Abstract
Background:
Relapse after allogeneic hematopoietic stem cell transplantation (HSCT) remains a major clinical challenge in higher-risk myelodysplastic neoplasms (MDS). Although hypomethylating agents have been evaluated as posttransplant maintenance therapy, their efficacy remains uncertain.
Objective:
We aimed to evaluate the long-term clinical outcomes of individualized decitabine maintenance therapy following HSCT in patients with MDS or secondary acute myeloid leukemia from MDS.
Design:
This was a retrospective comparative study based on a long-term follow-up of the phase I PODAC trial, which individualized posttransplant decitabine maintenance dosing.
Methods:
Nineteen PODAC participants were included in the treatment group and compared with 52 matched control patients selected through target trial emulation using identical eligibility criteria.
Results:
For the 19 PODAC participants, a median of 6 cycles was administered, starting at 5 mg/m2/day and escalating to a median maintenance dose of 7 mg/m2/day. After a median follow-up of 11.4 years in the PODAC group and 9.7 years in the control group (p = 0.90), no significant differences were observed between the two groups in overall survival, relapse-free survival, graft-versus-host disease (GVHD)-free relapse-free survival, or the incidence of relapse, nonrelapse mortality, and grades II–IV acute GVHD. However, the incidence of moderate-to-severe chronic GVHD was significantly lower in the PODAC group and decitabine maintenance emerged as an independent predictor for longer chronic GVHD-free relapse-free survival (cCRFS).
Conclusion:
Decitabine maintenance using an adaptive dosing approach enabled prolonged treatment and was associated with a lower incidence of chronic GVHD and improved cGRFS. However, it did not improve survival or relapse outcomes compared to the control group.
Plain language summary
Myelodysplastic neoplasms (MDS) are disorders in which the bone marrow does not make enough healthy blood cells, leading to problems such as anemia, infections, or bleeding, and in some patients the disease can progress to leukemia. A hematopoietic stem cell transplant from a donor is the only treatment that can potentially cure the disease. Even after transplant, however, many patients face serious complications or experience disease relapse. Doctors have been studying whether a drug called decitabine, which belongs to a group of medicines known as hypomethylating agents, can be given after transplant to reduce the risk of relapse. In this study, 19 patients with MDS or leukemia that developed from MDS received decitabine after transplant, with doses adjusted for each patient. Their outcomes were compared with those of 52 similar patients who did not receive the drug. Patients were followed for more than 10 years. The study showed that decitabine did not reduce the risk of disease relapse and did not improve survival. However, patients who received decitabine were less likely to develop a long-term complication called chronic graft-versus-host disease, in which donor immune cells attack the patient’s body. As a result, these patients spent more time free from serious immune-related complications. In summary, decitabine did not reduce the risk of disease relapse or improve survival; however, it was associated with a lower incidence of long-term immune complications after transplant.
Keywords
Introduction
Myelodysplastic neoplasms (MDS) represent a heterogeneous group of clonal hematopoietic stem cell disorders, with a spectrum that varies from lower-risk to higher-risk disease based on expected survival and the probability of progression to acute myeloid leukemia. 1 Still, the treatment and management of higher-risk MDS remain challenging, and proceeding to allogeneic hematopoietic stem cell transplantation (HSCT) is encouraged for medically fit patients as an only way to potentially achieve a cure.2,3
Over recent decades, there has been steady improvement in HSCT-related mortality with the introduction of reduced-intensity conditioning, novel anti-infective agents, and graft-versus-host disease (GVHD) therapeutics.4–10 However, there has been little progress in reducing the risk of post-HSCT relapse, which remains the major cause of transplant failure in MDS11,12; approximately 30%–40% of patients experience relapse, resulting in dismal outcomes for HSCT.13,14
The use of hypomethylating agents (HMAs), azacitidine (AZA), and decitabine (DEC), is primarily considered for patients with higher-risk MDS, either as standalone therapy or in conjunction with HSCT, to improve progression-free or overall survival (OS).15–18 In lower-risk MDS, it is used with the aim of improving cytopenia. 19 In addition to these conventional indications, HMAs have been investigated as a preemptive20,21 or maintenance22,23 strategies following HSCT to prevent overt relapse. The rationale for using HMAs in this setting includes their potential to enhance the graft-versus-leukemia effect via upregulation of tumor antigens and re-expression of silenced tumor suppressor genes, low toxicity, and the lack of targetable mutations in MDS.24,25 However, the efficacy of HMAs for this purpose, particularly as maintenance therapy, remains a subject of ongoing debate.26–28 This uncertainty may be attributed, apart from the intrinsic efficacy of HMAs, to heterogeneity in patient populations across trials, variations in treatment duration, and potential differences between AZA and DEC. Notably, our PODAC trial 29 —designed more than a decade ago—focused on patients with higher-risk MDS and secondary acute myeloid leukemia from MDS (sAML) employed DEC for post-HSCT maintenance, a regimen that has been relatively less studied than AZA. The trial adopted a pharmacokinetic/pharmacodynamic (PK/PD)-guided approach to establish a safe and individualized dosing schedule, a factor closely associated with treatment duration.
