Abstract
Objective
The albumin–globulin ratio (AGR) has been identified as a promising prognostic predictor of mortality in patients with hematological malignancies. This study investigated the prognostic significance of AGR in patients with multiple myeloma.
Methods
Two hundred patients diagnosed with multiple myeloma from January 2010 to October 2018 were retrospectively analyzed and followed up until December 2019. Kaplan–Meier curves and multivariate Cox regression analysis were applied to detect the prognostic value of AGR.
Results
The median follow-up period was 36 months. The optimal cutoff of AGR was 1.16 according to receiver operating characteristic curve analysis. High AGR was significantly correlated with better overall survival (OS) and progression-free survival (PFS). Multivariate analysis revealed that low AGR was an independent prognostic factor for worse OS (hazard ratio [HR] = 1.82, 95% confidence interval [CI] = 1.15–2.94) and PFS (HR = 1.53, 95% CI = 1.09–2.17).
Conclusions
AGR may represent a potential prognostic biomarker in patients with multiple myeloma.
Keywords
Introduction
Multiple myeloma (MM), which is characterized by a measurable immunoglobulin (Ig) peak in plasma or urine caused by the abnormal proliferation of atypical plasma cells in bone marrow, is a major hematological malignancy globally that is associated with high morbidity and mortality. Despite substantial advances in the diagnosis and treatment (chemotherapy and stem cell therapy) of MM, long-term survival remains unsatisfactory owing to ineffective drug treatment, disease-related complications, and local relapse. 1 At present, a number of prognostic models of clinical characteristics have considerably increased the available tools for screening patients with advanced disease and poor prognoses, including the Durie–Salmon Staging System, International Staging System (ISS), and Revised International Staging System (RISS).2,3 However, patients within similar prognostic groups display heterogeneous clinical outcomes. Therefore, it is imperative to apply critical MM markers together with traditional clinical parameters to improve the prediction of prognosis and survival.
It is accepted that malnutrition and inflammation are highly prevalent and related to the prognosis of patients with cancers. In general, serum albumin is used to reflect malnutrition, whereas serum globulin serves as an indicator of chronic inflammation. However, emerging evidence illustrated that hypoalbuminemia may represent a reliable indicator of an enhanced cancer-related inflammatory response, which is mainly associated with cytokine-induced impairment of albumin synthesis and excessive albumin degradation. 4 Recent studies identified the albumin–globulin ratio (AGR) as a valuable marker for predicting the survival of patients with different types of lymphoma5,6 and other diseases such as chronic kidney disease and chronic obstructive pulmonary disease.7,8 However, there is a lack of evidence regarding the predictive role of AGR and the albumin–globulin score (AGS) for mortality in patients with MM. The purpose of the current study was to evaluate the influence of AGR and AGS on clinical prognosis in patients with MM.
Patients and methods
Participants
The retrospective study enrolled patients with newly diagnosed MM between January 2010 and October 2018 in The Affiliated Jiangyin Hospital, School of Medicine, Southeast University. The diagnosis of MM was based on the criteria published by the International Myeloma Working Group. 9 The staging of MM was determined using the Durie–Salmon Staging System, ISS, and RISS.3,10 The study was approved by the Medical Ethics Committee of The Affiliated Jiangyin Hospital of Southeast University Medical College. The need for informed consent was waived because of the retrospective nature of this study.
Clinical and laboratory data collection
All blood samples were collected before treatment to measure the following variables: hemoglobin; mean corpuscular hemoglobin; mean corpuscular hemoglobin concentration (MCHC); mean corpuscular volume; red blood cell distribution width; white blood cell, neutrophil, and lymphocyte counts; and albumin, globulin, β2-microglobulin, and lactate dehydrogenase (LDH) levels. AGR was determined using the following formula: AGR = albumin/(total proteins − albumin). Patients with MM and low albumin levels (≤35 g/L) and high globulin levels (>35 g/L) were given an AGS of 2, those with only one abnormality were given an AGS of 1, and those with neither abnormality were given an AGS of 0. AGS of 1–2 was characterized as high, and AGS of 0 was characterized as low.
