Abstract
Background:
Gene therapy has emerged as a transformative treatment for hemophilia, offering the potential for durable factor expression after a single administration. Since 2019, multiple gene therapies have been approved for adults with hemophilia A and B. However, the adoption of gene therapy into clinical practice has been slow, and provider preparedness remains uncertain.
Objective:
To assess current knowledge, perceptions, and clinical preparedness regarding hemophilia gene therapy among international healthcare professionals.
Methods:
An anonymous online survey was distributed internationally from August 24, 2023 to January 24, 2024 through four professional organizations. The 24-question survey included items on demographics, perceptions of gene therapy, and knowledge-based questions. Descriptive statistics were used to analyze responses.
Results:
A total of 327 participants from 66 countries responded. Top concerns with current therapies included dosing burden, inhibitor risk, and venous access. Only 7.5% of respondents were extremely familiar with regulatory guidance on gene therapy, and 35.6% were not comfortable answering patient questions about trials. Perceived barriers to adoption included uncertainty about durability, immune responses, eligibility, and cost.
Conclusion:
While clinical integration of gene therapy for hemophilia is advancing, this survey highlights persistent gaps in provider knowledge and confidence, especially related to regulatory guidance and clinical trial data. Targeted education and training initiatives are needed to support informed implementation of gene therapy in diverse clinical settings.
Keywords
Introduction
The emergence of gene therapy has ushered in a new era of hemophilia treatment, offering the potential for long-term disease control with a single intravenous administration.1,2 Clinical studies have shown that hemophilia gene therapy can lead to a significant increase in clotting factor activity, with some patients achieving factor levels approaching normal or near-normal levels.1,2
In November 2022, etranacogene dezaparvovec was approved as the first gene therapy for hemophilia, and was indicated for adult patients with hemophilia B. 3 Later, in August 2023, the FDA approved valoctocogene roxaparvovec as the first gene therapy for severe hemophilia A in adults. 4 Finally, in April 2024, the FDA approved a second gene therapy, fidanacogene elaparvovec, for the treatment of adults with moderate-to-severe hemophilia B. 5 Each of these therapies has also received corresponding approval in the EU.
Despite the approval of novel gene therapies for hemophilia, adoption into clinical practice has been slow thus far. 6 Factors that may be contributing to the slow uptake include high costs, concerns about durability and safety, and the complexity of the treatment and reimbursement process. 7 Some patients have also experienced a loss of response or a return to prophylaxis treatment after receiving gene therapy, raising questions about the consistency of the therapeutic effect.7,8
Nevertheless, gene therapy has shown improved treatment durability relative to standard of care, and reduced treatment burden for patients due to a reduction or elimination of the need for infusions.1,2
In 2019, a survey was conducted to evaluate the knowledge and perceptions of healthcare practitioners and scientists about gene therapy for hemophilia. 9 Results from this survey highlighted significant knowledge gaps and educational needs related to hemophilia gene therapy among clinicians. 9 A significant proportion of clinicians, even those directly involved in hemophilia care, still lacked the ability to confidently explain the fundamentals of adeno-associated viral (AAV)-mediated gene therapy or discuss the latest clinical trial results with patients. 9
This follow-up survey aimed to further explore the current state of knowledge, attitudes, and preparedness of the broader healthcare team involved in the care of people with hemophilia. Understanding the educational needs across different provider roles, practice settings, and geographic regions will be critical to developing targeted training and resources to support the integration of gene therapy into clinical practice.
Materials and methods
This study was based on an international survey conducted between August 24, 2023 and January 24, 2024. The survey was designed by expert members of the ISTH Scientific Standardization Committee (SSC) for Gene Therapy based on the responses and feedback from the 2019 survey. Survey participants were all members of the International Society on Thrombosis and Haemostasis (ISTH), the European Haemophilia Consortium (EHC), the European Hematology Association, or the European Association for Haemophilia and Allied Disorders (EAHAD) with valid email contacts. The survey was validated through expert targeting, content replication from a prior instrument (Peyvandi et al.), and internal consistency in response patterns across provider roles and regions. 9
The survey was composed of 24 questions covering demographic information (9), perceptions about gene therapy for hemophilia (5), and knowledge of gene therapy (9 single correct answers, 1 polling). A variety of question formats were included: single-answer check box, single answer from a drop-down menu, “select all that apply” or multiple responses allowed, 5-point Likert-type scale questions with descriptors of the point rating, open text response, and ranking (Supplemental Table 1).