In light of the continued debate regarding the utility of HMA maintenance therapy following HSCT, we conducted a study to evaluate long-term transplant outcomes associated with DEC maintenance by comparing them with those of a control group. Although the number of patients included in the PODAC trial was limited, the extended follow-up period provided an opportunity to explore the impact of DEC maintenance beyond its original dose-finding objective.
Methods
Selection of patient
To evaluate the long-term impact of individualized DEC maintenance following HSCT, we analyzed a cohort of 19 patients enrolled in the PODAC trial along with 52 matched control patients. The PODAC study 29 aimed to refine the DEC dosing regimen through a phase I trial that incorporated adaptive dose individualization based on PK-PD modeling. In the original PODAC trial, patients who met the following inclusion criteria—age ⩽65 years; diagnosis of higher-risk MDS according to the International Prognostic Scoring System (IPSS; intermediate-2 or high risk) 30 or sAML in remission after HSCT; and absence of early relapse or severe acute GVHD (grade III/IV; Table S1)—received intravenous DEC at 5 mg/m2/day for five consecutive days every 4 weeks, beginning between days 42 and 90 post-HSCT. For cycles 2–4, doses were adaptively individualized using PK/PD simulations to determine the highest tolerated dose that avoided grade IV neutropenia (ANC <500/mm3) or thrombocytopenia (platelet <25,000/mm3). The individualized maintenance dose established at cycle 4 was continued for up to 12 cycles.
To establish a comparative analysis, we employed target-trial emulation methodology to select an external control cohort from St. Mary’s Hospital registry data. Specifically, control patients were identified among individuals who would have met the eligibility criteria for the PODAC trial but did not receive posttransplant DEC maintenance; the same inclusion and exclusion criteria as the original PODAC trial were applied to the control group (Table S1 and Figure S1). For both the PODAC and control cohorts, follow-up was aligned at a common post-HSCT time zero corresponding to the earliest time point at which DEC maintenance could have been initiated, in order to minimize immortal time bias.
For the original PODAC trial (ClinicalTrials.gov, identifier NCT01277484), the study protocol was approved by the independent Institutional Review Board (IRB) of Seoul St. Mary’s Hospital prior to the initiation of any study-related procedures, and written informed consent was obtained from all participants (KC10MISF0505). The retrospective comparative study was also approved by the IRB of Seoul St. Mary’s Hospital (KC25RISI0107). Both studies were designed and conducted in accordance with the principles of the Declaration of Helsinki and the good clinical practice guidelines of Korea. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement 31 (Supplemental File).
Outcome assessment
OS was defined as the duration from HSCT to death from any cause or to the date of the last follow-up. Relapse-free survival (RFS) was considered as the time from HSCT to the occurrence of relapse or death whichever came first. Nonrelapse mortality (NRM) was specified as death without evidence of disease relapse. GVHD-free relapse-free survival (GRFS) 32 was defined as the time from HSCT to the first occurrence of grades III–IV acute GVHD, 33 moderate-to-severe chronic GVHD, 34 relapse, or death from any cause. In contrast, chronic GVHD-free relapse-free survival (cGRFS) 35 was defined by excluding acute GVHD events and was measured as the time from HSCT to the first occurrence of moderate-to-severe chronic GVHD, relapse, or death.
Statistical methods
Descriptive statistics were employed to summarize baseline characteristics, treatment modalities, and clinical outcomes. Continuous variables were reported as mean ± standard deviation or median with interquartile range, based on the distribution. Categorical variables were presented as frequencies and percentages. Normality of distributions was assessed using the Shapiro–Wilk test, with the independent samples t test or the Mann–Whitney U test applied for continuous variables as appropriate. Categorical data were analyzed using the chi-square test or Fisher’s exact test, as indicated by expected frequencies. For the multivariate analyses, the assessment of multicollinearity among variables was conducted using the variance inflation factor, with a threshold of 10.
Estimations of OS, RFS, and GRFS were conducted using the Kaplan–Meier method, with comparisons across different groups made through the log-rank test. The evaluation of associations between various factors and OS, RFS, and GRFS, along with the estimation of adjusted probabilities, was performed using the Cox proportional hazards regression model. Statistical analyses for nonrelapse mortality, GVHD incidence, and relapse incidence were conducted using subdistribution hazard models to account for death as competing events. Factors that exhibited a p value of less than 0.2 in univariate analyses were considered for inclusion in the multivariate models. However, whether the subject was in the PODAC trial was included in all multivariate models regardless of its p value.
