Abstract
Background:
Epigenetic priming prior to chemotherapy represents a promising treatment strategy for refractory or relapsed diffuse large B-cell lymphoma (R/R DLBCL). We conducted a phase II trial to evaluate the efficacy and safety of azacitidine in combination with R-GDP (rituximab/gemcitabine/dexamethasone/cisplatin) in transplant-ineligible R/R DLBCL.
Methods:
Fifteen patients were enrolled and treated with azacitidine and R-GDP regimen (NCT03719989). Azacitidine was administered intravenously at a dose of 25 mg/m2/day for 5 days. Each cycle consisted of 21 days, with patients receiving up to a maximum of six cycles. The primary endpoint was the objective response rate, and the secondary objectives were toxicity, progression-free survival (PFS), and overall survival (OS).
Results:
Overall, 15 patients were enrolled in the study from March 2019 to August 2023, and the median age was 64 years (range: 41–75). The objective response rate was 66.7% with a complete response rate of 53.3%. The most common grade 3 or higher adverse events were hematologic toxicities, including neutropenia (66.7%) and thrombocytopenia (53.3%). Grade 3 or higher non-hematologic toxicities were rare, and most adverse events were transient and manageable. During a median follow-up of 15.8 months, five patients died, all from DLBCL. The median PFS was 12.6 months, while the median OS was not reached.
Conclusion:
Our study suggests that azacitidine followed by R-GDP is an effective and safe strategy for transplant-ineligible patients with R/R DLBCL. This represents the first phase II study to demonstrate the potential of epigenetic priming with azacitidine to enhance chemosensitivity in this patient population.
Keywords
Introduction
First-line standard treatment for diffuse large B-cell lymphoma (DLBCL) involves immunochemotherapy combining rituximab, which targets the CD20 antigen on the surface of B-cells, with conventional combination chemotherapy.1,2 Immunochemotherapy has shown curative potential even in CD20-positive tumors with a large tumor burden. 3 However, 30% of patients either fail to respond to R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, prednisolone) or experience relapse. 4 For these refractory or relapsed (R/R) patients, salvage chemotherapy and autologous stem cell transplantation (ASCT) are employed with curative intent. Nevertheless, the prognosis remains poor, particularly for patients who are ineligible for transplantation.5,6
R/R DLBCL is characterized by aberrant hypermethylation of cancer cell DNA, which is associated with disease progression and resistance to chemotherapy.7–9 Azacitidine, a nucleoside analog of cytosine, exerts its hypomethylating effects by incorporating it into DNA and RNA, resulting in the inactivation of DNA methyltransferase and subsequent inhibition of DNA methylation.10,11 A phase I clinical trial demonstrated that the regimen of azacitidine followed by R-CHOP is effective and safe in patients with newly diagnosed DLBCL. 12 In addition, Pera et al. found that azacitidine has the potential to induce epigenetic priming, thereby restoring delayed chemosensitivity to subsequent chemotherapy. 13 Hypomethylating agents can restore sensitivity to platinum-based chemotherapy and induce synergistic effects.14,15 However, clinical trials investigating the use of azacitidine-induced epigenetic priming followed by chemotherapy in patients with R/R DLBCL remain limited.
The R-GDP (rituximab, gemcitabine, dexamethasone, and cisplatin) regimen demonstrated lower toxicity compared to other regimens while achieving comparable efficacy in R/R DLBCL.16,17 Therefore, we conducted a prospective phase II trial to evaluate the efficacy and safety of azacitidine followed by R-GDP in transplant-ineligible patients with R/R DLBCL.
Methods
Participants and study design
This investigator-initiated, multicenter, open-label, single-arm, prospective phase II study started in December 2018 at seven university-affiliated hospitals in South Korea (ClinicalTrials.gov identifier, NCT03719989). The CONSORT 2025 checklist is provided in the Supplementary material.
Patients with histologically confirmed DLBCL who were either refractory to R-CHOP or relapsed after achieving complete response (CR) with R-CHOP were eligible. Eligible patients were aged 18–75 years, and had adequate organ function but were unsuitable for ASCT; an Eastern Cooperative Oncology Group (ECOG) performance score of 0–2; received one to four lines of previous therapy; and had measurable disease. The main exclusion criteria included leptomeningeal or central nervous system infiltration, DLBCL transformed from previously diagnosed indolent lymphoma, and intravascular DLBCL.
