Abstract
Background:
Ixazomib is an oral proteasome inhibitor for relapsed/refractory multiple myeloma (RRMM). Our study aimed to analyze the efficacy and tolerability of ixazomib-based combination therapies.
Methods:
We performed a single-center retrospective analysis of 126 patients with RRMM and other plasma cell neoplasms.
Results:
The median age was 65 years, with a median of two prior therapy lines, and 16.7% were triple-class refractory. The overall response rate (ORR) was 52.5%; triple-class refractory patients had a significantly lower ORR than non-refractory controls (10.5% vs 60.2%,
Conclusion:
Altogether, ixazomib regimens are potent in RRMM but are less effective in heavily pretreated patients and those with renal impairment, suggesting earlier use may yield greater benefits.
Introduction
The inhibition of the proteasome represents a major strategy in the therapy of multiple myeloma (MM).
1
Currently, the three proteasome inhibitors (PIs) bortezomib, carfilzomib and ixazomib are commercially available for the treatment of newly diagnosed (ND) and/or relapsed/refractory (RR) MM, and are already integrated into combination regimens for clinical routine.2–10 Ixazomib is the first oral PI, which reversibly inhibits the β5 subunit (chymotrypsin-like) of the 20S proteasome.
11
In the phase III TOURMALINE-MM1 trial, the addition of ixazomib to the combination of lenalidomide and dexamethasone significantly increased the overall response rate (ORR) and improved the progression-free survival (PFS) in RRMM (median PFS: 20.6 months in ixazomib vs 14.7 months in placebo group; hazard ratio (HR): 0.74,
Methods
Patient characteristics
We conducted a single-center retrospective study of 126 patients with RRMM and other plasma cell neoplasms, who were treated with ixazomib-containing therapies between March 2016 and January 2024. All procedures adhered to the Declaration of Helsinki and Good Clinical Practice guidelines. Informed consent was obtained from all patients included in the analysis. Given the retrospective design of this study, the Ethics Committee of the University of Würzburg waived the need for additional approval. Collected data included demographics, cytogenetics, prior treatments, therapy regimens, responses, adverse events, and survival outcomes. High-risk cytogenetics were determined by fluorescence in situ hybridization and defined as the presence of at least one of the following abnormalities: t(4;14), +1q21, or del17p. 18 Extramedullary disease (EMD) was assessed by positron emission tomography/computed tomography or whole-body diffusion-weighted magnetic resonance imaging.
Treatment schedules and dosing
Patients received ixazomib orally on days 1, 8, and 15 of a 4-week cycle, with cycles repeated on day 29. In regimens containing immunomodulatory drugs (IMiDs) such as Ixa-Rd (lenalidomide–dexamethasone), Ixa-Pd (pomalidomide–dexamethasone), and Ixa-Td (thalidomide–dexamethasone), IMiDs were administered orally once daily on days 1–21. Drug dosing was determined at the discretion of the treating physician. Alternative IMiD-free regimens in this study included Ixa-Ed (ixazomib–elotuzumab–dexamethasone), Ixa-Dd (ixazomib–daratumumab–dexamethasone), Ixa-Cd (ixazomib–cyclophosphamide–dexamethasone), or Ixa-d (ixazomib–dexamethasone). Elotuzumab and daratumumab were administered intravenously or subcutaneously as per prescribing information. Cyclophosphamide dosing is detailed in Table 1. Except for ixazomib monotherapy, dexamethasone was given orally or intravenously on ixazomib treatment days. Therapy continued until disease progression or intolerable toxicity. Patients received prophylaxis against
Therapy regimens.

Ixa-Cd, ixazomib–cyclophosphamide–dexamethasone; Ixa-d, ixazomib–dexamethasone; Ixa-Dd, ixazomib–daratumumab–dexamethasone; Ixa-Ed, ixazomib–elotuzumab–dexamethasone; Ixa-Pd, ixazomib–pomalidomide–dexamethasone; Ixa-Rd, ixazomib–lenalidomide–dexamethasone; Ixa-Td, ixazomib–thalidomide–dexamethasone.
