Abstract
Background:
Little is known about the real-world survival benefits and safety profiles of carfilzomib–lenalidomide–dexamethasone (KRd) and carfilzomib–dexamethasone (Kd).
Methods:
We performed a retrospective analysis to evaluate their efficacy and safety in 157 patients registered in the Kansai Myeloma Forum database.
Results:
A total of 107 patients received KRd. Before KRd, 99% of patients had received bortezomib (54% were refractory disease), and 82% had received lenalidomide (57% were refractory disease). The overall response rate (ORR) was 68.2%. The median progression-free survival (PFS) and overall survival (OS) were 8.8 and 29.3 months, respectively. Multivariate analysis showed that reduction of the carfilzomib dose and non-IgG M protein were significantly associated with lower PFS and reduction of the carfilzomib dose and refractoriness to prior bortezomib-based regimens were significantly associated with lower OS. A total of 50 patients received Kd. Before Kd, 96% of patients had received bortezomib (54% were refractory disease). The ORR was 62.0%. The median PFS and OS were 7.1 and 20.9 months, respectively. Based on the multivariate analysis, reduction of the carfilzomib dose and International Staging System Stage III (ISS III) were significantly associated with lower PFS. Grade III or higher adverse events were observed in 48% of KRd cases and 54% of Kd cases. Cardiovascular events, cytopenia, and infections were frequent, and 4 KRd patients died due to heart failure, arrhythmia, cerebral hemorrhage, and pneumonia.
Conclusion:
Our analysis showed that an adequate dose of carfilzomib is important for achieving the best survival benefits in a real-world setting. Adverse effects after KRd and Kd therapy should also be considered.
Keywords
Introduction
In recent years, multiple proteasome inhibitors, immunomodulators, and antibody drugs for multiple myeloma (MM) have been introduced, which have greatly improved the treatment response, overall survival (OS) and progression-free survival (PFS).1–9 However, the prognosis of relapsed and refractory multiple myeloma (RRMM) is still poor. Carfilzomib is a next-generation selective and irreversible proteasome inhibitor 10 that has been demonstrated to have robust and durable activity and a favorable safety and tolerability profile as a single-agent treatment in heavily pretreated patients with RRMM in phase I/II trials.11–15 The efficacy and safety of carfilzomib–lenalidomide–dexamethasone (KRd) and carfilzomib–dexamethasone (Kd) for RRMM were evaluated in phase III trials, ASPIRE and ENDEAVOR, respectively.16–19 In the ASPIRE and ENDEAVOR studies, KRd was significantly superior with respect to both of median PFS and OS to lenalidomide–dexamethasone (Rd), and Kd also gave a significantly better both median PFS and OS than bortezomib–dexamethasone (Vd), respectively. Adverse effects, including cardiovascular events, cytopenia, and infections, were reported and characterized by the relatively high frequency of cardiovascular events. The ASPIRE study showed heart failure in 6.4% of KRd participants, whereas the ENDEAVOR study showed congestive heart failure in 10.8% of Kd participants, and several patients died of cardiovascular events in the both studies. In real-world clinical practice, KRd and Kd have been used according to ASPIRE and ENDEAVOR, but the dose and frequency of administration have often been reduced as appropriate depending on the patient’s age and general condition. There is little evidence or information available on the real-world use of KRd and Kd therapy. Then, we retrospectively analyzed the real-world efficacy and safety of KRd and Kd in Japanese patients who were registered in the Kansai Myeloma Forum (KMF) database.
Methods
Study design and participants
The KMF, a study group consisting of 46 hematological facilities in the Kansai region of Japan, was established in 2012 to register patients with myeloma-related disease. The group members have created and share a clinical database with the aim of retrospectively analyzing treatment strategies and outcomes. In this study, we included 157 patients from 17 hospitals in the KMF database who received KRd (n = 107) or Kd (n = 50) therapy between March 2016 and June 2019. The observation period was terminated on June 30, 2019. We obtained patient consent to treatment from all patients. Patients were selected consecutively. Patients needed to have received at least 1 line of therapy before KRd or Kd. Relapsed and refractory MM were defined according to International Myeloma Working Group (IMWG) criteria. 20 Patients who achieved stringent complete response (sCR) through autologous hematopoietic stem cell transplantation (auto-HSCT) and received KRd as consolidation therapy were excluded. Further, we performed a secondary survey on detailed medical information.
