Abstract
Background:
The systemic immune-inflammation index (SII) represents the immunoinflammatory score and can be considered as a prognostic marker; however, its relevance to the prognosis in patients with diffuse large B-cell lymphoma (DLBCL) remains unclear.
Objectives:
The present meta-analysis was conducted to comprehensively evaluate the relationship between the SII and prognosis in patients with DLBCL.
Design:
This meta-analysis was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement.
Data sources and methods:
The PubMed, Web of Science, Embase, and Cochrane Library databases were comprehensively searched from inception to 16 March 2023. We calculated combined hazard ratios (HRs) and 95% confidence intervals (CIs) to estimate the prognostic significance of the SII for overall survival (OS) and progression-free survival (PFS) in DLBCL. In addition, this study determined odds ratios (ORs) and their 95% CIs to evaluate the correlation of SII with the clinicopathological features of DLBCL.
Results:
Five articles including 592 cases were enrolled in the current meta-analysis. According to our combined findings, the higher SII significantly predicted worse OS (HR = 3.87, 95% CI: 2.48–6.04, p < 0.001) together with inferior PFS (HR = 2.38, 95% CI: 1.12–5.08, p = 0.024) in DLBCL. Furthermore, a high SII was significantly correlated with B symptoms (OR = 2.52, 95% CI: 1.66–3.81, p < 0.001), III–IV Ann Arbor stage (OR = 2.86, 95% CI: 1.84–4.45, p < 0.001), high–intermediate/high National Comprehensive Cancer Network International Prognostic Index (OR = 2.25, 95% CI: 1.52–3.31, p < 0.001), increased neutrophil-to-lymphocyte ratio (OR = 33.76, 95% CI: 17.18–66.35, p < 0.001), and increased platelet-to-lymphocyte ratio (OR = 44.65, 95% CI: 5.80–343.59, p < 0.001). Nonetheless, the SII was not significantly related to sex, age, lactic dehydrogenase level, Eastern Cooperative Oncology Group performance status, or histology.
Conclusion:
According to this meta-analysis, the higher SII dramatically predicted inferior OS and PFS of DLBCL. Furthermore, an increased SII significantly correlated with some clinicopathological features representing the disease progression of DLBCL.
Trial registration:
The protocol was registered in INPLASY under the number INPLASY202380106.
Background
Diffuse large B-cell lymphoma (DLBCL) is the most frequently observed histological subtype of non-Hodgkin’s lymphoma (NHL), accounting for approximately 30% of NHL cases. 1 Symptoms of DLBCL typically are progressive lymphadenopathy and/or extranodal disorder. 2 Most DLBCL cases are diagnosed at an advanced stage; however, the R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) regimen of immunochemotherapy is effective in >60% of all patients with DLBCL. 2 However, approximately 40% of the patients with DLBCL fail to respond to or relapse following the R-CHOP regimen and have poor prognoses. Early identification helps identify high-risk patients and provides implications for therapeutic strategies. Currently, the International Prognostic Index (IPI) and National Comprehensive Cancer Network IPI (NCCN-IPI) are the most commonly used tools for predicting outcomes and stratifying cases in clinical studies.3,4 However, these parameters cannot be used to distinguish between extremely high-risk cases and those with heterogeneous biological profiles. Therefore, identifying novel and cheap biomarkers is necessary to predict DLBCL prognosis.
In the past decade, cancer-associated inflammation has been increasingly recognized to play a role in carcinogenesis, cancer progression, and cancer prognosis.5,6 Many inflammation-related indices, such as the lymphocyte-to-monocyte ratio, platelet-to-lymphocyte ratio (PLR), 7 neutrophil-to-lymphocyte ratio (NLR), 8 together with albumin-to-globulin ratio, 9 can be used to predict the prognosis of different tumors. The systemic immune-inflammatory index (SII) was first proposed as a prognostic index for hepatocellular carcinoma in 2014. 10 The SII can be determined as follows: platelet count × neutrophil count/lymphocyte count. Many studies have shown that SII can be used to predict the prognosis of various cancers including pancreatic cancer, 11 thymoma, 12 gastric cancer, 13 glioblastoma, 14 and nasopharyngeal carcinoma (NPC). 15 Previous studies have analyzed the SII in terms of its significance in predicting DLBCL prognosis; however, no consistent findings have been obtained.16–20 For instance, a high SII has been reported to significantly predict the prognosis of DLBCL in certain articles.18,20 Other researchers found that the SII is not significantly related to the survival of patients with DLBCL. 16 Therefore, we comprehensively searched the literature and performed a meta-analysis on the prognostic performance of the SII in DLBCL. Moreover, we explored the relationship between the SII and 10 clinicopathological characteristics of DLBCL.