All statistical evaluations were executed utilizing R version 4.1.3 (http://cran.r-project.org/). Detailed statistical settings are presented in Table S1.
Result
Patient characteristics
Baseline characteristics of the patients in the PODAC trial and the control group are detailed in Table 1. The median age at transplantation was similar between the PODAC group and matched controls (49 years vs 50 years), with no significant differences in sex distribution, Revised International Prognostic Scoring System (IPSS-R) risk, bone marrow blast percentage, cytogenetics, hematopoietic cell transplantation-specific comorbidity index (HCT-CI) score, or worst diagnosis before HSCT. Donor source, HLA matching status, and donor age were also comparable between groups. Sex mismatch between donor and recipient occurred in 18% of the overall cohort, with female-to-male mismatches observed in 11% of the PODAC group and 21% of controls (p = 0.49). However, a significant difference was noted in refractoriness to bridging therapy (p = 0.034), with a higher proportion of refractory patients in the control group (50% vs 37%) and no patients in the PODAC group undergoing upfront transplantation, compared to 17% in the control group.
Baseline characteristics of patients in PODAC trial and matched controls.

Continuous variables are presented as median (interquartile range or range, as indicated), and categorical variables as number (%). Comparisons between groups were performed using the Mann–Whitney U test for continuous variables and the chi-square test or Fisher’s exact test for categorical variables, as appropriate. p Values <0.05 were considered statistically significant.
Median (IQR) or frequency (%).
p < 0.05.
CI, confidence interval; HCT-CI, hematopoietic cell transplantation–comorbidity index; HLA, human leukocyte antigen; HMT, hypomethylating therapy; HSCT, hematopoietic stem cell transplantation; ICTX, intensive chemotherapy; IPSS-R, Revised International Prognostic Scoring System; MDS-EB-2, myelodysplastic syndrome with excess blasts-2; sAML, secondary acute myeloid leukemia evolving from MDS.
Adverse events in PODAC trial
The summary of the PODAC trial, as outlined in Table 2, shows that the first dose of maintenance therapy was initiated at a median of 12.1 weeks posttransplant, with subsequent cycles administered at a median interval of 35 days. Of the 19 participants, 10 (52.6%) completed 6 or more cycles and 7 (36.8%) completed the total planned cycles (12 cycles), with a median of 6 cycles per patient. The median dosage per cycle began at 5 mg/m2 and stabilized at approximately 7 mg/m2 from the 4th through the 12th cycle. Severe hematological toxicities post-baseline, grade IV or higher, occurred in proportions of 16% for leukopenia, 63% for neutropenia, and 26% for thrombocytopenia, with no cases of severe anemia reported.
Summary of long-term adverse event among patients enrolled in the PODAC trial.

This table summarizes treatment characteristics and adverse events observed only in patients who received posttransplant DEC maintenance as part of the PODAC trial (n = 19). Adverse events were assessed during the DEC maintenance period and graded according to the CTCAE. Hematologic and nonhematologic toxicities are reported as number (%).
CTCAE, Common Terminology Criteria for Adverse Events; DEC, decitabine.
Survival and relapse outcome
Overall survival
The median follow-up period for survivors was 11.4 years in the PODAC group and 9.7 years in the control group (p = 0.90). The median OS for both the PODAC group and the matched control was not reached during the observation period. The 2-year OS rate was 68% in the PODAC group compared with 75% in the control, and the 8-year OS rates were 53% versus 62%, respectively, with a HR of 1.38 and p value of 0.43, suggesting no statistically significant differnce (Figure 1(a)). Univariate analysis identified factors such as male sex, MDS/AML, adverse cytogenetics, HCT-CI high-risk group, advanced donor age, and related donor, as associated with poor OS (Table S2). Multivariate analysis, detailed in Table 3, revealed advanced age at transplant (hazard ratio (HR) = 1.06, p = 0.02), related donor (HR = 0.1, p < 0.01), and HCT-CI high risk (HR = 5.37, p < 0.01) as independent predictors of shorter OS. DEC maintenance did not emerge as a significant factor in either analysis.

Kaplan–Meier curves for (a) Overall survival and (b) Relapse-free survival. Cumulative incidence curves for (c) Relapse and (d) Nonrelapse mortality.
Multivariate analysis on survival outcome.
