinitis and four cases of CMV viremia. All remaining viral infections were cases of COVID-19.
Discussion
This is the first real-world study on tisa-cel treatment in patients with R/R DLBCL in Korea and the second in Asia. 25 Commercial use of tisa-cel in Korea is feasible and has yielded results comparable to those observed in pivotal trials. Among the 44 patients who received tisa-cel infusion, the ORR was 70.5%, with a CR rate of 47.7%. In addition, overall response at 3 months post-infusion and low baseline LDH were favorable indicators of survival outcomes.
CAR T-cell therapy represents a revolutionary new pillar in the treatment of hematologic malignancies and has emerged as a promising treatment option for R/R DLBCL, demonstrating a superior ORR compared to alternative therapies. 26 As of December 2024, four products (axicabtagene ciloleucel, brexucabtagene autoleucel, idecabtagene vicleucel, and tisa-cel) have been approved by the US Food and Drug Administration (FDA) for the treatment of lymphoma. 27 In Korea, several domestically developed CAR T-cell therapies are available but only within a clinical trial platform.28–30 Tisa-cel remains the only commercially available CD19 CAR T-cell therapy in Korea, 31 underscoring the importance of generating real-world evidence of its use. The findings of our study are consistent with those of the JULIET trial, which reported an ORR of 52% in R/R DLBCL over a 40.3 months follow-up period. 10 Notably, patients who achieved overall response or CR at 3 months post-infusion exhibited significantly longer survival compared to those who did not. The median PFS of over 12 months in patients who achieved CR at 3 months post-infusion suggests that a substantial proportion of this group can maintain a durable response. This finding was confirmed through multivariate analysis. Furthermore, we analyzed pre-infusion factors to identify those influencing survival outcomes, and found that patients with high baseline LDH levels and those who were heavily pretreated had poorer outcomes. Pre-infusion LDH has been identified as a prognostic biomarker in patients undergoing CAR T-cell therapy in previous studies.32,33 In addition to LDH, Dean et al. reported the prognostic value of metabolic tumor volume, calculated through imaging. 34 In summary, patients with R/R DLBCL who have a less extensive treatment history and a lower tumor burden are likely to derive the greatest benefit from tisa-cel.
CRS and ICANS are AEs of interest in CAR T-cell therapy. Although the total percentage of patients experiencing CRS is higher in our experience (70.5%) compared to the JULIET trial (58%), there were significantly less cases with grade ⩾3 events (4.5% vs 22%). 10 The median time to CRS onset was comparable to that observed in the pivotal trial. All patients who experienced grade ⩾3 CRS recovered following treatment with tocilizumab and dexamethasone. In our study, a higher proportion of patients received tocilizumab (70.5% vs 14%) and corticosteroids (43.2% vs 10%) compared to the JULIET study. The increased use of appropriate management strategies, along with the growing experience with CAR T-cell therapy, likely contributed to the improved safety profile observed compared to clinical trials, which is consistent with findings from previous studies.35–37 There was one patient with CAR-related HLH in our cohort. Sandler et al. reported that the incidence of secondary HLH following CAR T-cell therapy was higher than that observed after stem cell transplantation. 38 CAR-related HLH poses challenges in diagnosis due to the limited applicability of conventional HLH criteria, the difficulty in distinguishing it from CRS, and the potential for tocilizumab to mask symptoms; therefore, a high index of suspicion is required for diagnosis.39–41 Documented infections were identified in 27.3% of patients. A total of eight bacterial infections were observed, with five occurring within 30 days post-infusion. Neutropenia often develops at the onset of CRS following infusion, and it can be challenging to distinguish the fever caused by CRS from an underlying infection. Therefore, the possibility of sepsis should be considered alongside CRS when fever occurs post-infusion. With primary viral prophylaxis using acyclovir, five of the eight viral infections were attributed to CMV, while the remaining three were caused by COVID-19. Compared to Western data, where bacterial infections were the most common, 33 our study, along with a study conducted in Japan, 25 identified CMV as the predominant infection. The higher prevalence of CMV seropositivity in Asia, including Korea, is well-documented.42,43 The incidence of CMV reactivation and infection following CAR T-cell therapy varies across studies,44,45 which may also be attributed to the lack of standardized monitoring guidelines. Further research is warranted to establish optimal CMV surveillance strategies in this setting.