Treatment
KRd was performed under the following conditions and based on insurance coverage. Patients with RRMM receive intravenous carfilzomib at a dose of 20 mg/m2 on days 1 and 2, and then 27 mg/m2 on days 8, 9, 15, and 16 of the first cycle and days 1, 2, 8, 9, 15, and 16 of subsequent cycles, dexamethasone 20 mg on days 1, 2, 8, 9, 15, 16, 22, and 23, and lenalidomide 25 mg orally on days 1–21 of each 28-day cycle.
As for Kd, RRMM patients receive intravenous carfilzomib at a dose of 20 mg/m2 on days 1 and 2, and then 56 mg/m2 on days 8, 9, 15, and 16 of the first cycle and days 1, 2, 8, 9, 15 and 16 of subsequent cycles, and dexamethasone 20 mg on days 1, 2, 8, 9, 15, 16, 22, and 23 of each 28-day cycle. Administration of carfilzomib 70 mg/m2 once a week was allowed, although it had not yet been approved in Japan during the observation period.
The dosages of all drugs for KRd or Kd, antibacterial, antimycotic, and antiviral agents, and antiplatelet drugs were determined at the discretion of each physician. Responses were assessed according to the IMWG Uniform Response Criteria 21 and designated as sCR, complete response (CR), very good partial response (VGPR), partial response (PR), stable disease (SD), and progressive disease (PD).
Endpoints
The primary endpoint was PFS. Secondary endpoints were OS and the safety and efficacy of KRd and Kd in terms of overall response rate (ORR) and toxicity. Therapeutic response and disease progression were assessed according to the IMWG criteria. 22 All adverse events (AEs) were recorded using the Common Terminology Criteria for Adverse Events, version 5.0 of the National Cancer Institute Cancer Therapy Evaluation Program.
Statistical methods
Descriptive statistics were used to summarize variables related to patient characteristics. Comparisons between groups were performed with the chi-square statistic or extended Fisher exact test as appropriate for categorical variables. The probabilities of OS and PFS were estimated according to the Kaplan–Meier method, and the groups were compared with the log-rank test. The Cox proportional hazards model was used to evaluate the effect of variables on OS and PFS. Factors with p < 0.10 in the univariate analysis were added to the multivariate model. All tests were two-sided, and p < 0.05 was considered to indicate statistical significance. All statistical analyses were performed with Stata version 16.1 software (StataCorp, College Station, TX).
Results
The patient background is summarized in Table 1. In the KRd cohort, the median observation period of survivors was 23.1 (range, 0.8–33.5) months. The median age at the start of KRd was 67 (range, 41–84) years. Fourteen cases (13.1%) were above 75 years old, 21 (19.6%) had International Staging System Stage III (ISS III), and 31 (29.0%) had poor chromosomal risk, which was defined as the presence of either del(17p), t(4;14)(p16; q32), or t(14;16)(q32; q23) in the fluorescence in situ hybridization (FISH) or G-BAND. The median number of prior therapies was 3 (range, 1–14). A total of 106 cases (99.1%) had received prior bortezomib-based regimens, and 58 of these 106 cases were refractory. In addition, 88 cases (82.2%) had received lenalidomide-based regimens, and 61 of these 88 cases were refractory. The dose of carfilzomib was not reduced in 69 cases (64.5%), and the dose was reduced by more than 40% within the three courses in 14 cases (13.1%). The dose of lenalidomide was not reduced in 19 cases (17.8%), and the dose was reduced by more than 40% within the three courses in 62 cases (57.9%). The median number of KRd cycles was 3 (range, 1–19) (Supplement Table 1). KRd was discontinued in 104 cases, and the reasons for discontinuation were PD in 38 cases (35.5%), AEs in 37 cases (34.6%), a switch to auto-HSCT in 12 cases (11.2%), and achievement of a good therapeutic effect (CR or VGPR) in 3 cases (2.8%).
Patient demographics and baseline characteristics.

BJP, Bence-Jones protein; ECOG, Eastern Cooperative Oncology Group; FISH, fluorescence in situ hybridization; ISS, International Staging System; Kd, carfilzomib and dexamethasone; KRd, carfilzomib, lenalidomide, and dexamethasone.
