Abstract
Background:
Beta-thalassemias (BTs) are characterized by deficient or absent synthesis of the beta-globin subunit, leading to anemia. Patient characteristics and treatment patterns in these patients may vary.
Objective:
This retrospective study evaluated demographics, clinical characteristics, and treatment patterns in patients with transfusion-dependent BT (TDT) and non-transfusion-dependent BT (NTDT).
Methods:
Medical records of adults with TDT or NTDT in the United Kingdom, France, Germany, Spain, and Canada with ⩾5 years of history within the practice were evaluated.
Results:
Among patients with TDT (N = 118), mean (standard deviation (SD)) age was 36.1 (11.9) years, and 28.8% were female; among patients with NTDT (N = 96), mean (SD) age was 36.6 (9.8) years, and 38.5% were female. Among patients with TDT, 21.2% received transfusions every 2 weeks or more frequently, 28.8% every 3 weeks, 26.3% every 4 weeks, and 21.2% less frequently than 4 weeks. Patients with TDT had a mean (SD) of 2.4 (0.6) units of blood transfused per transfusion, with a pretransfusion hemoglobin (Hb) level of 6.9 (1.3). In total, 84.4% of patients with NTDT had at least one transfusion, and the mean (SD) number of transfusions among patients with NTDT was 15.9 (15.9). Among patients with NTDT, the mean (SD) units of blood per transfusion were 2.2 (0.6) units, and the mean (SD) Hb level prior to transfusion was 7.4 (1.2) g/dL. Iron chelation therapy was received by 70.3% of TDT patients and 45.8% of NTDT patients.
Conclusion:
This study found that both patients with NTDT and TDT have low pretransfusion Hb levels. A high number of patients, especially patients with TDT, were not treated according to the current recommendations on target hemoglobin level, thereby highlighting the importance of national reference centers for improving long-term outcomes and quality of life in these patients.
Introduction
Beta-thalassemia (BT) syndromes are genetic disorders characterized by defective beta-globin production.1,2 The main feature of the disease is ineffective erythropoiesis, which is associated with the development of severe complications such as cardiac and liver diseases, thromboembolic events, skeletal deformities, and endocrinopathies.3,4 The annual incidence of symptomatic BT is estimated to be 1 in 100,000 persons worldwide and 1 in 10,000 persons in the European Union. 5 The incidence of BT is higher in certain geographic areas; nearly 80% of BT cases worldwide occur in sub-Saharan Africa, the Mediterranean Basin, the Middle East, and South and Southeast Asia. 6
The main treatment modality for patients with BT is red blood cell transfusions (RBCTs), and patients are typically classified into one of two categories—transfusion-dependent BT (TDT) or non-transfusion-dependent BT (NTDT)—depending on the need for regular RBCTs. Patients with TDT require lifelong, regular RBCTs. 7 Patients with NTDT may require regular RBCTs during pregnancy and periods of infection, as well as when managing disease-associated symptoms and/or complications. 2 Of note, both patient groups require lifelong medical care. While regular RBCTs are generally not indicated in patients with NTDT, these patients still may experience significant morbidity related to BT, including complications from both anemia (e.g., bone deformities, hepatosplenomegaly) and iron overload (e.g., leg ulcers, cardiac complications, growth deficiency), as well as a reduced quality of life and expected overall survival. 8 Although effective in managing the disease, RBCT introduces the risk of complications, such as alloimmunization and infections, and can impose a significant burden on patients. 3
Additionally, regular RBCTs are associated with iron overload (especially in patients with TDT). 3 Iron overload contributes to the development of numerous complications, most commonly cardiac dysfunction (e.g., arrhythmia, heart failure), endocrine disorders, and liver disease. 4 Iron overload is typically managed using iron chelation therapy (ICT). 7 However, ICT introduces additional toxicities and patient monitoring requirements and is often associated with low adherence.8,9 Real-world data regarding the characteristics and treatment patterns of patients with BT are currently limited.