Response assessment and adverse events
Response to treatment was evaluated using the current International Myeloma Working Group (IMWG) Response Criteria. 20 Overall survival (OS) was measured from the start of ixazomib-containing therapy to death or last follow-up. PFS was defined as the time from therapy initiation to relapse, progression, or the last follow-up if no relapse or progression occurred. Adverse events were classified according to the Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0.
Statistical analysis
For descriptive statistics, unless otherwise specified, data were reported as absolute numbers with percentages or medians with ranges. Comparisons between subgroups were conducted using the Mann–Whitney
Results
Patient characteristics
Most patients were male (
Patient characteristics and response to treatment.
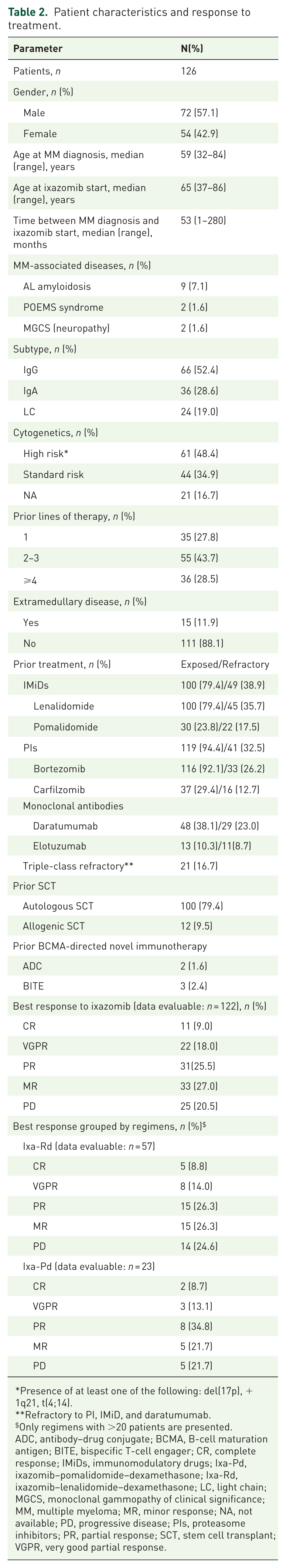
Presence of at least one of the following: del(17p), +1q21, t(4;14).
Refractory to PI, IMiD, and daratumumab.
Only regimens with >20 patients are presented.
ADC, antibody–drug conjugate; BCMA, B-cell maturation antigen; BITE, bispecific T-cell engager; CR, complete response; IMiDs, immunomodulatory drugs; Ixa-Pd, ixazomib–pomalidomide–dexamethasone; Ixa-Rd, ixazomib–lenalidomide–dexamethasone; LC, light chain; MGCS, monoclonal gammopathy of clinical significance; MM, multiple myeloma; MR, minor response; NA, not available; PD, progressive disease; PIs, proteasome inhibitors; PR, partial response; SCT, stem cell transplant; VGPR, very good partial response.
Regimens and dosing
In 112 (88.9%) patients, ixazomib was administered as a treatment for MM relapse. Most patients (