In the Kd cohort, the median observation period of survivors was 15.8 months (range, 1.0–61.3). The median age at the start of Kd was 69 (range, 35–88) years. Sixteen cases (32.0%) were above 75 years old, 13 (26.0%) had ISS III, and 12 (24.0%) had poor chromosomal risk. The median number of prior therapies was 3.5 (1–12), 48 cases (96.0%) had received prior bortezomib-based regimens, and 27 of these 48 cases were refractory. The dose of carfilzomib was not reduced within the three courses in 20 cases (40.0%), and the dose was reduced by more than 40% in 18 cases (36.0%). Nine cases (18.0%) received once-weekly carfilzomib administration. The median number of Kd cycles was 4 (range, 1–36, Supplement Table 1). Kd was discontinued in all 50 cases, and the reasons for discontinuation were PD in 26 cases (52.0%), AEs in 13 cases (26.0%), and a switch to auto-HSCT in 4 cases (8.0%).
Safety
Grade III or higher AEs with KRd or Kd are summarized in Table 2. In the KRd cohort, the most common AEs were hematological toxicities (anemia, thrombocytopenia, leukopenia), and these were observed in 25 cases (23.4%). Cardiovascular toxicities (heart failure, arrhythmia, hypertension, and pulmonary hypertension) occurred in 14 cases (13.1%), and infections (pneumonia, sepsis, CMV infection, etc.) were seen in 16 cases (15.0%). Four patients died of heart failure, arrhythmia, intracerebral hemorrhage, or pneumonia, respectively.
Adverse events.
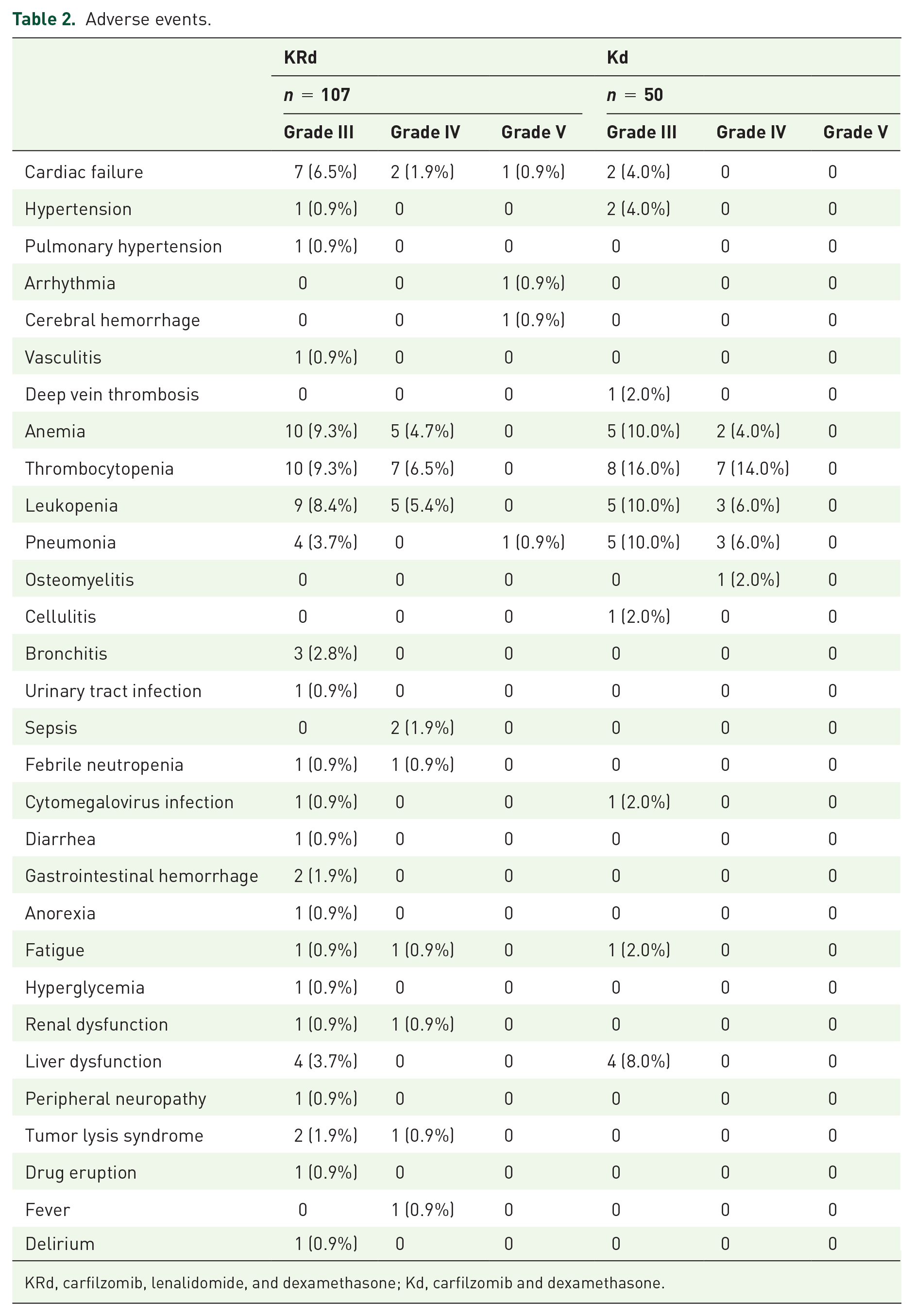
KRd, carfilzomib, lenalidomide, and dexamethasone; Kd, carfilzomib and dexamethasone.
The incidence of cardiovascular AEs was 28.6% for patients 75 years old and above, 14.6% for those aged 65–74, and 8.9% for those under 65 (p = 0.178). Non-cardiovascular AEs occurred in 57.1% of patients 75 years old and above, in 37.5% of those aged 65–74, and in 35.6% of those under 65 (p = 0.333) (Supplement Table 2).
