Abstract
Objective
The objective of this study was to examine the associations of the systemic immune-inflammation index and the systemic inflammation response index with all-cause and cardiovascular mortality in cancer and noncancer populations.
Methods
We analyzed data from 42,503 adults in the National Health and Nutrition Examination Survey using Cox models and restricted cubic spline analyses.
Results
Compared with the lowest tertile, the highest systemic immune-inflammation index tertile was associated with increased risks of all-cause mortality (hazard ratio, 1.24; 95% confidence interval: 1.16–1.32) and cardiovascular mortality (hazard ratio, 1.29; 95% confidence interval: 1.15–1.45). The highest systemic inflammation response index tertile demonstrated similar increases in all-cause mortality (hazard ratio, 1.33; 95% confidence interval: 1.24–1.43) and cardiovascular mortality (hazard ratio, 1.42; 95% confidence interval: 1.25–1.61). Risks increased across tertiles, and dose–response patterns were supported by spline analyses. Estimates were greater among participants with cancer than among those without cancer.
Conclusions
Elevated levels of systemic immune-inflammation index and systemic inflammation response index were associated with increased risks of all-cause and cardiovascular mortality, particularly among participants with cancer, supporting their potential use as low-cost screening tools for risk stratification. Given the observational design and single baseline measurement, residual confounding and measurement error remain possible; prospective validation is warranted.
Keywords
Introduction
The systemic immune-inflammation index (SII) and systemic inflammation response index (SIRI) have emerged as readily accessible, objective biomarkers reflecting levels of systemic inflammation. Both indices are derived from routine blood test parameters, including neutrophils, monocytes, lymphocytes, and platelets, each representing distinct inflammatory and immune mechanisms. 1 These indices have gained attention in clinical research because of their simplicity, cost-effectiveness, and reliability, enabling clinicians to effectively assess inflammatory states across diverse patient populations. Clinical studies consistently report that elevated levels of SII and SIRI are associated with poor prognostic outcomes across numerous diseases, particularly cancer and cardiovascular diseases (CVD).2–4 Moreover, the strength and nature of these associations may vary significantly across populations and demographic groups, with certain populations demonstrating more robust correlations, suggesting population-specific inflammatory responses. 5
Systemic inflammation plays a pivotal and complex role in the pathogenesis, progression, and prognosis of numerous chronic diseases. 6 It acts through diverse pathological pathways that influence the onset and severity of various health conditions. In the context of cancer, systemic inflammation often leads to immune dysregulation, creating an environment conducive to tumor growth, metastatic dissemination, and increased thrombotic risks. Elevated SII levels, in particular, are strongly associated with increased mortality risk in patients with cancer, mediated through mechanisms such as increased platelet activation, thrombosis, and immune suppression. 7 This association underscores the crucial interplay between inflammatory processes and tumor biology. Conversely, in noncancer populations, elevated SIRI levels have been strongly associated with an increased incidence of cardiovascular events, including myocardial infarction and stroke. 8 These associations suggest that inflammation-driven pathways significantly contribute to vascular damage and the progression of atherosclerosis, highlighting the broader impact of systemic inflammation beyond oncological contexts.9,10 Nevertheless, despite these clear associations, the precise biological mechanisms linking elevated SII and SIRI levels to increased mortality in these distinct patient populations remain inadequately characterized.
Despite growing evidence linking SII and SIRI to adverse outcomes, 5 critical knowledge gaps remain. Previous studies have largely examined these markers within single populations, 4 either cancer cohorts or general community samples, without directly comparing their prognostic utility across groups with fundamentally different inflammatory milieus. Notably, no large-scale study has simultaneously evaluated the performance of SII and SIRI in cancer compared with that in noncancer populations, despite marked differences between these groups in baseline immune activation, myelopoiesis, treatment exposures, and competing mortality risks. Recent studies have highlighted that inflammatory biomarkers may behave differently across demographic and clinical subgroups.5,7,8 However, it remains unclear whether these variations translate into distinct mortality risks across different populations. This study provides the first large-scale head-to-head evaluation of SII and SIRI across cancer and noncancer populations, examining specific inflammatory pathways and their differential prognostic implications for all-cause and cardiovascular mortality, with the aim of refining risk stratification and informing targeted anti-inflammatory strategies.
