Abstract
Background:
Renal impairment is one of the common characteristics of multiple myeloma (MM) and makes management of MM more complicated. Even though monoclonal antibodies targeting CD38 have wildly succeeded in treating MM, the addition of anti-CD38 monoclonal antibodies to standard therapy to treat MM patients with renal insufficiency is still poorly studied.
Objectives:
This study aims to evaluate whether using anti-CD38 monoclonal antibody-based immunotherapy would improve the prognosis of MM patients with renal insufficiency.
Design:
This is a systematic review and meta-analysis.
Data sources and methods:
We searched Scopus, PubMed, Cochrane Central Register of Controlled Trials (CENTRAL), Ovid MEDLINE, Ovid Embase, and Web of Science Core Collection for randomized controlled trials that enrolled patients with MM who received CD38-targeting monoclonal antibody regimens and reported the efficacy and survival of MM with renal insufficiency. We then performed a meta-analysis to estimate the efficacy of adding anti-CD38 monoclonal antibodies to backbone regimens in MM with renal insufficiency.
Results:
In 7594 studies screened, 12 phase III trials were eligible, including 5 trials for newly diagnosed MM (NDMM; 3194 patients; 1261 with renal insufficiency) and 7 trials for relapsed refractory MM (RRMM; 2657 patients; 648 with renal insufficiency). Among NDMM patients with renal insufficiency, the addition of anti-CD38 monoclonal antibody to backbone regimens was associated with improved progression-free survival (PFS; pooled HR, 0.50; 95% CI, 0.38–0.67; p < 0.001), with little evidence of heterogeneity (Cochran Q, p = 0.19; I2 = 34.7%). Similar results were seen among RRMM patients with renal insufficiency (pooled HR, 0.46; 95% CI, 0.37–0.57; p < 0.001), with no evidence of heterogeneity (Cochran Q, p = 0.89; I2 = 0%). Similarly, the addition of anti-CD38 monoclonal antibody in RRMM among patients with renal insufficiency was associated with improved overall survival (OS; pooled HR, 0.70; 95% CI, 0.57–0.88; p = 0.002), with no significant heterogeneity (Cochran Q, p = 0.69; I2 = 0%).
Conclusion:
This meta-analysis suggests that the addition of anti-CD38 monoclonal antibodies benefits PFS in both NDMM and RRMM with renal insufficiency and OS in RRMM patients with renal insufficiency.
Keywords
Introduction
Multiple myeloma (MM) is a hematologic malignancy characterized by the excessive proliferation of abnormal clonal plasma cells in the bone marrow, which could lead to bone damage, kidney impairment, anemia, and hypercalcemia. 1 Renal impairment is one of the main features of MM, with up to 20%–40% of myeloma patients having renal dysfunction at diagnosis and approximately 2%–4% of MM patients requiring renal dialysis treatment.2–5 Furthermore, the presence of renal impairment has been acknowledged to be associated with a poorer prognosis in MM. 6
The emergence of novel drugs such as immunotherapy has increased treatment options for MM patients. 7 However, renal insufficiency often complicates the treatment of MM and poses additional treatment barriers that require special consideration. 3 CD38 is a transmembrane glycoprotein highly expressed in myeloma cells. 8 Over the past few years, monoclonal antibodies targeting CD38 have shown significant efficacy and changed the treatment landscape for MM. 8 Daratumumab and isatuximab are two anti-CD38 monoclonal antibody drugs currently approved for extensive clinical use. Daratumumab is a humanized IgG1k monoclonal antibody targeting CD38, demonstrating efficacy in both monotherapy and combination therapy settings. 8 Isatuximab is a chimeric IgG1-kappa monoclonal antibody that binds to specific epitopes on the human cell surface antigen CD38 and has been approved for the treatment of newly diagnosed MM (NDMM) and relapsed refractory MM (RRMM). However, the efficacy and safety of these two anti-CD38 monoclonal antibodies in MM patients with renal insufficiency are still poorly studied.
In this situation, we conducted a systematic review and meta-analysis of randomized clinical trials to assess the impact of the anti-CD38 monoclonal antibody incorporation to widely used treatment protocols (for eligible and ineligible patients for autologous transplantation) on the survival outcomes in patients with NDMM or RRMM with renal impairment.
Methods
This study followed the reporting guidelines of Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA; Supplemental Material 1). 9 The systematic review and meta-analysis were conducted following a previously published protocol (CRD42024543018).
Search strategy and selection criteria
The search strategy was designed and conducted by (H.B. and W.T.) with input from study investigators using the following databases: Scopus, PubMed, Web of Science Core Collection, Ovid MEDLINE, Ovid Embase, and Cochrane Central Register of Controlled Trials (CENTRAL) from inception of each database to December 6, 2024. We employed a combination of controlled vocabulary (MeSH (Medical Subject Headings) and Emtree terms) and keywords with various synonyms that encompass the concepts: “multiple myeloma OR plasmacytoma” combined with “daratumumab OR Isatuximab” and “randomized controlled trial OR RCT.” Our search results were limited to studies of the English language. The search strategy was peer-reviewed by a second librarian using Peer Review for Electronic Search Strategies (PRESS). 10 Details of the search strategy are provided in Supplemental Material 2.