In the Kd cohort, the most common AEs were hematological toxicities (anemia, thrombocytopenia, and leukopenia), and these were observed in 20 cases (40.0%). Infections (pneumonia, osteomyelitis, etc.) occurred in 11 cases (22.0%), cardiovascular toxicities (heart failure, hypertension) were seen in 4 cases (8.0%), and liver dysfunction was seen in 4 cases (8.0%). None of the patients died of AEs. No association was found between the age of patients and either cardiovascular AEs (p = 0.294) or non-cardiovascular AEs (p = 0.913).
Efficacy and survival outcomes of KRd
In the KRd cohort, ORR was 68.2%, VGPR or better was 34.6%, and CR + sCR was 15.0% (Table 3). Relapse or PD was observed in 69 patients (64.5%), and 44 patients (41.1%) died during the observation period.
Response rates.

CR, complete response; Kd, carfilzomib and dexamethasone; KRd, carfilzomib, lenalidomide, and dexamethasone; ORR, overall response rate; PD, progressive disease; PR, partial response; sCR, stringent complete response; SD, stable disease; VGPR, very good partial response.
The median PFS was 8.8 months, and the 1-year PFS rate was 41.0% (Figure 1). Univariate analysis showed significant differences in PFS regarding age (1 year PFS: <65 years old, 46.6%; 65–74, 38.5%; ⩾75, 31.2%, p = 0.049), type of M protein (IgG, 52.9%; non-IgG, 23.2%; p < 0.001), number of prior lines of therapy (1 or 2 lines, 60.9%; ⩾3 lines, 30.5%; p = 0.011), refractoriness to bortezomib (sensitive, 58.3%; refractory, 26.4%; p < 0.001), refractoriness to lenalidomide (sensitive, 60.7%; refractory, 26.7%; p < 0.001), period from initial treatment to KRd (<2 years, 55.0%; ⩾2 years, 32.5%; p = 0.022), reduction in the dose of carfilzomib (100% dose, 48.4%; 60–99%, 30.5%; <60%, 21.4%; p = 0.007), and reduction in the dose of lenalidomide (100% dose, 57.4%; 60–99%, 46.6%; <60%, 33.4%; p = 0.004; Table 4, Figure 2, Supplement Figures 1 and 2). Multivariate analysis showed significantly shorter PFS in non-IgG M protein [hazard ratio (HR), 2.48, 95% confidence interval (CI): 1.42 – 4.33, p = 0.001] and reduction in the dose of carfilzomib to less than 60% (<60% vs 100%: HR, 3.05, 95% CI: 1.40–6.62, p = 0.005; Table 4).