Materials and methods
Study guideline
The current meta-analysis was performed and reported in line with the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 21 (Supplemental Material 1). This meta-analysis was registered on the INPLASY website (https://inplasy.com/) under the registration number INPLASY202380106. This protocol is available at https://inplasy.com/inplasy-2023-8-0106/.
Ethics statement
Ethics approval was not required owing to the meta-analysis nature of this study and the utilization of anonymized patient information.
Search strategy
PubMed, Web of Science, Embase, and Cochrane Library databases were comprehensively searched from inception to 16 March 2023 using the following terms: (systemic-immune-inflammation index OR SII OR systemic immune-inflammation index) AND (diffuse large B-cell lymphoma OR lymphoma large B-cell OR DLBCL OR lymphoma). The detailed literature strategies for each database are shown in Supplemental Material 2. Only publications in English were included. In addition, references in the included studies were scanned to identify other relevant reports.
Study eligibility criteria
Studies that met the following criteria were considered: (1) DLBCL was confirmed based on histology or pathology, (2) the relationship between SII and survival of patients with DLBCL was provided, (3) hazard ratios (HRs) and 95% confidence intervals (CIs) regarding survival outcomes were available, (4) the threshold of SII was provided, and (5) English studies. Following were the exclusion criteria: (1) reviews, conference abstracts, case reports, letters, and comments; (2) patients with any other cancer before DLBCL; (3) history of an active infectious or inflammatory disorder in the 30 days preceding DLBCL treatment; (4) duplicated cases; and (5) animal studies.
Data collection and quality evaluation
Two researchers (ZF and LS) independently collected information from each qualified study. All disagreements between the two investigators were resolved through discussion and based on consensus. The following data were collected: first author, publication year, country, sample size, study design, age, study period, Ann Arbor stage, treatment strategy, survival endpoint, follow-up, survival analysis type, threshold SII, threshold determination approach, and HRs with 95% CIs. Overall survival (OS) was considered as the primary outcome, whereas progression-free survival (PFS) was considered as the secondary outcome. Moreover, the Newcastle–Ottawa Scale (NOS), which considers the three aspects of selection, comparability, and outcome, was used to evaluate the enrolled study quality, 22 yielding a total score of 0–9 points. Studies with NOS scores ⩾6 are regarded as high-quality studies.
Statistical analysis
We determined the combined HRs and 95% CIs for estimating the SII regarding their significance in predicting the OS and PFS of DLBCL. Correlations between the SII and clinicopathological features of DLBCL were analyzed using odds ratios (ORs) and 95% CIs. Cochran’s Q test along with Higgins I2 statistic was used to detect heterogeneities across the enrolled articles. In the case of obvious heterogeneity (I2 > 50%), a random-effects model was used; otherwise, a fixed-effects model was applied. A subgroup analysis was performed to detect potential sources of heterogeneity and for further investigation. In addition, we used a funnel plot and Begg’s test to detect publication bias. Data were analyzed using the Stata version 12.0 software (Stata Corporation, College Station, TX, USA). Statistical significance was set at p < 0.05.
Results
Study screening
Originally, 72 records were obtained and duplicates were removed to obtain 38 articles (Figure 1). Through title and abstract screening, 32 studies were eliminated because they were irrelevant studies or animal studies. Subsequently, six studies were assessed by reading the full text. One study was excluded because it was not focused on patients with DLBCL. Ultimately, five studies involving 592 patients16–20 were enrolled in this meta-analysis (Figure 1 and Table 1).