Methods
This retrospective, noninterventional review of medical records evaluated patients diagnosed with either TDT or NTDT BT in real-world settings in the United Kingdom, France, Germany, Spain, and Canada; data from the review were then used to describe demographic and clinical characteristics, document overall treatment patterns related to anemia (including both transfusion and nontransfusion treatments), report on treatment patterns for iron overload, and characterize clinical outcomes. Physicians were recruited to participate if they were practicing hematologists, had treated at least five patients with BT since January 2016, acted as the main decision maker regarding treatment and follow-up, and had access to the records of both living and deceased patients. Recruitment was done via a fieldwork partner agency who identified potential participants using (1) physician directories maintained by local medical associations, (2) in-house databases, and (3) telephone directories (when supplemental recruitment efforts were needed). Physicians who participated in the study were compensated for their time spent on data abstraction according to a usual and customary rate.
Patients were identified for study inclusion if they had a confirmed diagnosis of TDT or NTDT (i.e., β-thalassemia or hemoglobin (Hb)) E/β-thalassemia; concomitant alpha globin mutation and/or multiplication was allowed) with at least one visit to a participating physician/center on or after January 2016. Patients were censored at the first observed occurrence of any of the following: gene therapy/gene editing, bone marrow or stem cell transplantation, initiation of luspatercept, participation in a clinical trial for the treatment of BT. Patients with any of these events before January 2016 were excluded from the study. Among patients with a censoring event, the date of the first occurrence of a censoring event defined their index date. Among patients without a censoring event, their index date was defined as their last observed visit to the physician/center. Patients were considered to have TDT if they received 6–20 units of packed RBC with no transfusion-free period of >35 days within 24 weeks preceding their index date. Patients were excluded from the study if they had BT minor (as reported in their medical record) or were known to be positive for human immunodeficiency virus. Furthermore, all patients were required to have at least 5 years of data history with the participating physician/center before the index date and to be aged at least 23 years on the index date (to guarantee that at least 5 years of history during adulthood were included).
In total, the target sample size was 50 patients per country (20 in Canada), with an even split between patients with TDT and NTDT. Only data existing in the medical record at the time of abstraction (i.e., October 2021 through February 2022) were used in the study. No prospective data collection or direct contact with patients occurred. Additionally, all data were de-identified.
From the medical record, the following information was extracted: patient demographics (e.g., age, sex, race/ethnicity, region of birth); clinical characteristics (transfusion dependency status, BT mutation status); BT-related complications and comorbidities recorded in the medical record at any point in time; treatment patterns for RBCTs (historical treatment frequency, average duration of transfusions, average units of blood transfused, average Hb concentration before transfusion); and treatment patterns other than RBCTs.
Patient demographics, clinical characteristics, BT-related complications and comorbidities, and treatment patterns were reported descriptively. Aggregated analyses were performed using pooled data from all countries and for the subgroups of patients with TDT and NTDT. Additionally, country-specific analyses were performed to further characterize geographic variations in treatment patterns. All analyses were performed in SAS statistical software, version 9.4 (SAS Institute, Cary, NC, USA).
Results
Demographics and clinical characteristics
In total, data for 214 patients were abstracted from medical records (118 patients with TDT and 96 patients with NTDT). Physicians had a mean (standard deviation (SD)) of 8.5 (3.8) years of history available for patients with TDT (range: 7.4 (2.9) in Germany to 10.6 (7.0) in Canada). Among patients with TDT, mean (SD) age was 36.1 (11.9) years (range: 31.6 (9.8) years in the United Kingdom to 41.7 (16.4) years in Spain) and 28.8% were female (range: 14.8% in France to 40.7% in Germany and the United Kingdom; Table 1). Approximately three-quarters of patients with TDT had their race/ethnic group listed as a Mediterranean country (range: 55.6% in the United Kingdom to 88.5% in Spain).
Baseline characteristics of patients with TDT.

Includes Italy, Spain, France, Greece, Turkey, and Albania.
Includes Iran, Iraq, and Afghanistan.
Includes India, Pakistan, and Sri Lanka.
Includes Morocco, Algeria, Tunisia, and Egypt.