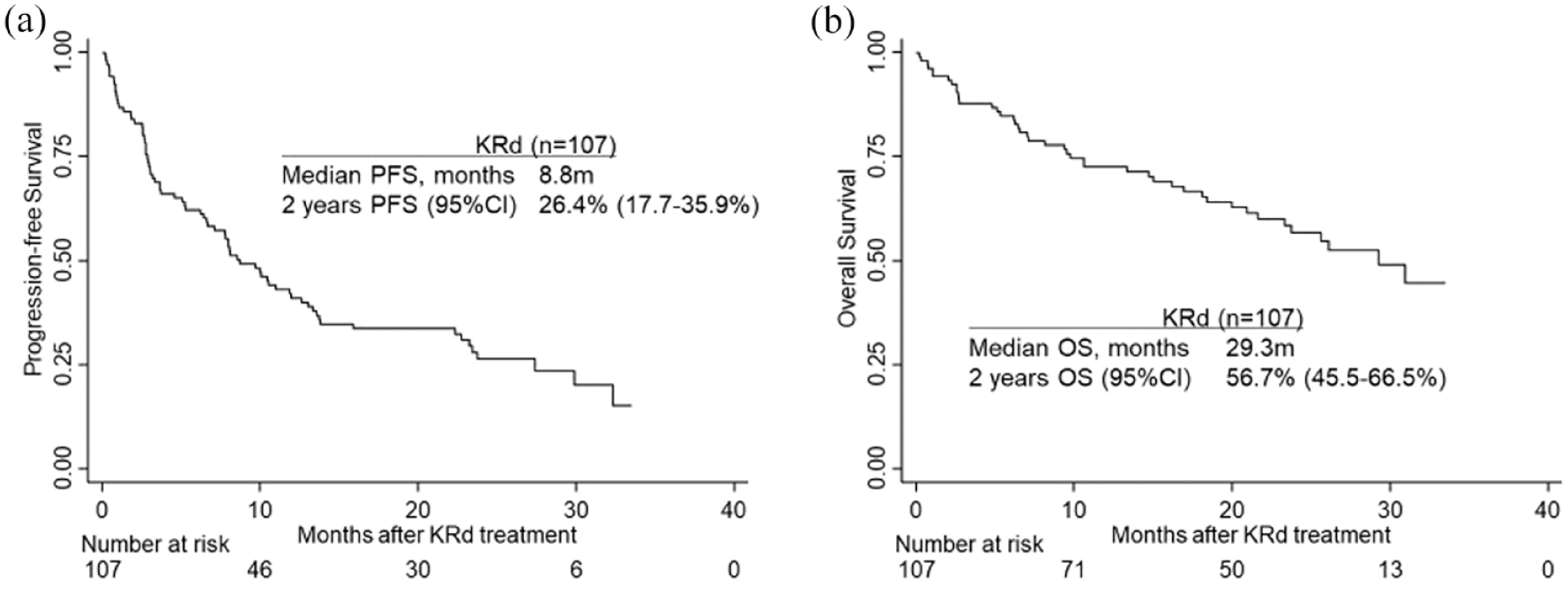
Kaplan-Meier curves of progression-free survival (PFS) (a) and overall survival (OS) (b) of 107 patients treated with carfilzomib–lenalidomide–dexamethasone (KRd).
KRd: Univariate and multivariate analysis of progression-free survival (PFS) and overall survival (OS).

BJP, Bence-Jones protein; ISS, International Staging System; Kd, carfilzomib and dexamethasone; KRd, carfilzomib, lenalidomide, and dexamethasone; PD, progressive disease; PS, performance status.

Kaplan-Meier curves of PFS (a) and OS (b) of patients treated with KRd based on the dosage of carfilzomib (100% vs. 60-99% vs. <60%).