Response to therapy
Among the 122 patients (96.8%) with available response data, the ORR was 52.5%, with 11 (9.0%), 22 (18.0%), and 31 (25.5%) patients achieving complete response (CR), very good partial response (VGPR), and partial response (PR), respectively. In addition, 33 (27.0%) patients achieved a minor response (MR), resulting in a clinical benefit rate (CBR) of 79.5%. Patients receiving ixazomib in the second line demonstrated a significantly higher ORR compared to those receiving ixazomib at later lines (77.1% vs 42.5%,
The ORR in Ixa-Rd-treated patients (
Survival analyses
At a median follow-up of 27.0 months, the PFS and OS for the entire cohort were 7.9 months (95% CI: 6.9–11.0) and 84.1 months (95% CI: 68.1–not reached), respectively (Figure 1(a) and (b)). No significant differences in PFS or OS were observed between patients receiving IMiD-containing versus IMiD-free combinations (both
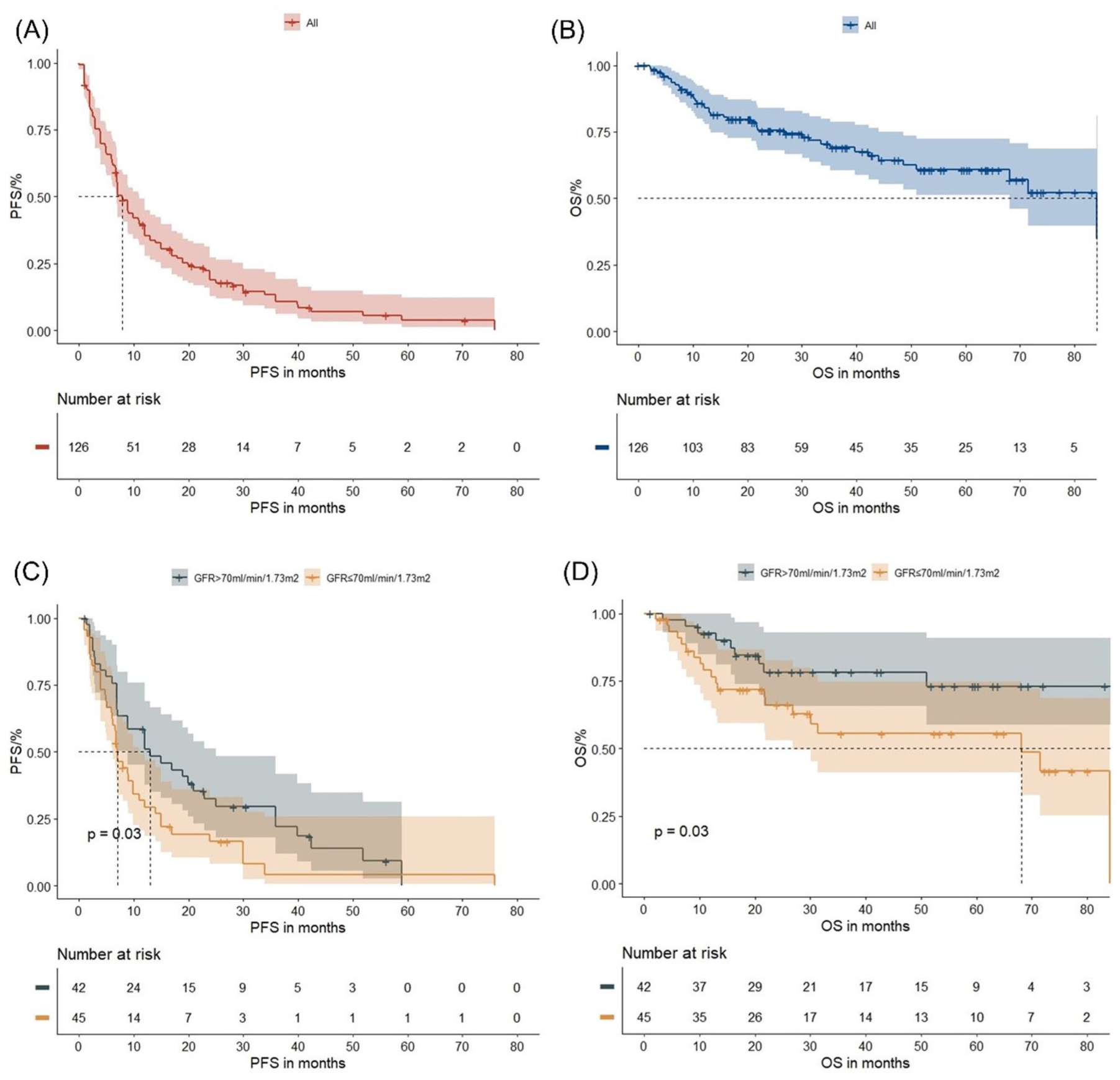
Survival outcomes and renal function in the entire group: The figures illustrate the PFS and OS in the entire group (a, b) and in patients with GFR >70 versus ⩽70 ml/min/1.73 m2 (c, d).
As expected, clinical responders (PR or better) had significantly longer PFS (18.9 months vs 3.5 months,
Patients with >1 prior line of therapy had significantly shorter PFS (6.9 months vs 17.0 months,

Survival outcomes and pretreatment in the entire group: The figures demonstrate the PFS and OS in PI-refractory versus non-PI-refractory patients (a, b), and in patients being triple-class refractory versus not triple-class refractory (c, d).