Patients with MM were routinely followed up every 3 months for the first 2 years and then every 6 months up to the fifth year. Overall survival (OS) was calculated from diagnosis to death. The date of last follow-up was applied for withdrawn patients. Progression-free survival (PFS) was calculated from diagnosis to disease progression or relapse or until the date of last follow-up.
Statistical analysis
Receiver operating characteristic (ROC) curves were applied to evaluate the sensitivity and specificity for OS and PFS, and the largest Youden’s index was estimated to determine the optimal cutoff points for AGR. 11 Comparisons of continuous variables were performed using the Mann–Whitney U test or Kruskal–Wallis test. Comparisons of categorical variables were performed using the chi-squared test or Fisher’s exact test. Survival rates in terms of OS and PFS were analyzed using the Kaplan–Meier method and log-rank test. The significant predictors of OS and PFS were evaluated by multivariate analysis using Cox’s proportional hazards model. Nomograms for OS and PFS were generated using R 3.0.3 software (Institute for Statistics and Mathematics, Vienna, Austria), and the predictive accuracy was evaluated using Harrell’s concordance index. All statistical analysis was performed using SPSS 20.0 software (IBM, Armonk, NY, USA).
Results
Patient characteristics
The clinicopathologic characteristics of all patients with MM stratified by AGS and AGR are described in Table 1. The study included 200 patients with MM, including 82 (41.0%) males and 118 (59%) females with a median age of 66 years (range, 29–88). The most prevalent MM type was IgG (49.4%). Twelve (6%) patients had bone marrow infiltration. The median follow-up periods for OS and PFS were 34 and 22 months, respectively. In the final follow-up, relapse or disease progression was identified in 183 (91.5%) patients, and 130 (65.0%) patients had died.
Baseline patient characteristics according to AGS and AGR at diagnosis.

AGR, albumin–globulin ratio; AGS, albumin–globulin score; Hb, hemoglobin; RBCs, red blood cells; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; RDW, red blood cell distribution width; WBCs, white blood cells; Ig, immunoglobulin; ISS, International Staging System; DS, Durie–Salmon; RISS, Revised International Staging System.
Optimal thresholds for AGR
The optimal cutoff for AGR was determined using ROC curve analysis. The area under the curve for AGR was 0.678 (95% confidence interval [CI] = 0.600–0.757,
Correlations of AGS and AGR with clinicopathologic parameters
To study the associations of AGS and AGR with clinicopathologic parameters, comparisons between the high and low groups for AGS and AGR were performed, as presented in Table 1. AGS and AGR were significantly associated with RDW, albumin, globulin, LDH, creatine, urea, Ig subtype, and MM stage. Moreover, AGR was closely associated with hemoglobin, red blood cell counts, MCHC, and β2-microglobulin.