Includes Southeast Asia (Thailand, Malaysia, Indonesia), Other Asia (South Korea, Japan, China), Other Africa, Other, and unknown.
BT, beta-thalassemia; Hb, hemoglobin; SD, standard deviation; TDT, transfusion-dependent beta-thalassemia; UK, United Kingdom.
Among patients with TDT, receipt of luspatercept was the most common reason for censoring in France, Germany, Spain, and Canada, whereas in the United Kingdom only one patient (with a bone marrow or stem cell transplant) was censored. Approximately 10% of patients with TDT had co-occurring alpha-thalassemia (range: no patients with alpha-thalassemia were identified in the United Kingdom to 19.2% in Spain). Nearly three-quarters of patients with TDT had BT mutation testing (range: 63.0% in Germany to 100.0% in Canada), with the most common mutation results including β0/β0 (35.6% overall), β+/β+ (26.4% overall), and β0/β+ (21.8% overall).
Patients with TDT had a mean (SD) of 1.8 (2.4) comorbid conditions (range: 0.4 (0.7) in Canada to 2.7 (3.0) in the United Kingdom). The most commonly reported comorbidities of interest among patients with TDT were hypothyroidism (19.5% overall), hepatosplenomegaly (12.7% overall), hypogonadism (11.9% overall), bone deformities (11.9% overall), growth hormone deficiency (11.0% overall), and mild liver disease (11.0% overall).
Physicians had an average of 8.6 (4.1) years of history available for patients with NTDT (range: 7.2 (3.2) in Germany to 11.7 (5.5) in the United Kingdom). Among patients with NTDT, mean (SD) age was 36.6 (9.8) years (range: 34.0 (9.8) years in Spain to 42.7 (10.8) years in Canada; Table 2) and 38.5% were female (range: 33.3% in France and Germany to 52.9% in the United Kingdom). Over half of patients with NTDT had their race/ethnic group listed as a Mediterranean country (range: 37.0% in France to 78.3% in Spain).
Baseline characteristics of patients with NTDT: overall and by country.

Includes Italy, Spain, France, Greece, Turkey, and Albania.
Includes Iran, Iraq, and Afghanistan.
Includes India, Pakistan, and Sri Lanka.
Includes Morocco, Algeria, Tunisia, and Egypt.
Includes Southeast Asia (Thailand, Malaysia, and Indonesia), Other Asia (South Korea, Japan, and China), Other Africa, Other, and unknown.
BT, beta-thalassemia; Hb, hemoglobin; NTDT, non-transfusion-dependent beta-thalassemia; SD, standard deviation; UK, United Kingdom.
Among patients with NTDT, the most common censoring criterium was receipt of luspatercept, with almost one-quarter of patients with NTDT receiving this medication (receipt of luspatercept was the most common reason for censoring in Germany and Canada, bone marrow or stem cell transplant was the most common reason for censoring in France and Spain, and no patients in the United Kingdom had a censoring criterium). In total, 12.5% of patients with NTDT had co-occurring alpha-thalassemia (range: 7.4% in France to 40.0% in Canada). Nearly two-thirds of patients with NTDT had BT mutation testing (range: 47.8% in Spain to 100.0% in Canada), with the most common mutation results including β+/β+ (31.8% overall), β0/β+ (30.2% overall), and HbE-β T/β+ (19.1% overall).
Patients with NTDT had an average of 1.0 (1.3) comorbid conditions (range: 0.4 (0.6) in Canada to 1.5 (1.4) in the United Kingdom). The most common comorbidities were cholelithiasis (13.5% overall) and hypersplenism (10.4% overall).
Transfusion patterns
Among patients with TDT, the most common transfusion frequency at the time of data abstraction was every 3 weeks (29% of patients), followed by every 4 weeks (26% of patients), every 6 weeks (18% of patients), and every 2 weeks (16% of patients; Table 3). However, transfusion frequency varied by country; in these patients, the most common transfusion frequency was every 2 weeks in the United Kingdom, every 3 weeks in Canada, every 4 weeks in France and Germany, and every 6 weeks in Spain.
