Abstract
Background:
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is crucial for treating high-risk hematological cancers. Posttransplant infections are a leading cause of mortality and morbidity, especially before engraftment. This study evaluated the effects of granulocyte transfusion (GT) therapy on neutrophil and platelet (PLT) engraftment in patients with febrile neutropenia during the pre-engraftment phase following allo-HSCT with a control group.
Methods:
We retrospectively analyzed 56 patients who underwent allo-HSCT from January 2019 to January 2024, dividing them into two groups: those who received GT (GTG) and those who did not (non-GTG).
Results:
A total of 76 GTs were administered to 28 patients with febrile neutropenia during the pre-engraftment period. The median granulocyte dose was 5.4 × 108/kg. Median engraftment times in the GTG were 13 days for both PLT and neutrophil engraftment, compared to 15.5 days (PLT) and 19 days (neutrophil) in the non-GTG (p < 0.001 for neutrophil and p = 0.007 for PLT). Additionally, 89.3% of patients in the GTG showed improved infection status. Overall survival (OS) at 2 year was 61.4% for GTG and 73.2% for non-GTG, respectively. No significant difference in OS between the groups (p > 0.05). In the non-GTG, the OS rate was 47.5%, with no significant difference in OS and mortality rates between the groups. GT did not affect the incidence of graft-versus-host disease or cytomegalovirus infection.
Conclusion:
This study is the first to include a control group and demonstrate a statistically significant association between GT therapy in the pre-engraftment period and shortened engraftment times in allo-HSCT recipients.
Plain language summary
This study evaluated the effects of granulocyte transfusion (GT) therapy on neutrophil and platelet (PLT) engraftment in patients with febrile neutropenia during the pre-engraftment phase following allo-HSCT with a control group and is the first to demonstrate a statistically significant association between GT therapy in the pre-engraftment period and shortened engraftment times in allo-HSCT recipients.
Keywords
Introduction
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) remains a cornerstone curative therapy for high-risk hematological malignancies and severe immune deficiencies. 1 Although this therapeutic approach has demonstrated good success rates for disease eradication, life-threatening complications such as infections remain major problems after HSCT.2,3 Despite the most appropriate antimicrobial and supportive treatment, bloodstream infections occur in 20%–40% of allo-HSCT recipients due to treatment-related neutropenia, and 12%–42% of these cases are fatal. 4 Three different phases of infection risk are described for post-HSCT patients: the early pre-engraftment phase, the early postengraftment phase, and the late phase. Neutropenia, delayed engraftment due to prolonged neutropenia, and mucosal damage are the leading risk factors in the early pre-engraftment phase for the development of severe bacterial and fungal infections in patients undergoing HSCT.5,6
For over 50 years, granulocyte transfusion (GT) therapy has been used as a supportive measure to bridge the interval until hematopoietic regeneration and to aid in the resolution of infection. Many studies have shown that it is a useful supportive therapy in cases of neutropenia.7–11 However, it remains unclear whether GT affects clinical outcomes. 12 Furthermore, the data on GT therapy, particularly in the context of the HSCT setting, are limited, and most studies have been conducted in very small patient populations and at any time point of allo-HSCT without specificating only to the pre-engraftment period.10,13–18
In our clinic, we apply GT therapy in cases of febrile neutropenia in patients who underwent allo-HSCT. With this application, we observed that these patients’ neutrophil recovery period was shortened. When we reviewed the literature on this subject, only Ikegawa et al. 13 mentioned the application of pre-engraftment period GT therapy in allo-HSCT patients and reported recovery of neutrophil values after GT in 17 of 20 allo-HSCT patients with active infection. Nikolajeva et al. 15 published their 10-year single-center experience with 28 pediatric patients with known bacterial and fungal infections but did not specify in their article on which day of allo-HSCT they applied GT treatment.
Based on this point, we retrospectively aimed to investigate the relationship between GT and the shortening of the duration of neutropenia and engraftment in patients who developed febrile neutropenia after undergoing allo-HSCT in the pre-engraftment period.
