Abstract
Background:
The economic burden of blood transfusions (BTs) in transfusion-dependent β-thalassemia (TDT) is not well characterized in adults from Brazil, Thailand, and India.
Objective:
To assess direct and indirect costs of BTs in adults with TDT in three geographically distinct countries: Brazil, Thailand, and India.
Design:
Healthcare professionals (HCPs) and administrative personnel completed a cross-sectional survey to assess transfusion-associated direct costs and indirect costs among patients with TDT.
Methods:
Direct (blood collection, BTs, iron chelation therapy (ICT), transfusion-related adverse events (AEs)), and indirect costs (blood supply shortages, waiting time, patient, and caregiver time) per patient per year (PPPY) were calculated from survey data, and publicly available cost data.
Results:
Between February 2, 2024, and March 12, 2024, 54, 104, and 125 participants in Brazil, Thailand, and India, respectively, were included, mostly hematologists or pharmacists. Median number of BTs PPPY was 12 (Brazil), 6 (Thailand), and 20 (India). Physicians and nurses conducted pretransfusion monitoring, whereas nurses monitored during BTs and posttransfusion appointments. AEs were estimated in 15% (Brazil), 5% (Thailand), and 10% (India) of BTs. In the past 12 months, 72%, 50%, and 54% of participants experienced blood supply shortages in Brazil, Thailand, and India, respectively; 40%, 25%, and 70% of patients experienced iron overload due to BTs, and 30%, 20%, and 70% of patients received ICT due to BTs. Total direct costs PPPY (USD) were 4438 in Brazil, 1775 in Thailand, and 1991 in India; total indirect costs PPPY (USD) were 831 in Brazil, 392 in Thailand, and 715 in India.
Conclusion:
Results showed a significant burden of BTs in TDT on HCPs, patients, and caregivers, along with shortages and delays in blood supplies. These findings underscore the importance of enhancing supportive care for BTs and exploring alternative approaches to alleviate this burden.
Keywords
Introduction
β-Thalassemia is an inherited blood disorder, characterized by chronic anemia. 1 Patients with transfusion-dependent β-thalassemia (TDT) need lifelong regular red blood cell (RBC) transfusions to manage anemia and prevent potential complications of ineffective erythropoiesis. 1 RBC transfusions also have a significant clinical, health-related quality of life, and economic burden on patients with TDT.2,3 For example, frequent RBC transfusions are associated with challenges such as iron overload, transfusion-transmitted infections, transfusion reactions, and antibody formation (alloimmunization). 4 Typically, within a year of receiving regular RBC transfusions, patients require iron chelation therapy (ICT) to prevent excess iron accumulation. 4 Regular RBC transfusions can also impose a significant burden on the healthcare system, as numerous resources are required for each step of the transfusion process, such as personnel, blood supply, and/or equipment for blood donations, transfusions, and posttransfusion appointments. Previous studies have shown that patients with TDT need an annual average of 17 RBC transfusions.5,6 As a result, RBC transfusions and ICT are the major contributors to the costs of TDT management.1,7 Although efforts have been made to improve the collection, preparation, testing, storage, transport, and administration of blood and blood components, inadequate transfusion therapy is reported frequently. 8
The burden of β-thalassemia on patients and healthcare systems is high, and globally, the World Health Organization has estimated that 40,000 infants are born with β-thalassemia annually, of which 25,500 are TDT. 9 A higher prevalence of β-thalassemia has been observed in certain regions, including the Mediterranean, the Middle East, and Asia, but is increasingly more common in other regions due to changing migration patterns. 10 Thailand has one of the highest rates of β-thalassemia worldwide, with 157.11 cases per 100,000 persons in 2021, and Brazil has experienced substantial increases in prevalence between 1990 and 2021. 11 In India, 1.25%–1.66% of the population, or around 20 million individuals, are estimated to be affected by β-thalassemia. 10 Despite the high prevalence worldwide, only about 12% of children born with TDT receive RBC transfusions, of whom fewer than 40% receive adequate ICT. 9 Access to care varies regionally: 52% of patients with TDT in the Americas receive RBC transfusions (58% of whom receive ICT), compared to 10% of patients with TDT in Southeast Asia (19% of whom receive adequate ICT). 9
A pragmatic literature review (PLR) revealed a need for further studies to understand healthcare resource utilization (HCRU) and costs associated with blood transfusions and ICT for adult patients with TDT. 12 Few studies evaluating costs of RBC transfusions in patients with TDT have been conducted in various countries, including (but not limited to) the United States and European countries.7,13–19 Costs of TDT management vary widely around the world; for instance, the yearly costs of TDT management are reportedly 130,000 US dollars (USD),5,6 while lifetime costs range from 360,000 USD in Taiwan to 600,000 USD in Malaysia.14,17 Information on TDT management and transfusion burden from other countries, such as Brazil, India, and Thailand, remains especially limited and economic data are lacking, highlighting the need for additional research. Factors contributing to costs can also differ per country; for example, patients in resource-deprived countries may encounter blood supply shortages or lack of access to regular and safe transfusions. 4 Moreover, existing studies have focused mainly on pediatric populations, leaving a gap in research estimating transfusion costs in adults.15,20–22 The current cross sectional survey and cost-modeling study aimed to assess the direct and indirect costs of blood transfusions and variation in burden, in adult patients with TDT in three geographically distinct countries, where data are currently lacking: Brazil, Thailand, and India.