Multivariate survival analysis identified a GFR ⩽70 ml/min/1.73 m2 as an independent prognostic risk factor for worse PFS (HR: 2.11, 95% CI: 1.21–3.70,

Multivariate survival analysis in the entire group: This figure displays the prognostic roles of refractoriness, renal function, EMD, cytogenetics, and age of patients in the entire cohort. Hazard ratio, 95% confidence interval, and
In the Ixa-Rd subgroup, median PFS and OS were 7.0 months (95% CI: 4.0–11.0) and 71.5 months (95% CI: 39.7–not reached), respectively (Figure 4(a) and (b)). Responders to Ixa-Rd had significantly longer PFS (19.9 months vs 3.0 months,
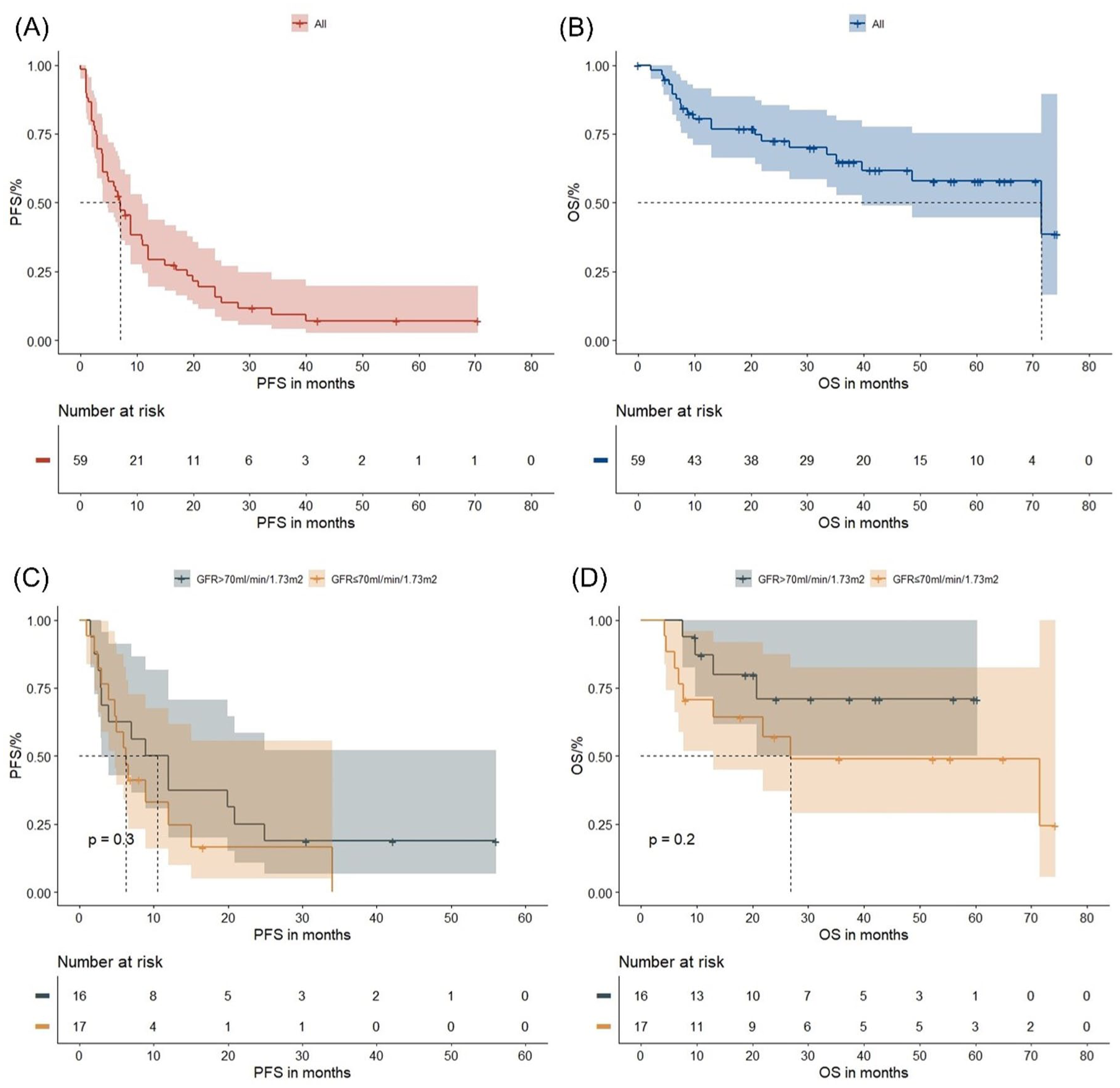
Survival outcomes and renal function in the subgroup Ixa-Rd: The figures illustrate the PFS and OS in the entire group (a, b), and in patients with GFR >70 versus ⩽70 ml/min/1.73 m2 (c, d).

