Abstract
Aims:
It was aimed at measuring the comorbidities of chronic myeloid leukemia (CML) patients at the time of diagnosis with different comorbidity indices and evaluating their effects on disease prognosis.
Methods:
The comorbidities of the patients were retrospectively screened and calculated in three different comorbidity indices: the ACE-27 Comorbidity Index, the Age-adjusted Charlson Comorbidity Index, and the Elixhauser Comorbidity Index. C-statistic was used to evaluate the ability of comorbidity indices to discriminate mortality. The relationship between the calculated scores and overall survival (OS) was evaluated with the Kaplan-Meier curve. Mortality risk was analyzed with a multivariate Cox regression model.
Results:
A total of 218 CML patients were evaluated, and 211 chronic-phase patients were included in this study. The median age of the patients was 56 years (21–89), and 53% were female. As initial tyrosine kinase inhibitors, 201 (95%) patients were treated with imatinib, 10 (5%) patients with nilotinib. The median follow-up was 94.50 (9–201) months. The median OS was not reached. The most common comorbid conditions were hypertension 23% (n = 48), weight loss 19% (n = 40), diabetes mellitus 13% (n = 27), and cardiovascular disease 9% (n = 19). C-statistic values were 0.76 for ACE-27, 0.41 for ACCI, and 0.32 for ECI scores. In the Cox regression model including comorbidity scores, mortality risk was higher in patients with moderate ACE-27 score (HR: 148.05; 95% CI: 7.89–2751.53; p = 0.012), severe ACE-27 score (HR: 232.36; 95% CI: 14.20–4793.20; p = 0.001), ECOG 3 score (HR: 34.62; 95% CI: 2.67–447.36; p = 0.007), and high ELTS score (HR: 27.52; 95% CI: 1.34–543.68; p = 0.031).
Conclusion:
This study showed that the ACE-27 Comorbidity Index is effective in predicting prognosis in CML patients. Therefore, comorbid conditions should be used more frequently as a prognostic marker at the time of diagnosis.
Introduction
The introduction of tyrosine kinase inhibitors (TKIs) in the treatment of chronic myeloid leukemia (CML) has led to a significant increase in patient survival. Currently, the life expectancy of CML patients is almost the same as that of the general population. 1 Different risk scores, such as Sokal, Hasford, and ELTS (EUTOS long-term survival), are used to determine prognosis. These prognostic scoring systems use different calculation models, including a combination of peripheral blast count, thrombocyte count, basophil count, eosinophil count, splenomegaly, and age variables.2,3 However, with the life expectancy of patients approaching that of the general population, the use of different prognostic markers has gained importance. In many studies conducted for this purpose, evaluation of the comorbid status of patients at the time of diagnosis with the comorbidity index has been accepted as a prognostic marker.4–6
Adult Comorbidity Evaluation 27 (ACE-27) is another comorbidity index evaluating 27 comorbid conditions in cancer patients. 7 In a study conducted on different cancer types, the ACE-27 index was shown to have a prognostic effect on patients. 8 The Age-adjusted Charlson Comorbidity Index (ACCI) was modified by Charlson et al. in 1994 and includes the evaluation of a total of 20 comorbid conditions. 9 Ono et al. showed that a high ACCI score was a negative prognostic factor for overall survival (OS) in a study conducted on 452 CML patients. 4 The Elixhauser Comorbidity Index (ECI) is a scoring system evaluating 30 different comorbid conditions that was first used in 1998 to predict in-hospital mortality. 10 ECI has been shown to be a reliable prognostic marker in many studies on cancer patients.11,12
In conclusion, it is important to define the relationship between comorbid conditions and OS in CML patients. The aim of this study was to evaluate the relationship between comorbidity scores calculated by the ECI, ACCI, and ACE-27 indices and OS outcomes in these patients.
Patients and methods
Patients who were followed up with a diagnosis of CML at Bursa Uludag University Faculty of Medicine Hospital between 2006 and 2021 were retrospectively analyzed. Chronic-phase CML patients older than 18 years were included in this study, and accelerated phase (AP) or blastic phase (BP) patients were excluded at diagnosis. Data on patient characteristics, comorbid conditions, and medications was collected from the hospital data processing system and patient files. Two standardized prognostic risk scores (Sokal and ELTS) were used to predict the prognosis. A complete cytogenetic response (CCyR) was defined as the absence of the Philadelphia chromosome (Ph) by conventional cytogenetic methods. Major molecular response (MMR, MR 3.0, or deeper response) was defined as a BCR-ABL1 transcript ratio ⩽0.1% according to the international scale. MMR and CCyR rates were evaluated at the 12th month of the use of TKIs. The Eastern Cooperative Oncology Group (ECOG) score and performance status at diagnosis were recorded.