PRISMA flow diagram outlining the literature search process.
The baseline characteristic of included studies.

CHOP, cyclophosphamide + doxorubicin + vincristine + prednisone; OS, overall survival; NOS, Newcastle–Ottawa Scale; PFS, progression-free survival; R-CEOP, rituximab + cyclophosphamide + etoposide + vincristine + prednisone; R-CHOP, rituximab + cyclophosphamide + doxorubicin + vincristine + prednisone; ROC, receiver operating characteristic.
Enrolled study features
Table 1 lists the basic features of the selected articles.16–20 All enrolled articles were published in English between 2019 and 2022. All five articles were conducted in China and had a retrospective design,16–20 with a sample size of 28–155 (median, 117). All included studies enrolled DLBCL cases of Ann Arbor stages I–IV. Two studies adopted the R-CHOP regimen for treatment16,19 and three studies used the CHOP/R-CHOP/rituximab, cyclophosphamide, etoposide, vincristine, and prednisone (R-CEOP) strategies.17,18,20 The threshold SII was 428.4–1684.09 (median, 521.5). All included studies adopted the receiver operating characteristic curve to determine the threshold.16–20 Four studies involving 524 patients demonstrated the significance of SII in predicting OS.16–18,20 Four studies involving 475 patients reported a relationship between SII and PFS.16,18–20 Three articles reported HRs and 95% CIs using univariate regression16–18 and two studies employed multivariate regression.19,20 Study quality according to NOS was ⩾6, indicating their high quality (Table 1).
SII and OS within DLBCL
Altogether, four articles involving 524 cases16–18,20 mentioned the association between the SII and OS. Considering the nonsignificant heterogeneity, we adopted the fixed-effects model (I2 = 29.7%, p = 0.234). Our combined data were HR = 3.87, 95% CI: 2.48–6.04, p < 0.001, suggesting the significant relation between a higher SII and worse OS of DLBCL (Figure 2 and Table 2). In addition, we performed a subgroup analysis based on sample size, survival analysis, treatment, and threshold. As shown in Table 2, the subgroup analysis identified a higher SII as an independent factor for predicting OS, regardless of the survival analysis and threshold. In addition, elevated SII still predicted poor OS in DLBCL in studies with sample size ⩾100 (p < 0.001) and studies using CHOP/R-CHOP/ R-CEOP regimen (p < 0.001) (Table 2).

Forest plots of the association between SII and OS in DLBCL patients.
Subgroup analysis of prognostic of SII for OS and PFS in patients with DLBCL.

CHOP, cyclophosphamide + doxorubicin + vincristine + prednisone; DLBCL, diffuse large B-cell lymphoma; OS, overall survival; PFS, progression-free survival; R-CEOP, rituximab + cyclophosphamide + etoposide + vincristine + prednisone; R-CHOP, rituximab + cyclophosphamide + doxorubicin + vincristine + prednisone.
SII and PFS in DLBCL
Four articles comprising 475 cases16,18–20 reported the significance of the SII in predicting PFS in patients with DLBCL. We used a random-effects model because of the obvious heterogeneity (I2 = 80.8%, p = 0.001). Our pooled findings suggested that the higher SII significantly predicted the inferior PFS in DLBCL (HR = 2.38, 95% CI: 1.12–5.08, p = 0.024; Table 2; Figure 3). Moreover, based on subgroup analysis, a higher SII significantly predicted dismal PFS in the following subgroups: sample size ⩾100 (p < 0.001), CHOP/R-CHOP/R-CEOP treatment (p < 0.001), univariate survival analysis (p = 0.001), and threshold SII < 500 (p = 0.005) (Table 2).