Association of AGS and AGR with patient outcomes
Kaplan–Meier analysis of OS and PFS was performed to determine the associations of AGS and AGR with clinical prognosis. Our results revealed that high AGS was associated with decreased OS (
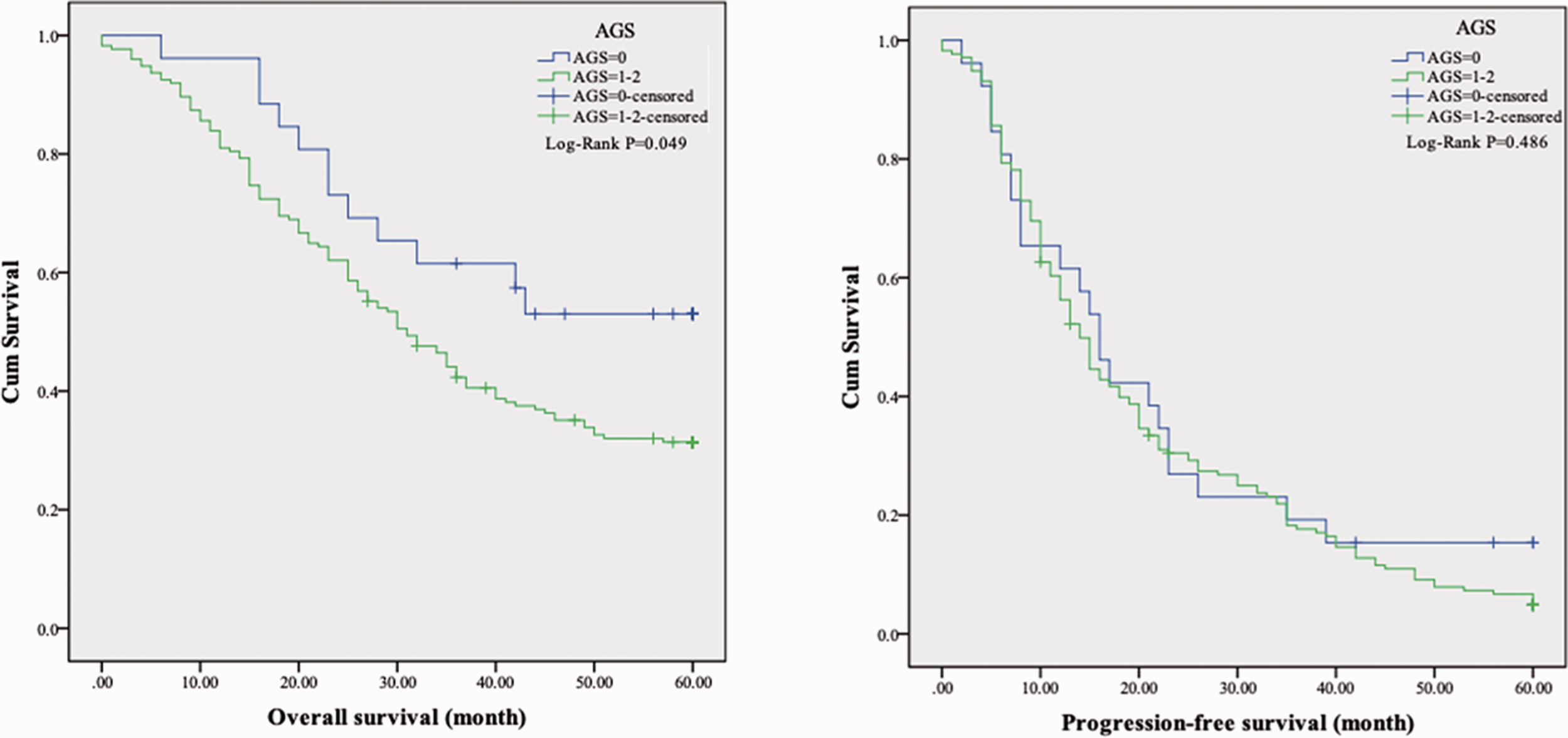
Kaplan–Meier curves of overall and progression-free survival according to the albumin–globulin score.

Kaplan–Meier curves of overall and progression-free survival according to the albumin–globulin ratio.
The influence of parameters identified as significant in univariate analysis on OS and PFS was investigated via multivariate analysis. Our results revealed that old age was an independent prognostic indicator of OS (hazard ratio [HR] = 1.02, 95% CI = 1.00–1.04,
Univariate and multivariate analysis of prognostic factors of overall and progression-free survival by Cox regression model.

AGR, albumin–globulin ratio; AGS, albumin–globulin score; Hb, hemoglobin; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; RDW, red cell distribution width; WBCs, white blood cells; Ig, immunoglobulin; ISS, International Staging System; DS, Durie–Salmon; R-ISS, Revised International Staging System; HR, hazard ratio; CI, confidence interval.
Discussion
Hypoalbuminemia is reported to be predictive of poor prognosis, and it is prevalent in patients with MM. 12 To the best of our knowledge, this is the first study to demonstrate the prognostic value of AGR in terms of OS and PFS in patients with MM. In our study, we demonstrated that high AGR was associated with favorable OS and PFS among patients with MM.