Treatment protocol
A first phase (lead-in safety cohort) was conducted with the following schedule: subcutaneous azacitidine 25 mg/m2 days (D)1–5, intravenous rituximab 375 mg/m2 on D8, intravenous gemcitabine 1000 mg/m2 on D8 and D15, intravenous or oral dexamethasone 40 mg on D8–11, and intravenous cisplatin 70 mg/m2 on D8, administered in cycles every 3 weeks. The administration of granulocyte colony-stimulating factor for neutropenia was permitted. Considering that dose-limiting toxicity (DLT) was observed at an azacitidine dose of 75 mg/m2 in a phase I study evaluating azacitidine followed by R-CHOP, 12 it was deemed unlikely that azacitidine 25 mg/m2 followed by R-GDP in this study would result in a significant increase in toxicity compared to R-GDP alone. However, given that this specific regimen has not been directly investigated, the initial 10% of the total target enrollment (N = 27), comprising three patients (up to a maximum of six), was designated as a lead-in safety cohort. After assessing the occurrence of DLTs and overall safety, the trial’s continuation was contingent upon a determination of tolerability by the Data Safety Monitoring Board (DSMB), which is operated independently by the Consortium for Improving Survival of Lymphoma (CISL), a national multicenter lymphoma clinical trial group.
Among the first three patients in the lead-in safety cohort, one case of DLT was observed. Subsequently, three additional patients were enrolled, resulting in the identification of one more DLT. Both DLTs were hematologic toxicities, specifically grade 4 thrombocytopenia and neutropenia. Based on the review by the DSMB, a second phase (run-in cohort) was conducted with a 20% reduction in the initial doses of gemcitabine (800 mg/m2 on D8 and D15) and cisplatin (56 mg/m2 on D8). For patients who did not experience grade 3 or higher adverse events (AEs) after the first cycle, dose escalation to gemcitabine 1000 mg/m2 and cisplatin 70 mg/m2 was permitted. Treatment could be administered for up to six cycles unless disease progression, unacceptable toxicity, or withdrawal of consent occurred. Dose modification of gemcitabine and cisplatin was performed based on the worst grade of toxicity, according to the approved protocol.
Outcomes
The primary endpoint was the objective response (CR + partial response (PR)) rate. The secondary endpoints included AEs, CR rate, progression-free survival (PFS), and overall survival (OS). Response evaluation was performed according to the Lugano classification, 18 utilizing 18F-fluorodeoxyglucose positron-emission tomography/computed tomography ( 18 F-FDG PET/CT) and spiral CT. Clinical evaluation at the time of study entry enrollment included medical history, physical examination, complete blood cell count, serum biochemistry, including lactate dehydrogenase, electrocardiogram, 18F-FDG PET/CT, and CT scan. 19 The stage of DLBCL was classified according to the Ann Arbor staging system. 20 For response evaluation, baseline assessments were conducted, followed by evaluations every two cycles. Patients who did not achieve a PR after four cycles were advised to discontinue participation in the study. A bone marrow (BM) examination was performed prior to the initiation of treatment. For patients with baseline BM involvement who demonstrated CR on imaging-based response evaluation after treatment completion, a follow-up BM examination was conducted for accurate response assessment. AEs were assessed according to the National Cancer Institute Common Terminology Criteria for AE v4.03. Following discontinuation of treatment, patients were monitored every 3 months.
Statistical analysis
This trial was designed with 80% power to reject the null hypothesis that the response rate is 40% or less, at a one-sided significance level of 0.05, assuming the true response rate to be 65% or higher. These hypotheses were based on the previously reported response rates of R-DHAP (rituximab, dexamethasone, cytarabine, and cisplatin) and R-GDP for R/R DLBCL.17,21 Fleming’s one-stage phase II design with an expected dropout rate of 10% indicated that 27 patients were required.
Descriptive statistics are presented as median values with ranges or numbers with percentiles. All efficacy and safety analyses included the intention-to-treat population (all enrolled patients). The Clopper-Pearson method was used for calculating the 95% confidence interval (CI) for the response rate. PFS and OS were defined as the time from enrollment to progressive disease (PD) or death from any cause, and the time from enrollment to death from any cause, respectively. Survival outcomes were estimated using the Kaplan-Meier method. The statistical software “R” version 4.3.1 (www.r-project.org) was used for all statistical analyses. p Values of <0.05 were considered statistically significant.
Results
Patient and disease characteristics
Overall, 15 patients were enrolled in the study from March 2019 to August 2023; follow-up continued until November 2024. Enrollment was terminated prior to reaching the target number because of an increasing number of competing clinical trials.