The median OS was 29.3 months, and the 1-year OS rate was 72.6% (Figure 1). Univariate analysis showed significant differences in OS regarding type of M protein (1-year OS: IgG, 79.2%; non-IgG, 64.3%; p = 0.013), number of prior lines of therapy (1 or 2 lines, 82.7%; ⩾3 lines, 67.3%; p = 0.008), period from initial treatment to KRd (<2 years, 82.1%; ⩾2 years, 66.7%; p = 0.025), refractoriness to bortezomib (sensitive, 83.1%; refractory 63.8%; p = 0.006), refractoriness to lenalidomide (sensitive, 83.4%; refractory 64.3%; p = 0.003), and reduction in the dose of carfilzomib (100% dose, 81.7%; 60–99%, 64.3%; <60%, 40.8%; p = 0.018; Figure 2, Supplement Figures 1 and 2). Multivariate analysis revealed significantly shorter OS in carfilzomib reduction (100% vs <60%: HR 3.66, 95% CI: 1.47–9.10, p = 0.005) and prior bortezomib sensitivity (sensitive vs refractory: HR 2.33, 95% CI: 1.01–5.37, p = 0.047; Table 4).
Seven patients received KRd after auto-HSCT as consolidation therapy, and their ORR was 100% (sCR: 2 cases, VGPR: 3 cases, and PR: 2 cases). In these patients, the median observation period of survivors was 28.0 months (range, 5.2–33.4). Twelve-month PFS, 24-month PFS, 12-month OS, and 24-month OS were 40.0%, 40.0%, 100.0%, and 100.0%, respectively. Twelve patients underwent planned auto-HSCT after KRd, and their ORR was 100% (sCR: 4 cases, VGPR: 5 cases and PR: 3 cases). In these patients, the median observation period of survivors was 20.5 (range, 5.0–33.5) months. Twelve-month PFS, 24-month PFS, 12-month OS, and 24-month OS were 82.5%, 82.5%, 100.0%, and 65.6%, respectively.
Efficacy and survival outcomes of Kd
In the Kd cohort, ORR was 62.0%, VGPR or better was 26.0%, and CR + sCR was 4.0% (Table 3). Relapse or PD was observed in 36 patients (72.0%), and 23 patients (46.0%) died during the observation period.
The median PFS was 7.1 months, and the 1-year PFS rate was 28.8% (Figure 3). Univariate analysis showed significantly shorter PFS in ISS III (1 year PFS: ISS I/II, 37.7%; ISS III, 7.7%; p = 0.007; Table 5, Supplement Figure 3). Multivariate analysis showed significantly shorter PFS in ISS III (ISS I + II vs ISS III: HR 3.16, 95% CI: 1.21–8.26, p = 0.019) and reduction in the dose of carfilzomib to less than 60% (100% vs <60%: HR, 2.89, 95% CI: 1.25–6.70, p = 0.013; Table 5).

Kaplan-Meier curves of PFS (a) and OS (b) of 50 patients treated with carfilzomib-dexamethasone (Kd).
Kd: Univariate and multivariate analysis of progression-free survival (PFS) and overall survival (OS).

BJP, Bence-Jones protein; ISS, International Staging System; Kd, carfilzomib and dexamethasone; KRd, carfilzomib, lenalidomide, and dexamethasone; PD, progressive disease; PS, performance status.
The median OS was 20.9 months, and the 1-year OS rate was 70.9% (Figure 1). Univariate analysis revealed significantly shorter OS in ISS III (1-year OS: ISS I/II, 37.7%; ISS III, 7.7%; p = 0.002; Table 5, Supplement Figure 3). Refractoriness to bortezomib and a reduction in the dose of carfilzomib to less than 60% did not significantly affect PFS or OS in a univariate analysis (Figure 4). None of the factors examined significantly shortened OS in a multivariate analysis (Table 5).

Kaplan-Meier curves of PFS (a) and OS (b) of patients treated with Kd based on the dosage of carfilzomib (100% vs. 60-99% vs. <60%).
Discussion
Based on the results of the phase III ASPIRE and ENDEAVOR trials, KRd and Kd therapies have been approved and performed in Japan. Clinical experience at the KMF member hematological facilities has been accumulated, and we collected medical information including actual drug dosages, number of administrations, and the details of AEs. In this study, we evaluated the effectiveness and safety of KRd and Kd by conducting an analysis that focused on real-world data and compared our findings with past reports.