Materials and methods
Study population
This prospective study utilized fully deidentified data from the National Health and Nutrition Examination Survey (NHANES), a comprehensive survey designed to assess the health and nutritional status of adults and children in the United States from 1999 to 2018. 11 NHANES employs a complex, multistage probability sampling method to generate data representative of the US population, encompassing various biomarkers, lifestyle factors, disease incidence, and mortality information. 12 All participants provided written informed consent, and NHANES protocols were reviewed and approved by the National Center for Health Statistics (NCHS) Ethics Review Board for each 2-year survey cycle. 11 The NHANES database is publicly available and contains fully deidentified data (https://www.cdc.gov/nchs/nhanes/). The study was conducted in accordance with the Declaration of Helsinki (1975, as revised in 2024) and reporting adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13
To ensure data validity, we first included individuals with complete information on SII, SIRI, and mortality (N = 53,199). We then excluded individuals without a cancer diagnosis at baseline (N = 3717) and those with missing covariate data (N = 6979). After applying these exclusion criteria, a total of 42,503 participants were included in the analysis. (Supplementary Figure 1).
Assessment of SII and SIRI
In the NHANES cohort, complete blood counts were measured using an automated blood analyzer (Coulter DxH 800), which provided platelet, neutrophil, and lymphocyte counts, reported in thousands of cells per microliter (1000 cells/μL). All hematologic measures used to compute SII and SIRI were collected at NHANES Mobile Examination Centers following standardized protocols. Specimen handling, instrument calibration, internal quality controls, and technician performance monitoring followed procedures outlined in the NHANES laboratory procedures manual and assay-specific laboratory method documents. Distributions and outliers were evaluated during each survey cycle prior to public release. The SII was calculated as the product of platelet and neutrophil counts divided by the lymphocyte count, whereas the SIRI was defined as the product of monocyte and neutrophil counts divided by the lymphocyte count.
Assessment of all-cause and cardiovascular mortality
In this study, the primary outcomes of interest were all-cause and cardiovascular mortality. To determine mortality outcomes, NCHS used a probabilistic matching algorithm to link publicly available NHANES mortality files with the National Death Index (NDI) data until 31 December 2019. Causes of death were classified according to the 10th revision of the International Classification of Diseases (ICD-10), with cardiovascular mortality defined as deaths due to heart disease (codes I00–I09, I11, I13, and I20–I51) or cerebrovascular disease (codes I60–I69). Follow-up time was calculated as the interval from the baseline examination date to either the last known date of survival or the date of death recorded in the mortality files.
Covariates
In this study, covariates included sex (male or female), age (modeled as a continuous variable), race/ethnicity (White or other races), marital status (married or partnered, separated/widowed/divorced, or never married), and educational level (<high school, high school, or >high school). Anthropometric and socioeconomic factors included body mass index (BMI; <18.5, 18.5–24.9, 25.0–29.9, and ≥30) and the poverty–income ratio (PIR; categorized into quartiles). Lifestyle factors included smoking status (never, former, or current). Baseline self-reported diseases included hypertension, diabetes, and CVD. Additionally, biomarkers were assessed as continuous variables, including glycated hemoglobin (HbA1c), alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatinine, triglycerides, and high-density lipoprotein (HDL) cholesterol.
Statistical analysis
All statistical analyses were conducted using R software (version 4.2.2), with a two-sided p value of <0.05 considered statistically significant. In accordance with previous studies on SII and SIRI, all analyses were performed without weighting.14–16 Descriptive statistics were used to summarize baseline characteristics of participants, stratified by cancer and noncancer status. Multivariable Cox proportional hazards models were employed to examine the associations between SII, SIRI, and risks of all-cause and cardiovascular mortality, with results reported as hazard ratios (HRs) and 95% confidence intervals (CIs). Model 1 was adjusted for age, sex, ethnicity, BMI, marital status, educational level, PIR, smoking status, and baseline comorbidities, including hypertension, diabetes, and CVD. Model 2 included additional adjustments for biochemical markers, including HbA1c, ALT, AST, creatinine, triglycerides, and HDL cholesterol. Restricted cubic spline (RCS) regression models were used to examine the potential nonlinear relationships between SII, SIRI, and all-cause or cardiovascular mortality. HRs for both outcomes were visualized based on Model 2, among participants with and without cancer.
