Abstract
Background:
Polycythemia vera (PV) patients often experience constitutional symptoms and are at risk of thromboembolism as well as disease progression to myelofibrosis or acute myeloid leukemia. Not only is PV often overlooked but treatment options are also limited, however.
Objectives:
To explore the patient characteristics and treatment pattern of PV patients in Taiwan, and compare with other countries reported in the literature.
Design:
This is a nationwide cross-sectional study.
Methods:
The National Health Insurance Research Database in Taiwan, which covers 99% of the population, was utilized. Patients were identified during the cross-sectional period between 2016 and 2017, and their retrospective data were retrieved from 2001 to 2017.
Results:
A total of 2647 PV patients were identified between 1 January 2016 and 31 December 2017. This study described the demographic information of these patients, including number of patients by risk stratification and by sex, age at diagnosis, age at cross-sectional period, rate of bone marrow aspiration/biopsy at diagnosis, comorbidities, number of postdiagnosis thrombosis, number of disease progression, and death. The mortality rate of PV patients (4.1%) over 60 of age was higher than the general population of the same age group (2.8%). This study also compared the different treatment patterns between sexes and risk groups. Hydroxyurea was deferred to an older age, but conversely was prescribed at higher dose to younger patients. Alarmingly, a high proportion of patients did not receive phlebotomy or hydroxyurea for at least 2 years. Furthermore, discrepancies in prevalence, age at diagnosis, sex ratio, incidence of thrombosis and mortality were also found when compared with data reported in other countries.
Conclusion:
The clinical landscape of PV in Taiwan between 2016 and 2017 was examined. Distinctive patterns of phlebotomy and hydroxyurea were identified. Overall, these findings highlight the importance of understanding the patient characteristics and treatment patterns of PV in different regions to better inform clinical practice and improve patient outcomes.
Keywords
Introduction
Polycythemia vera (PV) is one of the classical Philadelphia-negative chronic myeloproliferative neoplasms (MPNs). Common clinical manifestations of PV include primary erythrocytosis and subsequent hypervolemia and hyperviscosity as well as thrombocytosis and leukocytosis. Besides constitutional symptoms such as fatigue, headache, fever, night sweats, weight loss, and pruritus. PV patients are also at risk of thromboembolism, progression to myelofibrosis, and transformation to acute myeloid leukemia.
Patients with PV are divided into low- and high-risk groups of thrombosis occurrence based on age and history of thrombosis. 1 A low-risk patient is defined as age below 60 years with no history of thrombosis, whereas high-risk is defined as patients aged 60 years and above or with a history of thrombosis. Based on this risk classification, recommendations for treatment by the National Comprehensive Cancer Network vary slightly. 2 Recommendations for low-risk patients include monitoring for new thrombosis/bleeding events and managing cardiovascular risk factors as well as receiving low-dose aspirin, phlebotomy, and ropeginterferon alfa-2b if needed. In addition to the above, recommendations for high-risk patients also include the use of hydroxyurea (HU) or peginterferon alfa-2a at the time of confirmed diagnosis. Cytoreductive therapy (for low-risk patients) or an alternative therapeutic agent (for high-risk patients) is also recommended if patients develop new thrombosis or disease-related major bleeding, symptomatic or progressive splenomegaly, symptomatic thrombocytosis, progressive leukocytosis, or progressive disease–related symptoms, or if patients have a frequent and persistent need for but poor tolerance to phlebotomy. In addition, intolerance and resistance to cytoreductive agents also necessitate a switch of medications. Besides HU and interferons, ruxolitinib is indicated for HU-intolerant/resistant patients as a second-line treatment.
The treatment options for PV are limited, especially in Taiwan. Prior to 2022, the most common treatment modalities were aspirin, phlebotomy, and HU. At the time, ruxolitinib and ropeginterferon alfa-2b were not subsidized by the National Health Insurance (NHI), and other interferon agents were not readily available; hence, these therapeutic agents are not within the scope of this study.