Methods
Pragmatic literature review
A PLR was conducted in Thailand and Brazil (November 12, 2022) and India (June 29, 2023) to investigate the economic burden of TDT in these countries over a 10-year period. 12 The PLR used the PICOS (Patient, Intervention, Comparison, Outcomes, Study)-based search strategy to identify observational studies for adults with TDT published in English over the past 10 years from Embase®, MEDLINE®, evidence-based medicine (EBM) Reviews-Health Technology Assessment (HTA), and EconLit. Blood transfusions and ICT were consistently shown to be the standard of care for patients with TDT in these countries; however, inadequate transfusions or transfusion delays caused by blood supply shortages were commonly reported, highlighting the need for improving blood transfusion support and providing alternative solutions. The overall finding of a lack of literature to adequately understand HCRU and costs associated with blood transfusions provides clear rationale for conducting a full survey.
Study design
The cross-sectional survey was conducted among healthcare professionals (HCPs) and administrative personnel in Brazil, Thailand, and India, in conjunction with desk research to determine the direct and indirect costs associated with blood transfusions among patients with TDT. Key topics of the survey comprised clinical pathways, frequency of transfusions, blood supply shortages, and economic burden. 12 To ensure appropriateness of the questionnaire, one key expert per country reviewed the inclusion and exclusion criteria for selecting study respondents and provided clinical insights on the survey questions (e.g., a list of most common adverse events (AEs) related to transfusion). A pilot interview with 1 respondent per country was conducted with a moderator to ensure correct understanding of the survey. Specific instructions were given at the start of the survey, and to reduce bias in the survey results, survey respondents were asked to consider the previous 12-month period for their responses. The survey period was from February 2, 2024 to March 12, 2024. The objectives were to quantify the direct costs (primary objective) and the indirect costs (secondary objective) associated with blood transfusions among patients with TDT in each of the three countries. Direct costs included costs for blood collection, transfusion therapy, ICT, and transfusion-related AEs; indirect costs included costs for blood supply shortages, waiting time, and patient and caregiver time. Cost estimates were calculated using data derived from a prior PLR and respondent-reported frequencies of events from the survey. Further information can be found in Supplemental Material A.
STROBE statement
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 23 A STROBE checklist is included as Supplemental Material B.
Survey population
The survey was administered to eligible study participants; eligibility criteria had been verified through discussions with a local key opinion leader. Eligible participants had provided consent to having data processed as part of an anonymized dataset. The survey participants were HCPs (hematologists, hospital pharmacists, heads of pharmacy, and hematology unit nurses where available) and administrative personnel (hospital procurement managers, and blood unit administrators) in Brazil, Thailand, and India. All participants in Brazil worked in institutions that were part of the public healthcare system. Participants included in the study had 3–35 years of experience working in a hematology clinic or a hospital containing a hematology clinic. Additionally, hematologists and hematology unit nurses were included if they spent at least 60% of their time in direct patient care, primarily with patients with TDT, and were involved in managing at least five unique β-thalassemia or at least three patients with TDT in the last year. Hospital pharmacists, heads of pharmacy, hospital procurement managers, and blood unit administrators were included if they worked in a center that conducts at least three blood transfusions annually, or were responsible for monitoring the costs, and/or commissioning, of at least one (for hospital pharmacists, heads of pharmacy), two (for hospital procurement managers), or four items (for blood unit administrators) from the following: blood transportation, RBC unit procurement and storage, hemovigilance related to blood transfusions, transfusion, posttransfusion monitoring, drug acquisition, blood collection procedures, hemovigilance related to blood collection, or blood component separation. In addition, general practitioners responsible for conducting/monitoring blood transfusions were included in India. The survey was conducted in accordance with the regulatory and ethical requirements of each country.
HCPs were excluded if they or someone in their immediate family were employed and/or paid by a pharmaceutical or biopharmaceutical company or a healthcare manufacturer as a consultant or researcher (other than for participation in clinical trials), or if they had any other conflicts of interest with the survey (i.e., those employed or contracted by regulatory bodies or agencies deciding on reimbursements of medication).
Survey instrument
A questionnaire containing a series of multiple-choice questions intended to capture resource use was employed to quantify direct and indirect costs of transfusion (Supplemental Material A). The questionnaire was administered in an online survey, and each respondent completed the survey once. Reported median and mean values were calculated based on responses provided by the survey participants, in response to questions that asked them to estimate how often a given event occurred (unless otherwise specified).