With regard to KRd patients, KRd showed a relatively good therapeutic effect in the population, including in patients who were refractory to several treatment regimens. Almost all cases had a history of bortezomib treatment and 54.2% of them were refractory. Univariate and multivariate analysis showed that bortezomib-resistant cases had worse ORR than non-resistant cases, and OS and PFS tended to be poor. The results suggested cross-resistance between bortezomib and carfilzomib. The carfilzomib doses in the first three courses were associated with OS and PFS, and dose reduction to 60% or less was identified as a poor prognosis factor. In cases of planned auto-HSCT after KRd or KRd as a consolidation therapy after auto-HSCT, median PFS and median OS were not reached. Both bridging KRd and consolidation KRd for auto-HSCT were considered useful. Several studies have reported that carfilzomib frequently had AEs in the elderly.23–30 In this study, there was no significant correlation between the incidence of grade ⩾3 AEs and age, partly due to the small sample size. The doses of carfilzomib and lenalidomide tended to be reduced in elderly patients. It would be desirable to evaluate cardiac function in advance, and to carefully observe the appearance of blood pressure change, signs of heart failure, and cytopenia even after the start of carfilzomib treatment. A patient who died of heart failure had a history of Sjogren’s syndrome and a patient who died of arrhythmia had a history of bronchial asthma, which might have affected the onset of fatal AEs.
To compare the real-world data set with a clinical trial, we selected patients who had received one to three pretreatment lines, had Eastern Cooperative Oncology Group performance status (ECOG PS) 2 or better, and maintained SD or better reactivity for pretreatment lenalidomide and bortezomib (n = 19), according to the ASPIRE study (n = 396); ORR (this study vs ASPIRE = 89.5 vs 87.1%), median PFS (NR vs 26.3 months), and median OS (NR vs 48.3 months) in this study were not inferior to those in the ASPIRE trial. Furthermore, the side effect profile of cardiovascular events, cytopenia, and infection was similar.
In Kd patients, the bortezomib sensitivity of the previous treatment did not significantly affect Kd efficacy. In cases of ISS III, PFS and OS were significantly inferior, and in cases in which the dose of carfilzomib was reduced to less than 60%, OS was poor. There were nine cases of once-weekly carfilzomib administration, and the ORR was not inferior to that of twice-weekly cases (once a week vs twice a week = 77.8% vs 58.5%). Once a week cases tended to receive long-term administration of eight courses or more (once a week vs twice a week = 55.6% vs 24.4%). It is probable that the burden of commuting to the hospital for patients was reduced, and this helped them to continue Kd. Once-weekly carfilzomib 70 mg/two administration is synonymous with carfilzomib dose reduction to 62.5% compared to twice-weekly administration. However, because carfilzomib has been demonstrated to inhibit the proteasome dose dependently, the increase in proteasome inhibition associated with increasing carfilzomib 70 mg/two doses in 1 day may partly account for the improved clinical response. 31 The incidences of cardiovascular AEs (once a week vs twice a week = 11.1% vs 7.3%) and non-cardiovascular AEs (once a week vs twice a week = 44.4% vs 56.1%) were not significantly different. Once-a-week, Kd was considered to be a promising and safe option. As in previous reports, AEs were mainly cardiovascular side effects, cytopenia, and infection; however, there was no significant correlation between AEs and age of patients.16,32,33 No serious AE leading to death was observed.
To compare the real-world data set with a clinical trial, we selected patients who had received one to three lines of pretreatment, had ECOG PS 2 or better and were proteasome inhibitor-free for 6 months until the start of Kd (n = 12), according to the ENDEAVOR study (n = 464); ORR (this study vs ENDEAVOR = 66.7 vs 76.7%), median PFS (11.1 vs 18.7 months), and median OS (27.0 vs 47.6 months) in this study were inferior to those in the ENDEAVOR study. It is considered that this was because the ENDEAVOR study excluded patients with cytopenia, heart failure, and renal dysfunction at the start of Kd.
This study has several limitations. First, although chromosomal abnormalities have been reported to be an important factor that affects the prognosis of MM, all chromosomal abnormalities were not necessarily evaluated by FISH or G-BAND before the start of KRd or Kd in all patients in this study. In most of the cases, only one or two genetic abnormalities had been investigated by FISH at the diagnosis, and all risk factors had not been examined. Second, Kd was approved later than KRd (KRd: July 2016, Kd: May 2017), and the shorter observation period in the Kd group might have affected the evaluation of treatment outcomes. In recent years, other combinations of carfilzomib such as daratumumab, isatuximab, and pomalidomide are available and are expected to show their efficacy and safety.
