Abstract
Background:
Recombinant porcine factor VIII (rpFVIII; susoctocog alfa) is indicated for the treatment of bleeding events (BEs) in adults with acquired hemophilia A (AHA).
Objectives:
To assess the safety, utilization, and effectiveness of rpFVIII in clinical practice.
Design:
EU post-authorization safety study (PASS) (NCT03199794) was a multicenter, noninterventional, post-authorization safety study conducted in adults with AHA.
Methods:
Data were collected retrospectively or prospectively for up to 180 days after the last rpFVIII dose. The primary objective was safety, as assessed by adverse events (AEs), serious AEs (SAEs), and AEs of special interest (AESIs) (e.g. immunogenicity, hypersensitivity reactions, thromboembolic events). Secondary endpoints included immunogenicity, rpFVIII hemostatic effectiveness, and rpFVIII utilization.
Results:
Fifty patients were enrolled; 31 completed the study. The median (range) follow-up for patients who completed or discontinued the study was 178 (26–371) days. The median (range) first dose of rpFVIII was 54.0 (11–200) U/kg. Thirty patients reported 46 SAEs; 5 SAEs were considered probably related to rpFVIII, of which 1 was lack of rpFVIII efficacy, and 4 were AESIs: drug resistance due to FVIII inhibition (one patient), antibody test positive for anti-pFVIII inhibitors (one patient), and
Conclusion:
Results from this real-world study support the use of rpFVIII for AHA, aligning with findings from the clinical trial of rpFVIII (NCT01178294) in the treatment of BEs in adults with AHA.
Trial registration:
EUPAS16055; NCT03199794.
Introduction
Acquired hemophilia A (AHA) is a rare autoimmune disease characterized by inhibitory autoantibodies against endogenous factor VIII (FVIII). 1 Treatment of AHA is based on controlling bleeding and eradicating inhibiting antibodies. 2 Bypassing agents (BPAs) control bleeding by avoiding the need for FVIII to generate thrombin. 3 These agents carry a risk of thromboembolic events that may be enhanced by patients’ comorbid conditions.1,4 High-dose replacement therapy with human FVIII (hFVIII) concentrates is used to increase FVIII levels when inhibitor titers are low [<10 Bethesda units (BU)/mL]. Otherwise, BPAs are used combined with immunosuppression. 5 Rarely, hFVIII can be used for patients with inhibitors >10 BU/mL combined with plasmapheresis or immunoadsorption.4,6 Newer approaches are biochemical rebalancing agents such as emicizumab, a monoclonal antibody that mimics the function of activated FVIII by enhancing the interaction between FIX/FIXa and FX/FXa. 7 Immunosuppression is necessary to eradicate the autoantibodies in AHA alongside all treatments that stop bleeding, but relapses occur in 15–33% of cases after stopping treatment. 8
Recombinant porcine FVIII (rpFVIII; susoctocog alfa, Obizur®; Baxalta US Inc., a Takeda company, Lexington, MA, USA) is indicated for the treatment of bleeding events (BEs) in adults with AHA in the EU, UK, and USA.9,10 The rationale is that rpFVIII has sufficient sequence homology to hFVIII to be hemostatic but is different enough to be less susceptible to inactivation by circulating FVIII inhibitors. 11 Treatment guidelines for AHA recommend using BPAs or rpFVIII as first-line therapy, with the final choice determined by anti-porcine FVIII (pFVIII) inhibitor titer, treatment cost, and availability.12,13
The safety and hemostatic efficacy of rpFVIII in patients with AHA has been demonstrated in a phase II/III, prospective, multicenter, open-label study (NCT01178294). 14 In that study, a total of 86% (24/28) patients had successful treatment of the initial BE (complete cessation of bleeding). We present the results of an open-label, observational, post-authorization safety study (PASS) conducted in the EU, UK, and USA designed to assess the safety, effectiveness, and utilization of rpFVIII in real-world clinical practice.
Methods
Study design
This PASS (EUPAS16055; NCT03199794) was a multicenter, prospective and retrospective, uncontrolled, open-label, single-cohort, observational study. It was conducted between 14 December 2016 and 30 July 2021 at 15 sites in 8 countries (Austria, Germany, Switzerland, Netherlands, Italy, France, UK, and USA). Patients were followed for 180 days after the last administration of rpFVIII. In case of a new or rebleeding event treated with rpFVIII, the patient was followed for an additional 180 days (Supplemental Figure 1).