Comorbid conditions at the time of diagnosis were calculated with three different comorbidity indices. With the ECI score, risk groups of patients were defined as <0 points, 1–4 points, and >5 points. Patients evaluated with ACCI were divided into 0, 1–2, 3–4, and >4 risk groups according to their total scores, respectively. With the ACE-27 index, patients’ comorbid conditions were categorized into four different groups according to their severity: none, mild, moderate, or severe. In this study, we evaluated the comorbidity conditions of the patients at the time of diagnosis with three different comorbidity indices. We examined the effect of comorbidity indices on OS (Table 1).
Parameters questioned in each scoring system.

ACCI, Age-adjusted Charlson Comorbidity Index; ACE, Adult Comorbidity Evaluation; AIDS, acquired immune deficiency syndrome; ECI, Elixhauser Comorbidity Index; HIV, human immunodeficiency virus.
Statistical analysis
Statistical analyses were performed using “IBM SPSS Statistics for Windows Version 25.0 (Statistical Package for the Social Sciences, IBM Corp., Armonk, NY, USA).” Descriptive statistics are presented as n and % for categorical variables and mean ± SD, median for continuous variables.
The C-statistic was used to evaluate the ability of comorbidity indices to discriminate mortality. Values of the C-statistic between 0.5 and 1 are interpreted as follows: 0.5; the model performs no better than random guessing, 0.6–0.7; the model has weak discrimination, 0.7–0.8; the model performs reasonably well, 0.8–0.9; the model has strong discrimination, >0.9; the model performs exceptionally well, close to perfect prediction. The Kaplan-Meier method was used to compare survival times between various clinical variables. Lastly, multivariate Cox regression results of various clinical factors on mortality risk are given. p < 0.05 was considered statistically significant.
Results
This study included 211 chronic-phase CML patients. The median age of the patients was 56 years (21–89), and 53% were female. As initial TKIs, 201 (95%) patients received imatinib, and 10 (5%) patients received nilotinib. At 12 months, CCyR and MMR rates were 86.3% and 81%, respectively. The characteristics of the patients are shown in Table 2.
Demographic and clinical characteristics of patients (n = 211).

ACCI, Age-adjusted Charlson Comorbidity Index; ACE, Adult Comorbidity Evaluation; AIDS, acquired immune deficiency syndrome; CCyR, complete cytogenetic response; ECI, Elixhauser Comorbidity Index; ECOG, Eastern Cooperative Oncology Group; ELTS, EUTOS long-term survival; MMR, major molecular response.
The most common comorbid conditions were respectively hypertension 23% (n = 48), weight loss 19% (n = 40), diabetes mellitus 13% (n = 27), cardiovascular disease 9% (n = 19), hypothyroidism 6% (n = 13), iron deficiency 5% (n = 11), solid tumor 5% (n = 10), and chronic pulmonary disease 4% (n = 9; Figure 1). The median follow-up was 94.5 (9–201) months. The 2-year OS was 98.6%, while the 5-year OS was 94.8%. Median OS was not reached (Figure 2). ELTS and Sokal risk distributions among comorbidity indices are shown in Table 3.

Distribution of comorbid conditions in patients (n = 211).

Overall survival (OS) of patients (Kaplan-Meier curve). Median OS was not reached (n = 211).
Distribution of comorbidity indices according to Sokal risk score and ELTS score.

ACCI, Age-adjusted Charlson Comorbidity Index; ACE, Adult Comorbidity Evaluation; ELTS, EUTOS long-term survival.