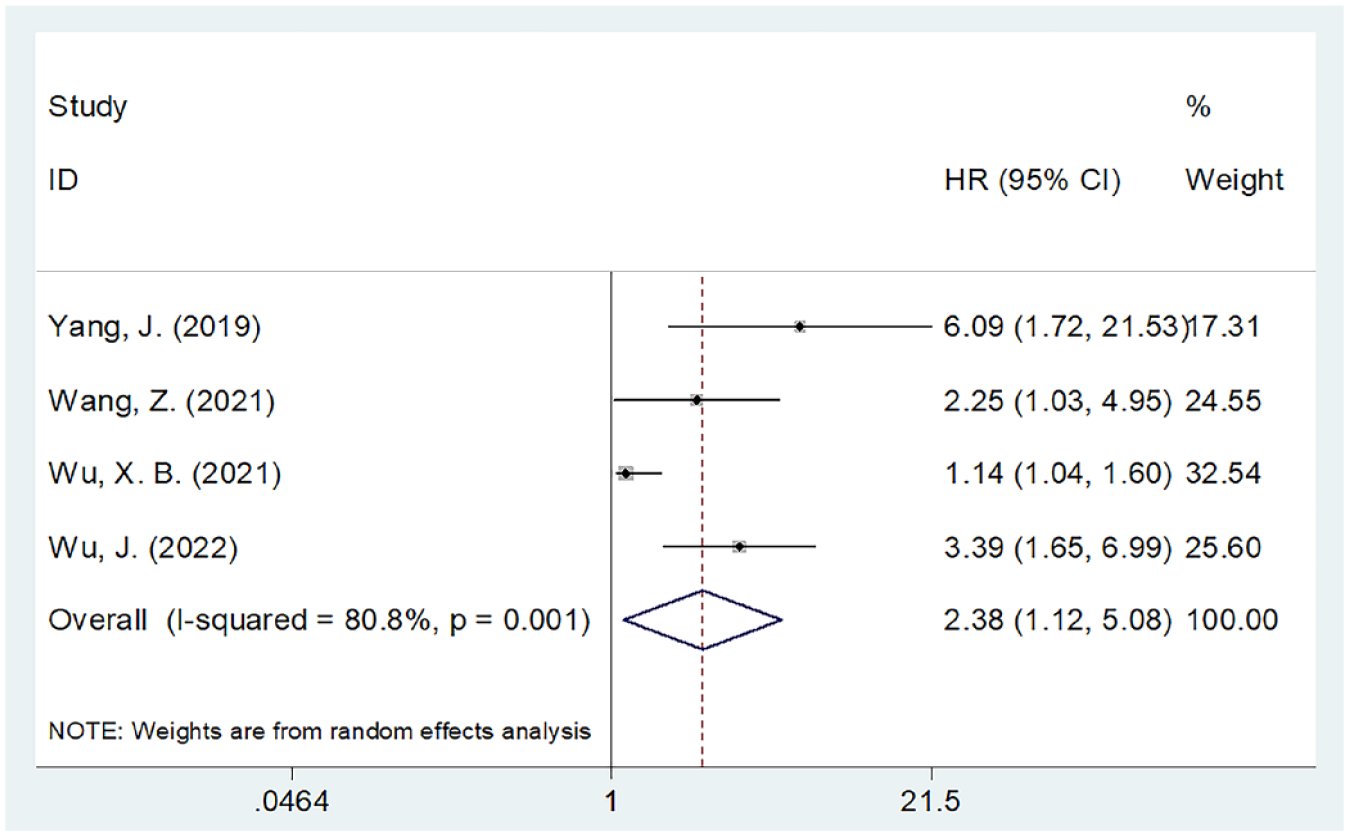
Forest plots of the association between SII and PFS in DLBCL patients.