RBC transfusion characteristics at the time of the abstraction: patients with TDT.

Estimation is limited to the number of years patients received follow-up.
RBC, red blood cell; SD, standard deviation; TDT, transfusion-dependent beta-thalassemia; UK, United Kingdom.
Among all patients with TDT, the mean (SD) average number of units of blood cells transfused per transfusion was 2.4 (0.6). Despite Spain having the highest percentage of patients with the longest interval between transfusions (i.e., 6 weeks), patients in Spain also had the lowest average number of units received per transfusion (mean (SD): 1.9 (0.6); with mean (SD) pretransfusion hemoglobin levels of 6.9 (1.2)), whereas patients with TDT in the United Kingdom—where there was the highest percentage of patients with the shortest interval (2 weeks) between transfusions—had the largest average number of units transfused (mean (SD): 2.7 (0.5); with mean (SD) pretransfusion hemoglobin levels of 6.5 (1.6)). Among all patients with TDT, the mean (SD) hemoglobin level before transfusion ranged from 5.9 (0.4) among patients in Canada to 7.3 (1.5) among patients in Germany. Additionally, patients with TDT tended to have the same transfusion frequency and units of blood transfused for several years (mean (SD) overall: 6.1 (6.0) years; range: 3.8 (2.2) years in Canada to 8.7 (5.8) years in the United Kingdom). Transfusion-related adverse events were infrequently reported in this study in patients with TDT, with allergic reactions being the most common (occurring in 2.5% of patients), followed by nonhemolytic febrile transfusion reactions (occurring in 1.7% of patients) and transfusion-associated overload (occurring in 0.9% of patients).
Among patients with NTDT, 84.4% of patients had detailed information for ⩾1 transfusion in the medical record (range: 66.7% in Germany to 100.0% in Canada), and on average patients had mean (SD) 15.9 (15.9) recorded transfusions (range: 7.0 (4.9) in Canada to 18.2 (14.5) in France; Table 4). Patients with NTDT had an average of 2.2 (0.6) units of blood transfused (range: 1.9 (0.3) in Spain to 2.7 (0.7) in France). The mean (SD) hemoglobin level among patients with NTDT before transfusion was 7.4 (1.2; range: 6.2 (0.3) in Canada to 8.2 (1.2) in Spain). Among patients with NTDT, the most common reason for the most recent transfusion was cited as hemoglobin level <8 g/dL (33.3% of patients), followed by hemoglobin level <7 g/dL (19.8% of patients), infection exacerbating anemia (16.1% of patients), maintaining hemoglobin levels above 10 g/dL (14.8% of patients), and hemoglobin level <6 g/dL (13.6% of patients). The most common reason for the most recent transfusion among patients with NTDT varied by country, with the most common reason in the United Kingdom listed as infection exacerbating anemia (46.7% of patients), in both France and Germany hemoglobin level <8 g/dL (58.3% and 31.3% of patients, respectively), in Spain maintaining hemoglobin level above 10 g/dL (38.1% of patients), and in Canada hemoglobin level <7 g/dL (100.0% of patients). The most common transfusion-associated adverse events experienced by patients with NTDT were allergic reactions (4.9% of patients), followed by nonhemolytic febrile transfusion reactions (1.2% of patients), alloimmunization (1.2% of patients), and autoimmune hemolytic anemia (1.2% of patients).
RBC transfusion characteristics: patients with NTDT.

Among patient with ⩾1 transfusion, represents all blood transfusions recorded in the medical record, regardless of the time period.
Numbers may sum to >100%, as multiple selections were allowed.
Across all available transfusions.
NTDT, non-transfusion-dependent beta-thalassemia; RBC, red blood cell; SD, standard deviation; UK, United Kingdom.