The ranking question format used a weighted approach, in which the overall rank was assigned by calculated score. Items ranked first by individual respondents are given higher value or weight than lower-ranked items; the sum of all weighted values is the score for a given item.
Each knowledge-based question included the answer option, “I’m not sure.” Participants accessed the questionnaire online using the SurveyGizmo (https://www.surveygizmo.com/) platform. Survey results were summarized using descriptive statistics. Participation in the survey was voluntary, without compensation, and responses were confidential and deidentified for summarization and analysis.
The results from this study and the results previously described in Peyvandi et al. (n = 194) are compared in the discussion. 9 The target population for both surveys was the same as described above. This survey questionnaire asked identical questions regarding demographic information. However, options for perceptions about gene therapy for hemophilia were updated from 2019, and seven out of nine of the knowledge of gene therapy questions remained the same. In addition, one opinion question about clinical endpoints was added to the 2023 survey. This replication supports consistency in measuring clinician understanding and evolving perceptions over time.
Results
Demographics
Of the 5117 survey recipients, 327 participants responded from 66 countries (6.4% response rate). Physicians constituted the largest group (69.7%), followed by researchers (10.4%) and other healthcare providers (9.2%; Figure 1(a)). A significant majority of respondents (124/157, 79%) were affiliated with hemophilia treatment centers (Figure 1(b)). Areas of clinical focus were primarily bleeding disorders (47.4%), hematology (27.3%), and thrombotic disorders (10.3%), with additional representation in pediatric hematology and other clinical or nonclinical specialties (Figure 1(c)). Respondents reported a range of roles in hemophilia care, including direct patient management (62.7%), clinical trial investigations (32.4%), and patient education or advocacy (22.3%; Figure 1(d)). Additional demographic data are further described in Figure S1.

Participant demographics overview of the demographic characteristics of the survey respondents (N = 327). (a) Distribution of professional categories among survey respondents (N = 327); (b) Affiliation with hemophilia treatment centers (N = 157); (c) Primary areas of clinical focus (N = 327); (d) Roles in care of individuals with hemophilia (N = 318).
Perceptions of current treatment approaches for hemophilia
Respondents were asked to select three of the greatest limitations associated with current treatment approaches (factor replacement therapy with standard or extended half-lives, bypassing therapy, and emicizumab) for patients with hemophilia. The most frequently selected concerns were the burden of dosing schedules and prophylaxis (108 mentions; 33.0%), risk of inhibitor development (91; 27.8%), and challenges with injections/venous access (87; 26.6%). Additional priorities included patient access to treatment (86; 26.3%), cost or reimbursement issues (82; 25.1%), and treatment adherence (75; 23.0%). Other notable limitations were the impact on overall quality of life (QoL; 62; 19.0%), development of joint disease (59; 18.0%), breakthrough bleeding (54; 16.5%), and pain associated with treatment (20; 6.1%; Figure 2).

Perceived limitations of current hemophilia treatments. Overview of participant ranked treatment limitations.
When analyzed by region (North America, South America, Europe, Africa, and Asia), the top three limitations were variable (Figure S2). North American respondents emphasized the impact on QoL over the risk of inhibitor development, whereas Europeans were more inclined to prioritize the development of joint disease as a limitation. Asian participants selected cost/reimbursement, while African participants selected patient access to treatment as one of the greatest limitations (Figure S2).
Familiarity with hemophilia gene therapy
Regarding regulatory guidance, 73 respondents (32.1%) reported a limited understanding, while 52 (22.9%) respondents were aware of documents under development but had not read them (Figure 3(a)). Fewer respondents (49, 21.5%) had reviewed available documents and could describe the content to colleagues, and only 17 (7.5%) considered themselves extremely familiar with current guidance. 16.2% (36) of all participants were unaware that there was regulatory guidance on gene therapy for hemophilia (Figure 3(a)).

Participant familiarity with regulatory guidance and ability to answer patient questions about gene therapy for hemophilia. Both metrics were evaluated on a 5-point Likert scale. (a) Self-reported familiarity with regulatory guidance on gene therapy for hemophilia (N = 227); (b) Self-assessed ability to answer patient questions about gene therapy for hemophilia (N = 227).