The mortality rate was 11%. Of the 23 patients with mortality, 52% (n = 12) had hypertension, 52% (n = 12) had weight loss, 26% (n = 6) had cardiovascular disease, and 22% (n = 5) had diabetes mellitus. The cause of death in seven patients was CML-related death (progression to AP/BP). In CML-unrelated death, five patients died due to pneumonia (four COVID-19 and one bacterial pneumonia), four patients died due to cardiovascular morbidity, two patients died due to sepsis (one soft tissue infection and one diabetic foot infection), two patients died due to cerebrovascular event, one died of pulmonary tuberculosis, one died of gastric cancer, and one died of acute pancreatitis. CML-unrelated death was significantly more frequent in patients with higher ECOG, Sokal, and ELTS scores (p < 0.001, 0.002, 0.001, respectively). Patients with higher ACE-27, ACCI, and ECI comorbidities scores were more frequent CML-unrelated deaths (p < 0.001, <0.001, 0.001, respectively; Table 4). While there was no significant relationship between CML-related death and ECOG, Sokal, ECI, and ACCI scores, CML-related death was more frequent in higher ELTS and ACE-27 scores (p = 0.032, 0.024, respectively; Table 4).
Cause of death according to performance status, prognostic scores, and comorbidity scores (n = 211).

p < 0.001; −2 Log Likelihood = 142.159.
ACCI, Age-adjusted Charlson Comorbidity Index; ACE, Adult Comorbidity Evaluation; ECI, Elixhauser Comorbidity Index; ECOG, Eastern Cooperative Oncology Group; ELTS, EUTOS long-term survival.
We evaluated the ability of three comorbidity indices (ACE-27, ACCI, and ECI) to discriminate mortality using C-statistics. C-statistic values were 0.76 (95% CI: 0.713–0.826) for ACE-27, 0.41 (95% CI: 0.367–0.465) for ACCI, and 0.32 (95% CI: 0.266–0.373) for ECI scores. When the C-statistic of the ACE-27 score was compared with ACCI and ECI, its ability to discriminate mortality was found to be more acceptable.
Overall survival
According to the ACE-27 Comorbidity Index, the median OS was significantly shorter in the moderate and severe groups. The median OS was 90.96 months (95% CI: 0.00–223.68) in the moderate group and 12.2 months (95% CI: 0.00–72.11) in the severe group (p < 0.001). The median OS was not reached in the none and mild groups. In the moderate group, 2-year survival was 85.7% and 5-year survival was 57.1%. In the severe group, 2-year survival was 50% and 5-year survival was 40%.
The median OS was significantly shorter in patients with ACCI scores 3–4 and >4. The median OS was 189.83 months (95% CI: 76.25–303.28) in the group with ACCI scores 3–4 and 103.3 months (95% CI: —) in the group with ACCI > 4 (p < 0.001). In the group with an ACCI of 3–4, 2-year survival was 100% and 5-year survival was 86.8%. In the ACCI >4 group, 2-year survival was 85.7% and 5-year survival was 71.4%.
The median OS in the group with ECI scores 1–5 was 189.83 months (95% CI: 62.18–317.48), while the median OS in the group with ECI scores >5 was not reached. In the group with ECI score 1–5, 2-year survival was 100% and 5-year survival was 95.5%. In the group with an ECI score >5, 2-year survival was 96.2% and 5-year survival was 86.1%.
The median OS decreased significantly with increasing ECOG scores. The median OS was 161.00 months (95% CI: 79.05–243.21) in the ECOG 1 group, was not reached in the ECOG 2 group, and 34.93 months (95% CI: —) in the ECOG 3 groups (p < 0.001). Median OS was not reached in patients with a high Sokal score, while median OS was significantly shorter in patients with high a ELTS score (p < 0.001). In patients with high Sokal scores, 2-year survival was 88.9% and 5-year survival was 84.9%. Median OS was determined as 26.2 months (95% CI: 0.00–71.89) in patients with high ELTS scores. In the group with a high ELTS score, 2-year survival was 75% and 5-year survival was 50% (Table 5 and Figure 3).
Comparison of patient variables with OS (n = 211).

Kaplan-Meier curve, Log rank test, p < 0.05 statistically significant.
ACCI, Age-adjusted Charlson Comorbidity Index; ACE, Adult Comorbidity Evaluation; ECI, Elixhauser Comorbidity Index; ECOG, Eastern Cooperative Oncology Group; ELTS, EUTOS long-term survival; OS, overall survival.