Survival outcomes and pretreatment in the subgroup Ixa-Rd: The figure demonstrates the PFS and OS in PI-refractory versus non-PI-refractory patients (a, b), and in patients being triple-class refractory versus not triple-class refractory (c, d).
Adverse events
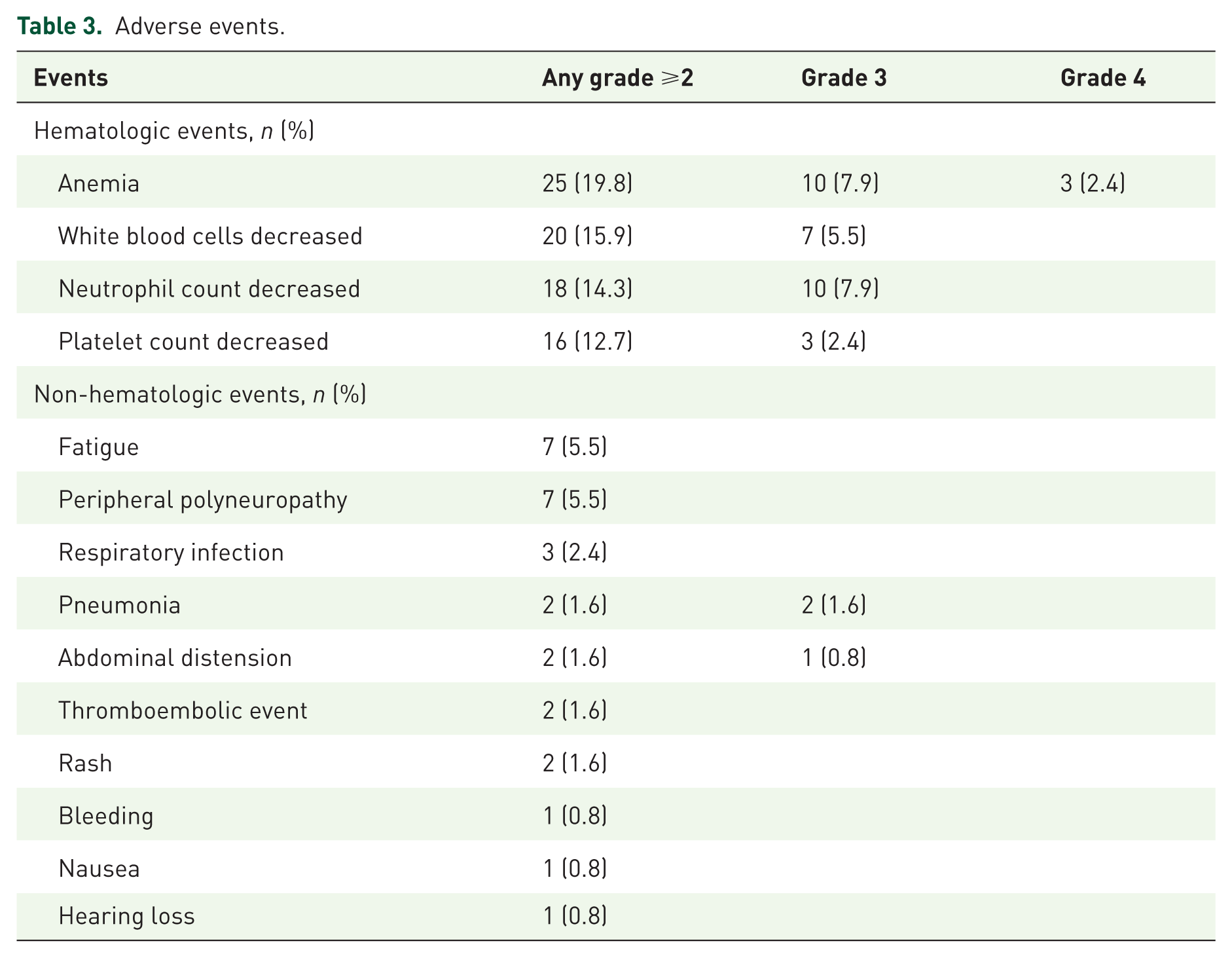
Overall, adverse events in this real-world cohort were mild and in line with previous reports on ixazomib toxicity.2–4 Grade ⩾2 hematologic adverse events were reported in 40 patients (31.7%), including grade ⩾3 in 19 (15.1%) patients (7.9% anemia, 5.5% leukopenia, 4.8% thrombocytopenia). Grade 4 thrombocytopenia was observed in three patients (2.4%), all of whom were treated with IMiD-containing combinations (Ixa-Rd:
Adverse events.