Study population
Eligible patients were adults with AHA who had received or were receiving rpFVIII in routine clinical practice. Patients with a known anaphylactic reaction to rpFVIII, excipients, or hamster protein were excluded. Patients who had participated, within 30 days of enrollment, in a clinical study of a medicinal product/device or were scheduled to participate in such a study during this study were also excluded, as were those participating in the US post-marketing study, NCT02610127.
Informed consent was obtained from the patient or legal representative at the earliest opportunity within 30 days (prospective patient) or >30 days (retrospective patient) after rpFVIII administration (Supplemental Figure 1). If the patient died before consent was obtained, informed consent was obtained from the next of kin in accordance with local regulations. If such consent could not be obtained, data were anonymized to ensure data privacy, if permitted by local regulations.
Outcomes and assessments
The primary objective was to document the safety of patients treated with rpFVIII. Secondary objectives were to monitor rpFVIII use in real-world clinical practice, including immunogenicity; hemostatic effectiveness of rpFVIII for resolution of BEs (bleeding stopped as assessed by the physician); rpFVIII treatment details; concomitant medication use; and complete remission rate (inhibitor eradication). An exploratory objective was the association of anti-pFVIII immunogenicity with a lack of therapeutic effect and/or other clinical manifestations.
Where available, the following data were collected: safety as assessed by adverse events (AEs), serious AEs (SAEs), and AEs of special interest (AESI; reported with the same priority as SAEs). AESIs that were primary endpoints were hypersensitivity reactions, thromboembolic events, and dose dispensing medication errors. Immunogenicity was a secondary endpoint AESI corresponding to the development of
SAEs and AEs were recorded as observed by physicians and categorized by Medical Dictionary for Regulatory Activities (MedDRA) preferred terms, with severity assessments based on physicians’ judgment. Data were extracted from existing patient medical records. Sites were recommended to enter prospective data into the electronic data capture system within 14 days of entering the patient chart, or retrospective data within 14 days of signing the informed consent form.
Statistical analysis
The full analysis set included all patients who met the eligibility criteria. Available data for prospectively and retrospectively enrolled patients were analyzed together, with the baseline defined as the time of the first rpFVIII infusion for a new BE. To reduce potential selection bias, inclusion criteria were left broad and exclusion criteria were limited; investigators were instructed to invite all eligible patients to participate in the study; and the decision to administer rpFVIII was made by the physician before, and independently of, the invitation to participate in this study.
A minimum target of 50 patients was calculated to provide substantial safety information on real-life use of rpFVIII based on the rate of anti-pFVIII inhibitors in the phase II/III study (NCT01178294). 14 Descriptive statistics were used to characterize the overall patient population, including BEs, treatment outcomes, and AEs. Incidence rates and 95% confidence intervals were estimated based on a Poisson regression model (intercept only). The association between rpFVIII immunogenicity and lack of therapeutic effect (BE not stopped) was assessed using Fisher’s exact test. Analyses were performed using SAS® version 9.4 (SAS Institute, Cary, NC, USA).
Results
Patients
A total of 50 patients were enrolled (prospective,

Patient disposition.
Baseline demographics.

⩾10 patients in total.
Percentages are based on the number of patients with available data (
MedDRA, Medical Dictionary for Regulatory Activities; rpFVIII, recombinant porcine factor VIII; SD, standard deviation.
At the start of rpFVIII treatment for the initial BE, almost all patients with available information had abnormal levels of hemoglobin [24/25 (96.0%)] and all patients had abnormal hematocrit [
Safety
Overall, 148 AEs were reported in 41 patients, of which 46 were SAEs (five SAEs were considered probably related to rpFVIII) (Table 2). In 11 patients, 12 AEs were reported as having a fatal outcome, none of which were assessed as being related to rpFVIII. Most AEs (
Overall incidence of AEs.