To investigate the clinical care landscape of PV treatment with limited therapeutic options, we conducted a nationwide database cross-sectional study to elucidate the demographics, clinical characteristics, as well as phlebotomy and HU treatment patterns of PV patients identified between 1 January 2016 and 31 December 2017.
Methods
Data source
The current NHI healthcare system in Taiwan is a single-payer compulsory social insurance that provides healthcare access to 99% of the population. We accessed the National Health Insurance Research Database (NHIRD), which provides de-identified data on the entire population from 2001 to 2017. Extracted data included dates of birth and death, sex, diagnostic codes [e.g. International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM); International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM)], procedure codes, and drug codes.
Study design
Unlike other studies that utilized the NHIRD, our investigation was limited to a cross-sectional study between 2016 and 2017 due to the ICD systems implemented in Taiwan. Prior to 2016, the ICD-9-CM classification system was in use, wherein no specific codes existed for essential thrombocythemia, which could have been miscoded as PV. Following the introduction of the ICD-10-CM classification system in 2016, all MPN subtypes now have their respective ICD-10-CM codes: PV (D45), essential thrombocythemia (D47.3), and myelofibrosis (D47.4). This change allowed us to more accurately investigate PV using the NHIRD for the first time.
The identification of PV patients was carried out by including cases between 2016 and 2017 with a D45 code and excluding those with a subsequent record of D47.3, which are believed to have been manually added later by physicians to indicate actual essential thrombocythemia cases. After the PV cohort had been identified, we reviewed the retrospective data from 2001 to 2017. We followed the cohort from the first appearance of the ICD code indicating PV until 31 December 2017 or death. Cases without biological sex records were excluded.
The study investigated several demographic and treatment pattern information related to PV patients, including total number of patients, sex, and risk group. The study also examined the age at diagnosis, age at enrollment, duration of disease, history of thrombosis prior to diagnosis, occurrence of thrombosis since diagnosis, and history of bone marrow aspirate/biopsy. Bone marrow aspirate/biopsy for diagnosis was defined as the occurrence of bone marrow aspirate/biopsy procedure codes 3 months before or after the very first ICD code of PV. Comorbidities such as type II diabetes, hypertension, hyperlipidemia, hepatitis B/C, ischemic heart disease, chronic kidney disease, and splenomegaly were included, which were confirmed by two outpatient records in a 365-day period or one inpatient record in the database. Treatment patterns during 2016–2017 were also investigated, such as the annualized frequency of phlebotomy and daily dose of HU received, in addition to treatment duration from 2001 to 2017. Thrombosis was defined by the ICD codes listed in Supplemental A. The reporting of this study conforms to the Strengthening The Reporting of Observational Studies in Epidemiology (STROBE) statement. 3 A STROBE checklist is included as Supplemental B.
Epidemiology definitions
Point prevalence was calculated as the total number of PV cases identified at the end of 2017 divided by the total population of Taiwan at the end of 2017. Incidence rate was calculated as new PV cases identified in a specific year divided by the total population of Taiwan in that specific year. Incidence rate of thrombotic events was calculated as PV cases with new thrombotic event between 2016 and 2017 divided by summed person-years between 2016 and 2017. Crude mortality rate was calculated as total number of deaths in PV patients between 2016 and 2017 divided by the total number of PV patients between 2016 and 2017.
Statistical analysis
Two-sample t tests or Mann–Whitney U tests were used to compare continuous variables between two groups (e.g. male versus female and low-risk versus high-risk). Chi-square tests or Fisher’s exact tests were used to test the association between two categorical variables. One-way analysis of variance was used to test the difference between continuous variables between more than two groups. Log-rank test was conducted to compare the Kaplan–Meier survival curves. A cohort of 10,000 non-PV patients from the same period of time was randomly selected for the comparison of survival probability. Statistical assumptions were fulfilled for all analyses. Differences between PV patients younger than 60 years of age with and without thrombosis, differences between PV patients aged 60 years or over with and without thrombosis, and differences between low-risk and high-risk group were examined. Treatment patterns between male and female patients and between low- and high-risk patients were also examined. SAS version 9.4 (SAS Inc, Cary, NC, USA) was used for analysis, in which a p value of less than 0.05 was considered as statistically significant.