The resulting citations from all databases were imported into an EndNote X9 database (Clarivate Analytics, New York, US). We removed the duplicates and screened the title and abstract of the remaining articles in EndNote. This screening process involved two independent reviewers conducting an initial review of titles and abstracts, with any discrepancies resolved by a third reviewer.
Selection criteria
After preliminary screening, the full text of potentially eligible studies was reviewed by two independent reviewers to ascertain final eligibility for inclusion in both qualitative and quantitative synthesis using the following selection criteria: (1) prospective randomized clinical trials comparing the efficacy of standard regimens with the same regimen plus daratumumab or isatuximab for NDMM or RRMM, thereby enabling a direct assessment of the comparative effectiveness, primarily attributed to the incorporation of the anti-CD38 monoclonal antibody and (2) studies reporting comparative effectiveness and survival data stratified by renal insufficiency in the primary or subgroup analysis. Exclusion criteria: (1) single-arm clinical trials or retrospective studies; (2) unavailable full text; (3) the study endpoints were unextractable.
Data extraction
Data extracted included study characteristics (first author, year of publication, journal, country of origin, study design, sample size, treatment regimens, and duration of follow-up), baseline characteristics of the participants (age, sex, race/ethnicity, and distribution by stage and performance status), and outcome data (effectiveness data and survival data). The assessment of quality was conducted using the Cochran risk of bias assessment tool. 11
Definition of outcomes
The primary outcome assessed in this study was progression-free survival (PFS), which is defined as the time from randomization to the date of first confirmed progression or death, whichever occurred first. We also evaluated the overall survival (OS) and adverse events in the meta-analysis. We quantified associations regarding hazard ratios (HRs) and 95% confidence intervals (CIs). If multiple publications were available from the same study, the publication with the most extended available follow-up results was used to extract the summary effect.
Statistical analysis
Data analysis was performed with RStudio version 4.3.2 (R Studio) using the meta packages. After extracting the PFS, OS, HRs, and 95% CI for each subgroup (MM with renal insufficiency vs MM without renal insufficiency), we pooled relative log-HRs using a DerSimonian-Laird random-effects model. 12 This model was chosen due to the anticipated variability in treatment regimens across eligible studies. We conducted separate analyses for MM cases with and without renal insufficiency, as well as for patients with newly diagnosed versus relapsed or refractory disease. In addition, separate analyses were performed for transplant-eligible and ineligible patients within NDMM. We conducted a sensitive analysis using alternative approaches to random-effects modeling, including the Knapp-Hartung method and restricted maximum-likelihood estimator. The heterogeneity among the results of the included studies was analyzed by Cochran Q and the I2 statistic and planned to explore evidence of any substantial heterogeneity with appropriate sensitivity and subgroup analysis. All statistical tests were two-sided, with a significance level of α = 0.05 for the meta-analysis.
Results
The bibliographic search resulted in 7594 citations. After removing duplicates, a total of 4455 articles underwent screening by the review of titles and abstracts. Among these, 202 articles met the criteria and were reviewed in full text, of which 12 randomized phase III clinical trials were identified, involving a collective cohort of 5851 patients, and deemed suitable for qualitative and quantitative analysis (Figure 1 and eTable 1 in Supplemental Material 3). This included five trials of patients with NDMM (ALCYONE, 13 MAIA, 14 CASSIOPEIA, 15 IMROZ, 16 and OCTANS 17 ; 3194 patients; 1261 patients with renal insufficiency) and seven trials of patients with RRMM (CASTOR, 18 POLLUX, 19 APOLLO, 20 LEPUS, 21 IKEMA, 22 ICARIA-MM, 23 and CANDOR 24 ; 2657 patients; 648 patients with renal insufficiency). The overall summary characteristics of these 12 studies are shown in Table 1.

PRISMA flow diagram.
Baseline characteristics of included trials.

Ctl, control group; DKd, daratumumab plus carfilzomib and dexamethasone; DPd, daratumumab plus pomalidomide and dexamethasone; DRd, daratumumab plus lenalidomide and dexamethasone; DVd, daratumumab plus bortezomib and dexamethasone; DVMP, daratumumab plus bortezomib, melphalan and prednisone; DVTd, daratumumab plus bortezomib, thalidomide and dexamethasone; Int, intervention group; Isa-Kd, isatuximab plus carfilzomib and dexamethasone; Isa-Pd, isatuximab plus pomalidomide and dexamethasone; Isa-VRd, isatuximab plus bortezomib, lenalidomide and dexamethasone; Kd, carfilzomib and dexamethasone; Pd, pomalidomide and dexamethasone; Rd, lenalidomide and dexamethasone; RRMM, relapsed/refractory multiple myeloma; TE-NDMM, transplant-eligible newly diagnosed multiple myeloma; TIE-NDMM, transplant-ineligible newly diagnosed multiple myeloma; Vd, bortezomib and dexamethasone; VMP, bortezomib, melphalan and prednisone; VRd, bortezomib, lenalidomide and dexamethasone; Vtd, bortezomib, thalidomide and dexamethasone.