Materials and methods
This retrospective study included 56 patients who developed febrile neutropenia in the pre-engraftment period after undergoing allo-HSCT at Erciyes University Bone Marrow Transplantation Unit from January 2019 to January 2024. Patients were divided into two groups: 28 received granulocyte transfusion (GTG) and 28 did not (non-GTG). Patient characteristics were recorded. GT was administered to patients with an absolute neutrophil count (ANC) <0.5 × 103/µL for at least 3 days, evidence of bacterial and/or fungal infection, and no response to appropriate antimicrobials for at least 48 h.
The study was approved by the Erciyes University Faculty of Medicine Ethics Committee (Date: 03.04.2024, Decision No.: 2024/250) and followed ethical guidelines and the Helsinki Declaration principles.
Granulocyte donors, collections, and transfusions
Donors aged 18–65 were selected based on compatible ABO and Rh blood groups, negative serological tests (HIV, cytomegalovirus (CMV), hepatitis B, hepatitis C, syphilis), and normal blood counts. Donors received 5 μg/kg granulocyte colony-stimulating factor (G-CSF) and 8 mg oral dexamethasone 8–16 h before apheresis. Granulocyte collection was performed using the Spectra Optia device, targeting a minimum of 1 × 1010 granulocytes per product. The granulocyte concentrate was irradiated (25 Gy) before transfusion. Patients received premedication with acetaminophen, hydrocortisone, and chlorpheniramine maleate before each GT, with vital signs monitored throughout the process. No adverse events requiring medical intervention were observed during the GTs.
GT was administered until the ANC was ⩾0.5 × 103/μL or until clinical signs of infection were controlled. Infections were categorized as bacterial, fungal, or viral. Clinical response to GT was defined by patient improvement, fever defervescence, infection control, and absence of proliferation in control cultures. Infection outcomes were assessed according to the Infectious Diseases Society of America and FDA guidelines.19–21
Engraftment criteria and data collection
Neutrophil engraftment was defined as the first of three consecutive days with an ANC ⩾0.5 × 103/μL. Platelet (PLT) engraftment was defined as a PLT count ⩾20,000/mm3 without PLT transfusions in the prior 7 days. 22 For non-GTG patients, PLT, total leukocyte count (TLC), and ANC values were recorded on days 5, 10, 15, and 20 post-allo-HSCT. For GTG patients, these values were recorded on days 5 and 10, before GT initiation, 4 days after GT, and 15 and 20. Data recorded included granulocyte dose, number of transfusions, infection improvement, C-reactive protein (CRP) value, type of infection, 30- and 100-day mortality, relapse, overall survival (OS), development of acute or chronic graft-versus-host disease (GVHD), and CMV infection.
Statistical analysis
Statistical analyses were conducted using IBM SPSS Statistics for Windows, Version 25.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics are presented as counts and percentages for categorical variables and as median (min–max) or median interquartile ranges (IQR) for continuous variables. Normality was tested using the Kolmogorov–Smirnov test, with p < 0.05 indicating a nonnormal distribution. The Mann–Whitney U test was used for comparisons between the two groups. The Spearman correlation test assessed the relationship between continuous variables. Pearson’s Chi square and Fisher’s exact tests compared categorical variables. Survival rates and nonrelapse mortality (NRM) were estimated using the Kaplan–Meier method, with comparisons conducted using the log-rank test. A p-value of <0.05 was considered statistically significant.
Results
The baseline characteristics of the 56 patients are summarized in Table 1. There were 28 patients in both the GTG and non-GTG. The median age of patients in GTG was 44 years (min–max, 20–66 years), and for non-GTG, it was 44 years (min–max, 19–66 years) (p = 0.72). In GTG, 46.4% of the patients were female and 53.6% were male. In non-GTG, 35.7% were female and 64.3% were male (p = 0.41). There was no statistical difference between the two groups regarding age and gender. The majority of patients in both groups were diagnosed with acute myeloid leukemia (60.7% for GTG and 42.9% for non-GTG; p = 0.18).
Baseline characteristics of the patients.

Pearson’s Chi square test.
Mann–Whitney-U test.
Fisher’s exact test.