Results
A total of 2647 PV patients were identified between 1 January 2016 and 31 December 2017 (Table 1). The prevalence rate was approximately 10.8 cases per 100,000 people (the denominator being 23,571,227, which was the population in Taiwan in 2017). The incidence rates were estimated to be 2.41 and 2.65 cases per 100,000 people in 2016 and 2017, respectively. The mean ± standard deviation (SD) age at diagnosis was 53.4 ± 15.8 years and at the cross-sectional period was 56.9 ± 16.6 years. There were 1840 (69.5%) male and 807 (30.5%) female patients. The mean age of male patients was 55.5 ± 16.3 years, in contrast to 60.1 ± 16.9 years for female patients. There were 1288 (48.7%) low-risk and 1359 (51.3%) high-risk patients. A Chi-square test of independence showed that there was a significant association between sex and risk group (p < 0.0001). Comorbidities including type II diabetes, hypertension, hyperlipidemia, ischemic heart disease, and chronic kidney disease were generally more common in patients aged 60 years or above. Of the patients below 60 years of age, comorbidities were more prevalent in those with a history of thrombosis. Splenomegaly was noted in 39 (3.0%) low-risk and 51 (3.8%) high-risk patients. The occurrence of thrombosis between 2016 and 2017 was higher in the high-risk group (n = 94, 6.9%) compared with the low-risk group (n = 8, 0.62%) (p < 0.0001). Seventeen patients (0.64%) were found to have progressed to myelofibrosis between 2016 and 2017, of which seven occurred in low-risk patients and ten occurred in high-risk patients. There were 103 deaths (7.6%) in the high-risk group and 21 deaths (1.6%) in the low-risk group. The overall crude mortality rate was 2.3 per 100 persons per year, while the crude mortality rate for patients over 60 years of age was 4.1 per 100 persons per year. The Kaplan–Meier survival curve analysis showed that the PV cohort has significantly lower survival probability than the non-PV cohort (p < 0.0001) (data not shown).
Characteristics of polycythemia vera patients between 2016 and 2017 in Taiwan.

Q1, lower quartile; Q3, higher quartile; SD, standard deviation.
High-risk is defined as aged 60 years or over and history of thrombosis. p values were derived from the Chi-square test, Fisher’s exact test, two-sample t test, and one-way analysis of variance, where applicable. Statistical significance is presented in bold.
Between 2016 and 2017, 647 (24.4%) patients received phlebotomy only, 489 (18.5%) patients received HU treatment only, and 528 (19.9%) patients received both phlebotomy and HU treatment, whereas the remaining 983 (37.1%) had no records of phlebotomy or HU prescription, of which 355 were high-risk patients. Furthermore, the type of treatment differed between sexes (p < 0.0001). Phlebotomy-only treatment was more common in males (29.4%), whereas HU-only treatment was more common in females (28.9%) (Table 2). Both male and female patients receiving only phlebotomy were on average younger than those receiving only HU and those receiving both phlebotomy and HU (Figure 1). Between risk groups, the type of treatment also varied (p < 0.0001). Phlebotomy alone (30.7%) was most common in low-risk patients, whereas HU alone (28.8%) and combination therapy (26.6%) were more common in high-risk patients (Table 2).
Treatment by sexes and by risk stratification presented as number of patients (percentage in the brackets).

Chi-square tests showed significant differences between male and female patients as well as between low- and high-risk patients. Significance was defined as a p value < 0.05.