Calculation of costs
Micro-costing of different elements of the transfusion pathway for patients with TDT was performed in accordance with the methods reported previously, which involves measuring costs of all individual components of resource use.24,25 Direct and indirect costs were derived using information collected from a prior PLR, and the median and mean values from survey responses were used as inputs for cost calculations. For Brazil, direct costs were calculated using data from the public healthcare system. Cost-analysis models for direct and indirect costs were developed separately, each of which combined the costs for blood collection, blood transfusions, and ICT, and applied to frequencies of events derived from the survey. Costs related to HCP time were calculated by multiplying time required by the average salary statistics for their role. The cost of donor, patient, or caregiver time spent was calculated by multiplying time required by the average salary statistics for working-age adults to calculate the salary loss incurred. The type and frequency of other healthcare inputs use (e.g., tests, blood bags, ICT drugs) or percentages/proportions reflecting demand for healthcare (e.g., proportion of patients with TDT receiving ICT) were used to provide granularity on the cost, where possible. The overall cost of AEs was estimated by multiplying the frequency of AEs by the time and cost associated with the AE. Country-specific cost units related to HCRU were used to quantify the time per episode of care associated with an AE, which was then multiplied by the frequency of the AE. Details of the calculations used to estimate costs are described further in Supplemental Material A.
All costs were expressed in the local currency and reported here in USD using the December 31, 2023, exchange rate and estimated per patient per year (PPPY), to enable comparisons of costs across countries. The exchange rates were 34.35 Thai baht = 1 USD, 26 4.85 Brazilian real = 1 USD, 27 and 83.20 Indian rupee = 1 USD. 28 For Brazil and India, the costs refer solely to the public sector; for Thailand, most centers were from the public sector.
Statistical analysis
All analyses were descriptive in nature, and no statistical comparisons or imputation of missing data were performed. To mitigate any issues caused by potential outliers, median values were reported (i.e., the middle value across all survey responses received for each question) throughout, along with interquartile ranges (IQRs). However, some questions asked respondents to estimate percentage distributions among prespecified options, where the sum was required to be 100%; here, mean values and standard deviations were reported. Based on a previous study, the target sample size for HCP respondents was about 55 in Brazil, 90–100 in Thailand, and 105–125 in India. 24 As part of data collection, the responses of each respondent type were reviewed to ensure that they were plausible.
Results
A total of 54 participants in Brazil, 104 in Thailand, and 125 in India were included in the cross-sectional study. The majority were hematologists or pharmacists across all countries; approximately 30%–50% were hematologists, and 40%–50% were pharmacists (Supplemental Table 1). All median and mean values reported below are estimates based on responses provided by the survey participants, in response to questions that asked them to estimate how often a given event occurred (unless otherwise specified).
Blood transfusion burden
The median number of blood transfusions PPPY was 12 in Brazil, 6 in Thailand, and 20 in India (Table 1). The median volume of packed red blood cells (pRBC) per transfusion was 250 mL in Brazil, 200 mL in Thailand, and 300 mL in India. In Brazil, 56% of participants and 87% in Thailand considered the 200 mL pRBC as the typical size for blood transfusions; in India, 46% of participants considered the 400 mL pRBC as the typical size (Table 1). Transfusions occurred mostly in the outpatient setting and blood banks in Brazil, whereas they occurred predominantly in inpatient or outpatient settings in Thailand and India (Table 1). The most common source for pRBC was internal, that is, from the blood banks of the hematology unit where the survey participant worked. Other sources were external (outside the hematology unit of survey participant) and funded by the government or by not-for-profit organizations across the three countries (Table 1).
Number of blood transfusions PPPY, and type, size, and costs of pRBC units in local currency.

Please note that the denominator for the survey responses may not equal the total number of participants as some questions were specific to respondent profile, or in some cases, respondents selected a response that would exclude them from answering a subsequent question.
IQR, interquartile range; N, number of respondents in total per country; n, number of respondents who provided an answer to the given question; PPPY, per person per year; pRBC, packed red blood cells; SD, standard deviation; USD, US dollars.
Pretransfusion tests and AEs due to blood transfusion
ABO blood group, Rhesus D, and crossmatching were the most frequently conducted pretransfusion tests in all countries. Pretransfusion monitoring was typically carried out by physicians and nurses, while nurses performed monitoring during and posttransfusions in all countries (Table 2). AEs occurred during a median of 15% of blood transfusions in Brazil, 5% in Thailand, and 10% in India. About a quarter of the transfusion-related AEs were allergic reactions (Table 3). Median hospitalization rates due to transfusion-related AEs were 5% in Brazil and 10% each in Thailand and India, with a median inpatient length of stay of 2–3 days (Table 3).
HCP time spent pre-, during, and posttransfusion.

Please note that the denominator for the survey responses may not equal the total number of participants as some questions were specific to respondent type, or in some cases, respondents selected a response that would exclude them from answering a subsequent question.
GP, general practitioner; HCP, healthcare professional; IQR, interquartile range; N, number of respondents in total per country; n, number of respondents who provided an answer to the given question.
Blood transfusions leading to AEs.