ALL, acute lymphoblastic leucemia; allo-HSCT, allogeneic hematopoietic stem cell transplantation; AML, acute myeloid leukemia; CMML, chronic myelomonocytic leucemia; CMV, cytomegalovirus; CNI, calcineurin inhibitor; CRP, C-reactive protein; GTG, granulocyte transfusion group; GVHD, graft versus host disease; HLA, human leukocyte antigen; ICU, intensive care unit; IGG, immunoglobulin G; MMF, mycophenolate mofetil; MRSA, methicillin-resistant Staphylococcus aureus; NA, not available; non-GTG, non-granulocyte transfusion group; PTCY, posttransplant cyclophosphamide; RIC, reduced intensity conditioning; sMTX, short-term methotrexate.
Haploidentical allo-HSCT and myeloablative conditioning regimens were more common in GTG (17.9% (n = 5), 57.1% (n = 16)) compared to non-GTG (3.6% (n = 1), 39.3% (n = 11)), but these differences were not statistically significant.
In GTG, the median CRP value was 119 mg/dL (min–max, 35–332 mg/dL), while in non-GTG, the median CRP value was 72.5 mg/dL (min–max, 30–240 mg/dL), with CRP values being statistically higher in GTG (p = 0.007).
The infection etiologies for GTG included pneumonia (n = 5), sepsis (n = 2), pneumonia and sepsis (n = 18), pneumonia + sepsis + catheter-associated infection (n = 1), urinary infection (n = 1), and BK virus + catheter-associated infection (n = 1). For non-GTG, the etiologies were pneumonia (n = 7), sepsis (n = 5), pneumonia and sepsis (n = 1), catheter-associated infection (n = 1), sepsis + catheter-associated infection (n = 1), urinary infection (n = 4), mucositis (n = 4), pneumonia + sepsis + BK virus (n = 2), perianal infection + BK virus (n = 1), pneumonia + BK virus (n = 1), and perianal infection + sepsis (n = 1).
In GTG patients, bacterial infections were observed in 78.6% (n = 22), fungal infections in 64.3% (n = 18), and viral infections in 7.1% of cases (n = 2). In non-GTG patients, these rates were 53.6% (n = 15), 46.4% (n = 13), and 14.3% (n = 4), respectively. The incidence of bacterial infections was significantly higher in GTG patients (p = 0.04), while the differences in fungal and viral infection rates were not statistically significant between GTG and non-GTG patients (p = 0.17 and p = 0.66, respectively).
Characteristics of the donor and GT
Among the 76 donors, the median age was 34 years (min–max, 18–59 years; Table 2). A total of 76 GTs were administered. Each patient received a median of 2.5 GTs (min–max, 1–6). The median transfused granulocyte dose per transfusion was 3.8 × 1010 (min–max, 1–9.4 × 1010). The median dose transfused, calculated based on the recipient’s body weight, was 5.4 × 108/kg (min–max, 1–18.1 × 108/kg). On average, the median number of granulocytes transfused per patient was 6.1 × 108/kg (min–max, 1.39–12.9 × 108/kg). The median time from HSCT to the first GT was 11.42 days (min–max, 8–14 days). The median duration of fever before GT was 3 days (min–max, 2–6 days), and the time until the fever defervescence was 1.5 days (min–max, 1–9 days).
The main characteristics of the granulocyte donor and the granulocyte product.

GT, granulocyte transfusion.
The TLC, ANC, and PLT counts were statistically lower in GTG patients on allo-HSCT day 0 (p-values: p = 0.024, p = 0.19, p = 0.004, respectively; Figures 1–3). There was no statistically significant difference in TLC, ANC, and PLT counts between GTG and non-GTG on the 5th and 10th days after allo-HSCT (p > 0.05).

Distribution of TLC of GTG, and non-GTG according to allo-HSCT days.

Distribution of ANC of GTG, and non-GTG according to allo-HSCT days.

Distribution of PLT values of GTG, and non-GTG according to allo-HSCT days.