Kaplan-Meier curves of factors affecting overall survival. (a) ACCI, (b) ECI, (c) ACE-27. ACE: Adult Comorbidity Evulation; ACCI: Age-adjusted Charlson Comorbidity Index; ECI: Elixhauser Comorbidity Index.
Ten-year survival was lower in patients with hypertension (68% vs 92%, p < 0.001), and cardiovascular disease (74% vs 88%, p = 0.01). Diabetes mellitus, hypothyroidism, iron deficiency, solid tumor, chronic pulmonary disease, weight loss, and other comorbid conditions were not found related to lower OS (p = 0.078, p = 0.132, p = 0.670, p = 0.321, p = 0.211, p = 0.454, and p = 0.743, respectively).
Multivariate Cox regression analysis
As a result of the univariate analysis of the variables in Table 3, ACCI, ACE-27, and ECI variables were determined to be statistically significant in terms of mortality risk (p < 0.05). These variables, which were found significant as a result of univariate analyses, were included in the multivariate Cox regression model. According to the results of the multivariate Cox regression model, moderate ACE-27 score (HR: 148.05; 95% CI: 7.89–2751.53; p = 0.012), severe ACE-27 score (HR: 232.36; 95% CI: 14.20–4793.20; p = 0.001), ECOG 3 score (HR: 34.62; 95% CI: 2.67–447.36; p = 0.007), and high ELTS score (HR: 27.52; 95% CI: 1.34–543.68; p = 0.031) increased mortality. According to this model, there was no statistically significant relationship between ACCI and ECI scores and mortality risk (Table 6).
Multivariate Cox regression results for variables found to be significant as a result of univariate analysis.
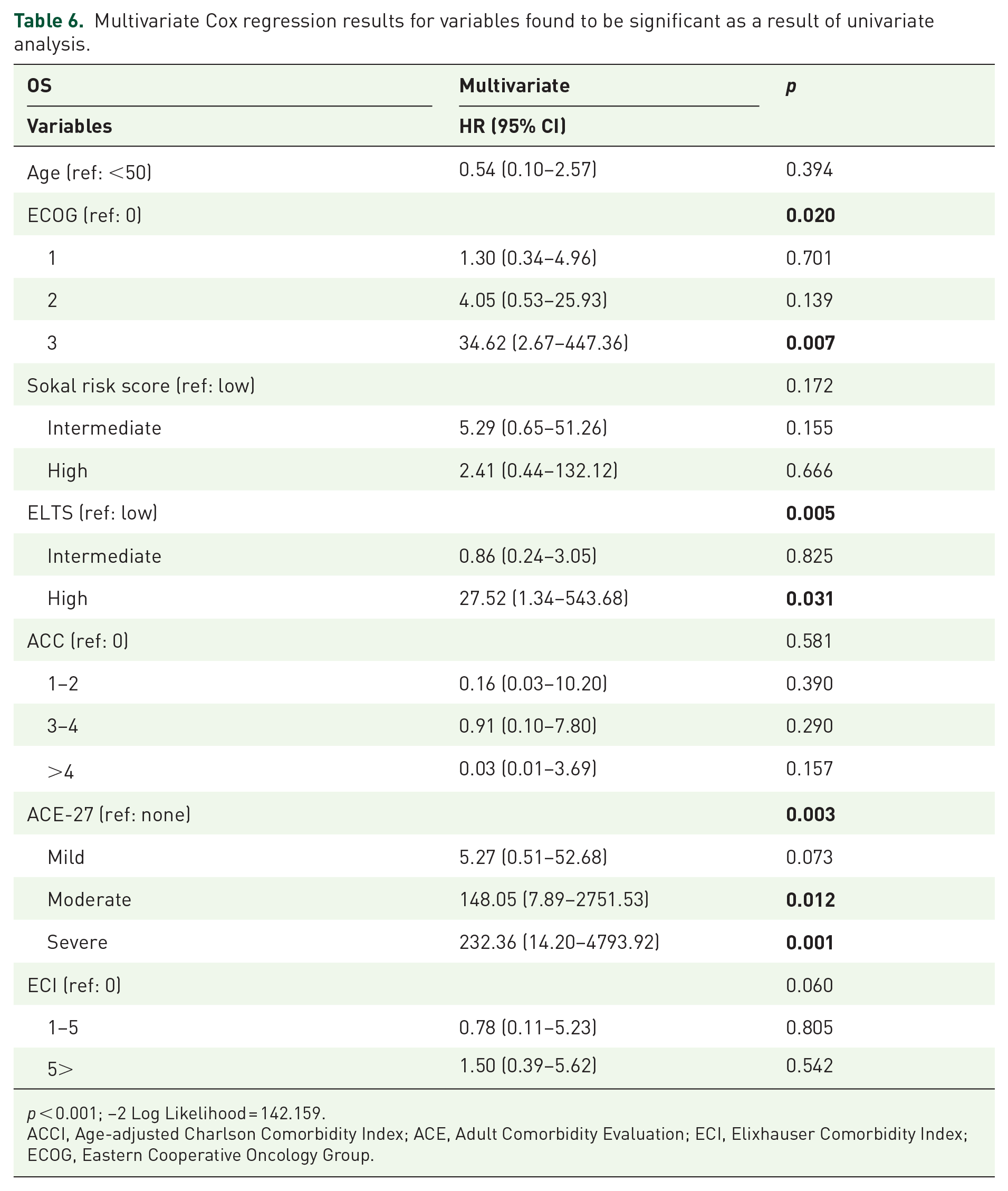
p < 0.001; −2 Log Likelihood = 142.159.
