Abstract
Myelofibrosis is one of the Philadelphia chromosome (Ph)-negative myeloproliferative neoplasms with heterogeneous clinical course. Though many treatment options, including Janus kinase (JAK) inhibitors, have provided clinical benefits and improved survival, allogeneic hematopoietic stem-cell transplantation (AHSCT) remains the only potentially curative therapy. Considering the significant transplant-related morbidity and mortality, it is crucial to decide who to proceed to AHSCT, and when. In this review, we discuss recent updates in patient selection, prior splenectomy, conditioning regimen, donor type, molecular mutation, and other factors affecting AHSCT outcomes. Relapse is a major cause of treatment failure; we also describe recent data on minimal residual disease monitoring and management of relapse. In addition, emerging studies have reported pretransplant therapy with ruxolitinib for myelofibrosis showing favorable results, and further research is needed to explore its use in the post-transplant setting.
Introduction
Myelofibrosis (MF) is a Philadelphia chromosome (Ph)-negative myeloproliferative neoplasm, characterized by bone marrow fibrosis, leukoerythroblastosis in blood, extramedullary hematopoiesis, splenomegaly, constitutional symptoms, and an increased risk of transformation to acute myeloid leukemia (AML). The disease can arise de novo as primary MF (PMF) or evolve from polycythemia vera (post PV-MF), or essential thrombocythemia (post ET-MF). It primarily affects the elderly, with a median age at diagnosis of 67 years. 1 Though Janus kinase (JAK) inhibitors have broadened the treatment options in MF, allogeneic hematopoietic stem-cell transplantation (AHSCT) remains the only known curative therapy. In this review, we describe the available data and summary current recommendations on transplantation for myelofibrosis.
Prognosis and risk stratification
The clinical course of MF is heterogeneous, ranging from an indolent course persisting for decades to rapidly progressive disease with a survival of a few months. Prognosis is currently assessed by the International Prognostic Scoring System (IPSS) at diagnosis, which incorporates the following five risk factors: age (>65 years), anemia (hemoglobin <10 g/dl), leukocyte count (>25 × 109/l), circulating blasts (⩾1%), and constitutional symptoms. 2 The presence of 0, 1, 2, and ⩾3 factors are categorized as low, intermediate-1, intermediate-2, and high-risk disease, with median survival of 135, 95, 48, and 27 months, respectively. The Dynamic International Prognostic Scoring System (DIPSS) uses the same risk factors as the IPSS, but more weight is assigned to anemia, and it allows for prognostic prediction at any time during the disease course. 3 DIPSS was later refined as DIPSS-plus with inclusion of three additional risk factors: transfusion dependence, platelet count <100 × 109/l, and unfavorable karyotype. 4 For patients with post-PV/ET MF, the MF secondary to PV, and ET prognostic model (MYSEC-PM) has been defined and validated for risk stratification.5–7 This new score assigns 2 points to hemoglobin level <11 g/dl, circulating blasts ⩾3% and CALR-unmutated genotype, 1 point to platelet count <150 × 109/l and constitutional symptoms, and 0.15 points to any year of age. Patients are allocated into four risk categories with different survival: low (median survival not reached), intermediate-1 (9.3 years), intermediate-2 (4.4 years), and high (2 years) risk. Very recently, a large study confirmed that the MYSEC-PM allowed a more accurate prediction of survival after AHSCT than the DIPSS for post-PV/ET MF. 8
Molecular risk stratification
JAK2, CALR, and MPL are considered mutually exclusive driver mutations in MF. In general, CALR mutated patients are of younger age, with higher platelet counts and lower leukocyte counts, and have a favorable overall survival (OS).9,10 MPL does not seem to be prognostically relevant. 11 The prognosis impact of JAK2V617F in MF remains controversial. Some studies found JAK2 mutated patients had inferior survival 12 and a higher risk of leukemic transformation (LT) 13 ; however, others did not observe such an impact.14,15 Besides, about 10% of MF patients have JAK2/CALR/MPL triple-negative disease,16,17 and usually have the worst prognosis.9,10
In addition, several researchers found ASXL1, EZH2, SRSF2, or IDH1/2 mutations predicted poor outcomes.18–21 In a study of 879 patients with PMF, Vannucchi and colleagues showed that ASXL1, SRSF2, and EZH2 mutations were associated with shorter OS, and mutations in IDH1 and SRSF2 with LT. 21 Moreover, the detrimental effects of these mutations were additive.22,23 With incorporation of molecular mutations, the mutation-enhanced IPSS (MIPSS70) for transplant-age patients have been described, 24 which highlights the importance of mutational profiling on refining patient risk.