On post-allo-HSCT day 15, TLC, ANC, and PLT counts were as follows: 3.90 × 103/μL (IQR, 2.98 × 103/μL), 2.54 × 103/μL (IQR, 2.54 × 103/μL), and 42 × 103/mm3 (IQR, 66 × 103/mm3) versus 1.05 × 103/μL (IQR, 0.84 × 103/μL), 0.37 × 103/μL (IQR, 0.33 × 103/μL), and 49 × 103/mm3 (IQR, 118.75 × 103/mm3), for GTG and non-GT, respectively. Although TLC and ANC values were significantly higher in GTG on post-allo-HSCT day 15 (p < 0.001 for both), there was no difference between the groups regarding PLT counts (p = 0.31).
TLC, ANC, and PLT counts were as follows: 5.16 × 103/μL (IQR, 4.08 × 103/μL), 2.9 × 103/μL (IQR, 2.84 × 103/μL), and 61 × 103/mm³ (IQR, 64 × 103/mm³) versus 2.68 × 103/μL (IQR, 2.39 × 103/μL), 1.23 × 103/μL (IQR, 1.71 × 103/μL), and 107 × 103/mm³ (IQR, 115.5 × 103/mm³), on post-allo-HSCT day 20, for GTG and non-GTG, respectively. TLC, ANC, and PLT counts were significantly higher in GTG on post-allo-HSCT day 20 (p = 0.004, p < 0.001, p = 0.002, respectively).
The median ANC, TLC, and PLT counts increased from 0.05 × 103/μL (IQR, 0.31 × 103/μL) to 1.84 × 103/μL (IQR, 1.4 × 103/μL), from 0.23 × 103/μL (IQR, 0.47 × 103/μL) to 2.11 × 103/μL (IQR, 1.54 × 103/μL), and 25 × 103/μL (IQR, 25 × 103/μL) to 60 × 103/mm³ (IQR, 51 × 103/mm³) on first-day post-GT, showing a statistically significant difference, respectively (p < 0.001). The median ANC, TLC, and PLT counts were 2.5 × 103/μL (IQR, 3.28 × 103/μL), 3.92 × 103/μL (IQR, 3.91 × 103/μL), and 60 × 103/mm³ (IQR, 51 × 103/mm³), respectively, on the fourth-day post-GT. A statistically significant difference was found between the time of GT initiation and the ANC, TLC, and PLT counts on the fourth-day post-GT (p < 0.001, separately for ANC, TLC, and PLT). Moreover, there was a significant correlation between the transfused granulocyte dosage and the ANC value (Spearman correlation coefficient: 0.439, p = 0.019; Figure 4).

The relationship between granulocyte dosage and the increment of neutrophil count 1 day after granulocyte transfusion.
Engraftment
For the GTG, the median day for PLT and neutrophil engraftment was 13 (min–max: 7–28, IQR: 2) days and 13 (min–max: 9–19, IQR: 2.7) days, respectively. For the non-GTG, the median day for PLT and neutrophil engraftment was 15.5 (min–max: 10–27, IQR: 4.5) days and 19 (min–max: 16–21, IQR: 2) days, respectively. There was a statistically significant difference between the GTG and non-GTG in terms of neutrophil and PLT engraftment time (p < 0.001 and p = 0.007, respectively), with engraftment being significantly shorter in the GTG compared to the non-GTG (Table 3).
Comparison of parameters affecting posttransplant outcomes in allo-HSCT recipients.

Pearson’s Chi square test.
Mann–Whitney U test.
Fisher’s exact test.
GVHD, graft-versus-host-disease; NA, not avaliable.
OS outcomes
For the GTG, the median follow-up was 17 months (min–max, 0.5–60 months). GTG general median OS (months) could not be obtained. The 30-day mortality rate was 3.6%, 100-day mortality was 3.6% and 1-year NRM was 19.8%. The 30-day, 100-day, 1-year, and 2-year OS rates were 96.4%, 88.4%, 80.2%, and 61.4%, respectively.
For the non-GTG, the median follow-up was 25.5 months (min–max, 4–57 months). The 30-day mortality rate was 0%, 100-day mortality was 0% and 1-year NRM was 18.2%. The 30-day, 100-day, 1-year, and 2-year OS rates were 100%, 100%, 81.8%, and 73.2%, respectively. There was no statistically significant difference between GTG and non-GTG in terms of survival and NRM at day 30 (p > 0.05; Figure 5).