Sensitivity analyses were conducted to assess the robustness of the findings. First, the associations between SII, SIRI, and all-cause and cardiovascular mortality were examined after incorporating sampling weights into models. Next, to address potential bias from missing data, missing covariate data were imputed using the multiple imputation by chained equations approach, with 100 iterations generating 5 imputed datasets. 17 Third, we employed competing risk regression models to evaluate the associations of SII and SIRI with cardiovascular mortality, treating noncardiovascular mortality as competing events. Fourth, subgroup analyses were performed to assess the associations across covariate-defined subgroups. Finally, we accessed the dose–response relationships between SII, SIRI, and the mortality outcomes after excluding participants who died within the first 2 years of follow-up.
Results
Baseline characteristics
This study included 42,503 participants (20,525 males and 21,978 females), whose baseline characteristics are presented in Table 1. Among them, 9.0% (N = 3822) had a history of cancer and differed significantly from those without cancer in terms of demographic and clinical characteristics. They were generally older; predominantly White; and more likely to be separated, widowed, or divorced. Participants with cancer exhibited markedly elevated systemic inflammation markers, as reflected by higher levels of SII and SIRI. Additionally, they had higher HbA1c and creatinine levels but lower ALT levels. The prevalence of hypertension, diabetes, and CVD was also higher among these participants. Baseline characteristics of participants across tertiles of SIRI and SII are presented in Supplementary Tables 1–2, and the missing proportion of these baseline characteristics is shown in Supplementary Table 3.
Characteristics of participants with or without cancer at baseline.
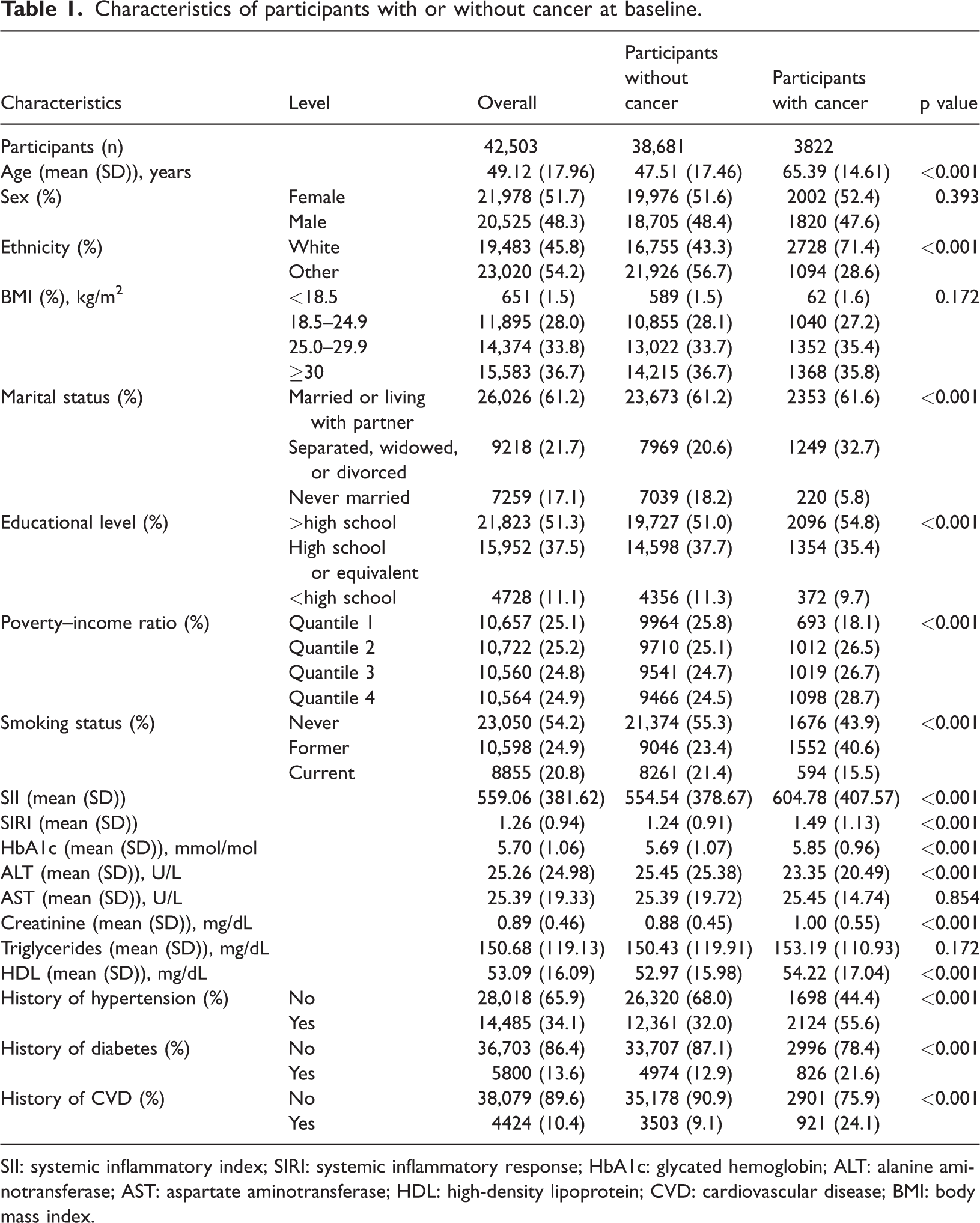
