Abstract
Background:
While carfilzomib has shown effectiveness in treating relapsed or refractory multiple myeloma (RRMM), the best frequency of dosing is still debated. This meta-analysis aims to investigate the differences in safety and effectiveness between once-weekly and twice-weekly carfilzomib treatment schedules for patients with RRMM.
Methods:
A thorough search of five databases was performed. We calculated pooled relative risks (RRs), hazard ratios (HRs), and 95% confidence intervals (95% CIs), and conducted heterogeneity and sensitivity analyses using StateMP 18 software.
Results:
Five studies met the inclusion criteria. Analysis indicated that once-weekly carfilzomib significantly enhanced progression-free survival (HR: 0.80, 95% CI: 0.69–0.94, p = 0.007). However, no statistical difference was observed in the pooled RRs for overall response rate (RR: 1.13, 95% CI: 0.94–1.38, p = 0.198) and complete response or better (RR: 2.08, 95% CI: 0.65–6.65, p = 0.217). The once-weekly regimen was notably associated with a reduction in adverse events (RR: 0.98, 95% CI: 0.96–1.00, p = 0.047) relative to the twice-weekly regimen.
Conclusion:
The results propose once-weekly carfilzomib as a viable alternative treatment option for RRMM.
Introduction
Multiple myeloma (MM) is a type of cancer that predominantly affects the bone marrow, characterized by the excessive growth of clonal plasma cells. Globally, MM affects approximately 4 individuals per 100,000 annually, representing about 10% of all hematologic cancers. 1 Despite significant advancements in treatment, including the advent of novel immunomodulatory drugs (IMiDs) and proteasome inhibitors (PIs), MM remains an incurable disease. In recent years, the validation of molecular targets in MM has provided substantial translational insights and facilitated the development of innovative therapeutic approaches. These include monoclonal antibodies, small molecules, bispecific antibodies, and cell-based autologous immunotherapies such as chimeric antigen receptor T-cell therapy. 2 These innovative therapies have undoubtedly extended patient survival and enhanced quality of life, yet the disease’s recurrence remains a formidable challenge, 3 often resulting in relapsed or refractory multiple myeloma (RRMM).
In the past two decades, the development and approval of various therapeutic agents for MM have led to improved overall survival (OS) rates for both transplant-ineligible and transplant-eligible patients. 4 Bortezomib, a first-generation PI, has shown significant efficacy against MM, initially in relapsed cases and later as a frontline therapy. However, the side effect of peripheral neuropathy limits its long-term use.5,6 Carfilzomib, a next-generation PI, has emerged as a potent agent in the treatment of RRMM. Its efficacy has been recognized by regulatory bodies, including the European Medicines Agency and the FDA, which have approved its use in combination with lenalidomide–dexamethasone or dexamethasone for RRMM patients. 7 Carfilzomib demonstrated significantly superior efficacy compared to bortezomib. The phase III ENDEAVOR trial (carfilzomib plus dexamethasone (KD) vs bortezomib plus dexamethasone (VD)) revealed improved progression-free survival (PFS; hazard ratio (HR): 0.53, 95% CI: 0.44–0.65) and OS (HR: 0.79, 95% CI: 0.65–0.96) with carfilzomib.8,9 Furthermore, Berenson et al. 10 reported an overall response rate of 22.9% for carfilzomib in bortezomib-refractory patients. The promising results observed in relapsed settings have prompted investigations into carfilzomib’s potential role in initial treatment regimens for newly diagnosed MM patients, often alongside IMiDs, alkylating agents, or CD38 monoclonal antibodies.11–15
Although carfilzomib, administered twice weekly, has shown a favorable benefit–risk profile, patients and healthcare professionals may find the frequency of doses to be problematic. Frequent treatment sessions can lead to non-compliance or early therapy termination from the patient. Using clinical data from the CHAMPION-1 trial, 16 Moreau et al. 17 conducted the ARROW study. They discovered that giving carfilzomib once weekly at a dose of 70 mg/m² significantly increased PFS in comparison to the biweekly regimen, all while maintaining a similar safety profile and a reduced rate of adverse events (AEs). While, carfilzomib administered twice weekly, has shown a favorable benefit–risk profile, the frequency of dosing may pose challenges for patients and healthcare providers. The requirement for frequent and consecutive visits to infusion centers can result in treatment non-adherence or premature discontinuation.