Risk of bias
The quality assessment of the included studies is shown in Figure 2. All 12 studies demonstrated a low risk for bias in random sequence generation (selection bias, 100%). Ten of 12 studies had a low risk for bias in allocation concealment (83%, LEPUS 21 and OCTANS 17 lack reporting in this regard). All included studies were open-label studies and none reported blinding of outcome assessment, suggesting a potential for detection bias. All included studies exhibited a low risk of bias concerning incomplete outcome data (attrition bias) and selective reporting (reporting bias). All studies reported survival analysis using intention-to-treat analysis and reported response rates and toxic effect results using per-protocol analysis.
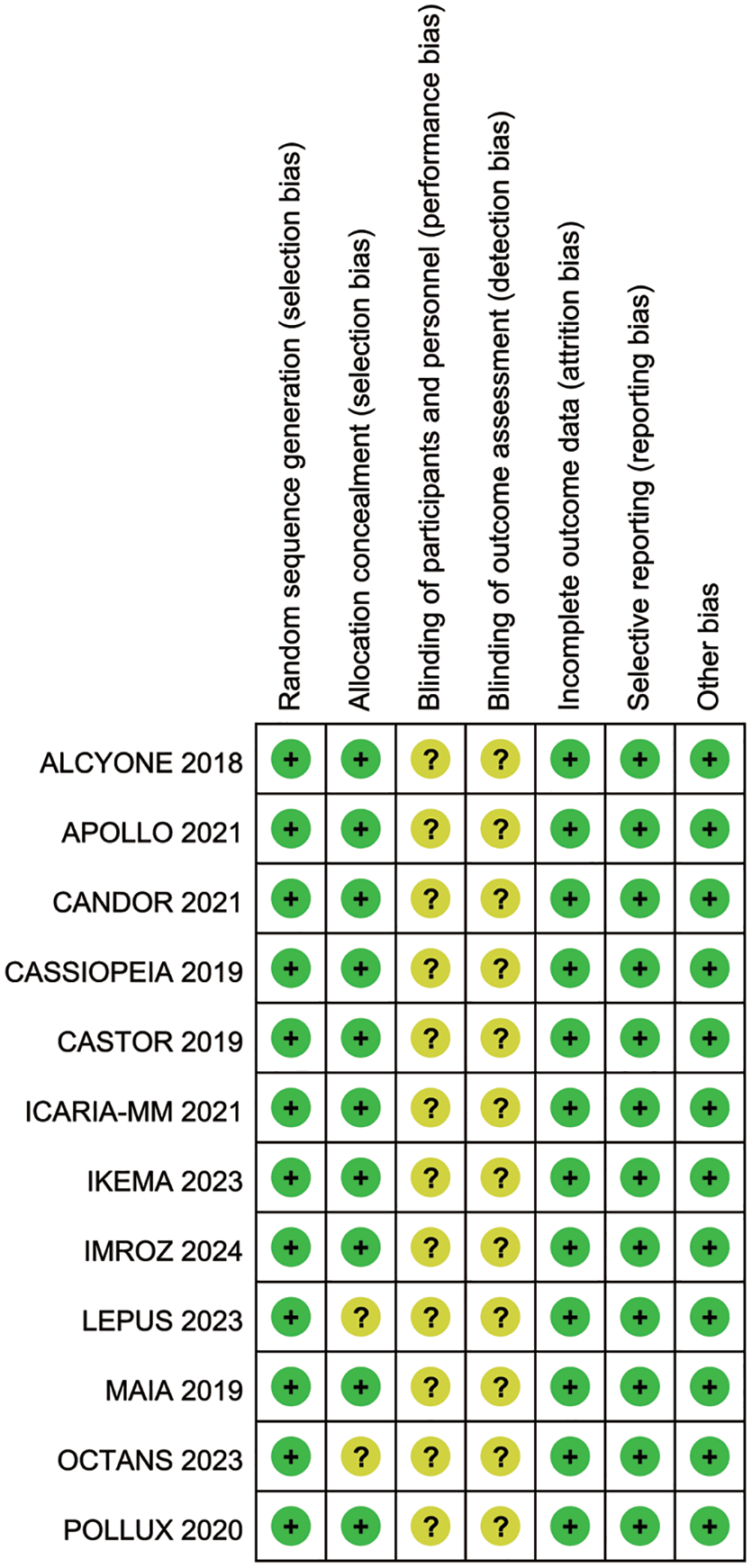
Quality assessment of included studies using the Cochrane risk of bias tool.