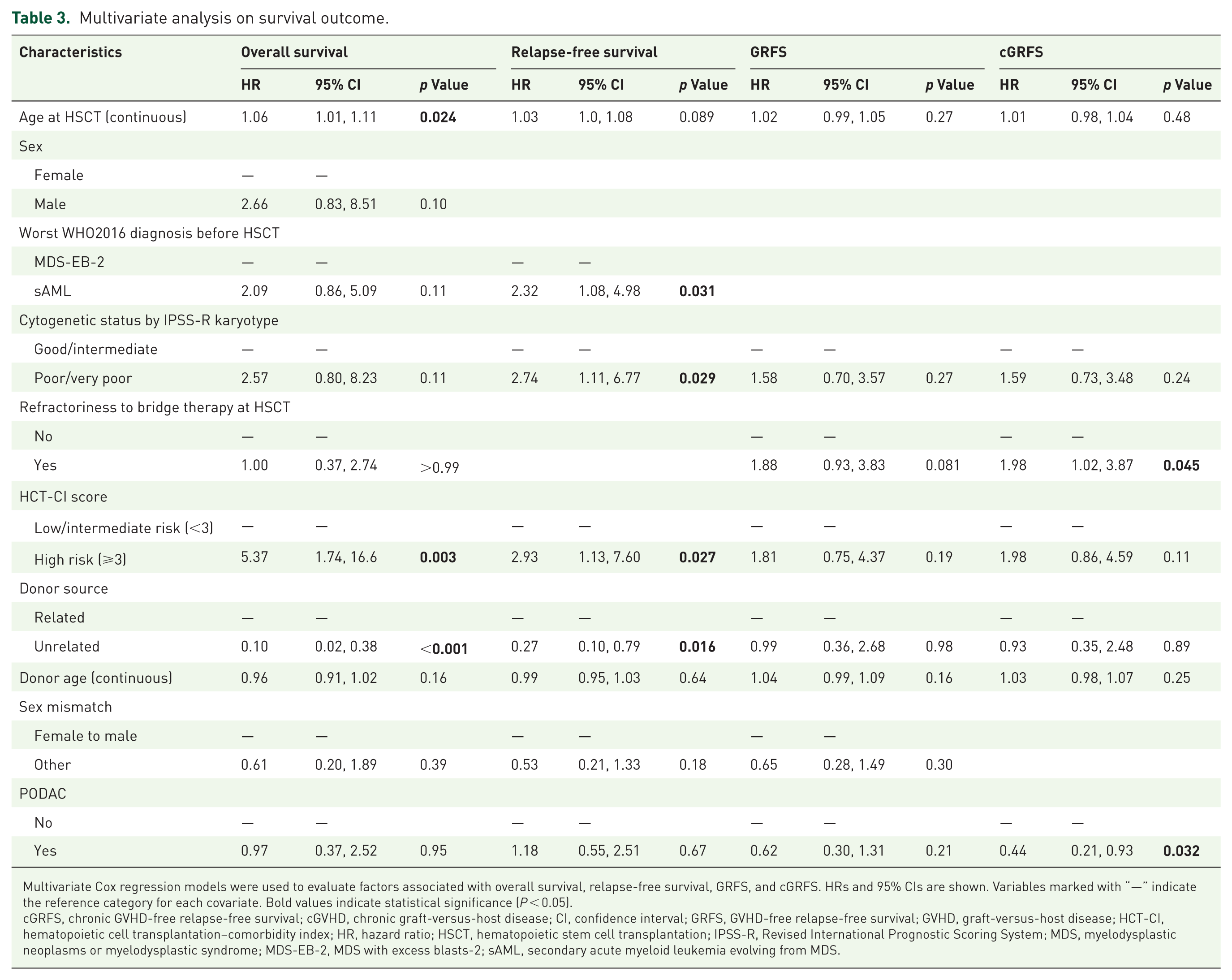
Multivariate Cox regression models were used to evaluate factors associated with overall survival, relapse-free survival, GRFS, and cGRFS. HRs and 95% CIs are shown. Variables marked with “—” indicate the reference category for each covariate. Bold values indicate statistical significance (P < 0.05).
cGRFS, chronic GVHD-free relapse-free survival; cGVHD, chronic graft-versus-host disease; CI, confidence interval; GRFS, GVHD-free relapse-free survival; GVHD, graft-versus-host disease; HCT-CI, hematopoietic cell transplantation–comorbidity index; HR, hazard ratio; HSCT, hematopoietic stem cell transplantation; IPSS-R, Revised International Prognostic Scoring System; MDS, myelodysplastic neoplasms or myelodysplastic syndrome; MDS-EB-2, MDS with excess blasts-2; sAML, secondary acute myeloid leukemia evolving from MDS.
