Abstract
Chronic myeloid leukemia (CML) in patients with β-thalassemia major is a rare and concerning occurrence. The longer life expectancy of thalassemia patients, resulting from improved treatment options and better healthcare facilities, has led to the emergence of various health-related issues and complications. We present the case of a 24-year-old female diagnosed with β-thalassemia major at six months of age, who presented with weakness and lethargy in clinic. Investigations, including a bone marrow biopsy, confirmed CML in the chronic phase. Initial management with imatinib resulted in severe thrombocytopenia and failure to achieve a molecular response. The treatment was then switched to Ponatinib, leading to a favorable outcome with early molecular response. This case report highlights the increased risk of hematological malignancies in thalassemia patients and underscores the importance of vigilant monitoring in them. Furthermore, it emphasizes the role and effectiveness of third-generation tyrosine kinase inhibitors in the management of such cases.
Introduction
Thalassemia is defined as a group of inherited blood disorders caused by a decrease or absence of globin chain synthesis in hemoglobin, resulting in reduced or defective production of erythrocytes. 1 Depending on the type of globin chain involved, it can be divided into alpha and beta thalassemia. 1
β-Thalassemia major is a genetic hemoglobinopathy characterized by point mutations of the β-globin chain resulting in the accumulation and deposition of unpaired β-globin chains in red blood cells (RBCs). 2 In recent years, thalassemia patients have been living longer because of better treatment options. With thalassemia, there is an emergence of various health issues, including different forms of malignancies. 3
Chronic myeloid leukemia (CML) is a myeloproliferative neoplasm resulting from a reciprocal translocation known as t(9;22) (q34;q11.2), which results in the merging of ABL1 gene sequences (located on chromosome 9q34) with BCR gene sequences (located on chromosome 22q11). This fusion event can be observed cytogenetically as the Philadelphia chromosome. 4
We report a unique case of CML occurring in a patient with beta thalassemia, which is a very rare presentation. However, there are a few cases that have reported hematological malignancies in thalassemia patients. Several factors are believed to potentially contribute to the emergence of hematological malignancies and myeloproliferative neoplasms in individuals with beta thalassemia.3,5
Case report
A 24-year-old girl presented in the Hematology Clinic in May 2023 with complaints of weakness and lethargy from last 2 weeks. She was diagnosed with β-thalassemia major at the age of 6 months and was on regular blood transfusions since then, in addition to dual iron chelation therapy including subcutaneous deferoxamine and oral deferasirox. On examination, the spleen was found to be palpable 6 cm below the left subcostal margin. There was no pallor or any lymph nodes palpable (Supplemental Material).
A complete blood count (CBC) was done, which showed low platelet count (25,000/µL), leukocytosis with a white blood cell count of 132.1 × 109/L, along with immature cells (myelocytes, metamyelocytes, promyelocytes) and blast cells. Peripheral blood film review revealed hyperleukocytosis, 3% nucleated RBCs, low platelets, leukoerythroblastic picture, basophilia, eosinophilia, and 2% blast cells (Table 1). Ultrasound abdomen showed hepatosplenomegaly with an increment in spleen size from 13.6 to 17.6 cm over the last two and a half years.
Comparison of complete blood count results in patient at diagnosis of CML and after starting treatment with tyrosine kinase inhibitors (imatinib and ponatinib).

Thus, bone marrow biopsy was performed which was suggestive of CML in chronic phase. Bone marrow aspirate revealed spicular hypercellular specimen showing granulocytic hyperplasia, suppressed erythropoiesis, myeloid to erythroid ratio of 19:1, adequate megakaryocytes, blast cells of less than 3%, and plasma cells of less than 3%. Bone trephine showed overall cellularity of around 90%–95% and cellular areas exhibiting granulocytic hyperplasia (Figures 1 and 2).