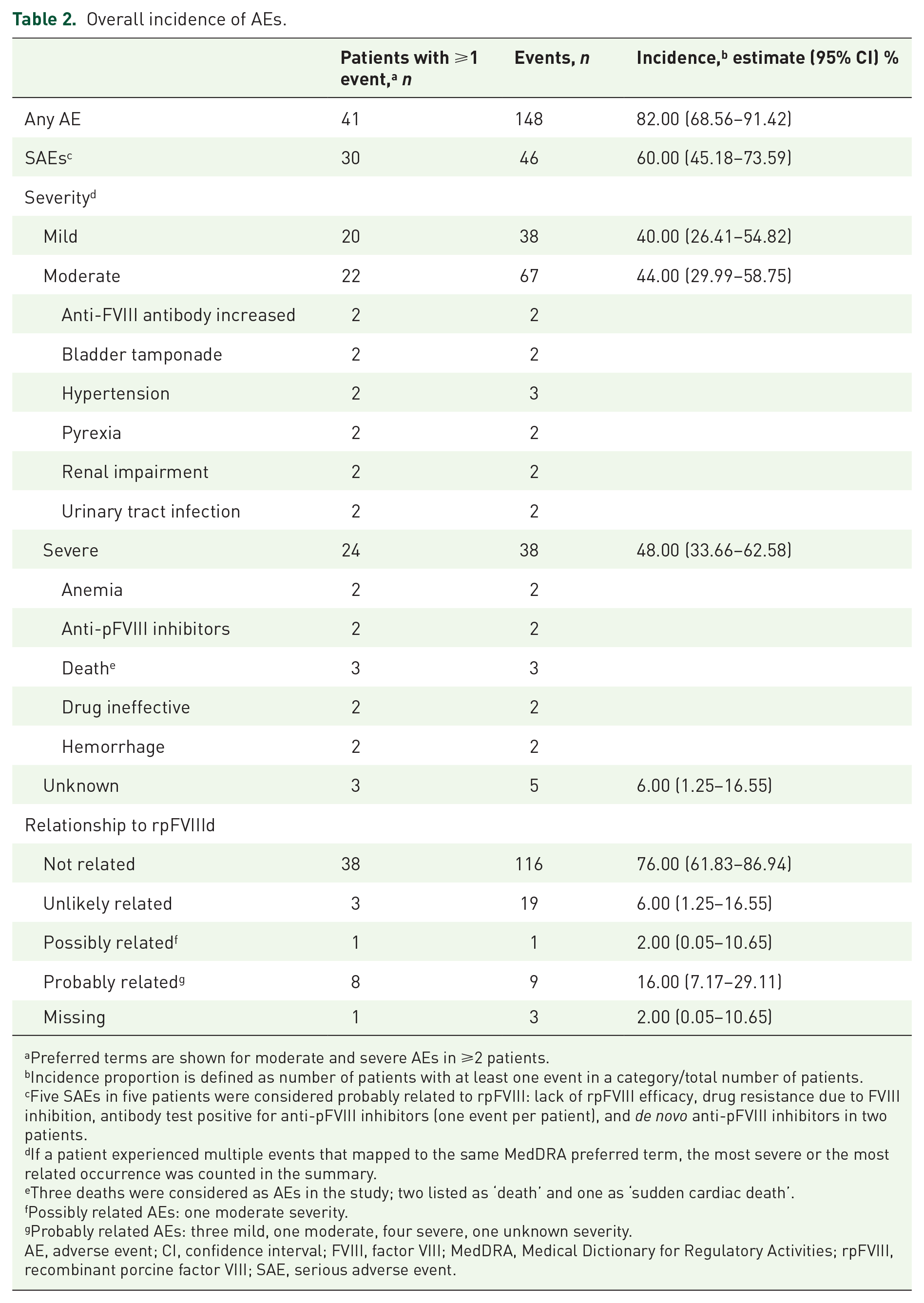
Preferred terms are shown for moderate and severe AEs in ⩾2 patients.
Incidence proportion is defined as number of patients with at least one event in a category/total number of patients.
Five SAEs in five patients were considered probably related to rpFVIII: lack of rpFVIII efficacy, drug resistance due to FVIII inhibition, antibody test positive for anti-pFVIII inhibitors (one event per patient), and
If a patient experienced multiple events that mapped to the same MedDRA preferred term, the most severe or the most related occurrence was counted in the summary.
Three deaths were considered as AEs in the study; two listed as ‘death’ and one as ‘sudden cardiac death’.
Possibly related AEs: one moderate severity.
Probably related AEs: three mild, one moderate, four severe, one unknown severity.
AE, adverse event; CI, confidence interval; FVIII, factor VIII; MedDRA, Medical Dictionary for Regulatory Activities; rpFVIII, recombinant porcine factor VIII; SAE, serious adverse event.
Among the 145 AEs with information on their relationship to rpFVIII, 9 were considered probably related to rpFVIII, 1 AE was considered possibly related, and 135 AEs were unrelated/unlikely related. Five patients had probably related AEs that were suggestive of a lack of efficacy/effect according to the standardized MedDRA query (SMQ) for lack of efficacy (SMQs are validated, standard sets of MedDRA terms,
15
Table 3). In these patients, the initial reported rpFVIII dose was lower than the recommended dose of 200 U/kg9,10 (between 54 and 180 U/kg). Four patients had probably related AEs that were classified as immunogenicity AESIs. These were drug resistance due to FVIII inhibition in one patient; antibody test positive for anti-pFVIII inhibitors in one patient (no increase in anti-pFVIII inhibitor titer from a measurable baseline); and
AEs that were possibly or probably related to rpFVIII.