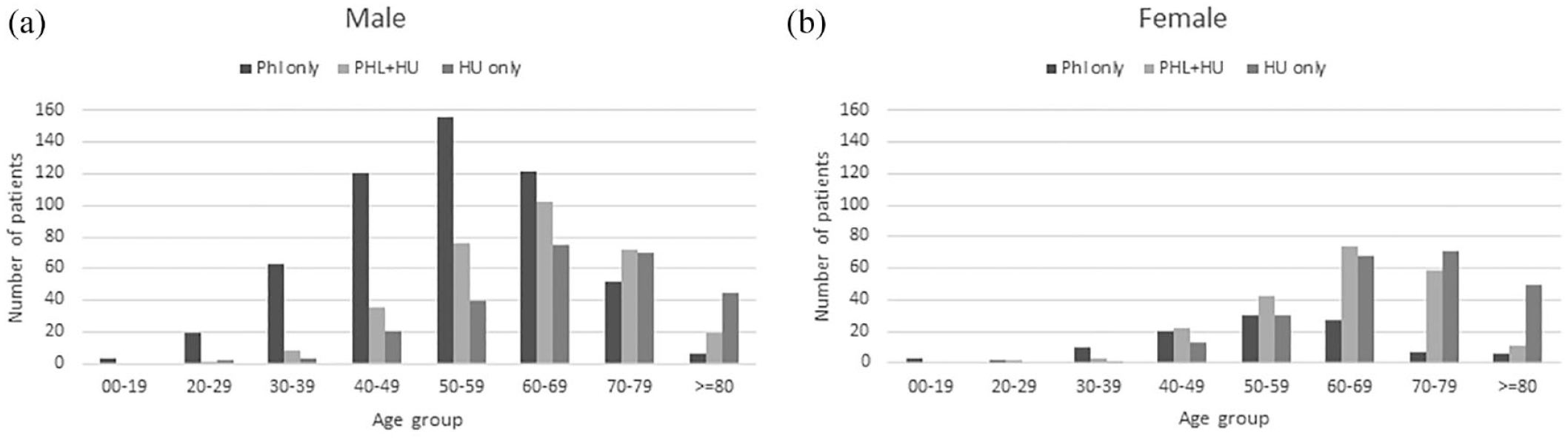
Age distribution of polycythemia vera patients by sex and treatment patterns.
After retrospectively reviewing the medical history of patients from 2001 to 2017, we identified 1504 patients who had previously received phlebotomy (Table 3), with a median duration of 3.4 years. Similarly, we identified 1080 patients who had previously received HU (Table 3), with a median duration of 4.6 years. The maximum durations for phlebotomy and HU were over 16 years. Notably, the duration of HU use was longer in high-risk patients compared with low-risk patients (p < 0.0001).
Treatment patterns in polycythemia vera patients in Taiwan between 2016 and 2017.

%, row percentage of subgroup; HU, hydroxyurea; Q1, lower quartile; Q3, higher quartile; SD, standard deviation.
Years of phlebotomy (n = 1504) or hydroxyurea (n = 1080) treatment in patients with polycythemia vera between 2001 and 2017. Frequency of phlebotomy within a complete 365-day period between 2016 and 2017 in polycythemia vera patients by sex and risk stratification (n = 1162). Average daily dose of hydroxyurea (in milligrams) within a complete 365-day period between 2016 and 2017 in polycythemia vera patients by sex and risk stratification (n = 663). Two-sample t tests were performed for statistical analysis. Statistical significance is presented in bold.
Of the 1162 patients with a complete 365-day period since the first record of phlebotomy between 2016 and 2017 (Table 3), we found that with concurrent HU treatment, male patients received more frequent phlebotomy than did female patients (p = 0.0017).
Of the 663 patients with a complete 365-day period since the first record of HU treatment between 2016 and 2017 (Table 3), we found that male patients received higher daily doses of HU than did female patients (p = 0.012) and that low-risk patients received higher daily doses of HU than did high-risk patients, irrespective of whether phlebotomy was (p = 0.002) or was not (p = 0.02) prescribed.