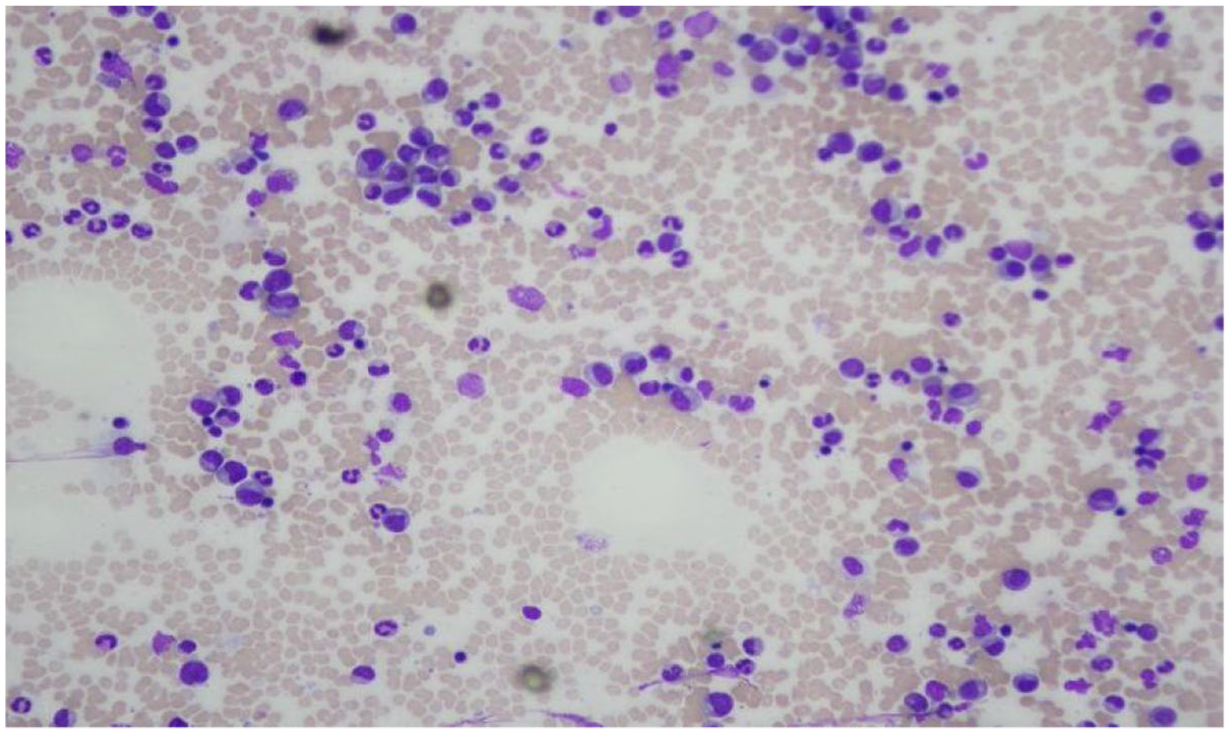
Bone marrow aspirate revealing spicular hypercellular specimen showing granulocytic hyperplasia, suppressed erythropoiesis, myeloid to erythroid ratio of 19:1, adequate megakaryocytes, blast cells of less than 3%, and plasma cells of less than 3%.

Bone trephine showing (a) overall cellularity of around 90%–95%, and (b) cellular areas exhibiting granulocytic hyperplasia.
Furthermore, bone marrow chromosome testing revealed a translocation between chromosomes 9q34 and 22q11.2, resulting in the Philadelphia chromosome, found in all cells. A FISH assay for BCR-ABL1 translocation was conducted, and BCR-ABL1 translocation was detected in 98% of the 200 nuclei examined. The EUTOS long-term survival score (ELTS) calculated for the patient was 3.2, indicating classification into the high-risk group.
She was managed on lines of CML with imatinib 400 mg once daily. Her platelet counts were monitored and were kept above 25 × 109/L. Imatinib was stopped whenever platelets dropped below 25 × 109/L. Regular blood transfusions for thalassemia were continued along with oral iron chelation therapy.
The assessment of treatment response involved molecular monitoring, in which the early molecular response (EMR) was evaluated by quantifying BCR-ABL levels using the Gene Xpert technique every 3 months. BCR-ABL1 levels after 3 months of initiation of imatinib were found to be 11.51%. However, with continued use of imatinib, she developed severe grade IV thrombocytopenia, and so the dose of imatinib was reduced accordingly with frequent drug holidays. Thus, we were unable to give the optimal dose of imatinib. Subsequent quantification of BCR-ABL1 after 3 months revealed a decrease to 9.77% only. Keeping in view the suboptimal response, other tyrosine kinase inhibitors were considered. Her echocardiogram showed trace pulmonary and tricuspid regurgitation. Nilotinib was not prescribed due to its cardiotoxic effects. Similarly, Dasatinib was also contraindicated in this case because of the risk of developing pulmonary hypertension. Consequently, ponatinib, a third-generation tyrosine kinase inhibitor, was initiated. She was then kept on Ponatinib 15 mg on alternate days; upon which she achieved early molecular response with BCR-ABL1 p210 mRNA transcripts of around 0.53% of total ABL1. Her last CBC on this treatment showed a hemoglobin level of 11.4 g/dL, a platelet count of 65 × 109/L, and a WBC of 8.3 × 109/L. Myelocytes, metamyelocytes, promyelocytes, and blast cells all are undetectable. Therefore, her treatment with Ponatinib 15 mg on alternate days is continued with frequent CBC monitoring. Her hemoglobin levels are also maintained around 10 g/dL with regular blood transfusions.