Patient selection and optimal timing of AHSCT
According to the IPSS, DIPSS, and DIPSS-plus scores, patients with intermediate-2 and high-risk disease have median survival of less than 5 years,2–4 and should be considered potential candidates for AHSCT.
25
A large retrospective study determined outcomes in 438 PMF patients who received AHSCT (
The optimal timing of AHSCT for MF is controversial, especially in the era of JAK inhibitors. Though not curative, the first JAK1/2 inhibitor Ruxolitinib has shown clinical benefits in patients with intermediate-2 and high-risk MF, including spleen size reduction and improvement of constitutional symptoms and also survival benefit.27–29 A major dilemma is when to proceed to AHSCT in a patient who responds well to JAK inhibitor. Early AHSCT may lead to significant morbidity and mortality, while delaying AHSCT may lead to a worse outcome due to advanced-stage disease, increasing age or LT. Some authors suggested that patients whose therapeutic goal was cure should still be referred for AHSCT, 30 even if they are responding to JAK1/2 inhibitors. This approach is not contradictory because there is a sound rationale for combining JAK1/2 inhibitor therapy with AHSCT, which we will discuss later.
Pretransplant splenectomy
Splenomegaly reflects an expansion of the underlying malignant clone in MF, and many reports have shown poor transplant outcomes in patients with splenomegaly.31,32 However, the role of splenectomy before AHSCT remains controversial. Some data observed faster engraftment in splenectomized patients.33–36 A retrospective study included 26 patients with MF, of them 11 patients had undergone splenectomy and they had faster granulocyte recovery than nonsplenectomized patients (18 days
Conditioning regimens
Many retrospective studies have demonstrated the curative potential of myeloablative conditioning (MAC) AHSCT in MF patients, with an OS of 30–60% and event-free survival (EFS) of 25–50%.34,42–47 However, the transplant-related mortality (TRM) is rather high, ranging from 30% to 48% at 1 year, which limits its applicability to young patients with good performance status (PS). The introduction of reduced intensity conditioning (RIC) has expanded the scope of AHSCT to older patients, or patients with significant comorbid conditions. Over the past decades, many studies have reported encouraging results of RIC AHSCT in patients with MF.42,48–53 In a prospective multicenter study, 103 patients received fludarabine/busulfan (FB)-based RIC regiment followed by AHSCT. The cumulative incidence of nonrelapse mortality (NRM) at 1 year was 16%, relapse at 3 years was 22%, and 5-year OS was 67%. 40
Among many available RIC regimens, FB or fludarabine/melphalan (FM) are most commonly used for MF.31,40,44,49,51,54 One study compared FB and FM regimens, and a total of 160 MF patients (FB group,
There is no prospective randomized trial comparing MAC with RIC, and all results were based on retrospective comparisons. Most studies found that patients who performed with RIC had similar outcomes to patients with MAC though they were older or with poor PS.36,45,54–56 However, a very large long-term study by Robin and colleagues observed poor results in patients with RIC AHSCT. 57 The study analyzed outcome in 1055 patients with MF undergoing transplant between 1995 and 2014, of which 645 patients received a RIC regimen. The 10-year OS, disease free survival (DFS), and relapse for 2-year survivors were 74%, 64%, and 21%, respectively. RIC was associated significantly with lower DFS and higher relapse. The consensus of the EBMT/ELN is that patients with older age or comorbidities should select a lower intensity regimen, while for patients with advanced disease and good PS a more intensive regimen is more appropriate. 25
Factors affecting AHSCT outcomes
Patient-related factors
Though older age has been considered as an independent risk factor for inferior transplant outcomes,40,42,58–60 it should not be a limiting factor, especially with the increasing use of RIC regimens. Samuelson and colleagues reported encouraging results of AHSCT in 30 patients aged 60–78 (median 65) years, with PMF or post-PV/ET MF. Conditioning regimens varied from very low intensity (fludarabine plus 2 Gy total body irradiation) to high dose (busulfan plus cyclophosphamide). Their 100-day mortality was 13%, 3-year OS and PFS were 45% and 40%, respectively.
61
In addition, a few studies identified no influence of age on post-transplant outcomes,38,50,62 what counted more were patient’s comorbidities and PS. Scott and colleagues showed in their study that higher Hematopoietic Cell Transplantation Comorbidity Index (HCT-CI) scores was associated with increased overall mortality and NRM.