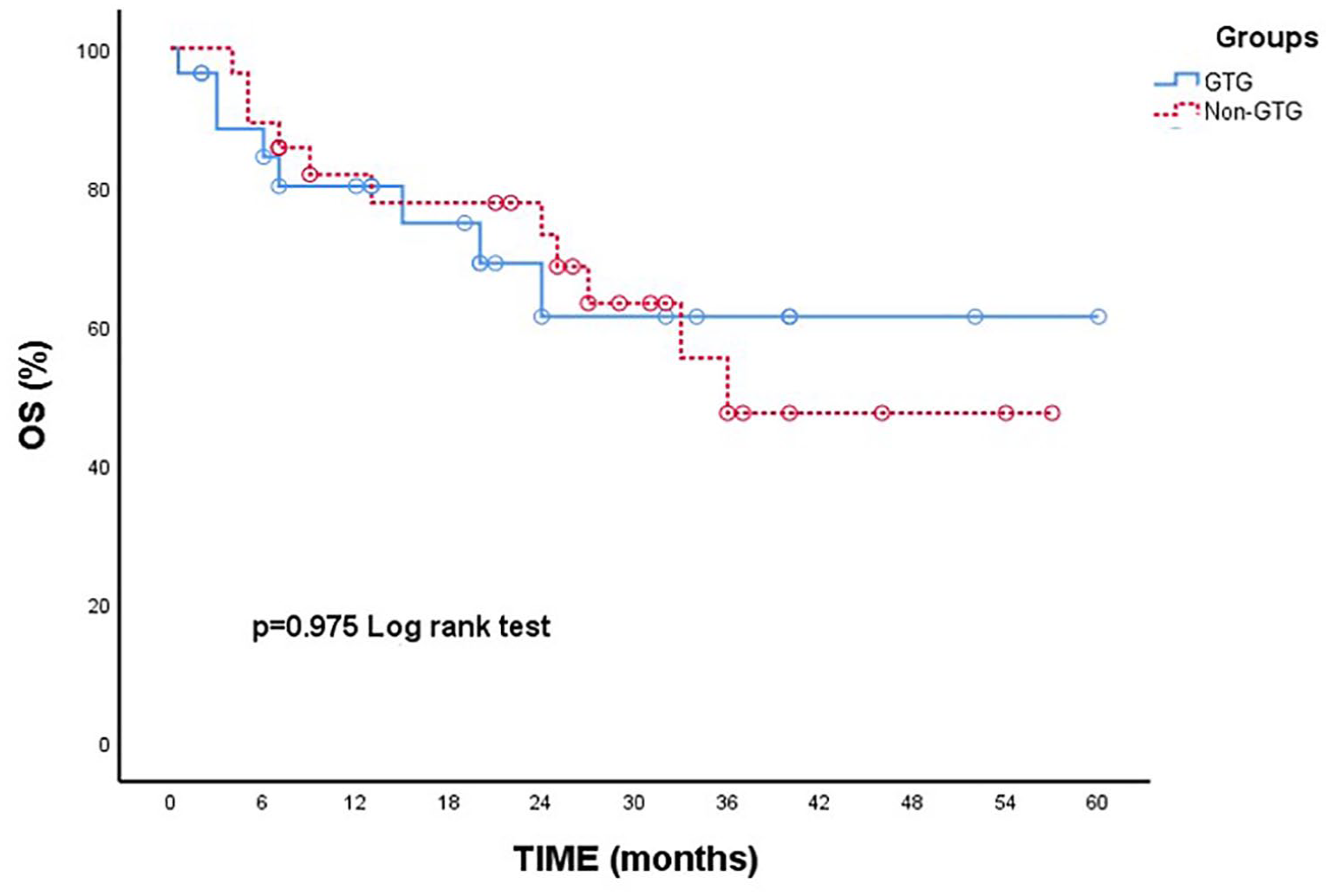
OS after allogeneic hematopoietic stem cell transplantation.
When we divided the recipients into two groups based on transfused granulocytes (GT-high group: ⩾5 × 108/kg and GT-low group: <5 × 108/kg), there was no significant difference in the 30-day survival, 100-day survival, 1-year OS, and 2-year OS between those who received a GT dose of ⩾5 × 108/kg, and those who received a dose of <5 × 108/kg (p > 0.05).