Meta-analysis for the association of anti-CD38 monoclonal antibody therapy with PFS in NDMM with renal insufficiency
Among the five trials studying the addition of anti-CD38 monoclonal antibody among patients with NDMM with renal insufficiency, a total of 1261 patients with renal insufficiency were included with 653 patients in the daratumumab-containing group and 608 patients in the standard therapy group respectively. The results of the meta-analysis showed 134 (20.5%) events of progression or death occurred in the anti-CD38 monoclonal antibody-containing group and 203 (40.0%) in the standard therapy group during the follow-up time. The HRs for PFS using the most recent follow-up data were 0.36 (95% CI, 0.24–0.56) in the ALCYONE 13 study, 0.37 (95% CI, 0.21–0.66) in the CASSIOPEIA 15 study, 0.60 (95% CI, 0.41–0.87) in the MAIA 14 study, 0.63 (95% CI, 0.37–1.07) in the IMROZ 16 study, and 0.85 (95% CI, 0.32–2.29) in the OCTANS 17 study. In our meta-analysis, incorporating anti-CD38 monoclonal antibody to backbone regimens among NDMM patients with renal insufficiency was associated with improved PFS (pooled HR, 0.50; 95% CI, 0.38–0.67; p < 0.001), with little heterogeneity (Cochran Q, p = 0.19; I2 = 34.7%). Among patients with NDMM without renal insufficiency, all five trials showed significant improvement in PFS when CD38 monoclonal antibody was added to backbone regimens (ALCYONE 13 : HR, 0.63 (95% CI, 0.45–0.88); CASSIOPEIA 15 : HR, 0.56 (95% CI, 0.35–0.89); MAIA 14 : HR, 0.52 (95% CI, 0.36–0.74); IMROZ 16 : HR, 0.60 (95% CI, 0.41–0.89); and OCTANS 17 : HR, 0.31 (95% CI, 0.15–0.62)). The meta-analysis showed that the addition of anti-CD38 monoclonal antibody in NDMM among patients without renal insufficiency was associated with improved PFS (pooled HR, 0.56; 95% CI, 0.46–0.67; p < 0. 001), with no evidence of heterogeneity (Cochran Q, p = 0.49; I2 = 0%). The subgroup analysis results showed no significant difference between NDMM patients with and without renal insufficiency (Figure 3).

Meta-analysis of the PFS associated with the addition of anti-CD38 monoclonal antibody for newly diagnosed multiple myeloma patients subgrouped by renal function.
Meta-analysis for the association of anti-CD38 monoclonal antibody therapy with PFS in transplant-ineligible NDMM with renal insufficiency
In the subgroup of NDMM, we further analyzed the results among patients ineligible for autologous stem cell transplantation, including 823 patients with renal insufficiency. In this subgroup analysis, incorporating anti-CD38 monoclonal antibody prolonged the PFS of transplant-ineligible NDMM patients with renal insufficiency (pooled HR, 0.54; 95% CI, 0.38–0.75; p < 0.001), with little heterogeneity (Cochran Q, p = 0.18; I2 = 38.9%). Similarly, the addition of anti-CD38 monoclonal antibody improved the PFS of transplant-ineligible NDMM patients without renal insufficiency (pooled HR, 0.56; 95% CI, 0.46–0.68; p < 0. 001), with little heterogeneity (Cochran Q, p = 0.33; I2 = 13%; Figure 4). The subgroup analysis results showed no significant difference between transplant-ineligible NDMM patients with and without renal insufficiency (Figure 4).