SII and clinicopathological features within DLBCL
Correlations between the SII and clinicopathological characteristics of DLBCL were analyzed in three studies with 496 patients.17,18,20 As presented in Table 3, Figure 4, and Figure 5, the pooled data showed that the higher SII significantly predicted B symptoms (OR = 2.52, 95% CI: 1.66–3.81, p < 0.001), III–IV Ann Arbor stage (OR = 2.86, 95% CI: 1.84–4.45, p < 0.001), high–intermediate/high NCCN-IPI (OR = 2.25, 95% CI: 1.52–3.31, p < 0.001), increased NLR (OR = 33.76, 95% CI: 17.18–66.35, p < 0.001), and increased PLR (OR = 44.65, 95% CI: 5.80–343.59, p < 0.001). Nonetheless, SII was not notably associated with sex (OR = 1.27, 95% CI: 0.87–1.86, p = 0.215), age (OR = 0.99, 95% CI: 0.67–1.45, p = 0.944), lactate dehydrogenase level (OR = 2.03, 95% CI: 0.34–12.22, p = 0.439), Eastern Cooperative Oncology Group performance status (ECOG PS) (OR = 2.31, 95% CI: 0.73–7.25, p = 0.153), or histology (OR = 1.06, 95% CI: 0.64–1.76, p = 0.820) (Table 3; Figures 4 and 5).
The correlation between SII and clinicopathological factors in patients with DLBCL.

DLBCL, diffuse large B-cell lymphoma; ECOG PS, Eastern Cooperative Oncology Group performance status; GCB, germinal center B-cell like; LDH, lactic dehydrogenase; NLR, neutrophil–lymphocyte ratio; non-GCB, non-germinal center B-cell like; PLR, platelet–lymphocyte ratio; SII, systemic immune-inflammation index.

Forest plots of the relationship between SII and clinicopathological factors in DLBCL patients. (a) Gender (male versus female); (b) age (years) (⩾60 versus <60), (c) B symptoms (presence versus absence), and (d) Ann Arbor stage (III–IV versus I–II).

Forest plots of the relationship between SII and clinicopathological factors in DLBCL patients. (a) LDH (increased versus normal), (b) NCCN-IPI (high–intermediate/high versus low/low-intermediate), (c) ECOG PS (⩾2 versus 0–1), (d) histology (GCB versus non-GCB), (e) NLR (increased versus normal), and (f) PLR (increased versus normal).
Publication bias
Funnel plots and Begg’s test were used to examine potential publication bias. As shown in Figure 6, the funnel plots for OS and PFS were approximately symmetrical. Moreover, Begg’s test suggested the absence of obvious publication bias for OS (p = 0.734) or PFS (p = 0.646).

Funnel plot to assess publication bias: (a) OS and (b) PFS.
Discussion
The SII is an inflammatory marker derived from blood tests and is readily available in the clinical setting. Previous studies have explored the significance of SII in predicting DLBCL prognosis, but their findings are conflicting. In this study, data were collected from five eligible studies with 592 cases, and the prognostic impact of SII on OS and PFS was quantitatively identified. According to our findings, a high SII significantly predicted OS and PFS in patients with DLBCL. Moreover, a higher SII was closely associated with the presence of B symptoms, Ann Arbor stage III–IV, high–intermediate/high NCCN-IPI, increased NLR, and increased PLR in patients with DLBCL. Considering that these clinicopathological features are well-established indicators of disease progression and poor prognosis, increased SII is also a marker for the highly malignant nature of DLBCL. Collectively, a high SII significantly predicted poor survival and was indicative of disease progression in patients with DLBCL. To our knowledge, the current study is the first meta-analysis to investigate the function of the SII in predicting the prognosis of DLBCL.
The SII can be determined according to the neutrophil, platelet, and lymphocyte counts. As a result, an elevated SII may reflect high neutrophil and platelet counts, and a low lymphocyte count, which may contribute to unfavorable outcomes in patients with DLBCL. The mechanisms underlying the role of SII in predicting patient prognosis are as follows: First, neutrophils are a critical part of the nonspecific immune system responsible for inducing inflammation. As neutrophils increase, inflammatory factors such as vascular endothelial growth factor (VEGF) interleukin-8 (IL-8), IL-16, and IL-20 are released, promoting tumor invasion by creating an inflammatory microenvironment. 23 Second, diverse cell factors, including VEGF, epidermal growth factor (EGF), and IL-1β, can be produced when platelets are activated, which promotes cancer development as well as angiogenesis. 24 Increased platelet count accompanies the onset and progression of malignancies, which interacted directly with the circulating tumor cells and facilitated their exosmosis of the tumor cells to the metastasis site. 25 In addition, platelets may prevent cancer cells from being lysed by natural killer cells when they aggregate around tumor cells. 26 Third, by causing cytotoxic cell death while generating cytokines, lymphocytes often act as tumor suppressors by inhibiting tumor cell growth and metastasis. 27 Systemic immune responses can cause lymphocytopenia, reduce lymphocyte activity, and impair innate cellular immunity, resulting in inferior survival. 28 Tumor-infiltrating CD4+ and CD8+ T lymphocytes are identified as factors indicating the dismal prognosis of several cancers.29,30 Therefore, the SII can represent both inflammation and the immune system and is a promising prognostic marker for DLBCL.