Nontransfusion therapies
Among patients with TDT, 21.2% had a splenectomy (range: 7.4% in the United Kingdom to 54.6% in Canada; Table 5) and mean (SD) age at splenectomy was 20.1 (10.4) years. Use of ICT was common among patients with TDT, with over 70% of patients receiving this treatment (range: 44.4% in the United Kingdom to 92.6% in Germany). The most common type of ICT received was deferoxamine (67.5% of patients), followed by deferasirox (38.6% of patients). In total, 72.9% of patients had at least one serum ferritin level test (range: 44.4% in the United Kingdom to 100.0% in Canada), and the mean (SD) test result was 668.7 (612.6) µg/L (range: 290.0 (26.5) in Canada to 1042.1 (798.6) in Germany). Hydroxyurea (HU) was received by less than one-quarter of patients with TDT (range: 3.7% in the United Kingdom to 45.5% in Canada). Among patients who received HU, mean (SD) average daily dose was 15.0 (5.4) mg/kg. Most (i.e., >80%) patients with TDT received a hepatitis B vaccine (range: 54.6% in Canada to 100.0% in the United Kingdom). Other medications commonly received by patients with TDT included oral supplements (33.9% overall), erythropoiesis-stimulating agents (29.7% overall), antibiotics (21.2% overall), and hypothyroidism treatment (11.9% overall).
Nontransfusion therapies: patients with TDT.

Received at any point in time, as available in the medical record.
Age of splenectomy was unknown for all patients in the United Kingdom with this procedure.
In the United Kingdom, France, and Spain, only one patient had information on hydroxyurea dose available.
Received since the patient was aged ⩾18 years.
SD, standard deviation; TDT, transfusion-dependent beta-thalassemia; UK, United Kingdom.
Among patients with NTDT, 19.8% had a splenectomy (range: 5.9% in the United Kingdom to 33.3% in Germany; Table 6). Across all countries, ICT was received by 45.8% of patients with NTDT (range: 37.0% in France to 80.0% in Canada). In total, 86.5% of patients had at least one serum ferritin level test (range: 75.0% in Germany to 100.0% in both the United Kingdom and Canada), and the mean (SD) test result was 372.1 (326.1) µg/L (range: 277.2 (31.7) in Canada to 466.2 (259.3) in the United Kingdom). Hydroxyurea was