38
In another study, PS was evaluable in 70 PMF patients, and PS ⩾2 (
Molecular mutations
Molecular mutations have prognostic value not only in the nontransplant setting, but also in the AHSCT setting. The presence of CALR mutations predicts a better post-transplant outcome for MF.59,64 A German group evaluated the impact of CALR mutations in 133 patients with MF who underwent AHSCT,
59
of whom 28 (21%) had mutated CALR. Compared with wild-type CALR patients, patients with mutated CALR had a significant higher 4-year OS (82%
Degree of bone marrow fibrosis
Bone marrow fibrosis (BMF) has been demonstrated not to be a barrier to AHSCT, and several reports have shown a complete reversal of fibrosis between 6 and 12 months after transplantation.66,67 In a series of 24 patients with MF who underwent RIC AHSCT, all had advanced fibrosis MF-2 (
Donor type
Some reports found similar outcomes between patients who received HLA-matched sibling donor (MSD) and unrelated donor (MUD) AHSCT for MF.38,40,42,43 Conversely, a prospective phase II clinical trial found poor survival in patients with MUD AHSCT. 51 In this study, 32 patients with sibling donors received FM conditioning regimen, and 34 patients with unrelated donors received FM plus antithymocyte globulin conditioning regimen. NRM was 22% in siblings and 59% in unrelated AHSCT. With a median follow-up of 25 months, the OS was 75% in the sibling group and only 32% in the unrelated group.
For patients who lack MSD or MUD, mismatched related donors (MMRD) provide an attractive donor source due to widespread availability. Bregante and colleagues conducted a retrospective analysis of 95 patients with MF who were allografted between 2001 and 2014, and they found haploidentical donor transplantations improved significantly with years.
69
Patients were studied in two time intervals: 2000–2010 (
Stem cell source
Published studies failed to show significant difference in outcomes of bone marrow (BM) and peripheral blood stem-cell (PBSC) grafts in patients with MF.35,38,44,63,71 However, PBSC had favorable impact on engraftment in some reports. A study by Robin and colleagues evaluated transplant outcomes in 147 patients with MF,
35
with 39 patients received BM source of stem cell and the others received PBSC. The probability of engraftment was higher in PBSC group than in BM group (93%
Due to its high graft failure, there are little data on the use of umbilical cord blood (UCB) in MF patients. A study from Japan included 224 patients to evaluated different donor sources for PMF, including 29 patients with unrelated UCB. Compared with MSD BM transplantation, the incidence of neutrophil and platelet recovery after unrelated UCB transplantation were significantly lower, and NRM at 1 year was higher (41%
Leukemic transformation
LT is observed in approximately 20% of patients with MF and the prognosis is dismal, with a median survival of less than 3 months.73,74 Risk factors for LT were unfavorable karyotype, thrombocytopenia, and increased circulating blasts.75,76 Molecular abnormalities such as IDH-1/2, SRSF2, EZH2, or ASXL1 also predicted higher risk of LT. 77 Patients with LT may achieve long-term remission after induction chemotherapy and AHSCT. Alchalby and colleagues conducted a study of 46 patients who received AHSCT for AML evolving from MF. Before AHSCT, 42 patients received induction chemotherapy, while only 9 achieved complete remission (CR), 10 achieved a minor or partial remission, and 19 were refractory or had progressive disease at the time of AHSCT. The 3-year PFS, OS, and relapse rates were 26%, 33%, and 47%, respectively, and the only significant factor for survival was CR before transplantation. 77 Hence, AHSCT after chemotherapy is associated with a potential survival benefit, especially for patients with CR before transplantation.
Role of JAK inhibitors
Ruxolitinib is the first JAK inhibitor approved by the US Food and Drug Administration for patients with intermediate- or high-risk MF and in Europe for symptomatic MF patients with splenomegaly, irrespective of disease status. The results of COMFORT-I and COMFORT-II showed that ruxolitinib rapidly reduced splenomegaly and improved MF-related symptoms and quality of life.27,28 Several studies78–83 suggested pretreatment with ruxolitinib was well tolerated and might improve outcome after AHSCT (Table 1). Besides, two studies found patients who responded well to ruxolitinib had better transplant results.79,80 In one study, 100 patients with MF were stratified into five groups based on clinical status and response to JAK1/2 inhibitors at the time of AHSCT. OS at 2 years was 61%, and this was 91% for those who experienced clinical improvement, while only 32% for those who developed LT on JAK1/2 inhibitors. 79
Major studies of ruxolitinib therapy prior to AHSCT for myelofibrosis.