To note, patients treated with IMiD combinations had significantly lower nadir white blood cell (WBC) counts (median: 3.6 vs 4.4 ×1000/µl,

Nadir blood count in IMiD-containing versus IMiD-free regimens: The figure displays the nadir blood counts, that is, WBC (a), Hb (b), PLT (c), and ANC (d), in patients treated with IMiD-containing versus IMiD-free ixazomib combinations. The dashed red lines show the cut-off values for grade 3 hematologic toxicities according to the CTCAE classification version 5.0.
Among patients receiving IMiD regimens, those on Ixa-Pd had significantly lower nadir ANC compared to Ixa-Rd (median: 0.9 vs 2.2 ×1000/µl,

Nadir blood counts in different IMiD-containing regimens: The figure displays the nadir blood counts, that is, WBC (a), Hb (b), PLT (c), and ANC (d), in patients treated with different IMiD-containing ixazomib combinations. The dashed red lines show the cut-off values for grade 3 hematotoxicities according to the CTCAE classification version 5.0.
Non-hematologic adverse events (any grade) were documented in 46 patients (36.5%), with fatigue (
Discussion
Real-world experience with ixazomib-containing therapies, especially combinations beyond Ixa-Rd, remains limited in relapsed/refractory multiple myeloma (RRMM). 11 In this study, we therefore analyzed clinical data from 126 RRMM patients treated with ixazomib-containing regimens.
The ORR in our study was 52.5% for the entire cohort and 49.1% for the Ixa-Rd subgroup, both significantly lower than those reported in the pivotal TOURMALINE-MM1 trial, which led to the approval of Ixa-Rd in RRMM. 12 Furthermore, other real-world studies on Ixa-Rd have reported ORRs as high as 85%.21,22 Notably, most patients in these studies, including TOURMALINE-MM1, had received only 1–3 prior lines of therapy. By contrast, our cohort included a substantial proportion of heavily pretreated patients, with 36 (28.6%) receiving >3 prior therapies, 41 (32.5%) being refractory to other PIs, and 21 (16.7%) being classified as triple-class refractory. Consistently, both in the entire cohort and the Ixa-Rd subgroup, we observed a significantly lower ORR among heavily pretreated patients (those with >3 prior therapies, PI refractory, or triple-class refractory) compared to those with just one prior therapy. In the REMIX study evaluating real-world outcomes of Ixa-Rd, the ORR was 73.1%, but dropped to 54.4% in patients treated in fourth-line or later settings. 23
To the best of our knowledge, no real-world studies on ixazomib-containing therapies other than Ixa-Rd have been published in RRMM. In our cohort, 25 patients received Ixa-Pd, all of whom had prior exposure to lenalidomide and a PI. The ORR in this subgroup was 56.5%, which is comparable to results from two phase I/II trials (ORRs of 48.0% and 51.7%) investigating Ixa-Pd in RRMM.14,24 Overall, ixazomib-containing regimens, including Ixa-Rd and Ixa-Pd, demonstrated efficacy in RRMM, but their response rates diminished in heavily pretreated patients with >3 prior lines of therapy, suggesting that these regimens should be administered earlier in the treatment course.