received by 32.3% of patients with NTDT (range: 0.0% in Canada to 50.0% in Germany). Most (i.e., >80%) patients with NTDT received a hepatitis B vaccine (range: 73.9% in Spain to 87.5% in Germany). Other medications commonly received by patients with NTDT included oral supplements (25.0% overall), erythropoiesis-stimulating agents (15.6% overall), and antibiotics (11.5% overall).
Nontransfusion therapies: patients with NTDT.
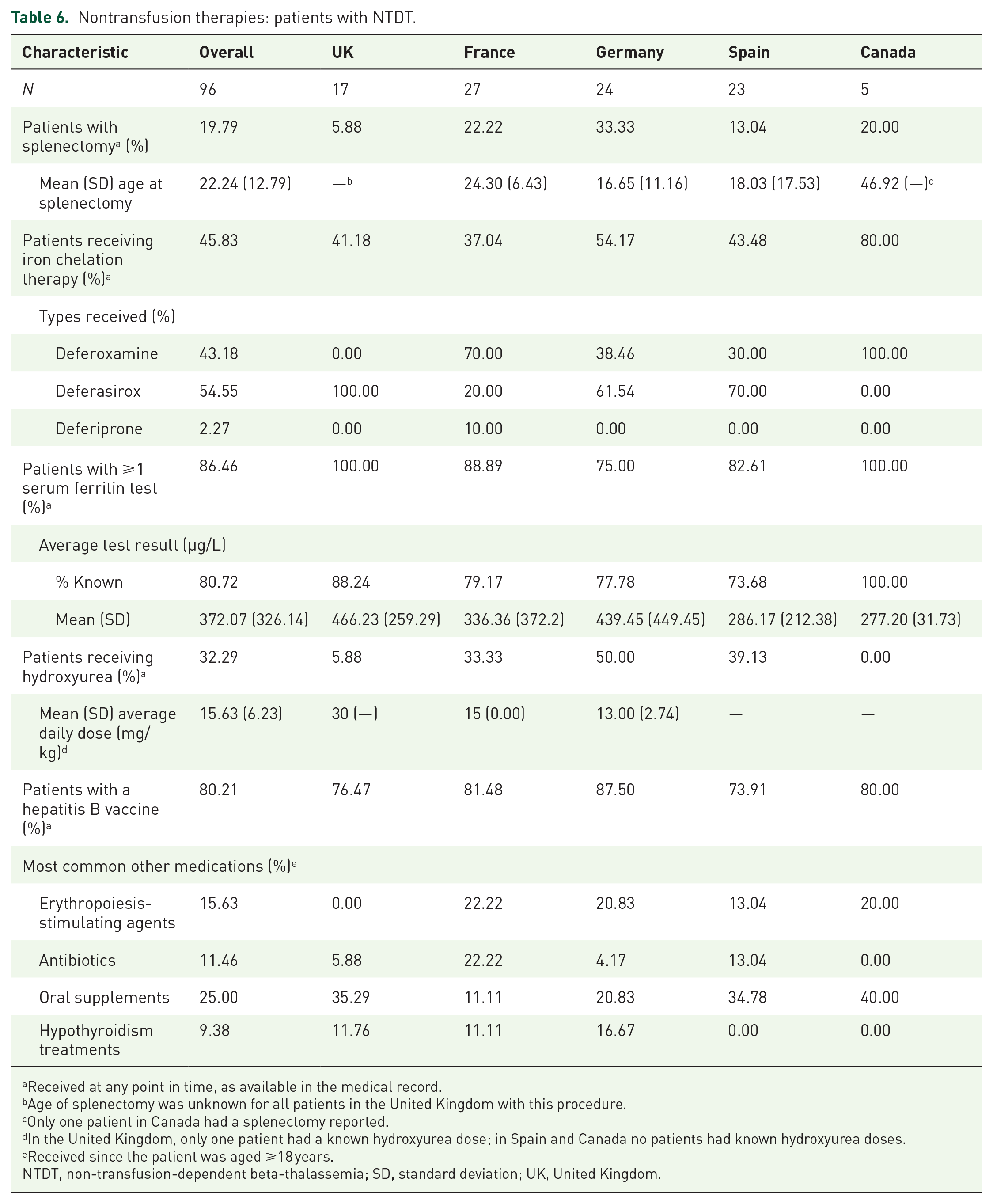
Received at any point in time, as available in the medical record.
Age of splenectomy was unknown for all patients in the United Kingdom with this procedure.
Only one patient in Canada had a splenectomy reported.
In the United Kingdom, only one patient had a known hydroxyurea dose; in Spain and Canada no patients had known hydroxyurea doses.
Received since the patient was aged ⩾18 years.
NTDT, non-transfusion-dependent beta-thalassemia; SD, standard deviation; UK, United Kingdom.
Discussion
This study evaluated 214 patients with BT across four countries in Europe and Canada and provides much-needed insight into the real-world clinical needs of patients with this disease. In this study, we observed that patients with TDT had a high transfusion burden, with over three-quarters of patients requiring transfusions at least every 4 weeks. Furthermore, patients with TDT had low average pretransfusion hemoglobin levels and required an average of 2.4 units of blood per transfusion.
Current treatment guidelines recommend that transfusions in patients with TDT be performed every 2–4 weeks to maintain pretransfusion hemoglobin levels above 9.5–10.5 g/dL (or up to 11.0–12.0 g/dL for patients with cardiac complications). 10 Additionally, a recent study by Musallam et al. found that risk of mortality decreased with increasing hemoglobin levels beginning at 9.5 g/dL. 11 However, this study found that average hemoglobin levels before transfusions among patients with TDT were 6.9 g/dL (range: 5.9 g/dL in Canada to 7.3 g/dL in Germany), which is well below the target range specified in the aforementioned guidelines. This highlights the need for better management of Hb levels to minimize disease-associated complications and increase quality of life in these patients.