SII: systemic inflammatory index; SIRI: systemic inflammatory response; HbA1c: glycated hemoglobin; ALT: alanine aminotransferase; AST: aspartate aminotransferase; HDL: high-density lipoprotein; CVD: cardiovascular disease; BMI: body mass index.
Association between SII and mortality
Higher SII levels were significantly associated with increased risks of all-cause and cardiovascular mortality. In Model 1 (unadjusted for biochemical markers), participants in the highest SII tertile (T3) showed modestly elevated risks compared to those in the lowest tertile (T1). After adjustment for biochemical markers in Model 2, these associations strengthened substantially. Particularly, participants in the highest SII tertile had a 24% increased risk of all-cause mortality (HR, 1.24; 95% CI: 1.16–1.32; p for trend < 0.001) and a 29% increased risk of cardiovascular mortality (HR, 1.29; 95% CI: 1.15–1.45; p for trend < 0.001). Moreover, incremental increases across SII tertiles were consistently associated with elevated risks of all-cause and cardiovascular mortality (all-cause mortality: HR, 1.12; 95% CI: 1.09–1.16; cardiovascular mortality: HR, 1.14; 95% CI: 1.08–1.21; both p < 0.001).
Figure 1 illustrates clear associations between SII levels and mortality outcomes in cancer and noncancer populations. Among participants with cancer, the association with all-cause mortality was nonlinear, characterized by sharply increasing risks at higher SII levels, whereas a linear association was observed for cardiovascular mortality. Participants without cancer demonstrated nonlinear trends for both outcomes, with marked risk escalation at higher SII levels. At comparable SII values, participants with cancer consistently exhibited higher mortality risks than their noncancer counterparts.
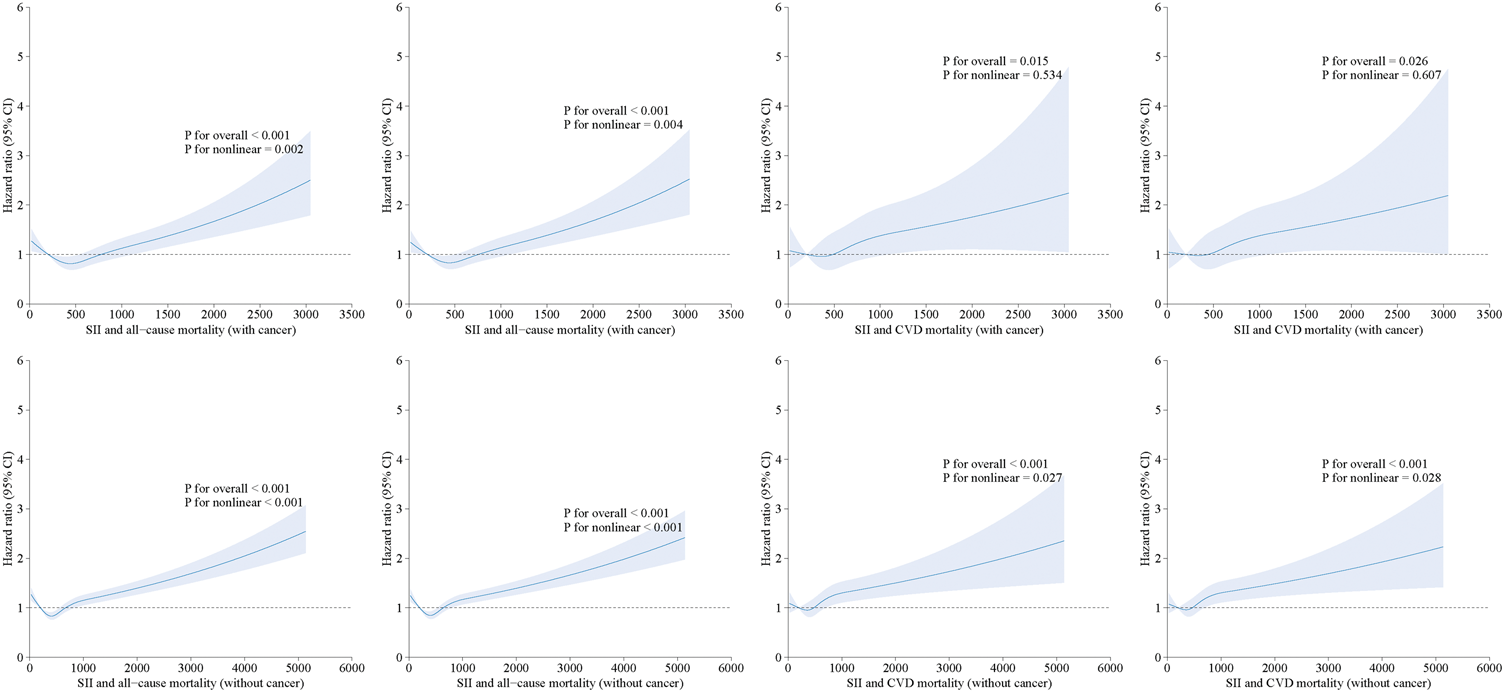