While the ARROW trial directly compared twice-weekly and once-weekly carfilzomib dosing in a single study, no thorough meta-analysis has yet synthesized data across multiple trials to evaluate these regimens comprehensively. There is a need for more research because no thorough analysis has yet directly compared twice-weekly and once-weekly carfilzomib dosage for RRMM. This research undertook a comprehensive review of the current literature, employing meta-analytical techniques to aggregate and interpret findings from pertinent investigations. It compared efficacy (such as PFS or response rates), safety (such as frequency and severity of AEs), and patient quality of life, aiming to provide clinicians with robust evidence to guide personalized treatment decisions.
Methods
Search strategy and selection criteria
The study was registered in the PROSPERO database with the ID CRD42024560605 and followed the requirements established by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 18 Using PubMed, Web of Science, Cochrane, and Embase databases, a thorough search was done for English-language publications published up until August 1, 2024. The search strategy employed various terms related to MM and carfilzomib, including but not limited to “Multiple Myeloma*,” “Myelomatos*,” “Plasma Cell Myeloma*,” “Kahler Disease,” “Myeloma-Multiple*,” “morbus kahler,” “myeloma multiplex,” “PR-171,” “fp 101,” “ono 7057,” and “carfilzomib.” Detailed search strategies are accessible in Supplemental Material 1. To ensure exhaustive coverage, the bibliographies of relevant publications were also meticulously examined through manual inspection.
Study selection
Subsequent to the elimination of duplicate records, two evaluators conducted an independent assessment of the remaining titles and abstracts. In instances of divergent opinions, a tertiary assessor was engaged to provide additional insight. Studies that satisfied the following requirements were included: (1) randomized controlled trail (RCT) studies or cohort studies; (2) focused on patients with RRMM; (3) carfilzomib regimens administered once or twice a week were compared; and (4) HRs were supplied for at least one of the survival endpoints (OS or PFS). Treatment-related AEs and the objective response rate (ORR) were secondary objectives. If there were several publications about the same trial, only the most up-to-date and useful ones were listed. Exclusion criteria encompassed: (1) reviews, comments, abstracts, case reports, and animal experiments; (2) non-English publications; (3) duplicate research; and (4) studies with fewer than five patients.
Risk of bias assessment
The quality of RCTs was assessed using the previously detailed 5-item Jadad scale, which examines aspects such as randomization, participant withdrawals, and double-blinding. For cohort studies, the Newcastle–Ottawa Scale was employed to appraise the quality of evidence.
Data extraction
Two investigators separately conducted a preliminary review of potentially relevant study titles and abstracts. Afterward, they retrieved and examined full-text publications of the chosen research. A third senior investigator was consulted to settle any disagreements. Studies with proven overlap were removed, and corresponding authors were contacted for clarification in cases where cohort overlap was suspected.
Data extraction was performed using standardized, pilot-tested forms by the same pair of investigators. A tertiary investigator subsequently corroborated all extracted data. The data encompassed patient age, treatment regimens, geographical origin, sample size, publication year, study design, author names, and pertinent outcomes. Primary outcomes of interest included AEs, the incidence of grade ⩾3 AEs, PFS, ORR, and complete response (CR) or better.
Statistical analysis
Results for PFS analyses were given as HRs with 95% confidence intervals (CIs). CR or better, AEs, and ORR were all assigned pooled relative risks (RRs) with 95% CIs. Forest plots were generated to visualize the effect size of each included study, and a summary effect was calculated using an appropriate effects model. The heterogeneity between trials was assessed using the I2 statistics based on χ2 and the Q statistic. For pooling effect sizes, random-effects or fixed-effects models were chosen according to the I2 value. A random-effects model was applied when tolerable heterogeneity (I2 < 50%) was noted; a fixed-effects model was applied when significant heterogeneity (I2 > 50%) was discovered. A sensitivity analysis was conducted to assess the stability of the combined effect sizes by systematically excluding individual studies. In instances where the number of included studies surpassed 10, Egger’s test was utilized to evaluate potential publication bias. All statistical analyses were performed using StateMP 18 software.
Results
Literature search outcomes
The systematic search identified a total of 7033 initial records. Following the removal of 2862 duplicates and the exclusion of 4122 records based on title and abstract screening, the remaining studies were subjected to a comprehensive full-text review. A stringent application of the inclusion and exclusion criteria led to the final selection of five studies. The detailed selection process is depicted in Figure 1.17,19–22

Study selection process.
Study characteristics and quality assessment
The five studies included in this review, published between 2018 and 2024, consisted of RCTs and cohort studies. Table 1 provides a summary of the key characteristics of these studies. The quality assessment indicated that the two RCTs were classified as having a low risk of bias, whereas the three cohort studies received quality scores exceeding 7, signifying high methodological quality. The detailed results of the quality assessment are presented in Figure 2(a) and (b) and Table 2.