ACCI, Age-adjusted Charlson Comorbidity Index; ACE, Adult Comorbidity Evaluation; ECI, Elixhauser Comorbidity Index; ECOG, Eastern Cooperative Oncology Group.
Discussion
The prognosis of cancer patients is influenced by comorbidities and low-performance status. Recently studies on many types of cancer have shown that comorbidities not only affect cancer but also negatively affect survival by limiting treatment.13,14 In this study, we evaluated the relationship between all three comorbidity indices and OS and showed that they have a prognostic impact on patients. We showed that the ACE-27 Comorbidity Index was predictive of prognosis in patients with CML. According to our risk model, a high ACE-27 score, ECOG 3 performance status, and high ELTS score were effective on OS. Hypertension and cardiovascular disease were found related to lower OS. Hypertension, one of the variables included in the ACE-27 index, is not included in the ACCI and has a score of 0 points in the ECI. While weight loss provides 6 points in ECI, it is not available in ACCI and ACE-27. While diabetes mellitus provides points in ACE-27 and ACCI, it also receives 0 points in ECI. In addition, while obesity has a negative score in the ECI, it is not included in the ACCI. These variables may be valuable in the relationship of the ACE-27 index with OS.
In a study by Kim et al. evaluating 524 CML patients, median OS was not reached in patients with ACE-27 scores of 0 and 1, while median OS was 12.5 months in patients with scores of 2–3. OS was worse in patients with an ACE-27 score of 2–3. 5 In our study, median OS was not reached in the none, and mild groups, while 5-year survival in moderate and severe groups was 62.5% and 40%, respectively. Similarly, in our results, OS decreased as the ACE-27 score increased. In addition, we have shown for the first time in this study that the mortality risk was increased in moderate and severe groups.
In a clinical study conducted on patients with chronic-phase CML, a significant relationship between high ACCI scores and low OS was shown. Eight-year survival was 94% in patients with ACCI score 2 and 46% in patients with ACCI ⩾ 7. 15 Another study reported that ACCI was significantly associated with OS in CML patients treated with a TKI. 16 In our results, the median OS decreased as the ACCI score increased. In the group with an ACCI of 3–4, 2-year survival was 100% and 5-year survival was 86.8%. In the group with ACCI >4, 2-year survival was 85.7% and 5-year survival was 71.4%.
Alam et al. found that an increase in ECI score was associated with increased in-hospital mortality in Hispanic patients over 60 years of age with acute myeloid leukemia. 17 Wang et al. showed that the probability of treatment with TKIs decreased as the ECI score increased in a study evaluating 1251 patients with CML. However, this study did not adequately explain the relationship between ECI score and OS in patients with CML. 18 In our study, 5-year survival decreased in patients with higher ECI scores. Both the ACCI and ECI indexes had a predictive effect in determining prognosis in patients with CML. However, the AACI and ECI indices were no better than the ACE-27 index in predicting increased mortality risk.