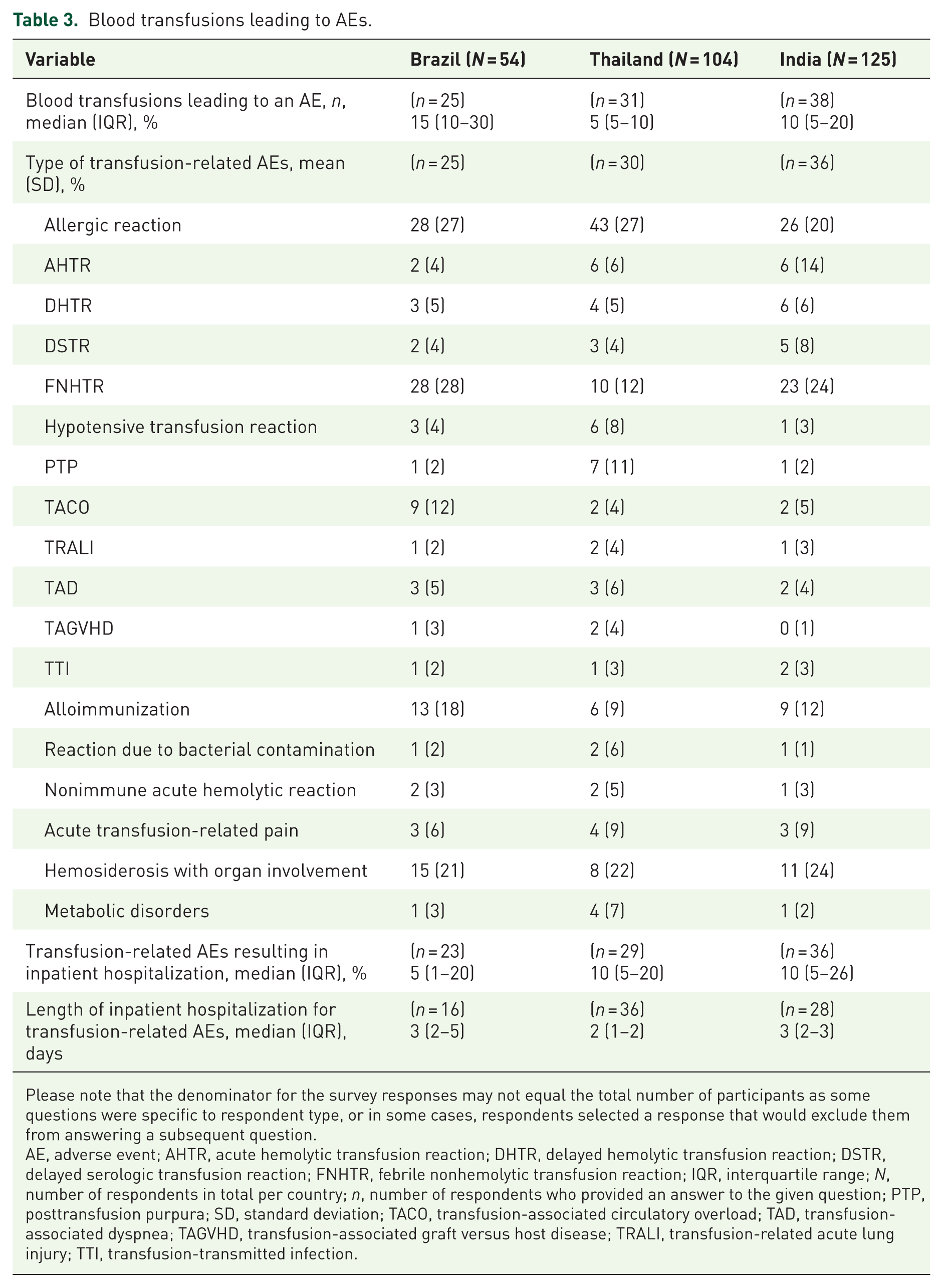
Please note that the denominator for the survey responses may not equal the total number of participants as some questions were specific to respondent type, or in some cases, respondents selected a response that would exclude them from answering a subsequent question.
AE, adverse event; AHTR, acute hemolytic transfusion reaction; DHTR, delayed hemolytic transfusion reaction; DSTR, delayed serologic transfusion reaction; FNHTR, febrile nonhemolytic transfusion reaction; IQR, interquartile range; N, number of respondents in total per country; n, number of respondents who provided an answer to the given question; PTP, posttransfusion purpura; SD, standard deviation; TACO, transfusion-associated circulatory overload; TAD, transfusion-associated dyspnea; TAGVHD, transfusion-associated graft versus host disease; TRALI, transfusion-related acute lung injury; TTI, transfusion-transmitted infection.