Discussion
The demographics of the current cohort were compared with the literature. The prevalence rate of PV in Taiwan (10.9 cases per 100,000 people) was higher than that in South Korea (2.8–5.4 cases per 100,000 people),4,5 similar to that in Norway (9.2 cases per 100,000 people), 6 and lower than that in the United States (22–57 cases per 100,000 people).7,8
The age of PV patients at diagnosis in Taiwan was similar to that of other Asian populations but younger than that of Western populations. The mean age at diagnosis (53.4 years) in Taiwan was very similar to that reported in China (54.0 years) 9 and South Korea (53.7 years). 5 Our observed age at diagnosis, however, was younger than the 60.4 years in the European Collaboration on Low-dose Aspirin (ECLAP) study, 10 64.3 years in Germany, 11 71.5 years in Canada, 12 and 73 years in the United Kingdom. 11 A wide age range (54.5–77 years) has been reported in the United States.13–15 An international study that enrolled 1545 patients in Italy, Austria, and the United States reported that the age of patients at diagnosis was 61 years. 16 A New Zealand study compared the age at diagnosis between Caucasian (71.7 years), Ma-ori (59.5 years), and Pacific Islander (56.8 years) patients, suggesting that age differences may exist perhaps due to genetic predisposition. 17 It is also important to consider the trend of population pyramids at the time of patient enrollment, as younger populations may contribute to the mean younger age at diagnosis.
Similar to previous reports, male patients were more prevalent in this study (70% male and 30% female). This difference in the proportion of sexes was greater than those in South Korea (60:40), 4 Japan (57:43), 18 China (54:46), 9 Denmark (56:44), 19 New Zealand (52:48), 17 and the United States (58:42). 20 Note that such a phenomenon was not observed in Norway (50:50). 5 On the contrary, a large cohort study (n = 931) in China found higher proportion of female patients (48:52). The reason for the higher proportion of male patients in this study remains unclear and warrants further investigation. One possible reason could be the miscoding of secondary erythrocytosis as PV, as secondary erythrocytosis is more common in male patients due to factors such as smoking habits. In addition, the age and sex distribution of PV patients were found to be congruent to those previously reported in South Korea, 5 in which male patients (Taiwan: 55.5 years, South Korea: 56.8 years) tended to be younger compared with female patients (Taiwan: 60.1 years, South Korea: 63.8 years).
The overall incidence rate of thrombotic events was lower than that in previous reports. This study estimated that the overall incidence rate of thrombotic events was 2.2%, while the incidence rate of high-risk patients was estimated to be 3.9%. These were lower than those reported in an international study that included PV patients from Austria, Italy, and the United States, where the incidence rates of postdiagnosis arterial and venous thrombosis were 12% and 9%, respectively. 21 The ECLAP study reported an incidence rate of cardiovascular events of 5.5%. 10 Such distinction is unlikely to have arisen due to age, as the mean age in the high-risk group in this study (64.5 ± 10 years) was similar to that in the ECLAP cohort (65.4 ± 12.7 years) and in a previous study (61 years). 21 One possible explanation for the lower observed incidence rate is genetic predisposition, as previous studies have reported lower incidence rates of thromboembolism in Asian populations compared with Caucasian populations.22,23
As expected, age and previous history of thrombosis were predictive of new thrombotic events. Between 2016 and 2017, thrombotic events occurred in 94 patients in the high-risk group but only in 8 patients in the low-risk group. Previous history of thrombosis seemed to play a more important role than old age, as a greater proportion of patients who had a history of thrombosis were at higher risk of new thrombosis (Table 1). The proportion of patients with hypertension and hyperlipidemia, which were previously reported to be predictive of new thrombotic events in PV patients,14,16 may in part explain such differences between the risk groups. As the conventional risk stratification model does not take into account disease progression, however, there seem to be minute differences in our data between low-risk and high-risk groups in terms of myelofibrotic progression and leukemic transformation. In 2022, the National Comprehensive Cancer Network guidelines included the Mutation-Enhanced International Prognostic System for Polycythemia Vera, which incorporates leucocyte count and adverse mutations into consideration. This system is expected to perform better in identifying patients at risk of disease progression.