Relapse-free survival
The median RFS of the PODAC group was 41.9 (95% CI: 27.0–NA) months and that of the matched control was not reached during the observation period. The 2-year RFS rate was 68% in the PODAC group compared with 69% in the control (p = 1.00), and the 8-year RFS rates were 42% versus 54% (p = 0.40), respectively, with a HR of 1.34 and p value of 0.42, suggesting no statistically significant difference (Figure 1(b)). Univariate analysis identified factors such as sAML before transplant, adverse cytogenetics, HCT-CI higher risk, advanced donor age, and related donor were associated with inferior RFS (Table S2). Multivariate analysis, detailed in Table 3, showed sAML before transplant (HR = 2.32, p = 0.03), adverse cytogenetics (HR = 2.74, p = 0.03), HCT-CI higher risk (HR = 2.93, p = 0.03), and related donor (HR = 0.27, p = 0.02) as independent predictors of inferior RFS. PODAC was not shown to be a significant factor in either analysis.
Relapse
The 2-year relapse rate was 16% in the PODAC group and 25% in the control group. At 8 years, the relapse rates were 42% for PODAC and 37% for the control, with a HR of 1.14, indicating no significant difference (p = 0.73; Figure 1(c)). In univariate analysis, advanced donor age, related donor, and sAML before transplant were associated with relapse (Table S3). Multivariate analysis, detailed in Table 4, identified sAML before transplant (HR = 2.2, p = 0.041), adverse cytogenetics (HR = 3.58, p = 0.002), and advanced donor age (HR = 1.05, p = 0.01) as independent predictors of relapse incidence. PODAC did not emerge as a significant factor in influencing relapse in either univariate or multivariate analysis.
Multivariate analysis on relapse, NRM, and GVHD incidence.
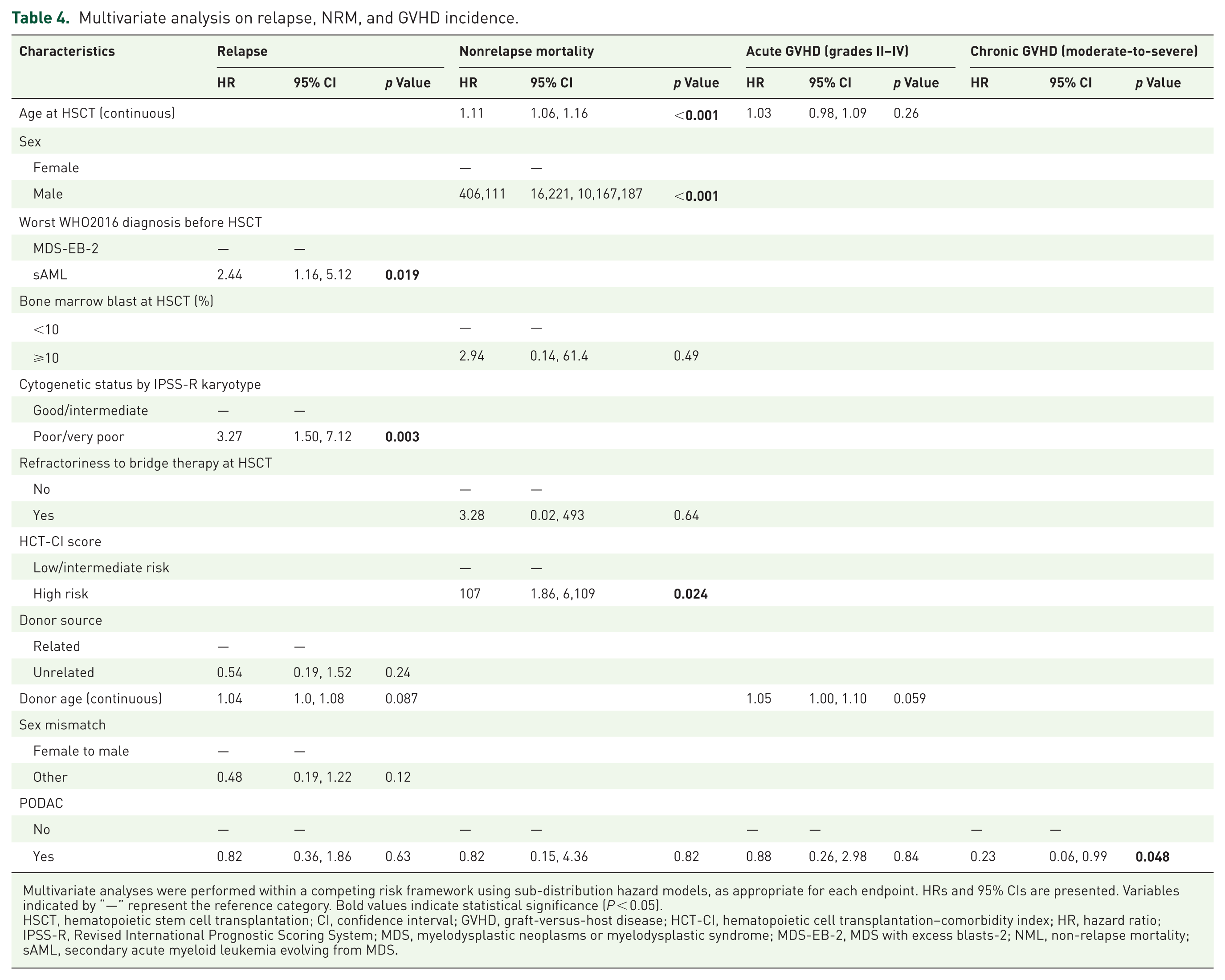
Multivariate analyses were performed within a competing risk framework using sub-distribution hazard models, as appropriate for each endpoint. HRs and 95% CIs are presented. Variables indicated by “—” represent the reference category. Bold values indicate statistical significance (P < 0.05).