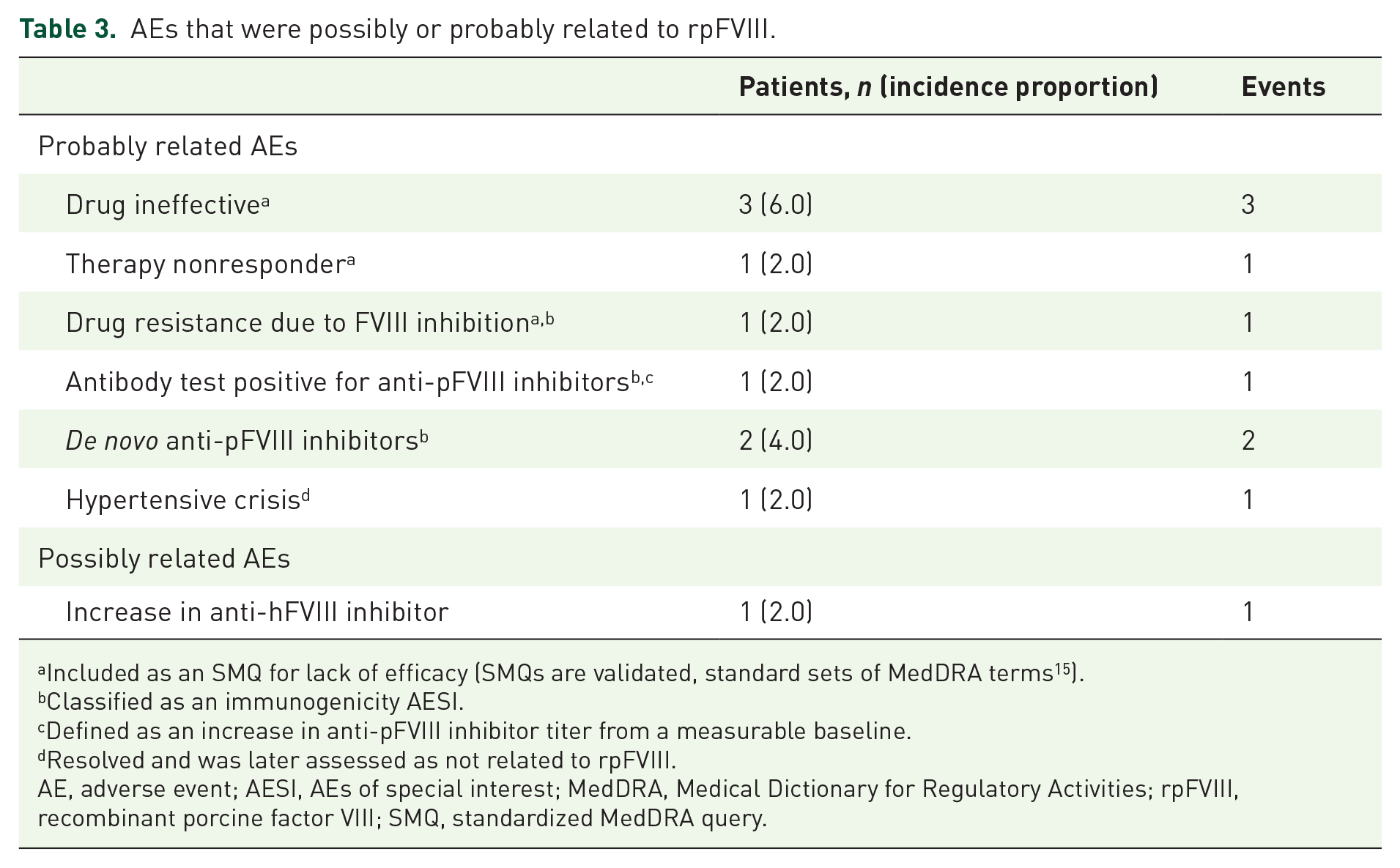
Included as an SMQ for lack of efficacy (SMQs are validated, standard sets of MedDRA terms 15 ).
Classified as an immunogenicity AESI.
Defined as an increase in anti-pFVIII inhibitor titer from a measurable baseline.
Resolved and was later assessed as not related to rpFVIII.
AE, adverse event; AESI, AEs of special interest; MedDRA, Medical Dictionary for Regulatory Activities; rpFVIII, recombinant porcine factor VIII; SMQ, standardized MedDRA query.