When it comes to answering patient questions on clinical trial results, 67 respondents (29.5%) felt capable of addressing basic inquiries, while 63 (27.7%) were comfortable discussing clinical trials, and 16 (7.0%) considered themselves experts (Figure 3(b)). However, over one-third of respondents (81, 35.6%) would not be able to, or did not feel comfortable answering patient questions (Figure 3(b)). Looking closer at the physician subgroup (n = 143), 41 physicians (28.6%) reported a limited understanding, while 35 physicians (24.4%) were aware of documents under development but had not read them (Figure S3(A)). Less than one-third of physicians (44, 30.7%) felt capable of addressing basic questions about clinical trials, while just over one-quarter (40, 27.9%) were comfortable discussing clinical trials, and 11 (7.7%) considered themselves experts (Figure S3(B)).
Knowledge of hemophilia gene therapy
One hundred ninety-two participants answered nine single-answer knowledge-based questions on aspects of hemophilia gene therapy. Overall, an average of 58% of respondents answered each question correctly (Supplemental Table 2). Results for the knowledge of gene therapy questions are described in more detail in the Supplemental Material.
Opinions on gene therapy
When asked about the most important efficacy outcome measure for gene therapy (n = 192), 44% of respondents prioritized factor activity levels, while 30% emphasized annualized bleeding rates (ABRs). Other measures, such as patient-reported outcomes (PROs; 10%) and factor concentrate usage (7%), were considered less critical, with 9% of respondents unsure of the best metric (Figure 4(a)).

Healthcare professionals’ opinions on gene therapy efficacy measures (N = 192) and professional society affiliations (N = 237). (a) Preferred outcome measures for gene therapy success; (b) Recommended professional organizations for sharing gene therapy resources.
The most frequently recommended resource for sharing information about gene therapy with patients was the ISTH, cited by 40% of respondents (Figure 4(b)). This was followed by the EAHAD at 21%, and the American Society of Gene and Cell Therapy at 12% (Figure 4(b)). Other organizations mentioned included the EHC at 11%, the European Society of Gene and Cell Therapy at 5%, and the Hemophilia Federation of America at 5% (Figure 4(b)).
Respondents provided rankings for the top 10 potential concerns or barriers regarding current clinical care of hemophilia related to gene therapy (Figure 5). The most frequently identified concerns included the durability of response, variability of response, and cost or reimbursement issues. Other significant barriers included immune response to AAV vectors, the potential need for immunosuppression, and patient eligibility. When analyzed by subgroup, the top three ranked items remained consistent with physicians.
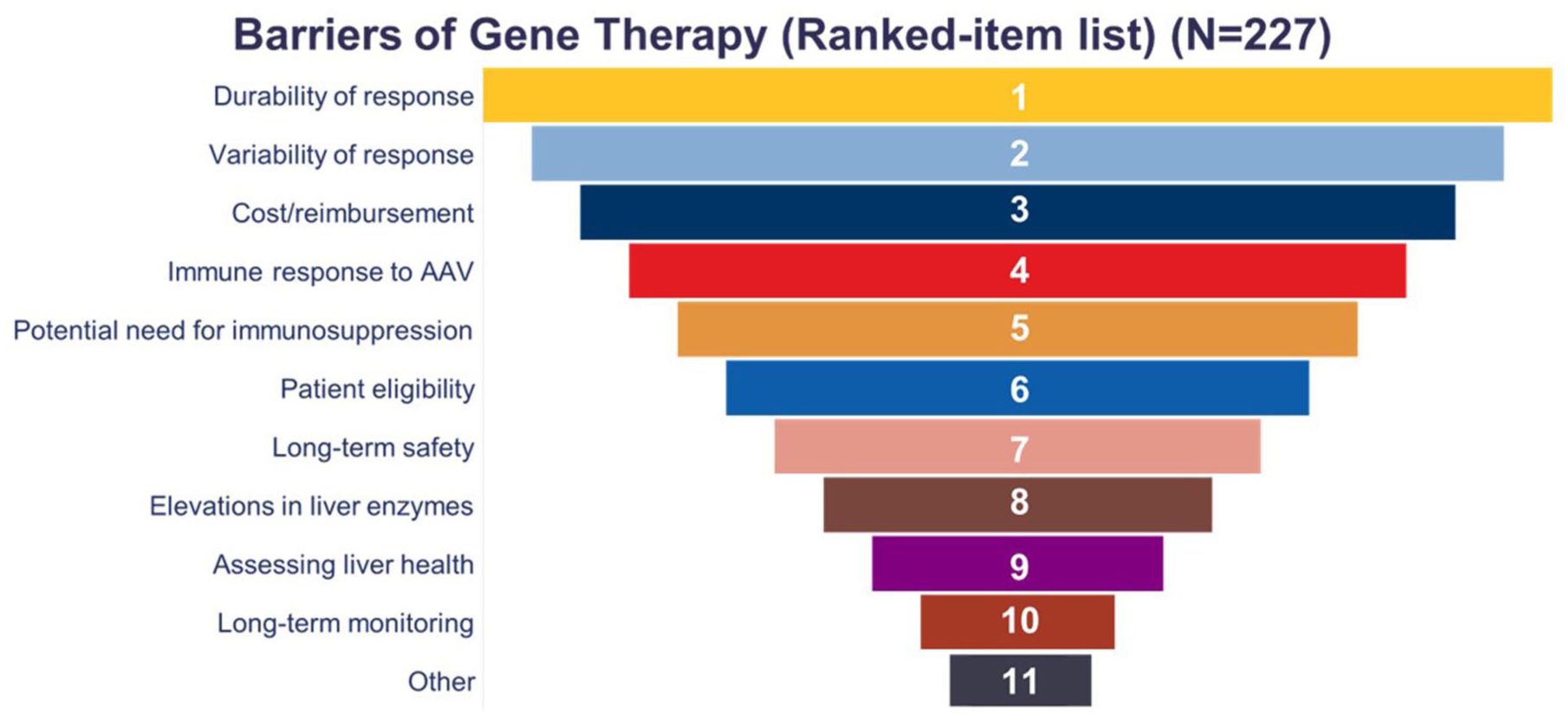
Top 10 concerns or barriers related to gene therapy for hemophilia as ranked by healthcare professionals (N = 227). Concerns are presented in descending order of priority.