Characteristics of the included studies.
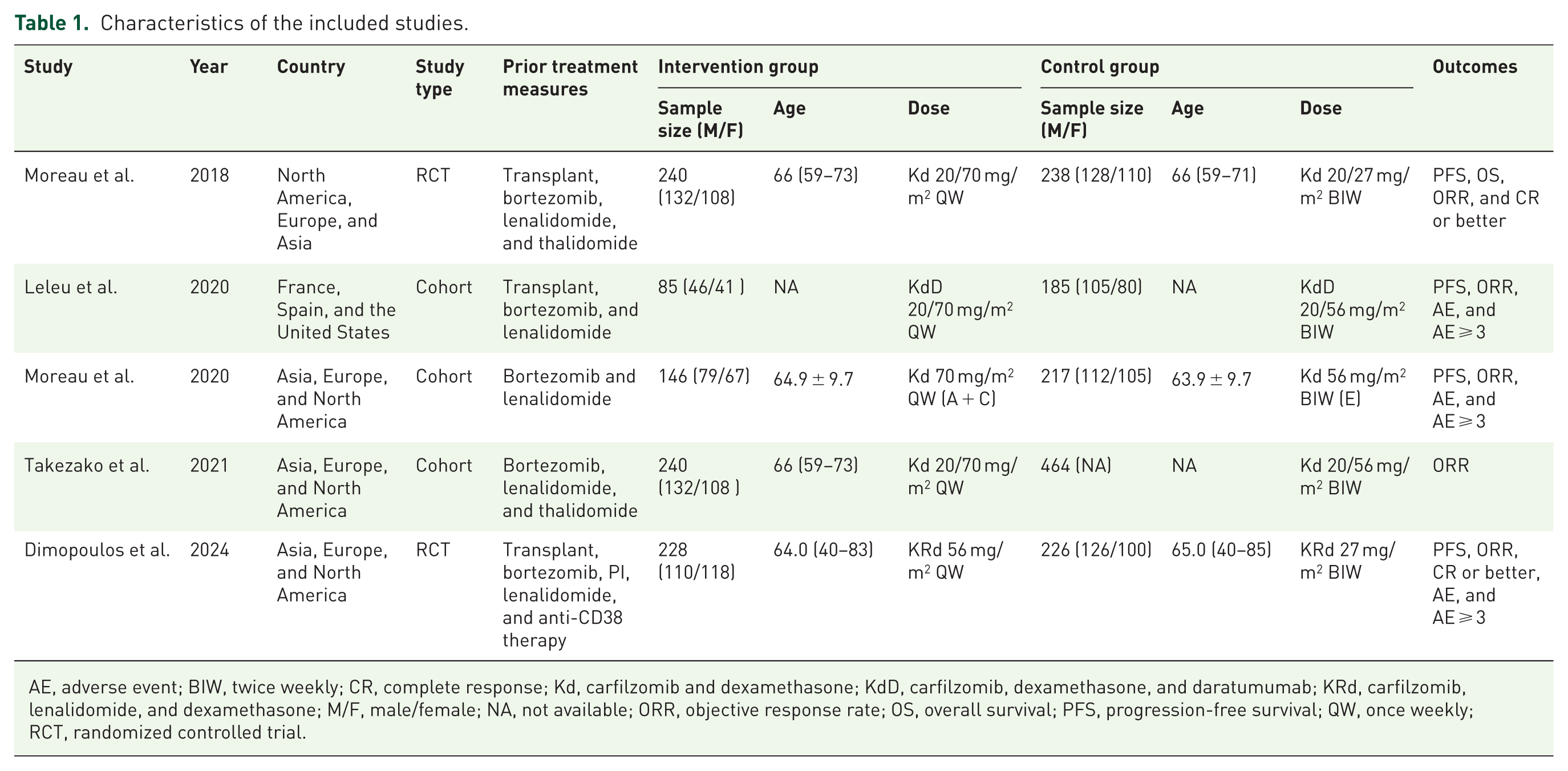
AE, adverse event; BIW, twice weekly; CR, complete response; Kd, carfilzomib and dexamethasone; KdD, carfilzomib, dexamethasone, and daratumumab; KRd, carfilzomib, lenalidomide, and dexamethasone; M/F, male/female; NA, not available; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; QW, once weekly; RCT, randomized controlled trial.

Summary of the risk of bias assessments for cohort studies.
Summary of the risk of bias assessments for RCT studies.