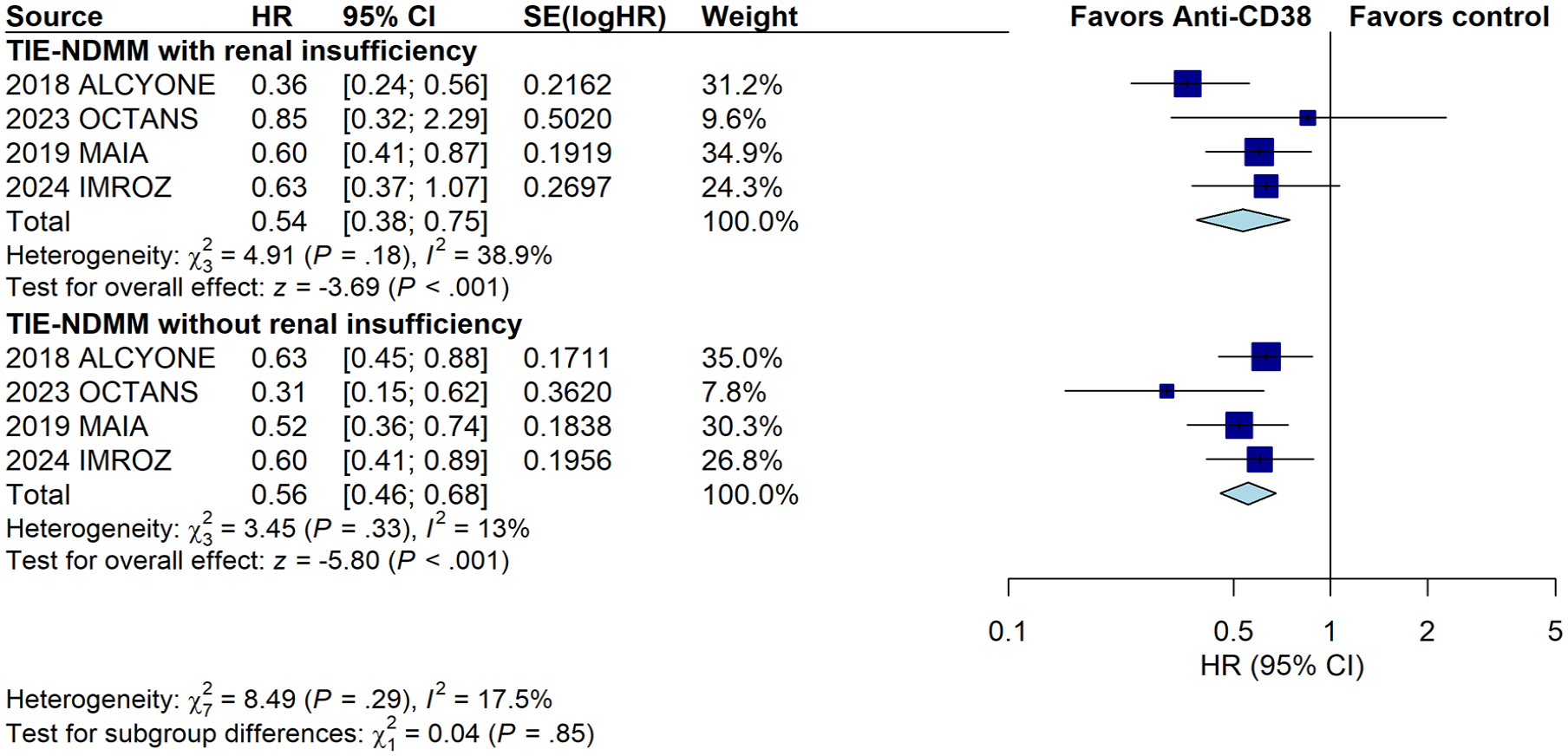
Meta-analysis of the PFS associated with the addition of anti-CD38 monoclonal antibody for transplant-ineligible newly diagnosed multiple myeloma patients subgrouped by renal function.
Meta-analysis for the association of anti-CD38 monoclonal antibody therapy with PFS in RRMM with renal insufficiency
Among the seven trials studying the addition of anti-CD38 monoclonal antibodies among patients with RRMM, a total of 648 patients with renal insufficiency were included with 354 patients in the anti-CD38 monoclonal antibody-containing group and 294 patients in the standard therapy group. The results of the meta-analysis showed 222 (62.7%) events of progression or death occurred in the anti-CD38 monoclonal antibody-containing group and 216 (73.5%) in the standard therapy group during the follow-up time. The HRs for PFS of the most recent data were 0.59 (95% CI, 0.35–0.99) in the APOLLO 20 study, 0.41 (95% CI, 0.19–0.90) in the CANDOR 24 study, 0.37 (95% CI, 0.22–0.61) in the CASTOR 18 study, 0.43 (95% CI, 0.23–0.82) in the LEPUS 21 study, 0.41 (95% CI, 0.26–0.65) in the POLLUX 19 study, 0.50 (95% CI, 0.30–0.85) in the ICARIA-MM 23 study, and 0.56 (95% CI, 0.27–1.19) in the IKEM 22 study. The meta-analysis showed the addition of anti-CD38 monoclonal antibody to backbone regimens prolonged PFS for RRMM, with no significant heterogeneity (pooled HR, 0.46; 95% CI, 0.37–0.57; p < 0.001; Cochran Q, p = 0.89; I2 = 0%; Figure 5). Similar results were seen among patients with RRMM without renal insufficiency, the significant PFS benefits in the CD38 monoclonal antibody group were reported in the APOLLO 20 (HR, 0.64; 95% CI, 0.45–0.90), CANDOR 24 (HR, 0.62; 95% CI, 0.46–0.84); CASTOR 18 (HR, 0.29; 95% CI, 0.22–0.38), ICARIA-MM 23 (HR, 0.58; 95% CI, 0.38–0.88); IKEMA 22 (HR, 0.55; 95% CI, 0.38–0.80), LEPUS 21 (HR, 0.37; 95% CI, 0.25–0.56), and POLLUX 19 (HR, 0.44; 95% CI, 0.33–0.57) studies. The meta-analysis resulted in improved PFS for RRMM without renal insufficiency (pooled HR, 0.48; 95% CI, 0.38–0.60; p < 0.001) but there was some evidence of heterogeneity (Cochran Q, p = 0.001; I2 = 72.1%; Figure 5). The subgroup analysis results showed no significant difference between RRMM patients with and without renal insufficiency (Figure 5).