Secondary objectives
Anti-FVIII inhibitors
Testing for anti-pFVIII inhibitors was performed in a small number of patients with initial BEs. Anti-pFVIII titers remained stable during and after rpFVIII treatment in comparison with baseline titers (Table 4). Using a quantification limit of 0.6 BU/mL as specified in the World Federation of Hemophilia guidelines,
16
11 patients had detectable anti-rpFVIII titers during or after rpFVIII treatment (Table 4), with a median [range, interquartile range (IQR)] treatment duration of 3.04 (0.4–60.9, 1.24–8.69) days. Anti-pFVIII immunogenicity data at baseline and after rpFVIII treatment were analyzed in relation to BE outcomes, but only 6 patients had available data for change from baseline. No specific trend or correlation between immunogenicity and bleed outcomes could be established due to the small number of patients for whom these data were available. Physicians were also asked about immunogenicity in the electronic case report form (eCRF) with the question: ‘Did the patient develop an inhibitor against pFVIII or increased inhibitor level from pretreatment values during or following OBIZUR treatment?’ Twenty-six responses were received [immunogenicity,
Inhibitor assessments for initial BEs treated with rpFVIII.
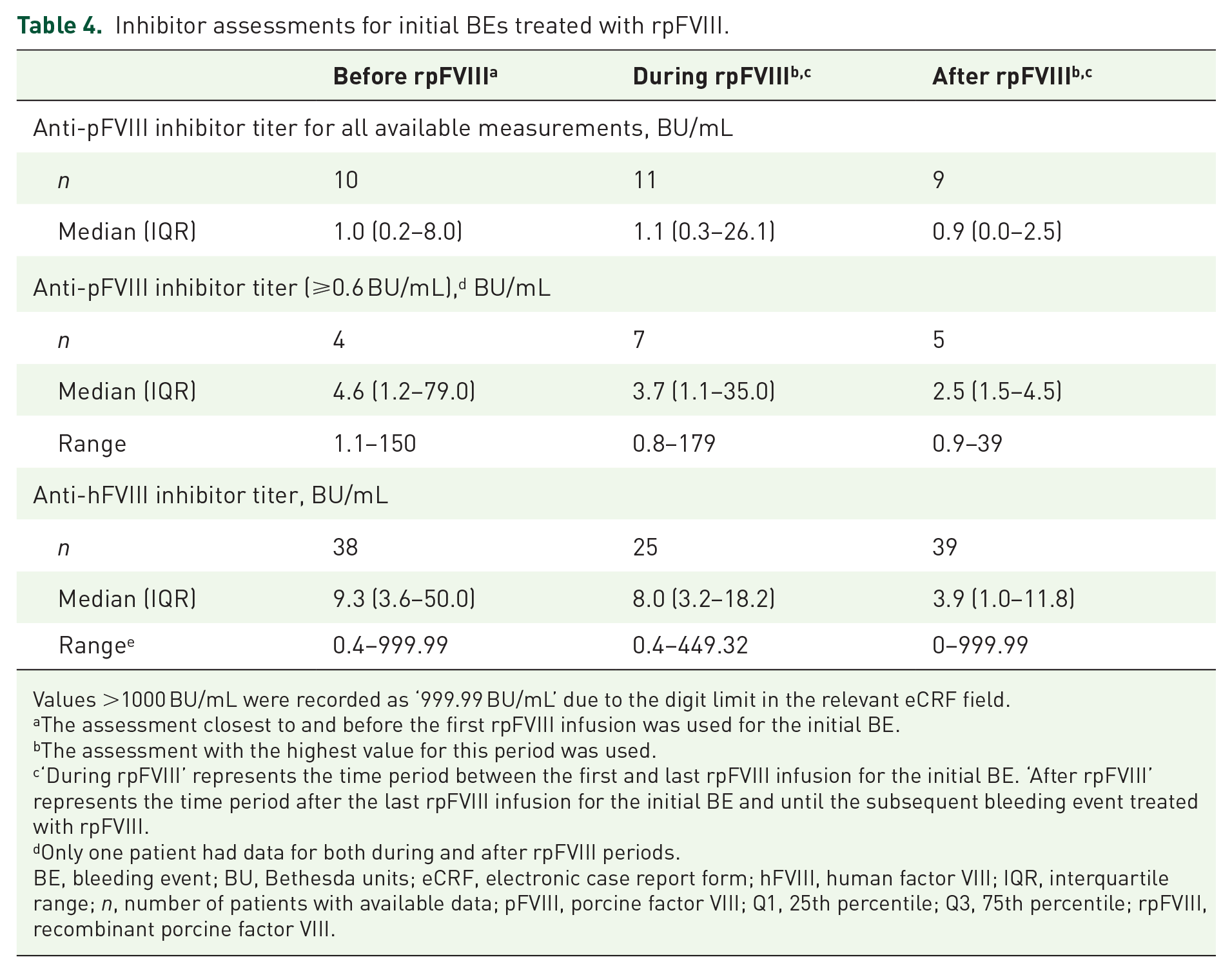
Values >1000 BU/mL were recorded as ‘999.99 BU/mL’ due to the digit limit in the relevant eCRF field.