In GTG, 8 out of 28 patients (28.6%) died. In non-GTG, 11 out of 28 patients (39.3%) died. Relapse was observed in 3.6% (1/28) of GTG patients, while it was 21.4% (6/28) in non-GTG patients. There was no statistical difference in terms of death and relapse between the groups (p = 0.39, p = 0.1, respectively; Table 3).
The causes of death for GTG patients were as follows: bacterial infection in three patients, organ failure in two patients, relapse of primary disease in one patient, COVID-19 in one patient, and chronic GVHD-related mortality in one patient. In two of the three patients who died due to infection, death occurred not from the infection attack for which GT treatment was applied but from another infection attack during their follow-up. In the remaining patient, death occurred as a result of an infection attack during the period when GT treatment was being applied. For non-GTG patients, the causes of death were as follows: bacterial infection in eight patients (a bacterial infection attack occurring sometime after the first 100 days after allo-HSCT), organ failure in one patient, COVID-19 in one patient, and recurrence of the primary disease in one patient.
Infection outcomes
Among the patients who received GT, 89.3% (25/28) showed improvement in their infection symptoms, while 10.7% (3/28) did not. Of the three patients whose infections did not improve, two received GT only once due to the unavailability of a donor, and GT treatment could not be continued. Consequently, one of these two patients required intensive care unit (ICU) admission and subsequently died. In the third patient, the myeloablative and haploidentical transplantation procedure was administered. In the GTG, 14.3% (4/28) of patients required ICU admission after infection, compared to 3.6% (1/28) in the non-GTG. Although the need for ICU was higher in the GTG, there was no statistical difference between the groups (p = 0.35). Of the four patients in the GTG who required ICU, three no longer needed ICU after GT.
Acute, chronic GVHD, and CMV infection
Acute GVHD was not observed in either the GTG or non-GTG. The rates of chronic GVHD at 1-year were 35.7% for GTG and 46.4% for non-GTG, respectively. Although chronic GVHD was more common in the non-GTG, the difference was not statistically significant (p > 0.05). According to our data, GT treatment did not increase the rate of chronic GVHD development.
The rate of CMV infection post-GTG was 25% (7/28), which was similar to the non-GTG rate of 25% (7/28). There was no statistically significant difference between the groups (p > 0.05).
Discussion
This study focused on the role of GT used for treating febrile neutropenia in the pre-engraftment period on engraftment outcomes in allo-HSCT patients. GT led to significantly earlier neutrophil and PLT engraftment (p = 0.001 and p = 0.007). Post-GT median ANC and PLT counts from days 1 to 4 were ⩾0.5 × 10³/μL and ⩾20,000/mm³, respectively. On day 15, post-allo-HSCT, ANC was significantly higher in the GT group than in the non-GT group (p < 0.001). This is the first statistically significant controlled study to evaluate GT’s effect on engraftment in allo-HSCT patients.
Administering G-CSF/dexamethasone for neutrophil collection increases circulating neutrophils and significantly alters gene expression, increasing mRNA expression of several toll-like receptors (TLRs) compared to normal neutrophils. 23 Increased TLR expression activates innate immunity and promotes myeloid lineage progression in primitive hematopoietic cells by increasing cytokine secretion upon cell stimulation.23–26 TLRs, especially TLR2 and TLR4, are expressed in neutrophils, stem cells, megakaryocytes, and early hematopoietic progenitors. TLR expression is crucial for thrombopoiesis and supports PLT formation and function via TLR2 and TLR4. 27 At the same time, TLR activation produces IL-6 and tumor necrosis factor-α. These two cytokines have a certain effect on PLT production. 28 Based on this information, In our study, we think that the reason why granulocyte transfusions shorten the engraftment period is that the increase in gene expression from mobilized granulocytes stimulates TLRs on immature stem cells, promoting myeloid lineage proliferation and shortening PLT and neutrophil times.