Serum albumin is generally regarded as a biological marker reflecting the nutritional status and systemic inflammatory response. Hypoalbuminemia is a common clinical feature of MM, and it includes multiple etiologies, such as malnutrition, renal dysfunction, and hepatic impairment.13–15 Despite great progress in understanding the association of serum albumin with the prognosis of several types of cancer,16–18 the influence of serum albumin on the prognosis of MM remains unclear. It has been reported that serum interleukin (IL)-6 levels are negatively correlated with serum albumin levels in patients with MM. 19 IL-6 was observed to be significantly overexpressed in various types of B-cell malignancies including MM, and it has been verified that IL-6 overproduction contributes to B-cell maturation and proliferation.20,21 Furthermore, the negative effects of hypoalbuminemia on patient prognosis might be partly attributable to its anti-oxidant function, which may induce cell growth stabilization and DNA replication. 22 Conversely, low serum albumin content may indicate malnutrition, which was found to be associated with poor prognosis.23,24 However, a recent study revealed that the increased mortality of hypoalbuminemia was partly attributed to inflammation opposed to malnutrition in patients undergoing peritoneal dialysis. 25 In contrast to albumin, globulin is a primary cortisol-binding protein containing several pro-inflammatory components such as Igs, ILs, and C-reactive protein. In fact, globulin has been widely reported as an independent prognostic indicator for various diseases.26–28 Therefore, combination variables such as AGR are considered better prognostic markers than individual markers alone.
AGR has been widely investigated as a potential prognostic factor in solid tumors, including breast cancer, 29 glioblastoma, 30 gastric cancer, 31 and renal cell carcinoma. 32 Recently, several studies found that higher AGR was correlated with reduced all-cause mortality in hematological malignancies, primarily lymphomas.5,6,33 The present study suggested that increased AGR was significantly associated with better OS and PFS in patients with MM, and the optimal AGR cutoff of 1.16 was a superior predictor in terms of HR, which achieved the best sensitivity and specificity. A low AGR could be the consequence of sustained protein loss, such as that associated with proteinuria. Proteinuria results in undernutrition and inflammation, which are associated with poor outcomes in patients with chronic disease, even in the general population. 34 Importantly, patients with MM and a low AGR should be assessed for the presence of a strong inflammatory response, malnourishment, or both. Treatment with nutritional support and anti-inflammatory agents might reduce the risk of mortality in patients with MM. Interestingly, AGR is also a predictor of chronic kidney disease [7], which may be a common manifestation of MM that is associated with an increased risk of all-cause death and cardiovascular disease. However, the applicable cutoff may be different in particular cohorts of patients with MM, and distinct results may be obtained regarding survival comparisons. Furthermore, our study revealed significant differences in OS among patients with MM stratified by AGS and AGR. Unfortunately, we failed to identify AGS as a positive predictor of outcomes for patients with MM in the present study. However, the predictive value of AGS needs further investigation.
Several limitations of the present study should be addressed. First, some CIs in univariate and multivariate Cox regression analysis were wide, which might be attributable to the retrospective, single-center nature of the study and its relatively small cohort of 200 patients, possibly resulting in bias for data collection and analysis. Second, other inflammatory parameters such as cytokines were not examined in our study because they were not routinely measured in our patients. Third, we only investigated the associations of baseline AGR with OS and PFS, and over time, treatment would lead to changes of AGR, which may reflect the dynamic changes of the nutritional and systemic inflammatory status and influence positive or negative clinical outcomes. Another potential limitation was that globulins are heterogeneous fractions that cannot be accurately measured, which may lead to different clinical prognoses.
In conclusion, our study demonstrated that AGR was an independent predictor of OS and PFS in patients with MM. However, further studies are required to identify the underlying mechanism linking inflammatory biomarkers and prognosis in these patients.
Footnotes
Authors’ contributions
WKX and XX developed the protocol; YC, YZ, QXD, and MZX collected the data; YZ and QXD analyzed the data; YC and WKX wrote and edited the manuscript; MZX revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