HSCT, hematopoietic stem cell transplantation; CI, confidence interval; GVHD, graft-versus-host disease; HCT-CI, hematopoietic cell transplantation–comorbidity index; HR, hazard ratio; IPSS-R, Revised International Prognostic Scoring System; MDS, myelodysplastic neoplasms or myelodysplastic syndrome; MDS-EB-2, MDS with excess blasts-2; NML, non-relapse mortality; sAML, secondary acute myeloid leukemia evolving from MDS.
Non-relapse mortality
The 1-year NRM rate was 5.3% in the PODAC group compared with 6.7% in the control, and the 8-year NRM rates were 16% versus 12%, respectively (HR = 1.39, p = 0.63; Figure 1(d)). For regression analysis, because no event occurred in patients with IPSS-R lower risk at transplant (Hazard = 0), IPSS-R at transplant was excluded from the analysis and was considered significantly associated with nonrelapse mortality. In univariate analysis, bone marrow blasts ⩾10% at transplant and high HCT-CI score at transplant were associated with increased NRM incidence (Table S3). Multivariate analysis results are shown in Table 4; HCT-CI score was identified as an independent factor associated with the incidence of NRM (HR = 13.7, p = 0.001). PODAC was not shown to be a significant factor in either analysis.
GVHD and GRFS
Acute GVHD
The 6-month acute GVHD rate was 11% in the PODAC group compared with 42% in the control (HR = 0.98, p = 0.98; Figure 2(a)). In univariate analysis, only donor age was associated with acute GVHD (Table S3). In multivariate analysis, shown in Table 4, no risk factor was significantly associated with the incidence of acute GVHD. PODAC was associated with lower acute GVHD with HR of 0.88 but not shown as a significant factor (p = 0.84)

Cumulative incidence curves for (a) Acute GVHD and (b) Chronic GVHD. Kaplan-Meir curves for (c) GRFS and (d) cGRFS.
Chronic GVHD
The 4-year chronic GVHD rate was 16% in the PODAC group compared with 47% in the control. (Figure 2(b)). The incidence of chronic GVHD was significantly lower in the PODAC group (HR = 0.23, p = 0.048), and among other relevant risk factors, none showed a significant association with chronic GVHD (Table 4).
GVHD-free relapse-free survival
The median GRFS of the PODAC group was 31.8 (95% CI: 16.1–NA) months and that of the matched control was 11.2 (95% CI: 5.0–55.3) months (p = 0.3). The 2-year GRFS rate was 63% in the PODAC group compared with 40% in the control (p = 0.3), and the 8-year GRFS rates were 37% versus 33% (p = 0.9), respectively, with a HR of 0.73 and p value of 0.35 (Figure 2(c)). Univariate analysis showed that older age, refractoriness to bridging therapy, related donor, advanced donor age, and female-to-male transplant were associated with inferior GRFS (Table S2). Multivariate analysis results are shown in Table 3. None of factors, including PODAC, were significantly associated with GRFS among the selected risk factors.
Chronic GVHD-free relapse-free survival
The median chronic GFRS of PODAC group was 41.9 (95% CI: 27.0–NA) months and that of the matched control was 11.2 (95% CI: 5.6–55.3) months. The 2-year chronic GRFS rate was 63% in the PODAC group compared with 35% in the control, and the 8-year GRFS rates were 37% versus 28%, respectively, with a HR of 0.63 and p value of 0.18 (Figure 2(d)). Univariate analysis showed that a high HCT-CI score and advanced donor age were associated with inferior cGRFS (Table S2). Multivariate analysis results are shown Table 3. Refractoriness to bridging therapy (HR = 1.98, p = 0.045) was associated with inferior cGRFS, and PODAC was associated with superior cGRFS (HR = 0.44, p = 0.03) as an independent factor. Thus, after adjusting for the potential risk factors, PODAC was shown to be a significant independent factor for the superior cGRFS.