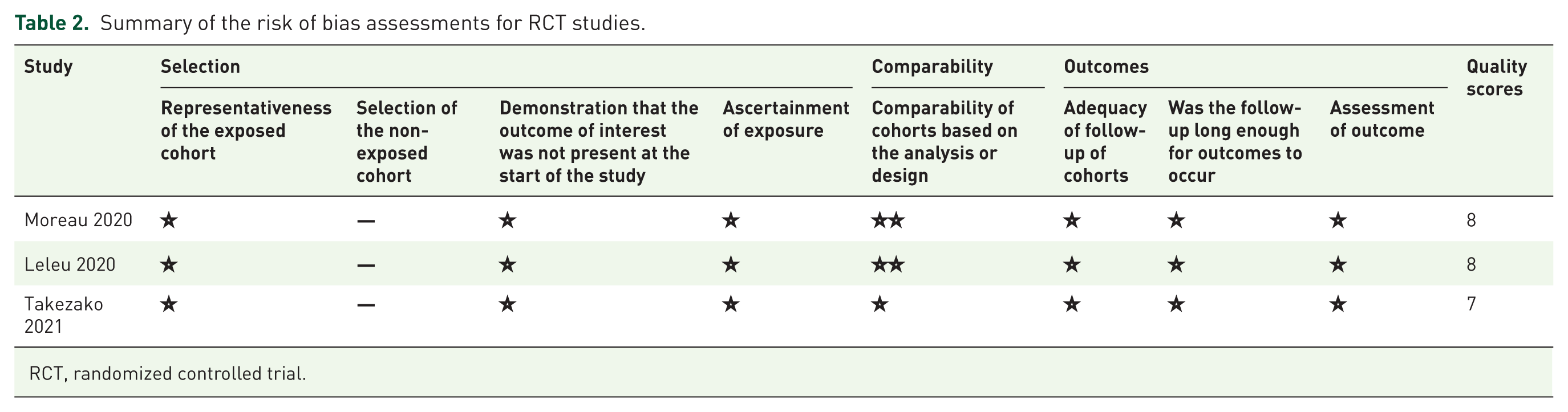
RCT, randomized controlled trial.
Meta-analysis results
Progression-free survival
There were four trials with PFS data available. Once-weekly carfilzomib administration showed a substantial improvement in PFS compared to twice-weekly administration (HR: 0.80, 95% CI: 0.69–0.94, p = 0.007, Figure 3), according to a fixed-effects model (I2 = 2.79%, p = 0.425).
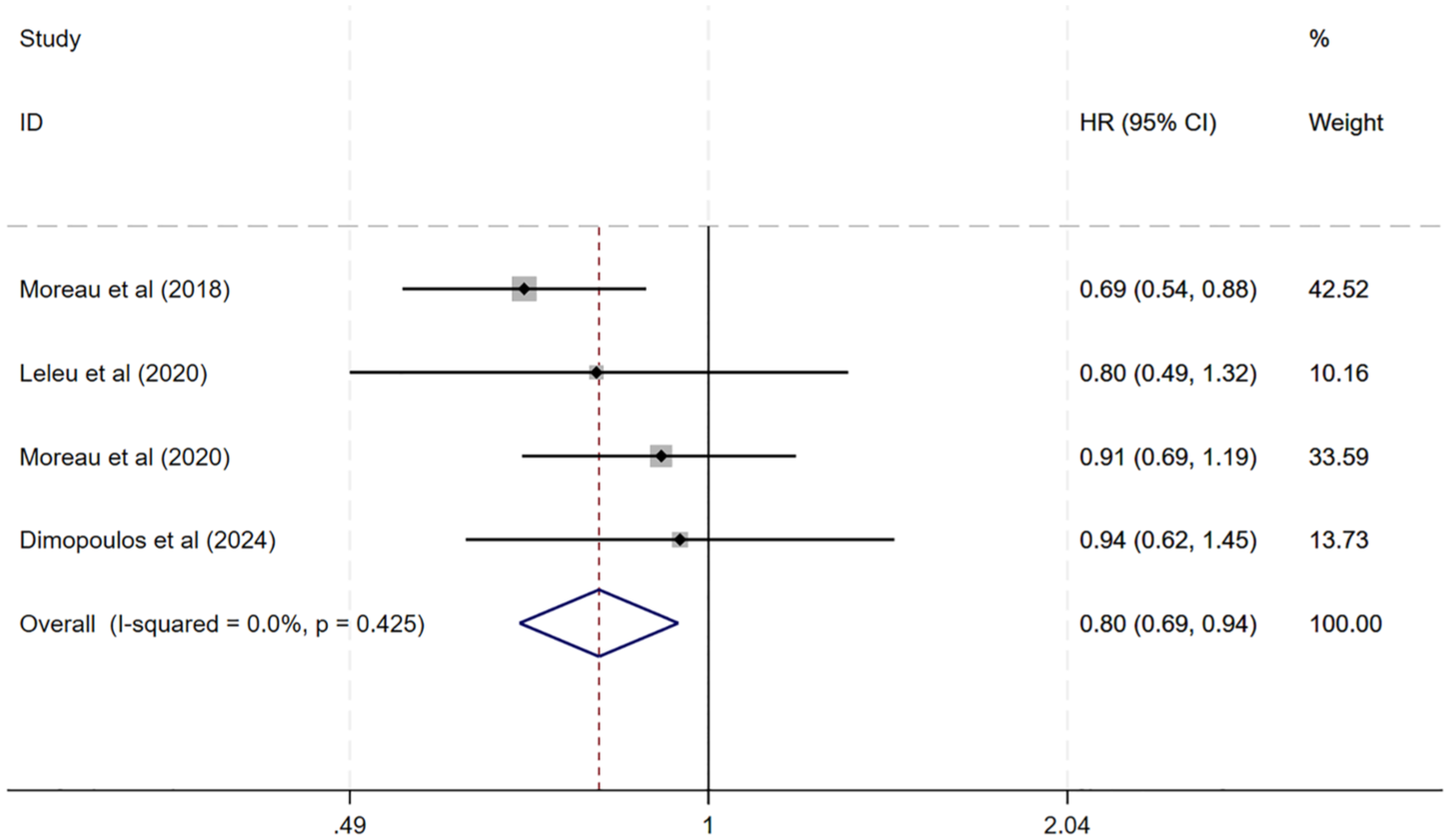
Forest plot of PFS.
ORR and CR or better
All five studies provided ORR results. No statistical difference between once-weekly and twice-weekly carfilzomib regimens was observed, according to the pooled RR for ORR, which was determined using a random-effects model (I2 = 35.79%, p < 0.001; RR: 1.14, 95% CI: 0.94–1.38, p = 0.198, Figure 4(a)).

Forest plots: (a) forest plot of ORR and (b) forest plot of CR or better.
Two clinical trials provided data regarding the rates of CR or better. The meta-analysis employing a random-effects model (I2 = 4.68%, p = 0.031) did not reveal a significant advantage for once-weekly administration of carfilzomib in achieving CR or better (RR: 2.08, 95% CI: 0.65–6.65, p = 0.217, Figure 4(b)).
Safety profile
The safety assessment of carfilzomib regimens primarily focused on AEs. Utilizing a fixed-effects model, our analysis revealed that twice-weekly carfilzomib administration was linked to a marginally higher AEs than once-weekly dosing (RR: 0.98, 95% CI: 0.96–1.00, p = 0.047, Figure 5). Nevertheless, no statistical difference was noted between the two dosing regimens when analyzing grade ⩾3 AEs in RRMM patients (RR: 0.98; 95% CI: 0.88–1.09, p = 0.713, Figure 6). The pooled RR for cardiac failure grade ⩾3 was 0.60 (95% CI: 0.30–1.22, p = 0.16, Figure 7), indicating no significant difference between regimens. The absence of a statistical difference in the total AE rates between the two regimens was further supported by a random-effects model analysis.