Blood supply shortages
Over 70% of respondents in all three countries reported blood supply shortages due to fewer pRBC units received; other reported issues included reduced availability of blood phenotypes and shipment delays (Table 4). Seventy-two percent of participants in Brazil, 50% in Thailand, and 54% in India experienced delay in receiving pRBC in the past 12 months (Table 4). Overall, a median of 18% of blood transfusions in Brazil, 10% in Thailand, and 5% in India, were affected by blood supply shortages in the past 12 months (Table 4). Blood transfusions were delayed due to blood supply shortages related to rare antibody or phenotype by a median of 3–5 days, and were delayed due to general blood supply shortages by a median of 2–3 days (Table 4). A median of 27% blood transfusions in Brazil, 8% in Thailand, and 31% in India impacted by blood supply shortages were not postponed in advance, requiring patients to schedule another visit (Table 4).
Blood supply shortages.

Please note that the denominator for the survey responses may not equal the total number of participants as some questions were specific to respondent type, or in some cases, respondents selected a response that would exclude them from answering a subsequent question.
IQR, interquartile range; N, number of respondents in total per country; n, number of respondents who provided an answer to the given question; pRBC, packed red blood cells.
Blood collection burden
A median of 400 mL of blood in Brazil, 250 mL in Thailand, and 350 mL in India was given per donation (Supplemental Table 2). Donors were reported to spend a median of 1 h per donation in Brazil and India, and 2 h per donation in Thailand (Supplemental Table 2). Most respondents indicated that nurses were largely involved in performing blood collections in all three countries (Supplemental Table 2). A median of 4% (Brazil), 5% (Thailand), and 4% (India) blood donations resulted in an AE (Supplemental Table 2); the most frequent AEs were hypotension, fainting, HIV, and hepatitis B or C (data not shown).
Monitoring of iron levels and ICT usage
A median of 100% of patients in Brazil and India, and 15% of patients in Thailand were assessed for iron overload over a period of 12 months, with the most frequently used test being serum ferritin (Table 5). A median of 40% of patients in Brazil, 25% in Thailand, and 70% in India were reported to have iron overload in the past 12 months, and a median of 30% of patients in Brazil, 20% in Thailand, and 70% in India received ICT (Table 5). The most frequently received ICT in Brazil (73%) and India (51%) was deferasirox monotherapy; in Thailand, it was deferiprone monotherapy (29%) (data not shown).
ICT patterns, administration, and assessment.
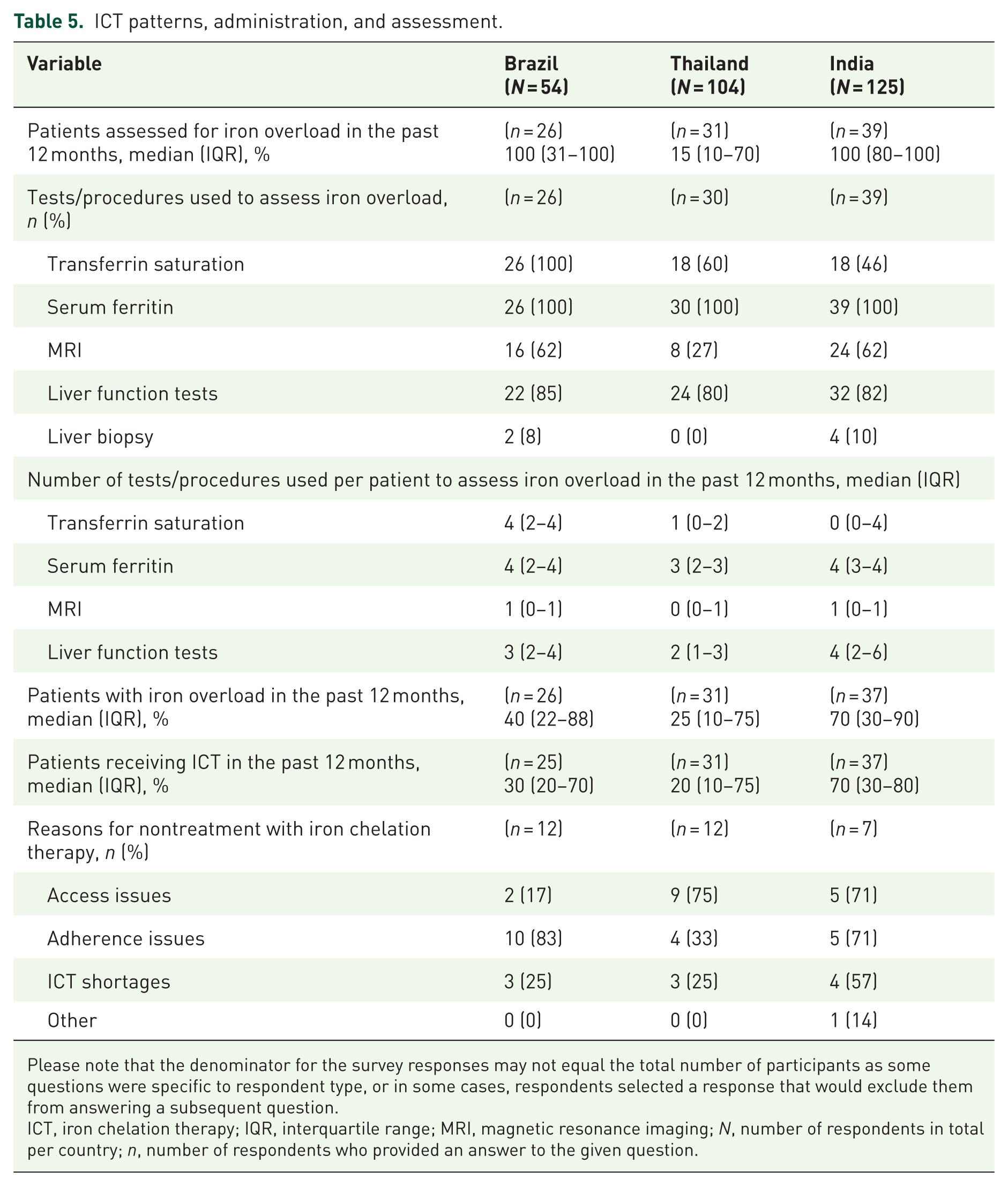
Please note that the denominator for the survey responses may not equal the total number of participants as some questions were specific to respondent type, or in some cases, respondents selected a response that would exclude them from answering a subsequent question.