In terms of survival outcomes, the median PFS for the entire cohort was 7.9 months, similar to the PFS observed in the Ixa-Rd subgroup (7.0 months). This is notably lower than the median PFS reported in the TOURMALINE-MM1 trial (20.6 months) and other real-world Ixa-Rd studies (up to 43 months), likely reflecting the more heavily pretreated nature of our cohort.12,21,22 In the Ixa-Pd subgroup, the median PFS was 7.0 months, aligning with phase I/II trial results from Krishnan et al. (8.6 months) and Voorhees et al. (4.4 months), where patients had also been exposed to PIs and were refractory to lenalidomide.14,24 Multivariate analysis identified impaired renal function (GFR ⩽70 ml/min/1.73 m2) and triple-class refractory disease as negative prognostic factors for both PFS and OS, highlighting the challenges of managing heavily pretreated, multi-refractory patients with renal impairment. In this context, ixazomib-based treatments do not appear to provide a substantial therapeutic advancement.
Overall, the treatment was well tolerated. Grade ⩾3 hematologic adverse events were observed in 19 patients (15.1%). Grade ⩾3 neutropenia and leukopenia were largely attributable to IMiD use, particularly pomalidomide, as evidenced by our finding that nadir WBC and ANCs were significantly lower in IMiD-containing ixazomib regimens compared to IMiD-free regimens. Importantly, the global rate of infection was very low in our study, even in patients treated with Ixa-Pd, most likely due to the universal use of co-trimoxazole prophylaxis. PN was the most common non-hematologic adverse event, occurring in 27 patients (21.4%) with grade 1 and in 6 patients (4.8%) with grade 2. Preexisting PN related to POEMS syndrome, thalidomide, or bortezomib treatment likely contributed to PN development, complicating the assessment of ixazomib’s role in PN. Interestingly, in three bortezomib-pretreated patients, PN symptoms resolved during ixazomib treatment. The underlying mechanisms of PN associated with PIs like bortezomib and ixazomib warrant further investigation.25,26 Comparative analyses between different regimens in this study were limited by small subgroup sizes, such as for Ixa-Td, Ixa-Cd, and Ixa-d.
Currently, the optimal positioning of ixazomib within the treatment landscape of MM has not been fully defined. Chimeric antigen-receptor (CAR) T-cell therapy and anti-CD38 antibodies are moving toward the earlier therapy lines, for example, ciltacabtagene autoleucel is now approved by EMA and FDA for MM patients at first relapse. In turn, an increasing number of lenalidomide- or anti-CD38 antibody-refractory patients are expected after first-line treatment. Therefore, PI-based regimens will be an option to bridge to CAR T-cell therapy in the second line. Due to its availability as an oral drug and given its manageable safety profile, ixazomib could be particularly attractive for elderly patients, also in later lines of therapy. In addition, ixazomib may depict another option as maintenance after first-line therapy, potentially also to provide an alternative to long-term lenalidomide and its detrimental effects on secondary primary malignancies, including secondary myelodysplastic neoplasm and acute myeloid leukemia.27,28
Our study has several limitations. First, this is a retrospective study based on a relatively small and heterogeneous patient cohort, including a drop-out rate and follow-up, which are intrinsically limited as typically seen in real-world studies. Second, high-risk cytogenetics were defined as per the R2-ISS classification and did not contain
Conclusion
Our real-world experience indicates that ixazomib-containing regimens are effective in RRMM. However, their efficacy decreases in heavily pretreated and multi-refractory patients, supporting the need for ixazomib combinations to be used earlier in treatment. PN remains one of the most common adverse events and warrants further investigation.
Supplemental Material
sj-docx-1-tah-10.1177_20406207261421841 – Supplemental material for Real-world efficacy and tolerability of ixazomib-based combination therapies in advanced multiple myeloma and other plasma cell neoplasms
Supplemental material, sj-docx-1-tah-10.1177_20406207261421841 for Real-world efficacy and tolerability of ixazomib-based combination therapies in advanced multiple myeloma and other plasma cell neoplasms by Xiang Zhou, Julia Mersi, Christine Riedhammer, Maximilian J. Steinhardt, Max Bittrich, Stefan Knop, Hermann Einsele, Leo Rasche, Klaus Martin Kortüm and Johannes M. Waldschmidt in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