Meta-analysis of the PFS associated with the addition of anti-CD38 monoclonal antibody for relapsed and refractory multiple myeloma patients subgrouped by renal function.
Meta-analysis of the association of anti-CD38 monoclonal antibody therapy with OS in RRMM with renal insufficiency
Among the included studies, mature OS data stratified by renal function were only available for six trials (MAIA 25 : HR, 0.67; 95% CI, 0.47–0.96; POLLUX 26 : HR, 0.60; 95% CI, 0.41–0.89; CASTOR 27 : HR, 0.81; 95% CI, 0.51–1.30; CANDOR 28 : HR, 0.54; 95% CI, 0.28–1.04; ICARIA-MM 29 : HR, 0.83; 95% CI, 0.54–1.27; IKEMA 30 : HR, 0.75; 95% CI, 0.37–1.58) in the renal insufficiency group. For patients without renal insufficiency, the HRs for OS using the most recent follow-up data were 0.36 (95% CI, 0.24–0.56) in the MAIA 25 study, 0.80 (95% CI, 0.61–1.04) in the POLLUX 26 study, 0.67 (95% CI, 0.51–0.88) in the CASTOR 27 study, 0.83 (95% CI, 0.61–1.28) in the CANDOR 28 study, 0.68 (95% CI, 0.47–0.98) in the ICARIA-MM 29 study, and 0.91 (95% CI, 0.60–1.38) in the IKEMA 30 study. In the meta-analysis, the addition of anti-CD38 monoclonal antibody in RRMM among patients with renal insufficiency was associated with improved OS (pooled HR, 0.70; 95% CI, 0.57–0.88; p = 0.002), with no significant heterogeneity (Cochran Q, p = 0.69; I2 = 0%). Similar results were seen for RRMM patients without renal insufficiency (pooled HR, 0.76; 95% CI, 0.66–0.88; p < 0.001), with no evidence of heterogeneity (Cochran Q, p = 0.68; I2 = 0%; Figure 6).

Meta-analysis of the OS associated with the addition of anti-CD38 monoclonal antibody for relapsed and refractory multiple myeloma patients with and without renal insufficiency.
Adverse effects associated with the addition of anti-CD38 monoclonal antibody for multiple myeloma patients
The studies we reviewed reported a range of treatment-emergent adverse events (TEAEs) for the intention-to-treat population. To evaluate the risk-benefit profile of anti-CD38 monoclonal antibody-based therapies. We performed a meta-analysis of relative risk (RR) for studies that reported relatively complete adverse event data, including neutropenia, thrombocytopenia, anemia, diarrhea, upper respiratory tract infection, and pneumonia. For NDMM patients, the results of the pooled analysis demonstrated that the addition of anti-CD38 monoclonal antibody was significantly associated with an increasing risk of diarrhea (RR, 1.14; 95% CI, 1.02–1.28), pneumonia (RR, 1.63; 95% CI, 1.35–1.97), and showed increased trends in the risks of neutropenia (RR, 1.16), thrombocytopenia (RR, 1.12), and upper respiratory tract infection (RR, 2.78; eFigures 1–5 in Supplemental Material 4). Meanwhile, anti-CD38 monoclonal antibody versus backbone regimens showed a lower trend of anemia (RR, 0.92) for NDMM patients (eFigure 6 in Supplemental Material 4). Similarly, for RRMM, the addition of an anti-CD38 monoclonal antibody significantly increased the risk of neutropenia (RR, 1.33; 95% CI, 1.15–1.56), thrombocytopenia (RR, 1.13; 95% CI, 1.04–1.22), diarrhea (RR, 1.51; 95% CI, 1.34–1.70), upper respiratory tract infection (RR, 1.58; 95% CI, 1.39–1.80), and pneumonia (RR, 1.37; 95% CI, 1.14–1.64; eFigures 1–5 in Supplemental Material 4).
Discussion
The presence of renal injury was usually associated with decreased survival outcomes and increased risk of premature mortality among individuals with MM. 6 There is still a lack of standard and effective treatment options and multiple anti-myeloma agents require dosage adjustment based on creatinine clearance. 6 Nowadays, bortezomib-based regimens are the preferred treatment for myeloma-related renal impairment due to the nonrenal metabolism of bortezomib, as well as its protective effects on kidney function.3,31,32 Several RCTs have shown that adding anti-CD38 monoclonal antibodies to the backbone regimen can improve the rate and depth of responses and PFS of MM patients with renal dysfunction.13–15,17–22,24,33 However, it still lacks further meta-analysis and higher-level evidence of the benefits of anti-CD38 monoclonal antibody on the survival outcome of MM patients with renal impairment. Meanwhile, due to the lack of head-to-head comparative trials on regimens with different drug combinations, it is difficult to determine which regimens are more advantageous for MM with kidney injury.