Association between systemic inflammatory index (SII) and all-cause mortality and cardiovascular mortality using a restricted cubic spline regression model. Graphs show HRs for cardiovascular mortality and all-cause mortality adjusted for age; sex; ethnicity; BMI; marital status; educational level; PIR; smoking status; and baseline prevalence of high blood pressure, diabetes, and cardiovascular disease (including congestive heart failure, coronary heart disease, angina, heart attack, and stroke), HbA1c, ALT, AST, creatinine (mg/dL), triglycerides (mg/dL; refrigerated samples), and HDL cholesterol (mg/dL). Data were fitted by a restricted cubic spline Cox proportional hazards regression model, and the model was conducted with four knots at the 5th, 35th, 65th, and 95th percentiles of SII (reference is the 5th percentile). Solid lines indicate HRs, and shadow shape indicate 95% CIs. ALT: alanine aminotransferase; AST: aspartate aminotransferase; BMI: body mass index; CIs: confidence intervals; HbA1c: glycated hemoglobin; HDL: high-density lipoprotein; HRs: hazard ratios; PIR: poverty–income ratio; SII: systemic inflammatory index; CVD: cardiovascular disease.
Association between SIRI and mortality
Similar results were observed for SIRI. In Model 1, which excluded biochemical markers, higher SIRI levels were associated with increased risks of all-cause and cardiovascular mortality. With the inclusion of biochemical markers in Model 2, these associations became more pronounced. Participants in the highest SIRI tertile (T3) had a 33% higher risk of all-cause mortality (HR, 1.33; 95% CI: 1.24–1.43; p for trend <0.001) and a 42% higher risk of cardiovascular mortality (HR, 1.42; 95% CI: 1.25–1.61; p for trend < 0.001) compared to those in the lowest tertile (Table 2). Moreover, each tertile increment in SIRI was associated with significantly increased risks of all-cause and cardiovascular mortality in Model 2 (all-cause mortality: HR, 1.17; 95% CI: 1.13–1.21; cardiovascular mortality: HR, 1.21; 95% CI: 1.13–1.28; both p < 0.001) (Table 2).
The association between systemic inflammatory index and systemic inflammatory response with all-cause mortality and cardiovascular mortality.

HR: hazard ratio; CI: confidence interval; SII: systemic inflammatory index; SIRI: systemic inflammatory response.