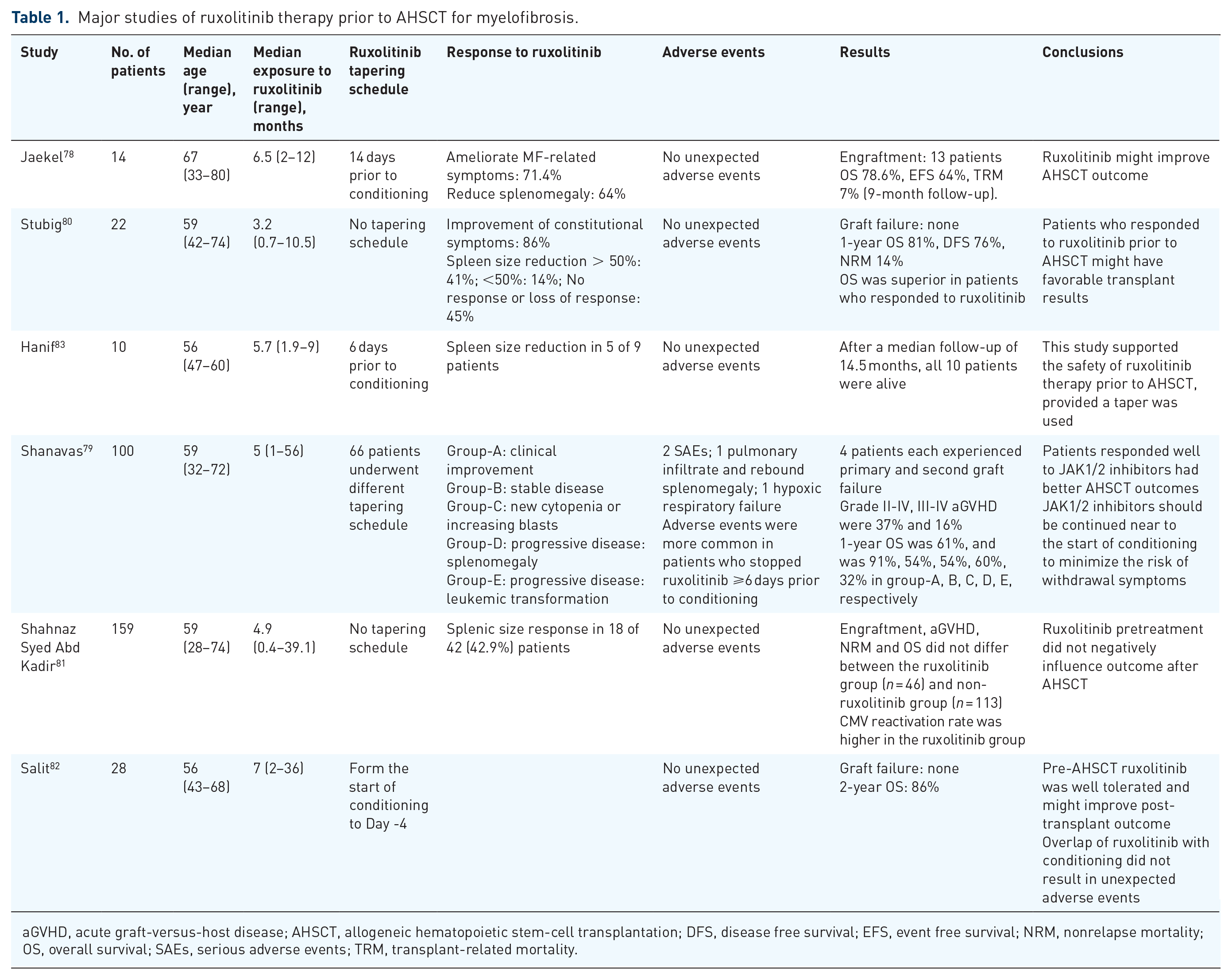
aGVHD, acute graft-versus-host disease; AHSCT, allogeneic hematopoietic stem-cell transplantation; DFS, disease free survival; EFS, event free survival; NRM, nonrelapse mortality; OS, overall survival; SAEs, serious adverse events; TRM, transplant-related mortality.