The current meta-analysis combined several studies with similar designs to increase the power to assess the impact of anti-CD38 monoclonal antibodies-based immunotherapy on MM patients with concurrent renal impairment. The selection of the studies for the meta-analysis guaranteed a consistent design in which the only difference between the control and experimental groups was the use of anti-CD38 monoclonal antibody. By focusing on MM patients with renal insufficiency and comparing the efficacy and survival outcome of anti-CD38 monoclonal antibody-based immunotherapy with standard treatment regimens, our study further confirmed the therapeutic benefits of anti-CD38 monoclonal antibody in MM patients with renal insufficiency and suggested incorporating anti-CD38 monoclonal antibody into backbone regimens is associated with significantly improved PFS for both NDMM and RRMM. These findings are of direct clinical relevance and may help clinicians choose an optimal MM regimen for patients with renal insufficiency.
The results of our meta-analysis supported the efficacy of anti-CD38 monoclonal antibody for NDMM patients with renal insufficiency. For transplant-ineligible MM patients with renal insufficiency, the results of the pooled analysis also indicated that the addition of an anti-CD38 monoclonal antibody improved the survival outcome. The benefits of anti-CD38 monoclonal antibodies appear consistent, irrespective of the backbone anti-myeloma regimens. In addition, the findings from our meta-analysis demonstrated the benefits of anti-CD38 monoclonal antibodies for both PFS and OS of RRMM patients with renal insufficiency. However, the management of patients with RRMM and impaired kidney function is notably more complicated in contrast to those with normal renal function due to frequent dose adjustment and diminished therapeutic efficacy. 7 The results from a UK-wide real-world dataset investigating the efficacy and tolerability of isatuximab with pomalidomide and dexamethasone in patients with RRMM showed encouraging efficacy outcomes. However, patients with renal impairment exhibited inferior PFS. 34 Therefore, the application of anti-CD38 monoclonal antibody in RRMM patients with renal insufficiency in real-world practice still needs further investigation.
Daratumumab-based regimens are generally well-tolerated. However, incorporating daratumumab into standard treatment protocols increases the frequency of infections, particularly respiratory infections, due to a higher incidence of neutropenia, induced hypogammaglobulinemia, and depletion of natural killer cells. 35 It is important to highlight the safety and adverse reaction profile of anti-CD38 monoclonal antibodies compared to the control group. We tried to fully utilize the data reported in the included studies and analyzed the available data on adverse effects. The results of pooled analysis for RR for TEAEs showed that the addition of anti-CD38 monoclonal antibody is associated with increased trends in the risks of several adverse effects such as neutropenia, thrombocytopenia, diarrhea, upper respiratory tract infections, and pneumonia. These side effects may occur more frequently than those in the control group, who typically received standard treatment. Although anti-CD38 monoclonal antibodies have demonstrated significant efficacy in treating MM, close monitoring and appropriate management strategies are essential to mitigate these risks.
In the past few years, a few clinical trials of daratumumab-based regimens have focused mainly on MM patients with kidney impairment. The prospective phase II GMMG-DANTE trial (NCT02977494) investigated daratumumab, bortezomib, and dexamethasone (DVd) in RRMM with severe renal impairment and exhibited promising results, with an overall response rate (ORR) of 67% (14/21). 36 Moreover, the GMMG-DANTE trial specifically examined the effect of DVd in MM patients with severe renal impairment, and its efficacy and safety were comparable to those without renal impairment. 36 In addition, the result of a non-comparative phase II trial (DARE, NCT03450057) demonstrated that the ORR achieved 47.4% (95% CI, 31.5–63.2) after the treatment of daratumumab with dexamethasone in RRMM patients with severe renal impairment or on dialysis. 37 A retrospective study conducted by Kuzume et al. examined 13 patients with severe renal insufficiency who received a minimum of eight doses of daratumumab and observed that the adverse effects of daratumumab in patients with severe renal insufficiency were comparable to those in patients without renal insufficiency. 38 Meanwhile, several retrospective studies and case reports showed similar results, indicating the safety and efficacy of anti-CD38 monoclonal antibodies in MM patients with renal insufficiency.38–42 Although the baseline of the included population was different, the results of these studies demonstrated the efficacy and safety of anti-CD38 monoclonal antibody for MM with renal impairment, which was consistent with the results of our study.