In conclusion, although this study tended to have a higher median age, more high-risk cases, more prior lines of therapy, and more cases of resistance to bortezomib and lenalidomide compared to other real-world reports (Supplement Tables 3 and 4), an equivalent therapeutic effect has been obtained, which suggested that KRd and Kd therapies are also effective in Japanese patients. It is desirable to avoid reducing the dose of carfilzomib as much as possible. Cytopenia, infections, and cardiovascular AEs were observed, and there were cases of death due to the AEs; therefore, careful attention should be paid with regard to the AEs.
Supplemental Material
sj-docx-1-tah-10.1177_20406207221104584 – Supplemental material for Real-world effectiveness and safety analysis of carfilzomib–lenalidomide–dexamethasone and carfilzomib–dexamethasone in relapsed/refractory multiple myeloma: a multicenter retrospective analysis
Supplemental material, sj-docx-1-tah-10.1177_20406207221104584 for Real-world effectiveness and safety analysis of carfilzomib–lenalidomide–dexamethasone and carfilzomib–dexamethasone in relapsed/refractory multiple myeloma: a multicenter retrospective analysis by Yoshiyuki Onda, Junya Kanda, Hitomi Kaneko, Yuji Shimura, Shin-ichi Fuchida, Aya Nakaya, Tomoki Itou, Ryosuke Yamamura, Hirokazu Tanaka, Hirohiko Shibayama, Yutaka Shimazu, Hitoji Uchiyama, Satoshi Yoshihara, Yoko Adachi, Mitsuhiro Matsuda, Hitoshi Hanamoto, Nobuhiko Uoshima, Satoru Kosugi, Kensuke Ohta, Hideo Yagi, Yuzuru Kanakura, Itaru Matsumura, Masayuki Hino, Shosaku Nomura, Chihiro Shimazaki, Akifumi Takaori-Kondo and Junya Kuroda in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
The authors are thankful to all the patients, their families, Ritsuko Okuyama, and KMF members who contributed to this study.
Ethics approval and consent to participate
This study was conducted in accordance with the Declaration of Helsinki, with the approval of the ethics committees of the KMF and each participating institution. Ethics/Review board approval: Kyoto University Certified Review Board, Yoshida-Konoe-cho, Sakyo-ku, Kyoto 606-8501, JAPAN, CRB5180002(R1856), March 19th, 2019. We have followed the guideline of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement. The checklist from the guideline is in Supplementary material. The informed consent requirement for this retrospective study was waived because the study was conducted retrospectively and the opportunity to refuse was guaranteed.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Ono Pharmaceutical Co., Ltd.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors received research funding from Ono Pharma. and Celgene (Current Bristol Myers Squibb). JKa, SF, TI, HT, HS, SY, KS, KO, MH, CS, and JKu received speaker’s bureau from Ono Pharma. JKa, SF, TI, HT, HS, SY, SK, KO, IM, MH, CS, AT-K, and JKu received speaker’s bureau from Celgene (Current Bristol Myers Squibb). SF, TI, TH, HS, SY, SK, KO, IM, MH, CS, and JKu received speaker’s bureau from Janssen. JKa, SF, TI, HT, HS, SY, SK, KO, IM, MH, CS, and JKu received speaker’s bureau from Takeda Pharma. JKa, SF, TI, HT, HS, SY, SK, MH, CS, and JKu received speaker’s bureau from Sanofi. JKa, TI, HT, HS, SY, SK, KO, IM, MH, AT-K, and JKu received speaker’s bureau from Novartis. SK and JKu received speaker’s bureau from Fujimoto Pharma. TI, HS, MH, and JKu received research funding from Celgene (Current Bristol Myers Squibb). HS received research funding from Janssen. IM and JKu received research funding from Takeda Pharma. HS, IM, and JKu received research funding from Sanofi. HS and MH received research funding from Novartis. JKu received research funding from Fujimoto Pharma. JKa received consulting fees from Janssen and Takeda Pharma. MH received scholarship donations from Takeda Pharma. and Ono Pharma. AT-K received scholarship donations from Takeda Pharma. and Sanofi.
Availability of data and materials
All data used in the analysis was anonymized, making it impossible for researchers to identify individuals.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