The TLC, ANC, and PLT counts were statistically lower in GTG patients on allo-HSCT day 0 (p-values: p = 0.024, p = 0.19, p = 0.004, respectively) This was attributed to the more frequent use of myeloablative conditioning regimens in the GT group, despite there being no statistical difference between the groups in terms of myeloablative conditioning regimen use. There is an increased risk of profound cytopenias in myeloablative allogeneic hematopoietic cell transplantation for hematologic malignancies.22,29
Our findings show significant benefits of GT in allo-HSCT patients. In our study, 89.3% (25 of 28) of patients who received GT showed improvement in their infection status. Ikegawa et al. 13 reported a 60% improvement in infection after GT in 20 neutropenic patients with peritransplant infections. Zhou et al. 10 found a cure rate of 72.3% in neutropenic patients with hematological malignancies and refractory infections. The granulocyte dose used in these studies was 4.8 × 108/kg and 2.55 × 108/kg, respectively, lower than in our study. Shrivastava et al. 30 reported a 78% improvement rate in infection status in a study involving cancer patients, mostly with hematological conditions, using a median granulocyte dose of 2.9 × 1010 cells. In our study, patients received a high granulocyte dose, with a median of 3.8 × 1010 per transfusion (5.44 × 108/kg), which may explain a high rate of improvement (89.3%) in infection status and highlight the importance of optimal granulocyte collection.13,15,31–33
Although there is no established evidence identifying the optimal granulocyte dose for predicting efficacy in treating underlying infections, several transfusion guidelines recommend that granulocyte products should contain at least 1.5–3 × 108/kg. Average doses vary widely among studies, ranging from 2.0 to 15.5 × 108 cells/kg, reflecting differences in recipient types (adults vs children), donor types, and mobilization protocols. Several previous studies have suggested improved infection control in patients receiving more than 10 × 109 granulocytes.34,35 In the RING trial, 36 a multicenter, prospective, randomized controlled clinical study, patients receiving the targeted dose (⩾6 × 108/kg) showed better treatment outcomes than those in the low-dose group. Another study observed that in neutropenic patients with hematological diseases, patients receiving higher doses of granulocytes (>2.55 × 108/kg) may have improved infection control than patients receiving lower doses of granulocytes (⩽2.55 × 108/kg). 10 The granulocyte dose in our study (median dose, 5.44 × 108/kg) was similar to the high granulocyte dose level in the RING study.
The timing of GTs initiation has been shown to play a crucial role in determining their efficacy. Another reason for the high rate of improvement in our infection outcomes may be the prompt administration of GT treatment, with a median application time of just 3 days. In Sachs et al.’s study, 8 where a very high response rate of 92.6% was observed, GTs were administered early after a median infection period of 6 days. Neutrophils produce various cytokines by upregulating TLRs in response to G-CSF and dexamethasone. IL-8, a key cytokine, helps control infections. Mobilized granulocytes also have an extended lifespan, so their increased numbers and longevity aid in overcoming infections.24,26
In our study, the bacterial infection rate was statistically significantly higher in the GTG compared to the non-GTG (p = 0.04). Generally, a higher response rate to GT has been reported in bacterial infections than in fungal infections.37–41 The high infection improvement rate in our study may be due to a higher rate of bacterial infections. Additionally, CRP levels significantly increased in the GTG compared to the non-GTG. Serum CRP levels rise within 24 h of infection and can predict fever and severe sepsis in neutropenic patients. 6 Most GT recipients in this study were on broad-spectrum antimicrobial therapies, with significantly more therapies used in the GTG than in the non-GTG (p = 0.003). Despite the high-risk population, 89.3% of infections resolved after GT, suggesting GT is valuable for stabilizing ANC and eliminating infection. In their study, Zhou et al. 10 reported 66% and 57.5% OS rates at 30 and 120 days in patients who received GT. Garg et al. 9 found that the 30-day OS was 67.7%. Additionally, Garg et al. 9 reported that administering GT within 7 days of neutropenic sepsis led to significantly higher 30-day OS (p = 0.01). Uppuluri et al. 42 observed that administration of granulocytes even earlier, within 48 h of a septic episode resulted in a significant improvement in OS, increasing from 41% to 54%. In our study, GT was performed after a median of 3 days of fever, and the 30- and 100-day OS rates were 96.4% and 88.4%, respectively. Our high OS rate in the first 3 months after allo-HSCT may be due to the early initiation of GTs. Further studies needed on the timing of GT administration.