The Sokal score was developed before the introduction of TKIs, while the ELTS scores were developed afterward. It has been shown that ELTS scores predict OS better than Sokal scores, especially in second-generation TKI (2GTKI) users. 19 In a study of 462 CML patients treated with imatinib, the ELTS score was able to effectively predict prognosis compared to Sokal and Hasford scores. 20 In our data, median OS was not reached in patients with a high Sokal score, whereas median OS decreased in patients with a high ELTS score. High ELTS scores were more effective than high Sokal scores in predicting mean OS. In addition, most of our patients were receiving imatinib as their initial TKI.
In a prospective study that included 1204 newly diagnosed CML patients (608 treated with imatinib, 598 treated with 2GTKI), a high ELTS score was associated with CML-related mortality. Age, high CCI score, high ELTS score, and patients treated with 2GTKI were associated with an increased risk of CML-unrelated mortality. 21 We examined three separate comorbidity indices, performance status, and prognosis scores in all causes of death in CML patients. Our findings showed that ACE-27 and ELTS were associated with increased CML-related mortality. Our findings showed that ELTS was more effective than the Sokal score in determining the risk of increased CML-related mortality.
A meta-analysis showed that second-generation and third-generation TKIs are better in newly diagnosed chronic-phase CML patients without comorbidities, while imatinib should be preferred in patients with comorbidities due to its toxicity profile. 22 In 125 chronic-phase CML patients treated with second-line dasatinib, the frequency of pleural effusion was shown to be increased in those with an ACCI score greater than 2 and an ACE-27 score of 2 or 3.
Of the 16 patients with CML-unrelated death, 11 patients were receiving imatinib and 5 patients were receiving 2GTKI (3 nilotinib, 2 dasatinib) at the time of death. The causes of death in patients receiving nilotinib were pneumonia, acute pancreatitis, and sepsis due to diabetic foot infection. Nilotinib has frequently been associated with cardiovascular toxicities such as ischemic heart disease, torsade de pointes/QT prolongation, and cardiac arrhythmias. Nilotinib can rarely cause pancreatitis.23,24 In this study, one patient receiving nilotinib died due to acute pancreatitis. COVID-19 pneumonia was the cause of death in two patients receiving dasatinib. Some studies in SARS-CoV-2-infected mice have shown that dasatinib reduced inflammation in lung disease and reduced COVID-19-related mortality.25,26
The ECOG performance score is used as a prognostic marker in patients with diffuse large B-cell and mantle-cell lymphoma.27,28 Our findings showed that median OS decreased as the ECOG score increased in patients with CML. Here, we determined that the ECOG status of the patients was predictive in terms of prognosis.
This study has some limitations. Firstly, the number of patients and mortality is low. Secondly, it was designed retrospectively.
Conclusion
CML prognostic scores, performance status, and comorbidity scores evaluate distinct aspects of patient health. The interplay between these scores highlights their complementary roles in patient management. Scoring systems such as Sokal and ELTS can only evaluate CML prognosis. However, these scoring systems alone are not sufficient to predict prognosis in patients with underlying comorbidities such as cardiovascular disease and hypertension. Our findings determined that comorbidities, lower performance, and high prognostic score have an impact on OS in chronic-phase CML patients. Nowadays, the importance of different prognostic markers is increasing with the increase in survival in CML. There is no standardized comorbidity index to evaluate the comorbid status of these patients at the time of diagnosis. This study showed that the ACE-27 Comorbidity Index is a predictive marker for prognosis. In addition, developing prognostic models that include these comorbidity conditions may be more useful in determining OS. These results can help the clinician determine the risk of mortality beforehand. Risk factors affecting mortality should be determined with different comorbidity indices, and appropriate prognostic models should be developed accordingly.
Supplemental Material
sj-pdf-1-tah-10.1177_20406207251323701 – Supplemental material for A comparison of the effect of three different comorbidity indices on overall survival in patients with chronic myeloid leukemia
Supplemental material, sj-pdf-1-tah-10.1177_20406207251323701 for A comparison of the effect of three different comorbidity indices on overall survival in patients with chronic myeloid leukemia by Cumali Yalçın, Bedrettin Orhan, Ömer Candar, Sinem Çubukçu, Tuba Güllü Koca, Fazıl Çağrı Hunutlu, Şeyma Yavuz, Merve Nur Akyol, Tuba Ersal, Vildan Özkocaman and Fahir Özkalemkaş in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