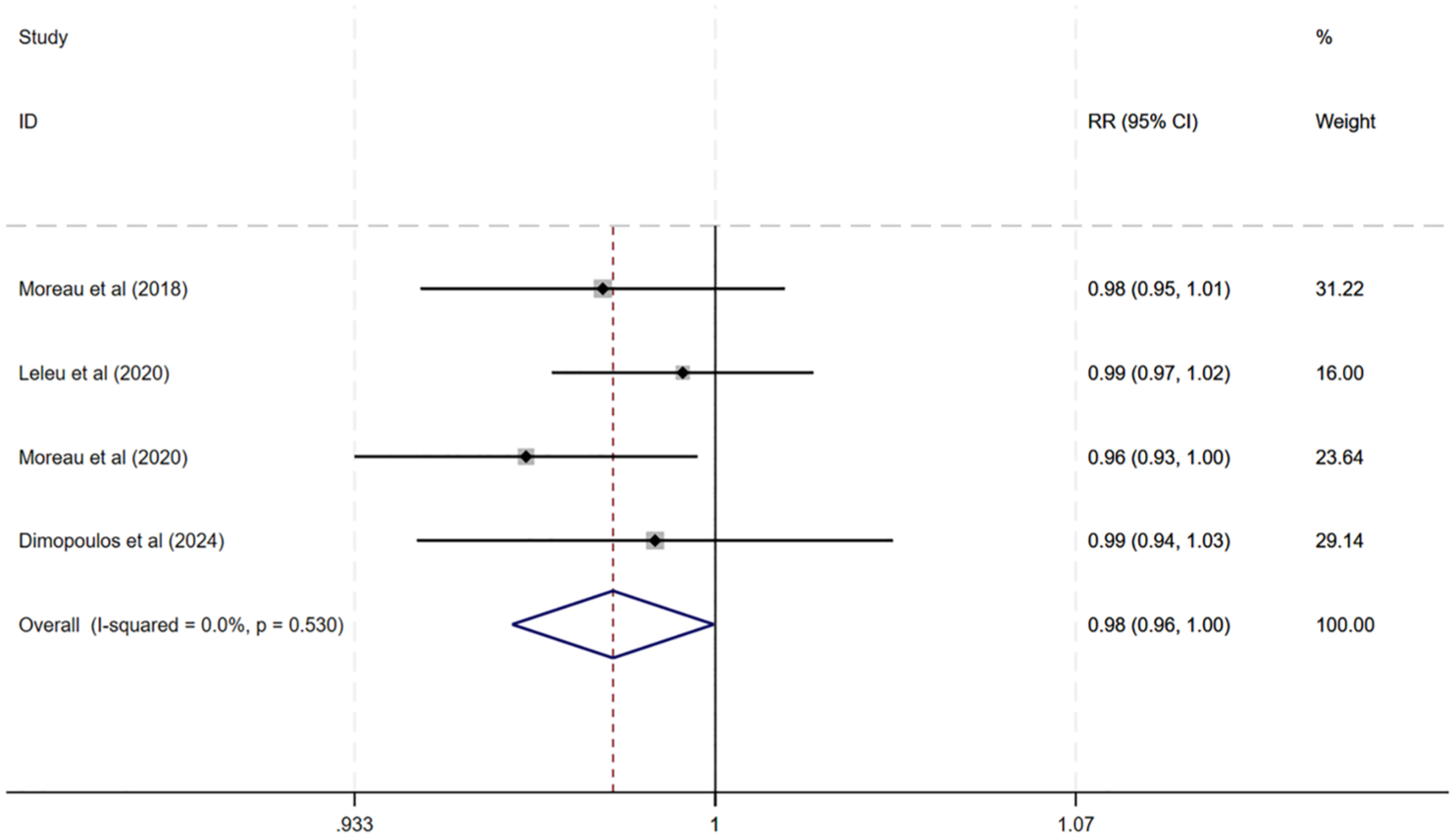
Forest plot of AE.

Forest plot of ⩾3 AEs.

Forest plot of ⩾3 cardiac failure.
Sensitivity analysis
We gradually removed specific studies from our findings to do a sensitivity analysis and assess their robustness. Significant heterogeneity among the included studies was not found by this procedure (Figure 8). There were not enough studies that fit our inclusion criteria; thus, we did not investigate publication bias.
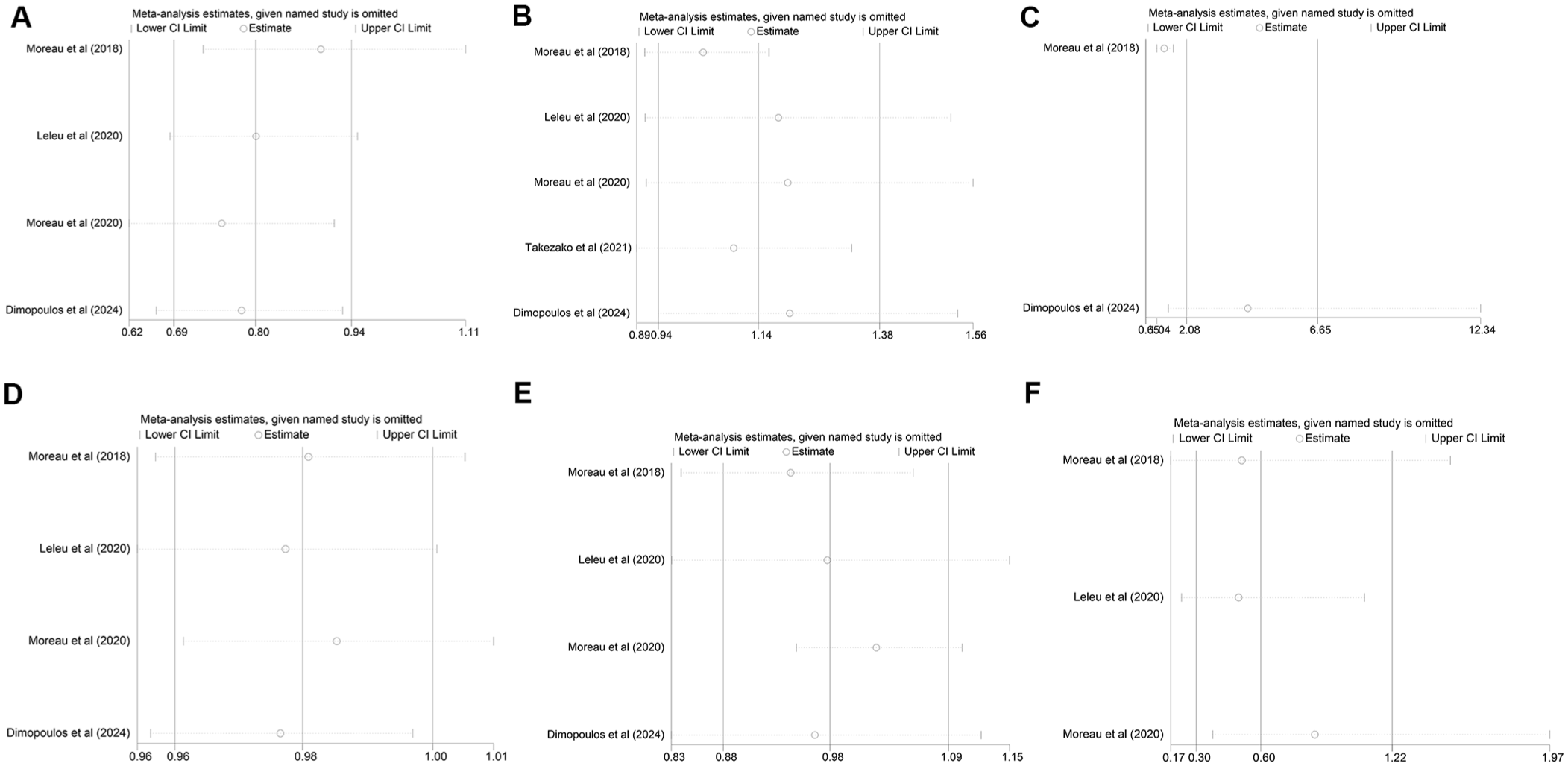
Sensitivity analysis of PFS (a), ORR (b), CR or better (c), AE (d), ⩾3 AEs (e), and ⩾3 cardiac failures (f).
Analysis of OS
Most studies did not evaluate OS as a primary endpoint. However, two studies specifically reported OS outcomes. In the interim analysis of the ARROW study 17 OS data were not yet mature, and patients continue to be followed up for mortality assessment until study completion. A trend toward improved survival was observed in the once-weekly dosing group compared to the twice-weekly dosing group, with a higher 12-month OS rate in the once-weekly group (76.6%) than in the twice-weekly group (71.9%). Similarly, findings by Bringhen et al. 23 indicated no significant difference in 3-year OS was observed between the once-weekly (70%) and twice-weekly (72%) groups (p = 0.5).
Discussion
To our knowledge, this is the first meta-analysis comprehensively comparing once-weekly and twice-weekly carfilzomib regimens in RRMM. According to our research, RRMM patients who receive carfilzomib once a week have noticeably longer PFS and fewer total AEs. Between the two dosing schedules, we did not find any appreciable variations in OS, ORR, PR, or the frequency of grade ⩾3 AEs.
The findings of Zhou et al. aligned with our results, demonstrating that a higher dose of carfilzomib achieved superior overall remission rates and prolonged PFS compared to low doses. 24 However, our analysis indicated that once-weekly high-dose carfilzomib did not significantly influence OS in heavily treated RRMM patients, suggesting that the regimen may not consistently impact long-term outcomes in these populations. It is plausible that disease progression under carfilzomib-based therapy may involve mechanisms beyond proteasome inhibition, such as adverse cytogenetics, extramedullary disease, and epigenetic alterations, highlighting the need for further investigation into potential resistance mechanisms.25–27 In addition, the absence of OS differences may reflect heterogeneity in subsequent therapies received by patients after progression, which were not systematically controlled across studies.