Adverse events associated with ruxolitinib in pretransplant setting including ruxolitinib withdrawal syndrome and increased risk for infections. Preliminary results of a prospective research from France reported unusual serious adverse effects such as tumor lysis syndrome, cardiogenic shock, and sepsis. 84 Of note, a large retrospective study observed two serious adverse events in MF patients who stopped JAK inhibitor ⩾6 days prior to conditioning therapy. 79 Rates of adverse symptoms were very low in patients who continued JAK inhibitor near to transplant conditioning therapy. Hanif and colleagues reported 10 patients who underwent AHSCT for MF and all patients were pretreated with ruxolitinib. 83 The steady state dose of ruxolitinib was 20 mg BID, and a standard tape schedule was employed starting 6 days prior to conditioning. At 6 days, 96 h, 72 h, 48 h, and 24 h prior to conditioning, ruxolitinib was reduced to 15 mg BID, 10 mg BID, 5 mg BID, 5 mg Daily, and None, respectively. No unexpected adverse events were observed upon ruxolitinib withdrawal. Therefore, it is recommended that the drug be initiated at least 2 months before transplant, and weaning start 5–7 days prior to conditioning, with the drug stopping the day before conditioning. 25 Ruxolitinib has been shown to inhibit in vitro and in vivo dendritic cell activation, migration, and antigen-specific T-cell response,85,86 resulting in an increased risk for infection, particularly viruses. In a small study of 12 patients with MF who were treated with ruxolitinib and underwent AHSCT, ruxolitinib was continued until stable engraftment. The authors demonstrated a higher incidence (41%) of cytomegalovirus reactivation and the onset was earlier compared with a historical group. 87 In summary, careful attention should be paid to these adverse events when received ruxolitinib prior to AHSCT.
There is little indication for JAK1/2 inhibitors after AHSCT for MF. However, they could be considered in patients who relapse to decrease the symptom burden. They may be also effective in the treatment of steroid-refractory GVHD by suppressing the production of pro-inflammatory cytokines, which needs prospective trials to validate it.
Post-transplant management
Graft failure
Graft failure is one of the major complications in patients with MF who undergo AHSCT, especially after RIC regimen, with the incidence ranging from 2% to 24%.40,51,88 Factors associated with engraftment are: donor type,51,71 stem cell source,35,89 intensity of conditioning regimen,44,90 spleen size,33,88 and age.
88
A study from Netherlands assessed the effect of conditioning regimens on graft failure in MF, and 53 patients with RIC or nonmyeloablative (NMA) regimens were included. The cumulative incidence of graft failure within 60 days was 28%, and this was higher in patients who received NMA conditioning (44%
Monitoring of residual disease and treatment of relapse
Relapse is a major cause of treatment failure for MF patients after AHSCT, with an incidence of 10–20% with MAC regimens and 29–43% with RIC regimens.40,42,43,50 As the three driver mutations, JAK2, MPL, and CALR, are detectable in approximately 90% of patients with MF, they can be used to monitor minimal residual disease (MRD) after AHSCT. Wolschke and colleagues screened 136 MF patients who underwent AHSCT for MRD for JAKV617F (
For patients with evidence of MRD or clinical relapse, discontinuation of immune-suppressive drugs, donor lymphocyte infusions (DLIs) or a second AHSCT are treatment strategies of choice. Klyuchnikov and colleagues reported their multicenter experience on the use of DLIs and a second RIC AHSCT in 30 patients with MF who relapsed (
Conclusion
AHSCT remains the only curative therapy for patients with MF. The optimal conditioning regimen has not been defined and should be based on patient age, comorbidity, and PS. MSD and MUD are preferred, mismatched related donor provides an attractive alternative donor source. JAK inhibitors have proved clinical benefits for MF patients, and further researches are needed to explore the safety and effectiveness of pretransplant therapy with ruxolitinib. Molecular mutations not only have prognostic value but also serve as MRD markers to predict early relapse. For patients with relapse, reduction of immune-suppressive drugs, DLIs, or a second AHSCT are effective treatment choices.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by grant from the National Megaproject on Key Infectious Diseases (grant number 2017ZX10202102); the CAMS Initiative for Innovative Medicine (grant number 2016-I2M-1-017), Tianjin Municipal Natural Science Foundation (grant number 18JCZDJC34400) and the Nonprofit Central Research Institute Fund of Chinese Academy of Medical Sciences (grant number 2018PT32034).
Conflict of interest statement
The author(s) declare that there is no conflict of interest.