The assessment closest to and before the first rpFVIII infusion was used for the initial BE.
The assessment with the highest value for this period was used.
‘During rpFVIII’ represents the time period between the first and last rpFVIII infusion for the initial BE. ‘After rpFVIII’ represents the time period after the last rpFVIII infusion for the initial BE and until the subsequent bleeding event treated with rpFVIII.
Only one patient had data for both during and after rpFVIII periods.
BE, bleeding event; BU, Bethesda units; eCRF, electronic case report form; hFVIII, human factor VIII; IQR, interquartile range;
Overall, lower median anti-hFVIII inhibitor titers for initial BEs were reported after rpFVIII treatment than at baseline (Table 4). Of 32 patients with anti-hFVIII inhibitor measurements available at baseline and after treatment, 10 had an increase in anti-hFVIII inhibitor titer after rpFVIII administration. However, the IQR indicated that the majority of anti-hFVIII titers were lower after rpFVIII, with an IQR of 1.0–11.8 BU/mL compared with 3.6–50.0 BU/mL at baseline.
FVIII assessments
For initial BEs, few patients had FVIII:C measured within 2 h and between 2 and 4 h after first rpFVIII administration (
FVIII:C after rpFVIII treatment for initial BEs.

The assessment closest to and before the first rpFVIII infusion was used for the initial BE.
The assessment with the highest value for this period was used.
The number of patients with missing data or without an assessment within the defined time period.
BE, bleeding event; FVIII, factor VIII; FVIII:C, factor VIII activity; IQR, interquartile range;
Hemostatic effectiveness
Bleeding characteristics
Most initial BEs and half of subsequent BEs were spontaneous. Rebleeds occurred with two initial and two subsequent BEs (one rebleed in each patient; Figure 2).

Bleeding characteristics.
Bleed resolution with rpFVIII
More than half of patients received rpFVIII as first-line treatment of initial BEs. Treatment details for initial and subsequent BEs are available in Table 6. Of the 50 initial BEs, 37 (74.0%) resolved after the rpFVIII treatment course [median (IQR) time from first infusion to bleeding resolution: 8.0 (4.0–16.0) days; Figure 3]. Among the 37 resolved initial BEs, 35 resolved after an initial rpFVIII treatment course and 2 after a subsequent rpFVIII treatment course. Most resolved initial BEs were spontaneous (70%,
rpFVIII treatment details for initial and subsequent BEs.

Data are median (range) unless otherwise stated.
First infusion date/time, minus initial bleeding date/time, plus 1.
Date/time of last infusion minus date/time of first infusion.
The median first dose of rpFVIII in this study was lower than that recommended in the current EMA and US FDA labels (200 U/kg). The EMA label also states that if testing of anti-pFVIII antibodies is negative at baseline, a rpFVIII dose lower than 200 U/kg may be used as the initial treatment dose.
The sum of reported doses across infusions per patient for the initial BE.
Total reported dose divided by number of infusions.
Count of first infusion plus subsequent infusions for the initial BE.