Regional differences in concerns and barriers were also notable. Across continents, the durability of response was a predominant concern, ranking highest in North America, Europe, and South America (Figure S4). The variability of response frequently appeared as the second or third most significant barrier across regions, while cost/reimbursement was a prominent concern, particularly in Africa, Asia, and South America (Figure S4). Patient eligibility also emerged as a barrier in some areas, reflecting concerns about restrictive criteria that could exclude many patients from accessing gene therapy based on age, liver health, or prior exposure to viral vectors (Figure S4).
Discussion
Since the publication of the 2019 survey, the hemophilia field has transitioned from reviewing perceptions and knowledge to implementing gene therapies in clinical practice. Recent studies have reported successful clinical outcomes in gene therapy for hemophilia, paving the way for multiple regulatory approvals.3–5,10,11 As new data emerges, gene therapies for hemophilia continue to move closer to becoming standard clinical practice.
This shift marks a new era in hemophilia treatment, where gene therapy is not only a topic of research but a viable therapeutic option for patients. The focus is now on refining these therapies, ensuring their long-term efficacy and safety, and making them accessible to a broader patient population.8,12 While gene therapy offers promising advancements in hemophilia treatment, its full potential can only be realized if the healthcare community is well-informed and prepared.
The present survey aimed to further explore the current state of knowledge, attitudes, and preparedness of the broader healthcare team involved in caring for people with hemophilia. The results demonstrated a general shift in knowledge and perceptions of gene therapy for hemophilia between 2019 and 2023. Since 2019, the therapeutic landscape for patients with hemophilia has expanded rapidly, with various novel options available.3–5 Thus, it was important to understand the implications of this expansion of treatment options on clinical perspectives for gene therapy.
While notable progress has been made since earlier assessments, persistent gaps and emerging challenges underscore the complexity of integrating this innovation into clinical practice. Although the participant demographics remained fairly similar, the number of participants increased from 201 in 2019 to 327 in 2023. 9 A prominent proportion of respondents in this survey reported limited familiarity with regulatory guidance and scientific advancements in gene therapy.
These gaps also extend to clinical application. In addition, more respondents in this survey (36%, n = 81) expressed discomfort in addressing patient questions about clinical trials or discussing the long-term implications of gene therapy relative to 2019 (30%, n = 44). This may be due in part to the proportion of self-identified experts in this survey relative to 2019. The additional discomfort is particularly concerning given the increasing interest among patients in exploring gene therapy as a treatment option.