Splenomegaly has been reported to affect 30–40% of patients with PV, and it is more common in advanced disease. 24 An international study that enrolled 1545 patients in Italy, Austria, and the United States reported 36% of patients with palpable spleen. 21 A single-center study that enrolled 587 patients in the United States also reported 31% of patients with palpable spleen. A lower prevalence of palpable spleen has also been reported, however. A multicenter observational study in the United States with 1601 patients who received spleen palpation at baseline showed that around 17.6% of patients had a palpable spleen. 25 In a multicenter cross-sectional study in Belgium that enrolled 343 patients, palpable spleen was reported in 12.3% of patients. 26 A small study in South Korea that enrolled 125 PV patients reported that only 8% had palpable splenomegaly. 27 This study presented records of splenomegaly in 3.0% of low-risk patients and 3.8% in high-risk patients. Although the lack of routine sonography and motivation to record could be the reasons for the low percentage of splenomegaly in Taiwan, possible genetic factors resulting in differential phenotypes remain an intriguing topic for future study.
The mortality rate in PV patients over 60 years of age was 4.1 per 100 persons per year in this study, which was higher than that of the general population in the same age group (2.8 per 100 persons per year). 28 The mortality rate in PV patients in this study was also similar to that of patients in the ECLAP study (3.7 per 100 persons per year) 29 but lower than that of patients in Norway (8.2 per 100 persons per year). 5
Treatment patterns of phlebotomy and HU were investigated in this study. Phlebotomy treatment was found in 43.7% and 45.1% of low- and high-risk patients, respectively, which were lower than the respective 70.4% and 53.4% in low- and high-risk patients in the REVEAL study. 25 Patients receiving only phlebotomy tended to be younger than those in the other treatment groups. HU treatment was found in 20.5% of low-risk and 55.4% of high-risk patients in this study, which were slightly higher than a US database study identifying that 18.9% of low-risk and 42.0% of high-risk patients received cytoreductive agents (mostly HU) 30 but slightly lower than the REVEAL study, which identified 33.9% of low-risk and 58.2% of high-risk patients on HU treatment. 25 A nationwide survey study in Japan also identified that one-third of PV patients below 60 years of age received HU. 18 Furthermore, this study also found that 13.0% of low-risk and 26.6% of high-risk patients received both phlebotomy and HU, which were similar to those in the REVEAL study (15.8% of low-risk and 26.0% of high-risk patients). 25 Overall, these findings suggest that Taiwanese physicians attempt to defer HU treatment for PV to older patients. Between 18.9% and 33.9% of low-risk patients received HU treatment, however, possibly due to uncontrollable leukocytosis and thrombocytosis.
In terms of the mean daily dose of HU, Taiwanese patients appeared to receive lower doses than Western populations. The Taiwanese patients in this study were prescribed lower doses (phlebotomy + HU: 654.1 mg, HU only: 633.1 mg) than patients in the REVEAL study (phlebotomy + HU: 769.5 mg, HU only: 834.9 mg). 25 A chart review study (n = 1080) reported a mean dose of 984.2 mg in US patients. 31 In addition, this study found that the median daily doses were higher in the low-risk groups compared with the high-risk groups, perhaps due to intolerance and greater comorbidities in older patients.
Our results in treatment pattern suggest that in Taiwan, (a) a large number of patients were passively monitored rather than receiving active treatment, (b) HU treatment was conservatively prescribed by physicians and deferred to older patients due possibly to the potential risks of mutagenicity and leukemogenesis, (c) limited treatment options existed for younger patients to address uncontrollable hematocrit, thrombocytosis, and leukocytosis, which necessitated the use of HU, and (d) patients did not tolerate HU as well compared with Western populations.