BE, bleeding event; EMA, European Medicines Agency; FDA, Food and Drug Administration; IQR, interquartile range;
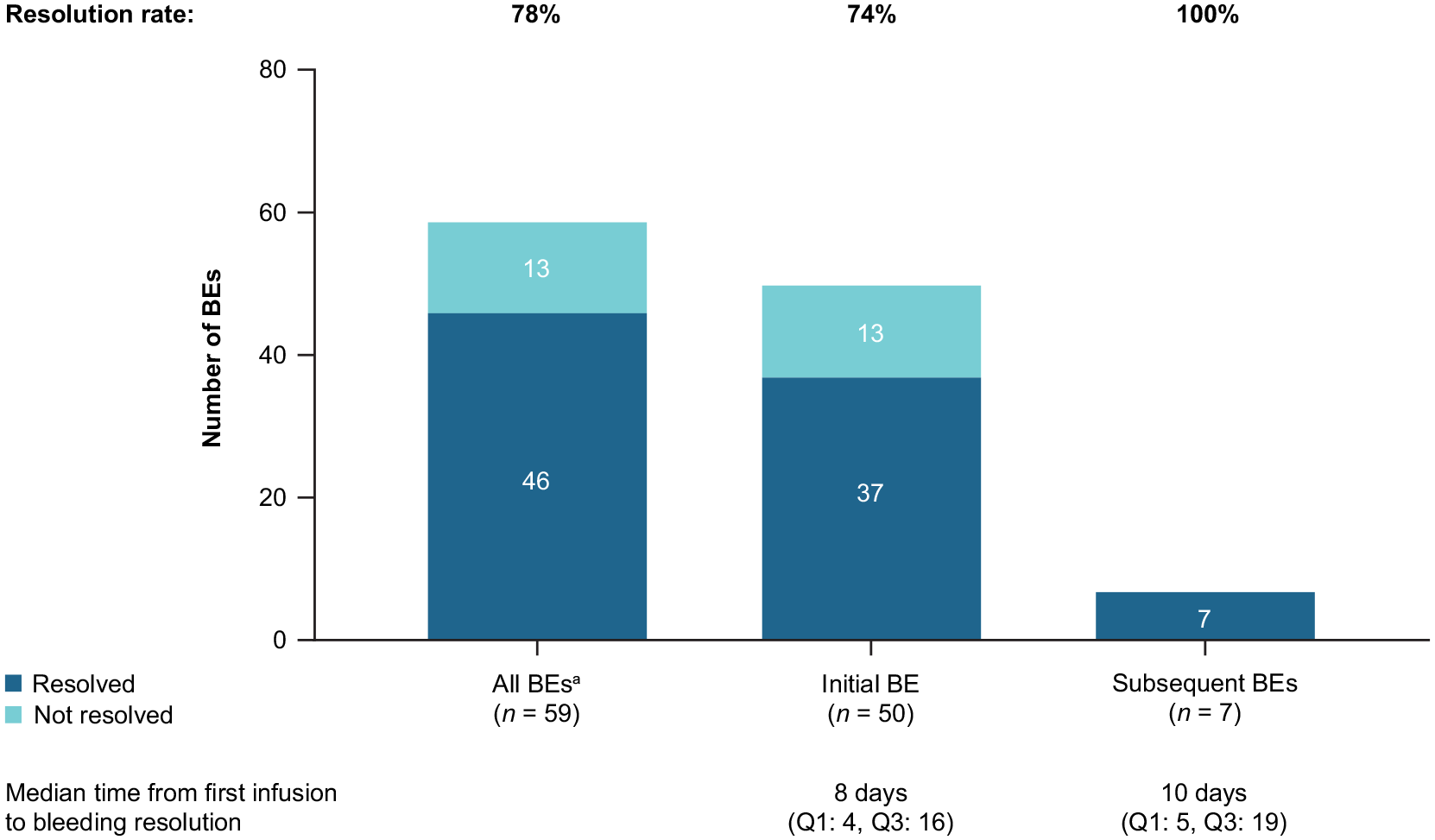
Bleed resolution with rpFVIII.
Some trends in the descriptive data were observed between the resolved and unresolved groups. Initial BEs resolved in a higher proportion of patients who received rpFVIII as first-line therapy (83%,
Concomitant medication
Concomitant treatment during the study was reported by all enrolled patients (Supplemental Table 1).
Inhibitor eradication
Data from patients with inhibitor eradication based on the physician’s judgment (none, partial, or complete remission) were available during or after rpFVIII treatment for initial and subsequent BEs. Of 46 initial BEs with available data, complete remission was achieved for 14 (30.4%) BEs, partial remission for 13 (28.3%) BEs, and no remission for 19 (41.3%) BEs. Six of the seven subsequent BEs treated with rpFVIII had information on inhibitor eradication. Three (50.0%) of these BEs achieved complete remission, one (16.7%) partial remission, and the remaining two (33.3%) no remission. Considering the response for each patient after the initial BE (as each patient had one initial BE), 58.7% of patients achieved partial or complete inhibitor eradication after their initial BE.