In this 2023 survey, only 7% of participants identified as gene therapy experts, which decreased from 11% in 2019. In both years, most participants were physicians (66% in 2019 vs 69.7% in 2023), with the majority actively involved in hemophilia care. However, the percentage of those directly involved in hemophilia care declined in our survey (59% in 2019 vs 52% in 2023). 9 Notably, pediatric clinicians also participated in the survey, despite no approved gene therapy for the pediatric population.
Directly comparing the knowledge across the surveys is not possible, as only respondents who self-reported at least some understanding of the science of AAV-mediated gene therapy answered the knowledge-based questions in 2019. In contrast, in 2023, these questions were available to all respondents. As expected, the average score for each question decreased due to the inclusion of participants with varying levels of expertise. Overall, the results indicated general competency among participants across different demographics, but there is still room for improvement.
About one-third of the total survey population either answered incorrectly or did not know the correct answers, highlighting significant knowledge gaps. These gaps included familiarity with AAV therapy, safety outcomes, and patient eligibility criteria for gene therapy. This underscores the need for targeted education and research to address these issues and improve patient outcomes. It will be interesting to see if knowledge of gene therapy shifts over the next few years as more patients are treated.
Previously, the three reported greatest limitations associated with current treatment approaches for patients with hemophilia were, in order of importance, risk of inhibitor development, dosing schedules/burden of prophylaxis, and impact on overall QoL. While issues such as dosing burden and inhibitor risk have persisted, newer concerns related to emerging therapies and healthcare system limitations have also become prominent. For instance, the emergence of gene therapies offers the potential for long-term relief from frequent infusions. However, it also brings uncertainties about long-term effects and the durability of the response. This creates a dilemma for clinicians and their patients who must weigh the known challenges of current treatments against the potential benefits and risks of newer, less established therapies. These ongoing concerns emphasize the need for continued research and development in hemophilia treatment, focusing not just on efficacy but also on improving the QoL for patients.
The durability and variability of response emerged as the top concerns regarding gene therapy, with cost and reimbursement cited as significant barriers, particularly in Asia and South America. These results mark a shift from the 2019 survey, when the top barriers were long-term safety and monitoring, durability of expression/response, and challenges in specific populations. 9 Although prior concerns about long-term safety have diminished due to favorable trial outcomes, the uncertainty surrounding the duration of therapeutic effects remains a critical issue.
Notably, clinicians also expressed concerns about restrictive eligibility criteria, which may exclude many patients from accessing these therapies. Presumably, this shift can be attributed to several factors, including increased awareness of gene therapy and patient-related considerations.
These results are consistent with the results of a survey of 20 Belgian patients with hemophilia A or B in 2021. For these patients, the most important considerations for gene therapy were ABR, factor level, long-term risks, impact on daily living, and probability of stopping prophylaxis. 13 The favorable clinical safety outcomes for patients treated with gene therapy for both hemophilia A and B, have alleviated some previous concerns about long-term safety.2,14,17 However, durability concerns remain a significant barrier for clinicians across surveys due to uncertainty about the duration of therapeutic effects.14,17
Clinical trials have shown that patient responses to gene therapy can vary widely.14,18 Not every patient responds to the treatment, and the degree of response differs significantly among those who do.14,18 Economic considerations have also become more apparent as gene therapies have moved to real-world clinical use, with high costs and questions about eligibility, accessibility, and healthcare system readiness. 19 Long-term data demonstrate the efficacy and durability of gene therapy for up to 10 years post-infusion, but the data remain limited, and the long-term effects beyond this period are still unknown. 20
There is also growing interest in gene therapy among patients with hemophilia. 15 A survey of French patients found that 64.2% were curious about hemophilia gene therapy, and 33.6% were interested in receiving it as soon as it became available. 15 Interestingly, in contrast, the same survey found 38.7% of patients would rather wait until more patients have received gene therapy before considering it for themselves. 15 This indicates an optimistic but cautious attitude toward this treatment. However, despite this interest, over 60% reported that they have never or almost never discussed gene therapy as a treatment option with their hemophilia care team. 14 This highlights the need for improved training and education for patients and professionals at hemophilia centers offering gene therapy.