Nikolajeva et al. 15 reported that in 28 allo-HSCT patients who received GT, only two deaths were due to progression of infection. Similarly, in our study, one patient died due to progression of infection, and the fact that only one GT could be administered to this patient was considered to have facilitated progression to death. Studies have shown that tandem GTs in neutropenic patients are effective. Seidel et al. 41 reported that repeated transfusions of granulocytes over 5 days, with a minimum of 3 × 108/kg neutrophils per concentrate, stabilized ANC, reduced neutropenic duration, and increased infection control. The number of successive GT administrations may be crucial for efficacy. In our study, continued GT administration could have stabilized ANC and potentially prevented the infection-related death.
When comparing the OS rate between the GTG and non-GTG in our study, no statistically significant difference was detected. Moreover, our study found no statistically significant difference in 30-day, 100-day, 1-year, and 2-year OS between patients receiving <0.5 × 109/kg and ⩾0.5 × 109/kg GT. Similarly, the RING study 36 observed no difference in OS between patients receiving GT and those in the control group. Ikegewa et al. 13 found no statistical difference in 1-year OS when comparing patients who received high-dose granulocytes (⩾0.5 × 109/kg) and low-dose granulocytes (<0.5 × 109/kg) for infection treatment. In addition, Garg et al. 9 found no statistical difference in 30-day survival between groups of patients treated with ⩾5 × 109 granulocytes per product and <5 × 109 granulocytes per product.
In contrast to the abovementioned studies, including ours, the RING study 36 demonstrated that patients treated with higher numbers of granulocytes per kg body weight (defined as >0.06 × 1010 cells/kg) had significantly better survival at 42 days compared to patients with less than 0.06 × 1010 transfused cells/kg. The reason for these different results across studies is that factors such as granulocyte dose, timing of GT initiation, and number of patients affect OS rates. Prospective studies with larger cohorts are needed to evaluate GT’s impact on OS thoroughly. Our study is limited by its single-center and retrospective design. In our study, none of the patients who received GT experienced acute GVHD. This is presumably because GTs maintained mucosal integrity, reducing bacterial translocation across the intestinal mucosa, which in turn lowered serum lipopolysaccharide and Tumor Necrosis Factor (TNF) production both integral in triggering acute GVHD. 43 Nikolajeva et al. 15 found a similarly low rate of grades 3–4 acute GVHD overall incidence (7%).
Regarding chronic GVHD, Zhou et al. 10 reported a 50% incidence of limited chronic GVHD in 10 allo-HSCT patients who underwent GT. In our study, although patients who received GT had a lower rate of chronic GVHD than those who did not (35.7% vs 46.4%), the difference was not statistically significant (p = 0.41). The rate of chronic GVHD in both GTG and non-GTG was similar to the rates reported in the literature for chronic GVHD developing after allo-HSCT. 44
Zhou et al. 10 reported that CMV infection did not develop in 10 patients who underwent allo-HSCT. In our study, there was no statistical difference between the GTG and non-GTG in terms of CMV infection development (p > 0.05). 8 Our primary data indicated that GT did not affect the incidence of GVHD or CMV infection.
Conclusion
Our study indicates the need for adequately powered prospective studies to confirm the significant impact of GT on engraftment in pre-engraftment febrile neutropenia among allo-HSCT recipients. GT may act as a bridge therapy, allowing time for marrow recovery and engraftment. Using GT therapy in profound neutropenia and sepsis in allo-HSCT recipients appears to be a safe and effective infection control approach. Further randomized studies are needed to confirm GT’s effectiveness, complications, and impact on engraftment.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251326765 – Supplemental material for Effective engraftment via granulocyte transfusion in pre-engraftment febrile neutropenia following allogeneic hematopoietic stem cell transplantation: granulocyte transfusion as bridge therapy
Supplemental material, sj-docx-1-tah-10.1177_20406207251326765 for Effective engraftment via granulocyte transfusion in pre-engraftment febrile neutropenia following allogeneic hematopoietic stem cell transplantation: granulocyte transfusion as bridge therapy by Neslihan Mandaci Şanli and Ali Ünal in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