Exploratory objective
The proportion of patients with inhibitors was numerically higher among patients whose initial bleeding did not resolve, compared with patients whose bleeding resolved, but patient numbers were small and the difference was not significant [2/10 (20.0%) and 2/16 (12.5%), respectively,
Discussion
The results of this PASS are informative of real-world treatment of AHA with rpFVIII. No new safety signals were observed when compared with the phase II/III clinical trial of rpFVIII in AHA (NCT01178294). 14 There were no reported AEs of hypersensitivity reactions or thromboembolic events, which are potential safety concerns with rpFVIII, and no dose dispensing medication errors were reported. These findings are consistent with several case-series studies reporting treatment of AHA with rpFVIII in real-world settings, in which no related AEs were reported.17–21
Of the 50 initial BEs treated with rpFVIII, 74.0% resolved, even when first doses were lower than recommended in the European Medicines Agency (EMA) and US Food and Drug Administration labels (200 U/kg).9,10 The median reported first dose was 54 U/kg, with 75% of patients receiving doses <100 U/kg. The administration of lower than recommended first doses has also been reported in other publications summarizing real-world experience in different centers.17,20–22 Of note, the EMA label has been updated to state that if anti-rpFVIII antibody testing is negative at baseline, a dose lower than the recommended 200 U/kg can be used as the initial treatment dose. 10
Some trends were noted regarding BE resolution, although no statistical testing was performed on these data. The data suggest that a shorter time from the initial bleed to the first rpFVIII infusion could favor bleed resolution. In addition, a higher proportion of patients receiving first-line rpFVIII achieved resolution of their initial BE than those receiving second-line therapy, suggesting that first-line rpFVIII is potentially better than second-line therapy for resolving initial BEs. Alternatively, this may reflect that BEs in patients requiring second-line therapy are more treatment-refractory than those requiring first-line therapy. Similarly, in the phase II/III clinical trial (NCT01178294), 14 patients treated with rpFVIII as first-line therapy had a higher rate of treatment success than patients treated with another hemostatic agent before rpFVIII treatment.
Based on a case-series study of four patients with AHA and one with congenital hemophilia with inhibitors, conducted by Abou-Ismail
FVIII:C was monitored for a limited number of patients before the first rpFVIII infusion and within the first 4 h after the infusion, despite the recommendation in the summary of product characteristics to monitor FVIII:C at 30 min after the first infusion and at 3 h after completing rpFVIII therapy. 10 This might reflect challenges encountered in real-world hospital settings. Time to initial bleeding resolution (8.0 days) was similar to the 6.5 days in the phase II/III clinical trial (NCT01178294). 14 However, in this PASS, BE control was assessed on the basis of bleeding resolution according to physician judgment, whereas in the clinical trial, it was measured using body system-specific evaluation criteria. 14
The mean age of the enrolled patients (76 years) is consistent with AHA mainly affecting older people,
2
and suggests that the data may be generalizable to the wider AHA population. The 60:40 ratio of males to females in the patient population is consistent with other AHA studies from Europe and Taiwan.23–25 There were several limitations, as expected given the study’s observational nature. These included the fact that not all assessments of interest were routinely collected or collected in a standardized manner. The availability of FVIII:C data and anti-pFVIII inhibitor assessments were limited, and
An additional study limitation was that the assessment of effectiveness and inhibitor eradication was based on the investigator’s judgment, and the analysis of inhibitor eradication data was linked to BEs rather than individual patients, thus limiting the analysis of follow-up to remission at the patient level. As race or ethnicity was not available for 22 of the 50 patients enrolled (44%) it is difficult to make any conclusions regarding applicability to other populations. Finally, this study did not capture clinician and patient satisfaction with the ability of rpFVIII to control bleeding quickly. Metrics are needed that capture the speed of BE control as well as satisfaction with delivery and receipt of a treatment that quickly stabilizes serious patient bleeding.
Conclusion
No new safety signals were observed in this real-world study. The findings of this study support the use of rpFVIII as first-line treatment for AHA and align with findings from the phase II/III clinical trial (NCT01178294) of rpFVIII in the treatment of BEs in adults with AHA. 14
Supplemental Material
sj-docx-1-tah-10.1177_20406207241260332 – Supplemental material for Real-world use of recombinant porcine sequence factor VIII in the treatment of acquired hemophilia A: EU PASS
Supplemental material, sj-docx-1-tah-10.1177_20406207241260332 for Real-world use of recombinant porcine sequence factor VIII in the treatment of acquired hemophilia A: EU PASS by Wolfgang Miesbach, Nicola Curry, Paul Knöbl, Charles Percy, Rita Santoro, Alvin H. Schmaier, Karolin Trautmann-Grill, Kayode Badejo, Jie Chen, Masoud Nouri, Pooja Oberai and Robert Klamroth in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
Medical writing support was provided by Nasser Malik, PhD, employee of Excel Medical Affairs (Fairfield, CT, USA), and was funded by Takeda Development Center Americas, Inc., Lexington, MA, USA.
Correction (September 2024):
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