Furthermore, in a study of PROs for those who received hemophilia gene therapy, patients reported favorable outcomes. 16 Prior to gene therapy, patients reported negative feelings related to treatment burden, pain, and barriers to physical activity. 16 Post-gene therapy patients experienced improvements in physical activity, work, lifestyle, and pain, although some expressed anxiety about the long-term efficacy of gene therapy and loss of control over treatment. 16 This further underscores the transformative potential gene therapy may hold for patients.
As the therapeutic options for hemophilia continue to expand and evolve, it is important that clinicians receive ongoing training and support to maintain awareness of the latest developments. This will enable them to make informed decisions, effectively communicate with patients about treatment options, and navigate the complex landscape of emerging therapies. Future efforts should focus on addressing the knowledge gaps noted above, providing comprehensive education on gene therapy, and supporting clinicians in managing the uncertainties associated with novel treatments. Only through such concerted efforts can we ensure that the potential benefits of advanced therapies, such as gene therapy, are fully realized for patients with hemophilia.
Limitations
The presented study has several limitations. While these outcomes support its use as a needs assessment tool, the absence of a formal psychometric evaluation limits its utility as a rigorously validated research instrument. In tandem, the knowledge domain was not analyzed using item-response theory or factor analysis, which would be required for establishing a validated knowledge scale. Furthermore, this survey had a small sample size, which may affect the robustness of the findings. Moreover, participants were not required to complete all questions, which resulted in variable response rates across items, with no data to clarify why a user may not have answered. Additionally, we did not distinguish between pediatric and adult providers, which may have influenced responses due to eligibility considerations. A potential selection bias may exist, as respondents with a particular interest in gene therapy may have been more likely to participate. The survey’s language availability in English only introduces an additional source of selection bias. These factors may limit the generalizability of the results across professional roles, practice settings, and geographic regions.
Conclusion
Healthcare professionals reported limited familiarity with regulatory guidance on hemophilia gene therapy, and many lacked confidence in discussing clinical trial data with patients
Knowledge assessments revealed gaps, particularly regarding the mechanism of AAV-based gene therapy, though awareness of common adverse events was relatively high
Top perceived limitations of current treatments included treatment burden, inhibitor risk, and access challenges, with regional variations in priority concerns
Durability and variability of gene therapy response, along with cost and eligibility restrictions, were identified as the most significant barriers to clinical adoption
Educational needs remain widespread across roles, regions, and experience levels, underscoring the importance of targeted training to support clinical integration of gene therapy
Supplemental Material
sj-docx-1-tah-10.1177_20406207251384802 – Supplemental material for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team
Supplemental material, sj-docx-1-tah-10.1177_20406207251384802 for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team by Melissa F. Glasner and Wolfgang Miesbach in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207251384802 – Supplemental material for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team
Supplemental material, sj-docx-2-tah-10.1177_20406207251384802 for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team by Melissa F. Glasner and Wolfgang Miesbach in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-3-tah-10.1177_20406207251384802 – Supplemental material for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team
Supplemental material, sj-docx-3-tah-10.1177_20406207251384802 for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team by Melissa F. Glasner and Wolfgang Miesbach in Therapeutic Advances in Hematology
Supplemental Material
sj-jpg-4-tah-10.1177_20406207251384802 – Supplemental material for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team
Supplemental material, sj-jpg-4-tah-10.1177_20406207251384802 for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team by Melissa F. Glasner and Wolfgang Miesbach in Therapeutic Advances in Hematology
Supplemental Material
sj-jpg-5-tah-10.1177_20406207251384802 – Supplemental material for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team
Supplemental material, sj-jpg-5-tah-10.1177_20406207251384802 for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team by Melissa F. Glasner and Wolfgang Miesbach in Therapeutic Advances in Hematology
Supplemental Material
sj-jpg-6-tah-10.1177_20406207251384802 – Supplemental material for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team
Supplemental material, sj-jpg-6-tah-10.1177_20406207251384802 for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team by Melissa F. Glasner and Wolfgang Miesbach in Therapeutic Advances in Hematology
Supplemental Material
sj-jpg-7-tah-10.1177_20406207251384802 – Supplemental material for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team
Supplemental material, sj-jpg-7-tah-10.1177_20406207251384802 for Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team by Melissa F. Glasner and Wolfgang Miesbach in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