Discussion
In this study, we evaluated the effect of prophylactic DEC following HSCT in patients with higher-risk MDS or sAML from MDS by comparing outcomes with those of a matched control group. In the DEC group, maintenance therapy was administered using an individualized dosing schedule. While there was no significant difference in OS, RFS, relapse, or NRM compared to matched controls, posttransplant DEC was associated with a significantly lower incidence of cGVHD. Notably, this translated into a marked improvement in cGRFS, highlighting a potential role for HMAs in modulating chronic GVHD and improving long-term quality of survival.
Starting with AZA, early attempts to evaluate post-HSCT HMA therapy focused on its potential for relapse prevention. In a preliminary report by Jabbour et al., low-dose AZA was administered as salvage or maintenance therapy in a small AML cohort (n = 17), suggesting potential for durable remissions in posttransplant relapse. In a phase I/II trial of AZA maintenance, de Lima et al. identified 32 mg/m2 IV for 5 days as the appropriate dose in the setting of reduced-intensity HSCT for high-risk AML and MDS patients (n = 45). 36 With 1-year event-free and OS rates of 58% and 77% after 20.5 months of follow-up, their study suggested that maintenance therapy may offer clinical benefits. Based on these findings, a randomized phase III trial was conducted to compare AZA maintenance at the same dose, for up to 12 cycles, with observation in patients with high-risk AML (n = 134) and MDS (n = 47). 22 The median time to initiation of AZA was 62 days after HSCT, and patients received a median of only four cycles. Despite the rationale and previous dose-finding data, AZA maintenance did not lead to significant improvement in relapse-free or OS. Moreover, the incidence of acute and chronic GVHD was comparable between the AZA and observation arms.
DEC has also been investigated as a post-HSCT maintenance strategy. In a phase I study by Pusic et al., 37 four dose levels (5, 7.5, 10, and 15 mg/m2/day) were tested, and a dose of 10 mg/m2 for 5 days every 6 weeks was suggested as optimal. DEC was initiated at a median of 95 days post-HSCT, with up to eight cycles planned; a median of five cycles was completed. After 26.7 months of follow-up, none of the MDS patients (n = 5) relapsed, while 3 of 14 AML patients in Complete remission (CR) and all 3 patients with persistent disease relapsed. More recently, a Chinese phase II randomized trial evaluated the prophylactic use of granulocyte colony-stimulating factor(G-CSF) combined with low-dose DEC (G-Dec) in high-risk AML patients (n = 202) who achieved MRD-negative CR post-HSCT. 38 With six planned cycles, 96% of patients in the intervention arm completed all cycles. After a median follow-up of 28.0 months, the G-Dec group showed a significantly lower 2-year relapse rate (15.0% vs 38.3%, p < 0.01), along with increased NK and CD8+ T-cells. Although regulatory T-cells also increased, there was no difference in chronic GVHD incidence.
Collectively, numerous studies have explored the feasibility and safety of HMAs as post-HSCT maintenance therapy; however, conclusive evidence regarding their efficacy in preventing relapse remains lacking. Across studies, it has been consistently noted that significantly reduced doses—compared to conventional regimens—are more tolerable and improve the likelihood of patients completing planned treatment cycles. Despite promising results from single-arm studies, AZA maintenance failed to demonstrate survival benefit in a randomized trial. 22 While DEC combined with recombinant human granulocyte colony-stimulating factor (rhG-CSF) showed a favorable impact on reducing post-HSCT relapse, only MRD-negative CR patients post-HSCT were included in this study. 38 Given that the incidence of relapse varied significantly among MDS, AML in CR, and AML with persistent disease in the study by Pusic et al., 37 HMAs alone may be insufficient to overcome the inherent aggressiveness of the disease, and the use of low-dose HMA in such settings may further limit their ability to prevent relapse. While studies such as RELAZA 20 and RELAZA2 21 demonstrated that preemptive HMA monotherapy can delay hematologic relapse in patients with molecular residual disease, relapse eventually occurred in a substantial proportion of patients. Moreover, real-world experience with preemptive or salvage treatment using HMAs—with or without donor lymphocyte infusion (DLI)—consistently indicates that HMA monotherapy provides only limited benefit, whereas immunotherapeutic approaches such as DLI or second allo-HSCT yield superior outcomes and should be considered when feasible.13,39 These findings suggest that combining maintenance HMA with immunotherapeutic strategies may enhance efficacy in MDS, although prospective trials comparing HMA alone, HMA plus DLI, or DLI alone are needed to clarify the contribution of each component. 40
Although DEC maintenance did not reduce post-HSCT relapse in our study, the PODAC group showed a lower incidence of chronic GVHD and significantly longer cGRFS. Due to the timing of drug administration and the exclusion of patients with acute GVHD at enrollment, the effect of HMA on acute GVHD could not be adequately assessed. Previous preclinical and clinical studies have suggested that HMAs increase the number of regulatory T-cells,41–43 potentially reducing the incidence of GVHD. This mechanism may explain the lower incidence of cGVHD observed in our study. However, the effects of HMAs on regulatory T-cells have not been consistently demonstrated. In our tumor-bearing murine transplant model, we did not observe any increase in regulatory T-cells following DEC treatment. 44 Moreover, in a randomized trial of AZA maintenance, no significant differences in the incidence of acute or chronic GVHD were observed between the AZA and non-AZA groups. 22 In addition, the increase in regulatory T-cells did not translate into a reduced incidence of chronic GVHD in the G-Dec prophylaxis study. 38 These altogether suggest a possible discrepancy between laboratory findings and clinical outcomes, which are often confounded by multiple clinical variables. Therefore, the lower incidence of chronic GVHD observed in our study should be interpreted with caution.