ICT, iron chelation therapy; IQR, interquartile range; MRI, magnetic resonance imaging; N, number of respondents in total per country; n, number of respondents who provided an answer to the given question.
Indirect burden related to wastage of blood, and patient and caregiver time
A median of 7% of pRBC units in Brazil, 8% in Thailand, and 2% of in India was wasted per month (Supplemental Table 3). Patients spent a median of 1–2 h traveling per transfusion, and at transfusion centers spent a median of 2–3 h pre-transfusion, 1.5–4 h during the transfusion, and 30–60 min posttransfusion (Supplemental Table 3). Additional pre-transfusion visits were required in a median of 65% of patients in India to 90% in Thailand, and additional post-transfusions visits were needed in a median of 20% of patients in Brazil to 91% in Thailand (Supplemental Table 3). Survey participants mentioned that a median of 60% of patients in Brazil, 45% in Thailand, and 90% in India were accompanied by a caregiver (Supplemental Table 3). In Brazil and India, the majority of patients reportedly traveled by public transport, whereas most traveled by car in Thailand.
Direct and indirect costs
The total direct costs PPPY were 4438 USD in Brazil, 1775 USD in Thailand, and 1991 USD in India (Table 6). In Brazil and India, the primary driver for direct costs was acquisition of ICT rather than transfusion, accounting for 61% and 58% of direct costs, respectively, whereas in Thailand, ICT accounted for 41% of direct costs. Transfusion costs were 1742 USD in Brazil, 1048 USD in Thailand, and 829 USD in India, and ICT costs were 2695 USD in Brazil, 729 USD in Thailand, and 1164 USD in India. The total indirect costs were 831 USD, 392 USD, and 715 USD in Brazil, Thailand, and India, respectively (Table 6).
Total direct and indirect costs.

Weighted by source and wastage.
Adjusted for wastage.
AE, adverse event; HCP, healthcare professional; ICT, iron chelation therapy; N, number of respondents in total per country; PPPY, per patient per year; USD, United States dollar.
Discussion
The present survey revealed a considerable burden of blood transfusions on patients with TDT, HCPs, and caregivers in Brazil, Thailand, and India. Patients received a median of 6–20 blood transfusions per year, 45%–90% were accompanied by caregivers, and patients were often required to make additional pre- and posttransfusion visits. Blood supply shortages, primarily due to fewer pRBC units received, were reported by over 70% of respondents in all three countries. The direct costs ranged from 1775 USD in Thailand up to 4438 USD in Brazil, with ICT being the primary driver of these costs in Brazil and India, and transfusion being the primary driver of costs in Thailand, while indirect costs ranged between 392 USD in Thailand to 831 USD in Brazil. Collectively, these findings highlight the need for improvement of services related to blood transfusions for patients with TDT across three distinct countries. The prevalence of β-thalassemia is already known to be high in regions such as Asia or the Mediterranean, but the results of this study shed light on specific factors contributing to the burden of TDT treatment, especially in regions/countries, where the disease burden has not been captured as thoroughly, such as Brazil. 10
The cost of treatment extends beyond the actual costs of a pRBC transfusion, involving multiple procedural steps and administration costs as well as the time commitment required by patients and caregivers. In this study, patients spent 5–6 h per blood transfusion plus 1–2 h travel time, and around half were accompanied by caregivers. This situation can disrupt patients’ and caregivers’ work routines and daily lives, and negatively impact their quality of life, leading to high indirect costs especially in developing nations. 29 As of 2024, the countries of interest in this study are all considered emerging markets and developing economies, with the International Monetary Fund estimating the gross domestic product (GDP) per capita as 10,300 USD in Brazil, 7530 USD in Thailand, and 2700 USD in India. 30 The estimated 2023 average salary PPPY for each is 7086 USD in Brazil, 5837 USD in Thailand, and 2853 USD in India.26–28,31 In our results, the annual total direct and indirect costs of TDT β-thalassemia were higher in India than Thailand; that India has roughly half the annual GDP and salary values of Thailand suggests that India may experience a relatively higher burden from this disease. Considering the total direct and indirect costs identified in this study in the context of the annual GDP and average salary data highlight the significant economic burden of TDT β-thalassemia in these countries. While previous studies have focused primarily on the challenges faced by children with β-thalassemia and their parents, the findings of this study indicate that adult patients and caregivers encounter similar difficulties. In terms of cost, a study of the economic burden of β-thalassemia in Thai children 2–18 years of age at three hospitals calculated mean annual total direct costs of 562.76 USD and indirect costs of 229.60 USD (both at 2005 prices). 15 While these values are lower than those for adults identified in our study, direct comparisons cannot be made, as increases in costs over time may account for some degree of the lower costs in the pediatric study.