SII tertiles: Tertile 1 ≤387.00; Tertile 2, 387.00–600.23; Tertile 3 ≥600.23; SIRI tertiles: Tertile 1 ≤0.81; Tertile 2, 0.81–1.33; Tertile 3 ≥1.33. Model 1 was adjusted for age; sex; ethnicity; body mass index (BMI); marital status; educational level; poverty–income ratio (PIR); smoking status; and baseline prevalence of high blood pressure, diabetes, and cardiovascular disease (CVD; including congestive heart failure, coronary heart disease, angina, heart attack, and stroke). Model 2 was further adjusted for glycated hemoglobin (HbA1c), alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatinine (mg/dL), triglycerides (mg/dL; refrigerated samples), and high-density lipoprotein (HDL) cholesterol (mg/dL).
Figure 2 illustrates the dose–response relationships between SIRI and the risks of all-cause and cardiovascular mortality in participants with and without cancer. Clear dose–response associations were observed for the two outcomes in both groups. Among participants with cancer, these associations were linear, with HRs increasing steadily as SIRI increased. In contrast, participants without cancer demonstrated nonlinear associations, with sharper risk increases at higher SIRI levels. At comparable SIRI values, participants with cancer generally exhibited higher HRs than those without cancer.

Association between systemic inflammatory response (SIRI) and all-cause mortality and cardiovascular mortality using a restricted cubic spline regression model. Graphs show HRs for cardiovascular mortality and all-cause mortality adjusted for age; sex; ethnicity; BMI; marital status; educational level; PIR; smoking status; and baseline prevalence of high blood pressure, diabetes, and cardiovascular disease (including congestive heart failure, coronary heart disease, angina, heart attack, and stroke), HbA1c, ALT, AST, creatinine (mg/dL), triglycerides (mg/dL; refrigerated samples), and HDL cholesterol (mg/dL). Data were fitted by a restricted cubic spline Cox proportional hazards regression model, and the model was conducted with four knots at the 5th, 35th, 65th, and 95th percentiles of SIRI (reference is the 5th percentile). Solid lines indicate HRs, and shadow shape indicate 95% CIs. ALT: alanine aminotransferase; AST: aspartate aminotransferase; BMI: body mass index; CIs: confidence intervals; CVD: cardiovascular disease; HbA1c: glycated hemoglobin; HDL: high-density lipoprotein; HRs: hazard ratios; PIR: poverty–income ratio; SII: systemic inflammatory index.
Sensitivity analyses
Sensitivity analyses were conducted to evaluate the robustness of our findings. Survey-weighted Cox models provided similar directions and magnitudes for the associations of SII and SIRI with all-cause and cardiovascular mortality, with wider 95% CIs as expected under the complex design (Supplementary Table 4). To address potential bias due to missing data, multiple imputation using chained equations was performed; the reanalyzed associations between SII, SIRI, and mortality outcomes are presented in Supplementary Table 5. Competing risk models further supported the associations between inflammatory markers and mortality outcomes (Supplementary Table 6). Subgroup analyses stratified by cancer and noncancer populations demonstrated consistent associations across outcomes (Supplementary Tables 7–14). To correct for multiple testing, the Benjamin–Hochberg method was applied to adjust the p values using the false discovery rate (FDR). 18 Finally, exclusion of participants who died within the first 2 years of follow-up did not materially alter the results, and RCS analyses confirmed the stability of nonlinear associations (Supplementary Figures 2–3).
Discussion
This study examined the associations of SII and SIRI with all-cause and cardiovascular mortality in participants with and without cancer. The results indicate that higher levels of SII and SIRI were significantly associated with increased risks of both outcomes across the entire study population. Notably, participants with cancer exhibited markedly higher risks than those without cancer, highlighting their greater vulnerability to systemic inflammation. These findings underscore that SII and SIRI can serve as effective predictors of cardiovascular and all-cause mortality, aiding clinicians in identifying high-risk populations.