The baseline characteristics of the 15 patients are presented in Table 1. The median age was 64 years (range: 41–75), and eight patients (53.3%) were male. All patients had received six cycles of R-CHOP as first-line treatment, with a median time to progression of 11.4 months. Overall, 13 patients (86.7%) were categorized as stage III/IV, and 5 (33.3%) were classified as germinal B-cell type according to the Hans algorithm. Seven patients (46.7%) had received more than one prior line of treatment.
Baseline characteristics.

BMI, body mass index; ECOG, Eastern Cooperative Oncology Group; GCB, germinal center B-cell; LDH, lactate dehydrogenase.
Treatment outcomes
Overall, 10 patients (66.7%) were able to complete the treatment protocol and 6 (40.0%) completed all six cycles (Table 2). Five patients were withdrawn from the trial: two due to AEs, two due to withdrawal of consent, and one due to protocol violation.
Treatment outcomes.

CI, confidence interval; CR, complete response; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease.
Both efficacy and safety analyses were conducted in all 15 patients who received at least one cycle of azacitidine combined with R-GDP. Based on the best response, the objective response rate was 66.7% (95% CI: 38.4–88.2), with a CR rate of 53.3% (8/15). PD rate was 26.7% (4/15). Of 15 patients, 11 (73.3%) showed a reduction in tumor size from baseline (Figure 1). The response was stable among the responders, with a median duration of response of 13.4 months. Among the eight complete responders, the median duration of CR was not reached, and six had ongoing CR at the time of this analysis.

Waterfall plot showing the best percentage change in SPD from baseline.
During a median follow-up of 15.8 months (95% CI: 10.0–not reached), a total of five patients died, all due to disease progression. The median PFS was 12.6 months (Figure 2(a)), with a 2-year PFS rate of 36.7% (95% CI: 16.6–80.9). The median OS was not reached (Figure 2(b)), with 1- and 3-year OS rates of 77.0% (95% CI: 57.0–100.0) and 53.9% (95% CI: 29.8–97.5), respectively.
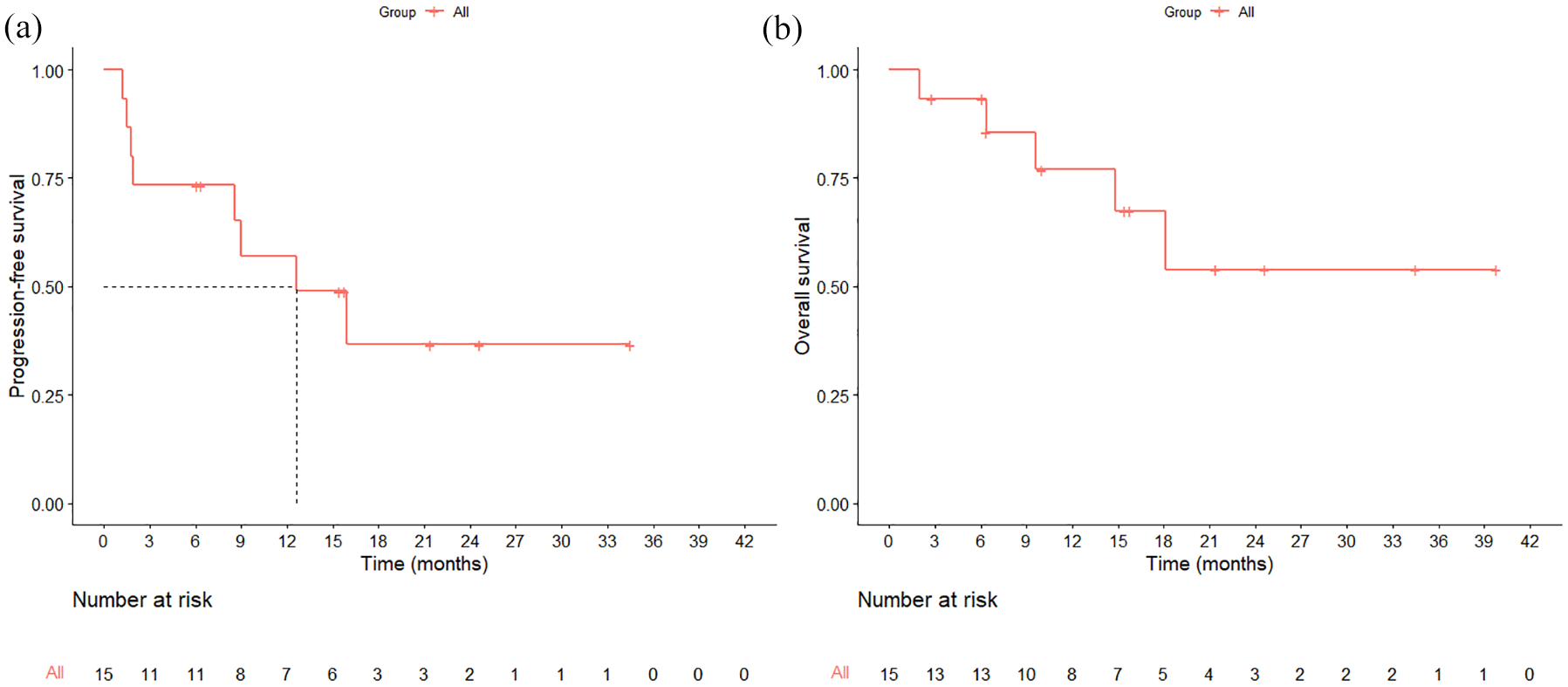
Kaplan-Meier curve of survival outcomes: (a) progression-free survival and (b) overall survival.