When treating RRMM, a once-weekly carfilzomib regimen has demonstrated a good tolerance and safety profile. The once-weekly and twice-weekly regimens showed similar incidence rates of grade ⩾3 treatment-emergent AEs, according to our analysis. Interestingly, compared to individuals on the twice-weekly regimen, patients on a once-weekly carfilzomib regimen experienced fewer overall AEs. Considering the possible effects of AEs on the benefit–risk ratio and the quality of life of patients, this discovery is especially important. Besides, our findings underscore that the once-weekly schedule demonstrated a statistically significant reduction in total AEs, while no significant difference was observed in the incidence of grade ⩾3 AEs. No significant differences were observed in the incidence of grade ⩾3 cardiac failure either.
The discrepancies observed between Moreau et al.’s 2018 and 2020 studies may be attributed to variations in control group design, particularly in dose intensity. These studies compared Kd 27 mg/m2 administered twice weekly with Kd 56 mg/m2 administered twice weekly, which may explain the divergent outcomes. Notwithstanding these discrepancies, our results clearly advocate for the administration of once-weekly carfilzomib therapy to patients with RRMM, as it demonstrates improved PFS while maintaining a tolerable safety profile.
Notably, the once-weekly regimen (70 mg/m2 on days 1, 8, and 15 of a 28-day cycle) delivers a higher cumulative dose per cycle (210 mg/m2) compared to the twice-weekly regimen (27 mg/m2 on days 1, 2, 8, 9, 15, and 16; 162 mg/m2 total). Despite this increased dose intensity, the once-weekly schedule demonstrated improved tolerability, enhancing therapeutic efficacy while reducing toxicity.
The safety and effectiveness of once-weekly carfilzomib for patients with RRMM have been repeatedly confirmed by prior clinical trials.17,28 The phase III ARROW study’s interim results were noteworthy since they showed a median PFS of 7.6 months within twice-weekly dosing, compared to 11.2 months with once-weekly scheduling. 17 Moreover, in line with our results, phase Ib EQUULEUS research examined a once-weekly regimen of daratumumab–carfilzomib–dexamethasone and showed acceptable tolerability and durable responses. 28 These collective results substantiate the therapeutic efficacy of once-weekly carfilzomib in RRMM, underscoring its potential to enhance treatment efficacy and safety, improve patient quality of life, and potentially extend survival. Based on its safety, tolerability, and effectiveness, we advocate for the adoption of once-weekly carfilzomib as a preferred therapeutic strategy for RRMM patients.
Despite evaluating combination regimens from multiple perspectives, our study still has some limitations. First, the limited number of included articles, particularly RCTs featuring direct data comparisons, might introduce bias, despite attempts at population matching. Second, the diversity in patient populations—ranging from those having undergone one to three prior therapies—may contribute to increased heterogeneity among trials. Third, discrepancies in follow-up durations and immature survival data at the time of analysis hinder the feasibility of comprehensive meta-analyses for some endpoints. Lastly, the limited number of available studies precluded a formal publication bias analysis. Further prospective clinical trials are necessary to strengthen the evidence base and validate these findings.
Conclusion
This meta-analysis confirms that once-weekly carfilzomib improves PFS compared to its twice-weekly counterpart, offering enhanced treatment convenience without compromising efficacy, though showing no significant differences in OS, ORR, or AEs. Notably, the once-weekly regimen demonstrated improved tolerability with fewer AEs despite higher cumulative dose intensity, further supporting its clinical utility. Our findings support the selection of once-weekly carfilzomib to maximize proteasome inhibition and optimize anti-MM effects. Besides, future research should include larger, multicenter RCTs to validate these findings and explore strategies to minimize and manage AEs effectively.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251359650 – Supplemental material for Comparative analysis of once-weekly versus twice-weekly carfilzomib in relapsed and refractory multiple myeloma: a meta-analysis
Supplemental material, sj-docx-1-tah-10.1177_20406207251359650 for Comparative analysis of once-weekly versus twice-weekly carfilzomib in relapsed and refractory multiple myeloma: a meta-analysis by Wenhao Yang, Xiangtu Kong, Hui Yu, Xiaosu Feng and Haiwen Ni in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207251359650 – Supplemental material for Comparative analysis of once-weekly versus twice-weekly carfilzomib in relapsed and refractory multiple myeloma: a meta-analysis
Supplemental material, sj-docx-2-tah-10.1177_20406207251359650 for Comparative analysis of once-weekly versus twice-weekly carfilzomib in relapsed and refractory multiple myeloma: a meta-analysis by Wenhao Yang, Xiangtu Kong, Hui Yu, Xiaosu Feng and Haiwen Ni in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