Our results extend and refine prior observations linking inflammatory markers to adverse outcomes in cancer and noncancer populations. Numerous previous studies have confirmed close associations of SII and SIRI with the risk of cardiovascular mortality,2,3 highlighting that higher levels of these indices are strongly associated with increased cardiovascular and all-cause mortality in the general population. 4 In line with these reports, we observed that elevated SII and SIRI were associated with mortality risk, reinforcing the role of inflammation as a common risk substrate for adverse outcomes. Moreover, our findings in participants with cancer are consistent with those of oncology studies showing that elevated SII is indicative of a worse prognosis across multiple cancer types. For example, a meta-analysis reported that patients with cancer and high SII had a higher hazard of overall mortality compared to those with low SII. 7 Similarly, a prior study suggested that elevated SII predicts poorer survival and treatment response among patients with cancer receiving immunotherapies. 5 However, evidence directly comparing the prognostic value of these markers between participants with and without cancer remains limited. By leveraging a large, nationally representative sample from the NHANES database, our study extends the existing knowledge base and underscores the need for population-specific risk assessment and intervention strategies.
One of the key observations from our comparative analysis is that, at comparable SII and SIRI levels, participants with cancer exhibited significantly higher mortality risks than those without cancer. Several complementary pathways may account for the stronger associations observed among participants with cancer. Chronic tumor–driven inflammation reshapes myelopoiesis and skews circulating leukocytes toward neutrophils and immunosuppressive monocyte–derived populations as well as enhances platelet activation and tumor-associated thrombosis. 19
These processes amplify the thromboinflammatory axis captured by SII and the innate immune axis captured by SIRI, thereby magnifying mortality risk at comparable index levels. 20 Tumor-derived cytokines such as interleukin-6 (IL-6) and granulocyte–macrophage colony-stimulating factor (GM-CSF) promote emergency myelopoiesis and the expansion of myeloid-derived suppressor cells, which dampen antitumor immunity and sustain systemic inflammation. 21 In participants with cancer, anticancer therapies can modify hematologic profiles through cytopenia and prothrombotic states and may interact with preexisting cardiometabolic risk, thereby increasing vulnerability to cancer progression and cardiovascular events. 22 In participants without cancer, elevated values of both indices are consistent with low-grade systemic inflammation that contributes to atherosclerosis progression and atherothrombotic events. Higher SII, which integrates neutrophils and platelets relative to lymphocytes, likely reflects a prothrombotic and proinflammatory milieu characterized by platelet activation, platelet–leukocyte aggregates, impaired endothelial repair, and a greater propensity to microthrombosis, all of which are associated with acute coronary and cerebrovascular events. 23 Higher SIRI, which emphasizes the neutrophil–monocyte axis relative to lymphocytes, aligns with innate immune activation in the vessel wall, including monocyte recruitment and differentiation into foam cells, activation of the NLRP3 inflammasome, IL-1β and IL-6 signaling, neutrophil extracellular trap formation, and plaque destabilization.23,24 Taken together, SII may be more sensitive to platelet-driven atherothrombotic activity, whereas SIRI may better capture myeloid-driven vascular inflammation. Their complementary biological profiles provide a coherent explanation for the observed dose–response relationships and support their use for risk stratification in the general population.6,25 These findings highlight that although SII and SIRI are robust markers of inflammation and mortality risk, the biological processes they reflect may manifest differently in cancer versus noncancer settings. Moreover, our results emphasize the multifactorial nature of systemic inflammation. Chronic low-grade inflammation is recognized as a common substrate across diverse chronic conditions, with potential effects on immune homeostasis, metabolic regulation, and cardiovascular health. 6 Therefore, assessment of SII and SIRI can help clinicians identify individuals with heightened inflammatory burden and prioritize personalized management strategies, including anti-inflammatory interventions, lifestyle modifications, or closer surveillance for cardiovascular complications.
To the best of our knowledge, this study is among the first large-scale investigations to evaluate the comparative prognostic value of SII and SIRI in cancer and noncancer populations using longitudinal data. Our dose–response analyses and stratified models further strengthen the inference that SII and SIRI, even at moderately elevated levels, are associated with worse survival outcomes. As inexpensive indices derived from routine blood counts, SII and SIRI may function as cost-effective tools for risk stratification and prioritization of monitoring, particularly in high-risk groups such as patients with cancer, and complement established assessment frameworks.