Several potential mechanisms are associated with improved PFS in MM with renal dysfunction treated with anti-CD38 monoclonal antibodies. Kidney damage in patients with MM is primarily caused by the nephrotoxic effects of monoclonal free light chains (FLCs) on the glomeruli and renal tubules.3,43 Previous studies have documented that daratumumab can significantly improve the rapidity and depth of hematologic responses in MM,15,18,44,45 which suggests the potential of the anti-CD38 monoclonal antibody to promote the clearance of FLCs and represents a promising tool for the therapy of light chain cast nephropathy. 46 Furthermore, incorporating anti-CD38 monoclonal antibodies is expected to optimize the therapeutic benefit-toxicity ratio of chemotherapy, and ultimately promote the recovery of kidney function, which is closely related to the morbidity and mortality of MM patients. 46
This study has some limitations. First, due to the limitation of raw data, this meta-analysis was based on PFS and OS. Meanwhile, the treatment response rates and renal change of subgroups were not assessed. Second, our study only provided limited data on the safety and adverse events of anti-CD38 monoclonal antibodies due to the variety in reporting of adverse events and inadequate data concerning renal injury, which represents a limitation for our study. Third, the heterogeneity in study designs, patient populations, and treatment protocols across the included trials poses a challenge to the comparability and overall robustness of the findings. Such variability could impact the accuracy and generalizability of the conclusions drawn from the meta-analysis. Therefore, alternative approaches to random-effects modeling including the Knapp-Hartung method and restricted maximum-likelihood estimator were used to assess the robustness of our results (eFigures 1 and 2 in Supplemental Material 5). Notably, daratumumab was the used anti-CD38 monoclonal antibody in the vast majority of the included studies. Thus, more studies with isatuximab are still needed to draw accurate conclusions regarding anti-CD38 monoclonal antibodies. Furthermore, we evaluated the potential bias using funnel plot, radial plot, and baujat plot, along with a leave-one-out sensitivity analysis (eFigures 3–6 in Supplemental Material 5). Despite efforts being made to minimize such imbalances through randomization and stratification for variables that might affect the risk of progression or death, residual confounding effects cannot be entirely ruled out. It is important to note that there is a lack of clinical study data on MM with severe renal insufficiency, as MM with severe renal insufficiency is often excluded from clinical trials. It is of great importance to include patients with extremely low renal function in future studies to ensure that the findings are applicable to all subgroups of MM patients with renal insufficiency in real-world studies.
Conclusion
In summary, our study suggests that anti-CD38 monoclonal antibody treatments could improve PFS in both NDMM and RRMM with renal insufficiency. The addition of an anti-CD38 monoclonal antibody was associated with improved OS in RRMM patients with renal insufficiency. Our findings highlight the efficacy of anti-CD38 monoclonal antibody therapy-based regimens in patients with renal insufficiency. Further research is needed to confirm these findings in real-world settings and understand their underlying mechanisms. Overall, our study contributes valuable insights into MM therapeutics.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251319593 – Supplemental material for Evaluation of anti-CD38 monoclonal antibody-based immunotherapy in multiple myeloma with renal insufficiency: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tah-10.1177_20406207251319593 for Evaluation of anti-CD38 monoclonal antibody-based immunotherapy in multiple myeloma with renal insufficiency: a systematic review and meta-analysis by Hexiang Bai, Chunlan Zhang, Ailin Zhao, Wenjiao Tang and Li Zhang in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207251319593 – Supplemental material for Evaluation of anti-CD38 monoclonal antibody-based immunotherapy in multiple myeloma with renal insufficiency: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tah-10.1177_20406207251319593 for Evaluation of anti-CD38 monoclonal antibody-based immunotherapy in multiple myeloma with renal insufficiency: a systematic review and meta-analysis by Hexiang Bai, Chunlan Zhang, Ailin Zhao, Wenjiao Tang and Li Zhang in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-3-tah-10.1177_20406207251319593 – Supplemental material for Evaluation of anti-CD38 monoclonal antibody-based immunotherapy in multiple myeloma with renal insufficiency: a systematic review and meta-analysis
Supplemental material, sj-docx-3-tah-10.1177_20406207251319593 for Evaluation of anti-CD38 monoclonal antibody-based immunotherapy in multiple myeloma with renal insufficiency: a systematic review and meta-analysis by Hexiang Bai, Chunlan Zhang, Ailin Zhao, Wenjiao Tang and Li Zhang in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-4-tah-10.1177_20406207251319593 – Supplemental material for Evaluation of anti-CD38 monoclonal antibody-based immunotherapy in multiple myeloma with renal insufficiency: a systematic review and meta-analysis
Supplemental material, sj-docx-4-tah-10.1177_20406207251319593 for Evaluation of anti-CD38 monoclonal antibody-based immunotherapy in multiple myeloma with renal insufficiency: a systematic review and meta-analysis by Hexiang Bai, Chunlan Zhang, Ailin Zhao, Wenjiao Tang and Li Zhang in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-5-tah-10.1177_20406207251319593 – Supplemental material for Evaluation of anti-CD38 monoclonal antibody-based immunotherapy in multiple myeloma with renal insufficiency: a systematic review and meta-analysis
Supplemental material, sj-docx-5-tah-10.1177_20406207251319593 for Evaluation of anti-CD38 monoclonal antibody-based immunotherapy in multiple myeloma with renal insufficiency: a systematic review and meta-analysis by Hexiang Bai, Chunlan Zhang, Ailin Zhao, Wenjiao Tang and Li Zhang in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