More than half of the participants in all three countries reported blood supply shortages in the past 12 months, attributed to inadequate blood supply (fewer pRBC or unavailability of blood phenotype). As patients wait to receive a blood transfusion, their quality of life can be compromised due to long waiting times and worsening of disease symptoms. A study conducted in 42 countries by the Thalassemia International Federation found that the COVID-19 pandemic led to long-term shortages in blood supply worldwide, resulting in moderate-to-severe drops in hemoglobin levels of patients with β-thalassemia, particularly in developing countries. 32 The findings confirm that blood supply shortages remain challenging, especially in countries where infrastructure for maintaining blood supply may be limited.
ICT is vital for managing iron overload resulting from blood transfusions, but less than 40% of patients receive adequate ICT, and access to ICT is known to vary per region. 4 In the present study, 70% of patients in India received ICT, similar to a previous study where 67% of eligible patients with TDT in Western India received ICT. 33 In Thailand, 15% of patients were assessed for iron overload and 20% of patients received ICT. Southeast Asia has previously reported high mortality rates due to insufficient ICT, and the region has accounted for nearly 50% of deaths per year due to iron overload. 9 Despite the availability of cost-effective forms of ICT such as deferiprone, the costs of ICT still contributes to one-third of treatment costs per patient in Thailand. 10 Significant variation was observed in patients being assessed for iron overload between countries which may reflect different practices across different geographic locations and highlights the difficulty in generalizing results to other countries.
The overall economic burden associated with blood transfusions in TDT from both healthcare and societal perspectives described in prior studies limits direct comparisons with the current study. The cost of treatment can vary widely per country; in the United States, the yearly cost of managing TDT has been reported as 130,000 USD.5,6 In comparison, lifetime costs of TDT management can range from to 360,000 USD in Taiwan 17 to 600,000 USD in Malaysia; 14 GDP estimates also vary between these countries, with 89,680 USD in the United States, 14,420 USD in Malaysia, and 39,320 in Taiwan in 2024. 30 Nevertheless, the total transfusion-related costs and ICT acquisition costs from the current study appear to be consistent with previous studies on costs in India and Thailand.15,20 ICT accounted for a major share of the direct costs of blood transfusions in all three countries, also consistent with previous literature. 34
These findings highlight the necessity for the development of alternative treatment approaches, to reduce the burden of blood transfusions. Given that patients with TDT have high HCRU and costs, there is an unmet need for effective, tolerable, and affordable novel treatments that can minimize the clinical and economic impact of TDT on healthcare systems. Stem cell transplantation is currently the only curative treatment for patients with TDT, and its utility is limited to the availability of matching donors. 1 Researchers are exploring alternative treatments that target correction of globin chain imbalance, ineffective erythropoiesis, or enhancement of the body’s iron-handling mechanisms. 35
Study limitations and strengths
The findings of the current study should be interpreted taking into account some limitations. The study is purely descriptive, lacking advanced analytical methods or covariate adjustments. The number of survey responses may not equal the total number of participants from each country as some questions were specific to respondent type, or in some cases, respondents selected a response that would exclude them from answering a subsequent question. The present data were solely reliant on HCP knowledge of blood transfusion processes. For example, in Thailand, half of the HCPs who responded in this survey were pharmacists who do not associate directly with patients with TDT. Variability in the results due to misunderstanding of the question could not be ruled out. While HCRU costs were derived from the survey results combined with prior desk research, cost estimates related to blood transfusions relied mostly on the estimates provided by respondents. Consequently, costs might have potentially been underestimated due to survey-derived responses, assumptions about procedure prices, and omission of certain expenses like ambulance callouts and long-term effects. Generalizability to the country level and across other countries is limited. Some data may be missing, requiring cautious interpretation.
Despite these limitations, our study provided detailed insights into direct and indirect costs for Brazil, Thailand, and India, information that was so far lacking in the literature. We quantitatively described the burden on patients, caregivers, and HCPs in clinical practice, considering their time and travel commitments. Our findings, together with existing literature, show a considerable economic burden associated with blood transfusions and emphasize an urgent need for alternative treatment approaches to reduce blood transfusion requirements.