Nonetheless, several limitations must be noted. First, although we applied extensive covariate adjustments, the possibility of residual confounding cannot be entirely excluded because of unmeasured or incompletely measured variables. Second, we only examined the independent effects of SII and SIRI using a single baseline measurement. Future studies should assess their combined predictive value and evaluate repeated measurements to capture temporal changes. Third, the majority of participants were White, which may limit the generalizability of these findings to more diverse populations. Fourth, we did not apply the NHANES sampling weights. Although our associations are internally valid under model assumptions, generalizability to the national population may be limited. Future analyses in other large-scale cohorts can complement the present estimates. Fifth, detailed information on cancer types, cardiovascular subtypes, disease stage, and treatment history was unavailable. Future studies using other registry datasets can provide more accurate risk estimation. Sixth, misclassification of cause of death may occur even with Fine–Gray competing risk modeling, particularly among patients with cancer. Any such error is likely nondifferential outcome misclassification with respect to SII and SIRI, which would bias associations toward the null and is unlikely to materially alter our conclusions. Seventh, immortal time bias may occur because participants must survive from diagnosis to cohort entry. However, this nondifferential selection is expected to bias estimates toward the null and is unlikely to influence our main results. Finally, detailed information on therapies that modulate circulating counts (e.g. corticosteroids, immunosuppressants, cytotoxic chemotherapy, and radiotherapy) was not available; therefore, these exposures were not fully controlled. This limitation may introduce residual confounding and nondifferential exposure misclassification of SII and SIRI. The measurement-related component would be expected to attenuate true associations, whereas confounding could bias estimates in either direction. Future studies incorporating treatment timing and dosing information will be necessary to quantify these effects.
Future prospective cohort studies and clinical trials should aim to elucidate the underlying mechanisms by which SII and SIRI contribute to heightened mortality risk, particularly in oncologic settings. Investigations integrating repeated inflammatory markers, genetic predispositions, and treatment modalities can offer a more comprehensive understanding of the interplay between chronic inflammation and disease progression. Additionally, determining whether interventions, such as targeted anti-inflammatory therapies, immune checkpoint inhibitors, or dietary modifications, can effectively modulate SII and SIRI and thereby improve survival remains a critical research gap. Addressing these questions will enhance our capacity to translate these markers into actionable strategies for cardiovascular risk reduction and cancer management.
Conclusion
In this study, elevated SII and SIRI were associated with higher risks of all-cause and cardiovascular mortality among participants with and without cancer, with stronger associations observed among those with cancer. These findings reinforce systemic inflammation as a common risk substrate and indicate that SII and SIRI may help identify individuals at heightened risk. However, as this was an observational analysis based on a single baseline measurement and lacking detailed tumor and treatment information, residual confounding, measurement error, and possible misclassification of cause of death cannot be excluded. The findings require validation in prospective, registry-linked, and mechanistic studies. Given that SII and SIRI are inexpensive indices derived from routine blood counts, they may serve as cost-effective screening tools to facilitate early identification and prioritization of high-risk individuals in clinical and public health settings.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251412972 - Supplemental material for Systemic immune-inflammation index and systemic inflammation response index as predictors of mortality in participants with and without cancer
Supplemental material, sj-pdf-1-imr-10.1177_03000605251412972 for Systemic immune-inflammation index and systemic inflammation response index as predictors of mortality in participants with and without cancer by Xiaomei Hu, Hemei Wu, Tongling Liu, Ying Liao, Hui Zeng, Fengming Zou, Jing Zhou and Xiao Liu in Journal of International Medical Research
Footnotes
Acknowledgments
We thank the NHANES staff, investigators, and participants.
Author contributions
XH conducted research and drafted the manuscript. HW and TL participated in data analysis. YL and HZ drafted the initial manuscript. FZ reviewed and revised subsequent drafts of the manuscript. All authors critically reviewed and approved the final manuscript, agreeing to take full responsibility for the integrity and accuracy of the work. All authors contributed to the article and approved the submitted version.
Additional information
Declaration of conflicting interests
The authors declare no competing interests.
Ethics approval and consent to participate
Clinical trial number: not applicable. The NHANES obtained approval from the National Centre for Health Statistics Research Ethics Review Board and was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Each participant gave written informed consent agreement when they were enrolled in the NHANES, and the National Center for Health Statistics’ ethics review board approved the study.
Funding
This work has received the financial support provided by the Joint Science and Technology Funding Project of the Zunyi Bureau of Industry and Science and Technology and The Second Affiliated Hospital of Zunyi Medical University (HZ(2024)427), as well as the Guizhou Provincial Science and Technology Project (QKHCG-LC[2024]037). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