Limitations
We acknowledge several limitations in our study. First, the comparative analysis was constrained by the small PODAC cohort (n = 19) and the retrospective design using external controls (n = 52) from our institutional registry, as the original single-arm trial was primarily designed to establish individualized post-HSCT DEC dosing through PK-PD modeling rather than to assess the efficacy of HMAs. This small-sample size and unequal group distribution limited statistical power and introduced potential small-sample bias. Second, although propensity score matching could theoretically improve covariate balance, we could not apply this method because the small-sample size resulted in substantial attrition and the inability to achieve a stable, balanced matching set, which would risk discarding valuable observations and reducing precision.45,46 Therefore, we employed multivariable time-dependent Cox regression with covariate adjustment, aligned a common post-HSCT time zero, and followed WHO real-world evidence principles 47 to emulate an intention-to-treat comparison while avoiding the unstable weights inherent to propensity-based methods in very small cohorts. Third, despite these adjustments, residual confounding from unmeasured variables (e.g., minimal residual disease status, detailed comorbidities, or variations in post-transplant supportive care) and limited positivity cannot be entirely excluded. Fourth, the single-center setting restricts generalizability across diverse populations and practice patterns. These factors collectively underscore that our findings should be interpreted as hypothesis-generating rather than confirmatory and warrant validation in larger, prospectively designed, multicenter randomized or pragmatic trials with balanced treatment allocation. In addition, although none of the patients demonstrated morphologic evidence of relapse at the time of DEC initiation or at the corresponding time point in the control cohort, the depth of response based on MRD assessment was not evaluated. Consequently, it remains uncertain whether DEC contributed to prolonging remission in patients who may have been in MRD-positive morphologic remission. Finally, although we observed a lower incidence of cGVHD and improved cGRFS, no patient samples were collected for correlative immune monitoring. Therefore, we were unable to evaluate whether DEC maintenance influenced posttransplant immune reconstitution, including regulatory T-cell dynamics, thereby precluding a mechanistic interpretation of these findings.
Conclusion
Although HMA maintenance is particularly appealing for high-risk MDS or AML as a strategy to reduce posttransplant relapse, results from previous studies and our analysis suggest that HMAs alone may not be sufficient to overcome the intrinsic aggressiveness of the disease. The observed reduction in chronic GVHD and the improvement in cGRFS with posttransplant DEC in this study suggest a potential immunomodulatory effect; however, these findings require confirmation in adequately powered prospective studies.
Supplemental Material
sj-docx-1-tah-10.1177_20406207261432568 – Supplemental material for Individualized decitabine dosing for post-HSCT maintenance in MDS and secondary AML from MDS: long-term outcomes from the PODAC trial and matched controls
Supplemental material, sj-docx-1-tah-10.1177_20406207261432568 for Individualized decitabine dosing for post-HSCT maintenance in MDS and secondary AML from MDS: long-term outcomes from the PODAC trial and matched controls by Silvia Park, Suein Choi, Seunghoon Han and Yoo-Jin Kim in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207261432568 – Supplemental material for Individualized decitabine dosing for post-HSCT maintenance in MDS and secondary AML from MDS: long-term outcomes from the PODAC trial and matched controls
Supplemental material, sj-docx-2-tah-10.1177_20406207261432568 for Individualized decitabine dosing for post-HSCT maintenance in MDS and secondary AML from MDS: long-term outcomes from the PODAC trial and matched controls by Silvia Park, Suein Choi, Seunghoon Han and Yoo-Jin Kim in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