Discussion
CML occurring in a patient with beta thalassemia is very rare. We report the first such case from Pakistan. However, there are few cases reported to show co-existence between thalassemia and hematological malignancies.
Alavi et al. reported a similar case in which a 13-year-old girl, a known case of thalassemia intermedia, was found to develop chronic myelogenous leukemia as she presented with hyperleukocytosis in the clinic. Although timely treatment with imatinib was also started in this case, she developed an accelerated blast crisis, which eventually progressed to acute myeloid leukemia (AML). She failed to exhibit a positive response to chemotherapy and eventually succumbed to her disease. 2 Response to tyrosine kinase inhibitors and long-term survival assessment is done by using the ELTS calculator. 6 Voskaridou et al. 7 also reported a case of a 32-year-old man diagnosed with thalassemia major who experienced a gradual rise in leukocyte and platelet counts, eventually leading to a diagnosis of chronic myelogenous leukemia. Myeloproliferative neoplasms (MPNs) and thalassemia are distinct hematological disorders that seldom occur together. 8 Myeloproliferative disorders like polycythemia vera have also been reported to occur in patients with thalassemia.5,8 In a case reported by Martinez-Lopez, a young lady with thalassemia minor who presented with complaints of fatigue and abdominal pain was evaluated for leukocytosis, thrombocytosis, and painful splenomegaly. She was diagnosed with polycythemia vera on bone marrow biopsy and was then treated accordingly. 5
The presence of hematological malignancies in individuals with thalassemia may be a coincidence or may result from a combination of genetic and environmental factors. There are various factors that can contribute to the emergence of hematological malignancies in individuals with beta thalassemia.
The study by Hodroj et al. revolves around the emerging concern of a potential association between thalassemia and an increased risk of malignancies. There are a few hypotheses to suggest the evolution of malignancies in thalassemia, which includes repeated blood transfusions, iron overload, viral infections, increased bone marrow stimulation, and the use of hydroxyurea. 3 Increased cellular iron levels due to chronic transfusions can generate reactive oxygen species, which can cause cell death and mutations. 9 Nontransfusion-dependent thalassemia patients have ineffective erythropoiesis and hypoxia, which can enhance bone marrow activity, leading to the development of hematologic malignancies, particularly myeloproliferative disorders. 10 Furthermore, hydroxyurea, which is used to reduce transfusion needs in thalassemic patients, can cause DNA mutations and affect chromosomal repair. As of now, there is no evidence that hydroxyurea is really carcinogenic.3,10 Moreover, blood transfusions pose a potential risk of transmitting oncogenic viruses, which can contribute to the development of hematologic malignancies. Some specific viruses, such as Hepatitis C, cytomegalovirus, Epstein–Barr virus, and human T-lymphotropic virus type-1 (HTLV-1), have been detected in patients with thalassemia and may be associated with conditions like Hodgkin’s or non-Hodgkin’s lymphoma, as well as adult T-cell leukemia/lymphoma. 10
Conclusion
In conclusion, thalassemia patients are at increased risk of developing hematological malignancies. This emphasizes the need to create screening, diagnostic, and treatment plans to avoid additional health problems in thalassemia patients. Therefore, it is crucial not to overlook patients presenting with worsening symptoms. Timely recognition of malignancy in thalassemia patients, like in our case, can help in improving the overall quality of life of patients.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251353194 – Supplemental material for Chronic myeloid leukemia in a patient with beta thalassemia major: a rare presentation
Supplemental material, sj-docx-1-tah-10.1177_20406207251353194 for Chronic myeloid leukemia in a patient with beta thalassemia major: a rare presentation by Alyna Hafeez, Muhammad Usman Shaikh, Abdul Hadi Mallick, Nouman Nathani and Junaid Alam in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