The efficacy and safety profiles observed in our cohort were within the range of real-world data reported from the United States and Europe (Table 6).17,33,37,46 These studies reported an ORR of about 60%–70%, with grade ⩾3 CRS occurring in around 5%, demonstrating favorable efficacy and safety outcomes compared to the JULIET trial. In addition, Nasta et al. demonstrated that tisa-cel treatment can be employed effectively in an outpatient setting. 47 The use of commercial tisa-cel in an outpatient setting is practical, with most patients avoiding the need for hospitalization. While real-world data on tisa-cel in East Asian populations remain scarce, Goto et al. reported the first Japanese cohort, demonstrating an ORR of 73%, an 89.9% incidence of any grade CRS, and grade ⩾3 CRS in 6.7% of patients, 25 which is comparable to our findings. The incidence and severity of ICANS seem to vary between populations, in contrast to the relatively stable response rates observed across studies. Predicting which patients will develop ICANS remains challenging, and both prophylaxis and treatment strategies for ICANS are largely based on expert opinion. 48 A recent multi-center survey across 20 countries reported variations in the incidence and severity of ICANS among centers, as well as heterogeneity in prophylaxis and treatment protocols. 49 While response rates appear to be consistent, variations in AE management and supportive care practices may contribute to differences in the safety profile. Further research is needed to evaluate the impact of standardized protocols on the prevention of AEs.
Summary of outcome from the literature regarding the real-world experience of CAR T-cell therapy.

B-NHL, non-Hodgkin B-cell lymphoma; CR, complete response; CRS, cytokine release syndrome; DLBCL, diffuse large B-cell lymphoma; ICANS, immune effector cell-associated neurotoxicity syndrome; LBCL, large B-cell lymphoma; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; R/R, relapsed or refractory; SCT, stem cell transplantation.
This study had several limitations. First, it was a retrospective study with a small sample size from a single center. Small subgroup sizes in some comparisons may have affected the statistical power. However, due to several factors, the number of centers capable of administering CAR T-cell therapy in Korea remains limited, leading to a relatively slow and insufficient accumulation of clinical experience. Therefore, given the rarity of real-world data on tisa-cel in East Asia, we believe that the single-center experience does not diminish the significance of the present study. Second, the use of various bridging therapies prior to infusion introduced heterogeneity, potentially confounding the interpretation of treatment outcomes. Lastly, the follow-up period may have been insufficient to fully assess long-term outcomes.
Despite these limitations, our study is the first to evaluate the logistics, efficacy, and safety of commercial tisa-cel in Korean patients with R/R DLBCL, filling a gap in the literature for East Asian populations. The treatment is feasible, with most toxicities being manageable. In contrast, achieving outcomes that surpass the efficacy reported in pivotal trials remains challenging in real-world settings. Moreover, other CAR T-cell therapies, such as axicabtagene ciloleucel and domestically developed CAR T-cell therapies, appear to have superior efficacy compared to tisa-cel. Therefore, further research on CAR T-cell therapy and immunotherapy for patients with R/R DLBCL remains essential.
Conclusion
In conclusion, treatment with commercial tisa-cel in Korea yielded results comparable to those observed in pivotal studies. Achieving an overall response or CR at 3 months post-infusion was associated with favorable survival outcomes. In addition, low pre-infusion LDH was a strong predictor of survival. Given the unresolved questions (e.g., selecting the “right” patients, predictors for long-term responders, efficacy in difficult to treat subpopulations, including those with central nervous system involvement), 50 further research is needed to optimize the use of CAR T-cell therapy in real-world settings for patients with R/R DLBCL.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251406461 – Supplemental material for Real-world experience with tisagenlecleucel for the treatment of relapsed or refractory diffuse large B-cell lymphoma in Korea
Supplemental material, sj-docx-1-tah-10.1177_20406207251406461 for Real-world experience with tisagenlecleucel for the treatment of relapsed or refractory diffuse large B-cell lymphoma in Korea by Dong Hyun Kim, Soyean Kwon, Seungah Cha, Rabia Seyma Öz, Junshik Hong, Dong-Yeop Shin, Youngil Koh, Sung-Soo Yoon and Ja Min Byun in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207251406461 – Supplemental material for Real-world experience with tisagenlecleucel for the treatment of relapsed or refractory diffuse large B-cell lymphoma in Korea
Supplemental material, sj-docx-2-tah-10.1177_20406207251406461 for Real-world experience with tisagenlecleucel for the treatment of relapsed or refractory diffuse large B-cell lymphoma in Korea by Dong Hyun Kim, Soyean Kwon, Seungah Cha, Rabia Seyma Öz, Junshik Hong, Dong-Yeop Shin, Youngil Koh, Sung-Soo Yoon and Ja Min Byun in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
Parts of this study was presented at the 73rd Annual Meeting of the Korean Association of Internal Medicine, 2022, Seoul, Republic of Korea. We would like to thank all the medical staff at Seoul National University Hospital, especially Hyangseon Kim, Saemi Han, Jihye Lee, Nam Hee Koo, and Yeonji Kim, for their devotion and unwavering support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