Hematologic and non-hematologic AEs
During treatment, a total of 10 patients (66.7%) underwent dose reductions; 7 for both gemcitabine and cisplatin due to cytopenia, and 3 for cisplatin alone due to impaired renal function per protocol. In cycle 1, all but one patient received the full dose of R-GDP; one received a 25% reduced dose of cisplatin. In cycle 2, a total of six patients underwent dose reductions. Four patients received a 20% reduced dose of gemcitabine and cisplatin, and two patients had a dose reduction of cisplatin alone.
Hematologic AEs for each cycle are presented in Figure 3. The most common AEs of all grades were neutropenia (73.3%), followed by thrombocytopenia (60.0%; Table 3). The most common grade 3 or higher AEs were neutropenia, impacting 10 patients (66.7%). Except for two patients, dose delays and reductions allowed treatment to proceed according to the protocol. However, the two patients who could not complete the study due to AEs experienced neutropenia as the primary cause.

Hematologic adverse events across treatment cycles. The percentile of each adverse event in each cycle was calculated based on the number of patients who received that specific cycle.
Adverse events.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma-glutamyltransferase.
Treatment-emergent non-hematologic AEs in the 15 patients under study were summarized in Table 3. The most common AEs were fatigue (33.3%) and nausea (33.3%), followed by vomiting (26.7%) and skin rash (20.0%; Figure 4). Two patients (13.3%) had serious AEs. One patient (6.7%) developed grade 2 cerebral infarction during cycle 1, but was able to continue treatment. This patient achieved a CR and completed all six cycles of treatment. Another patient (6.7%) developed grade 3 pneumonia and grade 2 pulmonary thromboembolism at cycle 5, necessitating a dose reduction. There were no treatment-related deaths in this study.
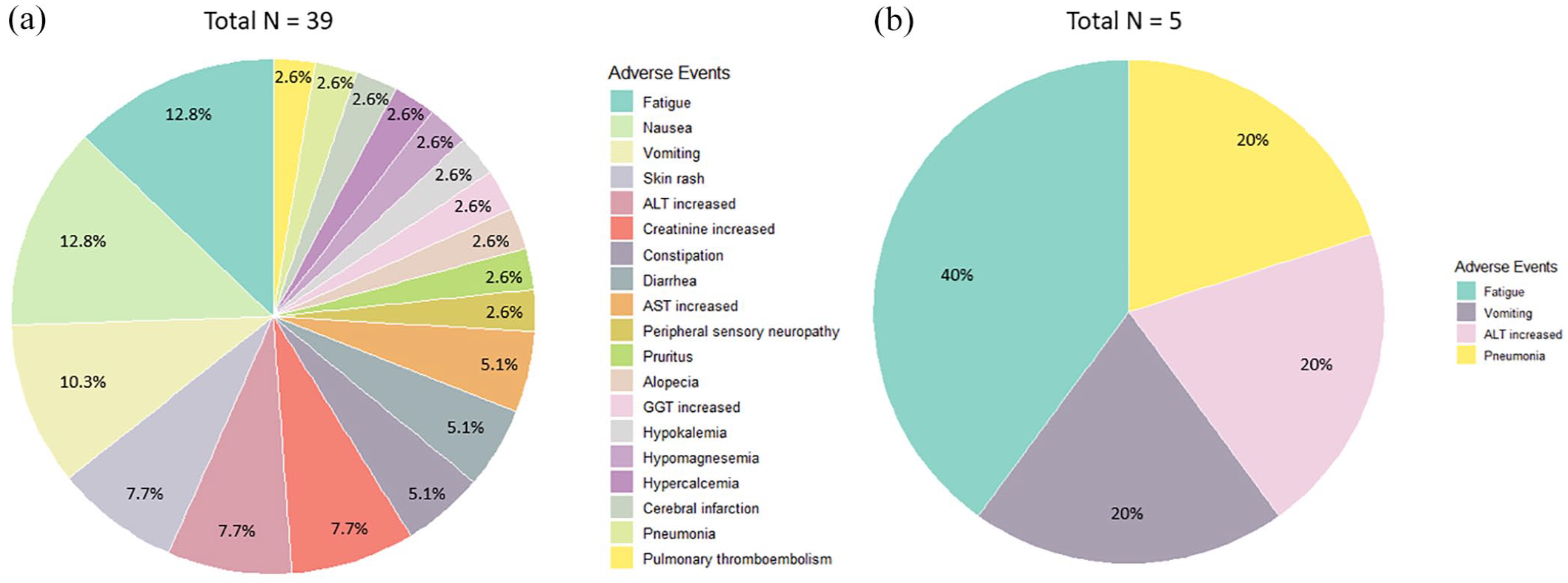
Distribution of non-hematologic adverse events (%): (a) any grade and (b) grade ⩾3.
Discussion