Recommendations and future direction
Given the considerable economic burden associated with blood transfusions, there is an urgent need for alternative treatment approaches to reduce blood transfusion requirements. In the meantime, there is also a need to improve services related to blood transfusions, including ensuring adequate blood supply, for patients with TDT in the countries studied.
Conclusion
The findings of this study show that there is a significant burden of blood transfusions in patients with TDT, HCPs, and caregivers, in three geographically distinct countries: Brazil, Thailand, and India. The study provided insight into the factors contributing to the burden of blood transfusions as well as the management of TDT in clinical practice. The direct and indirect transfusion-related costs were substantial across all three countries, with direct transfusion costs ranging from 829 USD PPPY in India to 1742 USD PPPY in Brazil, and one of the primary drivers of overall direct costs was the acquisition of ICT. These findings highlight the importance of improving services related to blood transfusions and to explore alternative approaches to alleviate transfusion-related burden.
Supplemental Material
sj-docx-1-tah-10.1177_20406207261416795 – Supplemental material for Direct and indirect medical costs associated with transfusion-dependent beta-thalassemia in Brazil, Thailand, and India
Supplemental material, sj-docx-1-tah-10.1177_20406207261416795 for Direct and indirect medical costs associated with transfusion-dependent beta-thalassemia in Brazil, Thailand, and India by Tulika Seth, Adisak Tantiworawit, Sandra R. Loggetto, Mrudula B. Glassberg, Pimwadee Khaikham, Paola Marinheiro, Jason Williams, Camila F. Roubik, Christopher Lee, Nelly F. Ly, Pooja Hindocha, Anita Mallya, Dorothea von Bredow, Yogesh Punekar and Ahmed Hnoosh in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207261416795 – Supplemental material for Direct and indirect medical costs associated with transfusion-dependent beta-thalassemia in Brazil, Thailand, and India
Supplemental material, sj-docx-2-tah-10.1177_20406207261416795 for Direct and indirect medical costs associated with transfusion-dependent beta-thalassemia in Brazil, Thailand, and India by Tulika Seth, Adisak Tantiworawit, Sandra R. Loggetto, Mrudula B. Glassberg, Pimwadee Khaikham, Paola Marinheiro, Jason Williams, Camila F. Roubik, Christopher Lee, Nelly F. Ly, Pooja Hindocha, Anita Mallya, Dorothea von Bredow, Yogesh Punekar and Ahmed Hnoosh in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-3-tah-10.1177_20406207261416795 – Supplemental material for Direct and indirect medical costs associated with transfusion-dependent beta-thalassemia in Brazil, Thailand, and India
Supplemental material, sj-docx-3-tah-10.1177_20406207261416795 for Direct and indirect medical costs associated with transfusion-dependent beta-thalassemia in Brazil, Thailand, and India by Tulika Seth, Adisak Tantiworawit, Sandra R. Loggetto, Mrudula B. Glassberg, Pimwadee Khaikham, Paola Marinheiro, Jason Williams, Camila F. Roubik, Christopher Lee, Nelly F. Ly, Pooja Hindocha, Anita Mallya, Dorothea von Bredow, Yogesh Punekar and Ahmed Hnoosh in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-4-tah-10.1177_20406207261416795 – Supplemental material for Direct and indirect medical costs associated with transfusion-dependent beta-thalassemia in Brazil, Thailand, and India
Supplemental material, sj-docx-4-tah-10.1177_20406207261416795 for Direct and indirect medical costs associated with transfusion-dependent beta-thalassemia in Brazil, Thailand, and India by Tulika Seth, Adisak Tantiworawit, Sandra R. Loggetto, Mrudula B. Glassberg, Pimwadee Khaikham, Paola Marinheiro, Jason Williams, Camila F. Roubik, Christopher Lee, Nelly F. Ly, Pooja Hindocha, Anita Mallya, Dorothea von Bredow, Yogesh Punekar and Ahmed Hnoosh in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-5-tah-10.1177_20406207261416795 – Supplemental material for Direct and indirect medical costs associated with transfusion-dependent beta-thalassemia in Brazil, Thailand, and India
Supplemental material, sj-docx-5-tah-10.1177_20406207261416795 for Direct and indirect medical costs associated with transfusion-dependent beta-thalassemia in Brazil, Thailand, and India by Tulika Seth, Adisak Tantiworawit, Sandra R. Loggetto, Mrudula B. Glassberg, Pimwadee Khaikham, Paola Marinheiro, Jason Williams, Camila F. Roubik, Christopher Lee, Nelly F. Ly, Pooja Hindocha, Anita Mallya, Dorothea von Bredow, Yogesh Punekar and Ahmed Hnoosh in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
Support for medical writing and editing were provided by Leena Patel, PhD, Chrysi Petraki, PhD, Jessica Warren, BS, of IQVIA; and Shruti Ullas, PhD, and Emma Rathbone, PhD, of Excerpta Medica, funded by Bristol Myers Squibb.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