To date, the published data regarding real-world treatment patterns and clinical characteristics of patients with BT are limited. Angelucci et al. 12 evaluated BT prevalence and treatment in Italy using hospital audit data combined with a medical record review. In total, 162 medical records of patients with TDT and 43 medical records of patients with NTDT were evaluated. Their study found that among patients with TDT, approximately half received ⩾3 units of red blood cells per month. Additionally, endocrine pathologies were the most commonly reported comorbidity among both TDT and NTDT patients. Similarly, our analysis found that, on average, patients with TDT received 2.9 units of red blood cells per month, whereas 84.4% of patients with NTDT had at least one transfusion recorded in their medical record. Our analysis also observed that endocrine pathologies, including hypothyroidism and hepatosplenomegaly, were the most common comorbidities in patients with TDT, whereas cholelithiasis and hypersplenism were the most common comorbidities in patients with NTDT.
Two studies based on retrospective administrative claims data have been published. Tang et al. evaluated health insurance claims data from Taiwan’s National Health Research Database and reported on complications stratified by RBCT units received. 13 They observed that the percentages of patients with complications were higher among patients receiving >0 RBCT units than among patients receiving 0 RBCT units—specifically, 13.3% versus 34.4% for cardiac complications, 16.1% versus 37.4% for endocrine complications, 21.0% versus 39.0% for liver disease, and 6.2% versus 23.0% for renal complications for patients receiving 0 RBCT units versus >0 RBCT units, respectively. Maserejian et al. 14 evaluated children and adult patients with BT using two large administrative claims databases in the United States to report on treatment patterns and health outcomes. They found that the most common comorbidities were nutritional, endocrine, and metabolic disorders (observed in 76.0%–80.0% of TDT patients), followed by diseases of the heart (observed in 46.4%–52.0% of TDT patients). While neither of these analyses is directly comparable to our study in terms of design or geographic location, our analysis observed a similarly high rate of comorbidities, with endocrinopathies the most common comorbid conditions observed.
This study has several limitations common to observational studies using data abstracted from medical records. Information on the number of patients in the physician practices who met each of the study inclusion and exclusion criteria were not available. Rather, the data were collected from a convenience sample that may not be generalizable to the overall population of patients with BT in each country. Furthermore, patients with bone marrow or stem cell transplantation were censored at the point of receipt of bone marrow/stem cell transplantation. This was done as patients with potentially curative treatment would likely have different treatment patterns and subsequent outcomes than patients without receipt of these therapies. Additionally, this study was focused on countries with a low incidence of BT. Accordingly, results may be different for other geographical regions. Although the electronic data collection form included numerous data checks to assess and maximize internal consistency, data collection may have been subject to data entry errors, potentially resulting in inaccuracies in reporting. In order to maintain patient deidentification, responses were not validated against patients’ medical records by an independent reviewer external to the study site. However, a key strength of this study was the use of a customized electronic data collection form that allowed for the abstraction of data in a uniform structure across all countries. Measures that may be subject to a clinician’s interpretation and are not typically available in pre-existing coded data sources were also collected as part of this study, contributing to the robustness of the analysis and adding another dimension of insight to the published literature on this topic. Identification of patients as TD or NTD was based on the expertise of the physician. There may have been some occasions when patients were falsely classified as NTDT even though their hemoglobin levels required transfusions, and they should therefore have been classified as TDT. Guidelines to aid in identification of these patients may be helpful to ensure that patients receive the appropriate treatment as soon as possible and thereby avoid complications.
Conclusion
In conclusion, this study provides much-needed insights on clinical characteristics and treatment patterns among patients with either NTDT or TDT in multiple countries. Both NTDT and TDT patients were found to have similarly low pretransfusion hemoglobin levels, with patients with TDT requiring RBCTs every 2–4 weeks with 2–3 units of blood per transfusion, on average. Furthermore, many patients, especially TDT patients, are not treated according to the current recommendations (e.g., lower than recommended pretransfusion hemoglobin levels), highlighting the importance of national reference centers to improve outcomes and quality of life in these patients. New therapeutic options that can reduce RBCT dependency for patients with TDT and maintain transfusion independence for patients with NTDT are needed.