In this prospective study, we combined epigenetic priming with azacitidine and conventional salvage immunochemotherapy R-GDP for the treatment of transplant-ineligible R/R DLBCL. The objective response rate was 66.7%, with a CR rate of 53.3%. The response was durable, and the toxicity profile was manageable. The results of the present study suggest that azacitidine priming prior to R-GDP is an effective treatment approach for transplant-ineligible patients with R/R DLBCL.
Advancements in human genome analysis have revealed that epigenetic abnormalities play a significant role in cancer progression and treatment resistance across various cancer types, including DLBCL.22–26 In DLBCL, aberrant hypermethylation activates BCL6 and suppresses the function of tumor suppressor genes. 27 Azacitidine and decitabine, as hypomethylating agents, are widely used in the treatment of hematologic malignancies and exert various effects, including cytotoxic and immune-modulating activities.28,29 These hypomethylating agents exhibit dose-dependent effects. 30 Previous clinical trials have demonstrated that low-dose decitabine at 0.2 mg/kg/day leads to DNA methyltransferase 1 (DNMT1) depletion or a reduction in DNMT1 protein levels.31,32 These findings suggest that even relatively low doses of hypomethylating agents may effectively correct hypermethylation. We determined the azacitidine dose to be 25 mg/m2/day based on a previous phase I study evaluating the combination of azacitidine and R-CHOP in DLBCL. This study enrolled 12 patients, and azacitidine was administered on D1–5 with escalating doses of 25, 50, and 75 mg/m2/day. DLT occurred in two patients treated with 75 mg/m2/day, while effective demethylation was achieved even at a dose of 25 mg/m2/day, 12 suggesting that low-dose azacitidine prior to chemotherapy can safely induce hypomethylation without increasing cytotoxicity. Our previous study demonstrated that low-dose azacitidine pretreatment can induce DNA demethylation without causing DNA damage and resensitize cisplatin-resistant DLBCL to cisplatin. 33 This epigenetic priming strategy can be significant in R/R DLBCL, as it enhances sensitivity to salvage chemotherapy without increasing toxicity.