We discovered a difference in treatment patterns between sexes in Taiwan. On one hand, a higher proportion of male patients (46.5%) received phlebotomy compared with female patients (39.4%), wherein a distinguishably high proportion of male patients relied solely on phlebotomy only (29.4%) in contrast to female patients (13.0%). Although male patients also received phlebotomy more frequently than did female patients, a significant difference was only found in those concomitantly treated with HU. No difference was found in the duration of phlebotomy treatment. On the other hand, a higher proportion of female patients (55.3%) received HU compared with male patients (31.0%), wherein a distinguishably high proportion of female patients received HU only (28.9%) compared with male patients (13.9%). Female patients, however, received a lower daily dose of HU compared with male patients when they also received concomitant phlebotomy. Note that the duration of HU treatment was longer in female patients.
A concerning finding was the high proportion of patients who did not receive treatment in Taiwan. Approximately 48.8% of low-risk patients and 26.1% of high-risk patients did not receive phlebotomy or HU, compared with only 4.9% of low-risk and 5.7% of high-risk patients reported in the REVEAL study. 25 Another veteran data set study 32 in the United States, however, found that 53% of patients did not receive phlebotomy or HU for treatment. Since the REVEAL study enrolled patients between 2014 and 2016 and the veteran data set study included data between 2005 and 2012, this could indicate a change in treatment behavior from passive to active.
This study had a number of limitations. First, a strict definition of thrombosis was adopted, such that the relevant results presented in this study may have underestimated the occurrence of thrombosis. Second, the absence of laboratory data in the NHI database limited our investigation on the disease and treatment patterns in PV patients in Taiwan. An important laboratory data missing in the database is the results of JAK2V617F mutation testing, which is readily available in clinical practice in Taiwan. Third, we were unable to identify cases of secondary erythrocytosis miscoded as PV, which may have affected our results. As the prevalence of PV cases found in this study is consistent with previous literature, however, the impact of secondary erythrocytosis is expected to be minimal.
Conclusion
This nationwide cross-sectional study provides a snapshot of the real-world clinical landscape of PV in Taiwan. This study reported a number of patient characteristics that were found to be different to the Caucasian populations reported in previous studies, which may provide some evidence that warrants further investigation into the genetics of PV among racial groups. This study also reported several similarities and differences of treatment patterns compared with other countries reported in previous literature.
Supplemental Material
sj-docx-1-tah-10.1177_20406207231179331 – Supplemental material for Real-world patient characteristics and treatment patterns of polycythemia vera in Taiwan between 2016 and 2017: a nationwide cross-sectional study
Supplemental material, sj-docx-1-tah-10.1177_20406207231179331 for Real-world patient characteristics and treatment patterns of polycythemia vera in Taiwan between 2016 and 2017: a nationwide cross-sectional study by Tsung-Hsien Tsai, Lennex Hsueh-Lin Yu, Ming-Sun Yu, Shih-Hao Huang, Alex Jia-Hong Lin, Kuan-Der Lee and Min-Chi Chen in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207231179331 – Supplemental material for Real-world patient characteristics and treatment patterns of polycythemia vera in Taiwan between 2016 and 2017: a nationwide cross-sectional study
Supplemental material, sj-docx-2-tah-10.1177_20406207231179331 for Real-world patient characteristics and treatment patterns of polycythemia vera in Taiwan between 2016 and 2017: a nationwide cross-sectional study by Tsung-Hsien Tsai, Lennex Hsueh-Lin Yu, Ming-Sun Yu, Shih-Hao Huang, Alex Jia-Hong Lin, Kuan-Der Lee and Min-Chi Chen in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
Access to the National Health Insurance Research Database was made possible by the Services Center for Health Information, Chang Gung University. The authors thank Dr Justin L. Chen for proofreading this work. K.-D.L. and M.-C.C. contributed equally to this work.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