Notably, SII is a composite measure that is not routinely used in patients with DLBCL. Furthermore, they are inexpensive and may be significantly affected by comorbidities or acute medical conditions unrelated to DLBCL. Therefore, SII seems to be affected by the immune status and comorbidities of the host. The following points require special attention: First, the patients with DLBCL involved in this meta-analysis did not have a history of another cancer or an active infectious or inflammatory disorder. Second, in clinical practice, SII should be measured in the absence of complications or inflammation in patients with DLBCL. Third, for individual patients with DLBCL, regular detection of the SII is helpful for the timely detection of tumor recurrence and monitoring of prognosis.
In recent years, numerous meta-analyses have demonstrated that the SII is of great significance in predicting the prognosis of various solid tumors.31–35 Zhang et al. 31 demonstrated that a higher SII indicated poor OS and PFS in glioma cases in their meta-analysis that included eight articles. As revealed by Zeng et al., 35 a high SII remarkably predicted poor OS and PFS in patients with NPC in a meta-analysis comprising 2169 subjects. According to a meta-analysis comprising 2132 patients, a higher pretreatment SII predicted poor OS and inferior cancer-specific survival/disease-free survival (DFS)/PFS and progression-free cancer. 36 Moreover, as indicated by Wang and Ni, 33 a higher SII predicts inferior OS and PFS in patients with cancer undergoing treatment with immune checkpoint inhibitors. In addition, according to a meta-analysis comprising 6925 patients, an increased SII remarkably predicted poor OS and worse DFS in gastric cancer. 37
Limitations
Some limitations of this study should be noted. First, the sample size is relatively small. Although we performed a comprehensive literature search, only five eligible articles were included, with an overall sample size of 592. Second, all included studies were performed in China. Although we restricted the publication language to English, each of our enrolled articles was from China. Third, all of the enrolled articles had a retrospective design, possibly introducing a selection bias. Consequently, large-scale cross-regional prospective trials are still required in the future to validate our findings.
Conclusion
In conclusion, according to this meta-analysis, a higher SII significantly predicted inferior OS and PFS in patients with DLBCL. Furthermore, an increased SII significantly correlated with some clinicopathological features representing the disease progression of DLBCL. SII can be used to predict the prognosis of patients with DLBCL in clinical practice.
Supplemental Material
sj-doc-1-tah-10.1177_20406207231208973 – Supplemental material for Prognostic and clinicopathological impacts of systemic immune-inflammation index on patients with diffuse large B-cell lymphoma: a meta-analysis
Supplemental material, sj-doc-1-tah-10.1177_20406207231208973 for Prognostic and clinicopathological impacts of systemic immune-inflammation index on patients with diffuse large B-cell lymphoma: a meta-analysis by Zaijing Fan and Lihong Shou in Therapeutic Advances in Hematology
Supplemental Material
sj-doc-1-tah-10.1177_20406207231208973 – Supplemental material for Prognostic and clinicopathological impacts of systemic immune-inflammation index on patients with diffuse large B-cell lymphoma: a meta-analysis
Supplemental material, sj-doc-1-tah-10.1177_20406207231208973 for Prognostic and clinicopathological impacts of systemic immune-inflammation index on patients with diffuse large B-cell lymphoma: a meta-analysis by Zaijing Fan and Lihong Shou in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