To date, several studies have investigated epigenetic priming prior to chemotherapy in DLBCL. Recently, Chen et al. reported an objective response rate of 79.1% and a CR rate of 45.8% in a phase II study evaluating chidamide followed by R-GDP in patients with R/R DLBCL. 34 Chidamide, a histone deacetylase inhibitor, has demonstrated efficacy in DLBCL when used either as monotherapy 35 or in combination with other agents.36,37 Pera et al. conducted a phase I study evaluating the combination of azacitidine and vorinostat, another histone deacetylase inhibitor, in 18 patients with R/R DLBCL. 13 While the toxicity was manageable, the outcomes were suboptimal, with no CR and only one PR. Notably, subsequent cytotoxic chemotherapy yielded favorable outcomes, suggesting that epigenetic priming has induced a chemosensitization effect in R/R DLBCL. In previous clinical trials evaluating the combination of DNMT inhibitors with R-CHOP in treatment-naïve DLBCL, objective response rates exceeding 90% were reported.38,39 In patients with R/R DLBCL, previous trials reported objective response rates of 50% and 40% for decitabine followed by DHAP 40 or R-DHAP, 41 respectively, with a median PFS of 7 months for both regimens. In this study, we combined azacitidine with the R-GDP regimen in transplant-ineligible patients with R/R DLBCL. We achieved the primary endpoint of the trial, with an objective response rate of 66.7% and an impressive CR rate of 53.3% in patients with R/R DLBCL. The median PFS exceeded 1 year, while the median OS was not reached. Among salvage chemotherapy regimens such as R-GDP and R-DHAP for R/R DLBCL, the efficacy is comparable, while R-GDP is more tolerable.21,42 In transplant-ineligible patients, the combination of azacitidine with R-GDP resulted in rare non-hematologic toxicities, which were mostly transient and manageable. In addition, no treatment-related deaths were observed. The results of this study demonstrate that low-dose azacitidine is safe and active, with a potential synergistic effect with R-GDP in R/R DLBCL.
Cytopenia is an expected toxicity of both azacitidine and cytotoxic chemotherapy, particularly in R/R patients with prior exposure to chemotherapy. The most common AE was neutropenia, which occurred at grade 3 or higher in 66.7% of our study. This aligns with the results of two clinical trials evaluating azacitidine combined with R-CHOP in previously untreated DLBCL, where the incidence of grade 3 or higher neutropenia was reported as 62.7% and 100%, respectively.12,38 Although hematologic toxicity appeared higher compared to R-GDP alone, most AEs were transient and reversible through dose reductions, with treatment discontinuation due to toxicity being infrequent. Furthermore, non-hematologic AEs were also manageable, with grade 3 or higher occurrences being rare.
Recently, significant advancements have been made in the treatment of B-cell lymphoma with advanced CD20 and CD79a inhibitors, bispecific T-cell engager, chimeric antigen receptor T-cell (CAR-T) therapies, and Bruton’s tyrosine kinase inhibitors.43–46 Consequently, this trial was terminated early due to poor accrual. However, this study, along with recent findings, suggests that treatment approaches incorporating epigenetic priming show promising efficacy and manageable toxicity in the R/R setting. Although our study was conducted in ASCT-ineligible patients, its high response rate and PFS suggest potential applicability in the salvage setting, either as bridging therapy prior to CAR-T or as a later-line treatment following relapse after CAR-T or ASCT. Further research is needed to explore this approach, with a focus on improving treatment convenience, such as the use of oral hypomethylating agents.
This study had several limitations. The first was the small sample size due to the early termination of the trial. We were unable to identify indicators associated with response or survival outcomes. Second, this trial was designed as a single-arm study. Therefore, large-scale comparative studies are required to validate our findings. In addition, a biomarker analysis of epigenetic changes was not conducted. Although previous studies have reported effective demethylation even at a low dose of azacitidine, we were unable to validate changes in methylation before and after treatment in our study. In addition, while we previously reported the biochemical effects of azacitidine pretreatment in DLBCL, 33 further research is warranted to identify predictive biomarkers that could guide clinical practice in epigenetic priming strategies.
Conclusion
Our study demonstrated that the combination of azacitidine with the R-GDP regimen is an effective treatment option for patients with R/R DLBCL following R-CHOP failure, achieving a CR rate of over 50%. To the best of our knowledge, this is the first phase II study to evaluate the efficacy and safety of epigenetic priming with azacitidine, followed by R-GDP in transplant-ineligible patients with R/R DLBCL. The use of hypomethylating agents for chemosensitization can be a promising approach when employing cytotoxic agents. Efforts to improve the outcomes of conventional treatments remain important, and further studies with larger sample sizes and translational analyses are warranted to validate this combination in R/R DLBCL.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251349361 – Supplemental material for Azacitidine followed by R-GDP in transplant-ineligible relapsed/refractory diffuse large B-cell lymphoma: preliminary results from a multicenter, phase II study
Supplemental material, sj-docx-1-tah-10.1177_20406207251349361 for Azacitidine followed by R-GDP in transplant-ineligible relapsed/refractory diffuse large B-cell lymphoma: preliminary results from a multicenter, phase II study by Dong Hyun Kim, Jee Hyun Kong, Junshik Hong, Ja Min Byun, Dong-Yeop Shin, Youngil Koh, Inho Kim, Jinny Park, Young Rok Do, Jeong-A Kim, Won Seog Kim, Ho-Jin Shin and Sung-Soo Yoon in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
